Abstract
Advances in mechanobiology have suggested that physiological and pathological angiogenesis may be differentiated based on the ways in which the cells interact with the extracellular matrix (ECM) that exhibits partially different mechanical properties. This warrants investigating the regulation of ECM stiffness on cell behavior using angiogenesis assays. In this article, we report the application of the technique of active manipulation of ECM stiffness to study in vitro angiogenic sprouting of human microvascular endothelial cells (HMVECs) in a microfluidic device. Magnetic beads were embedded in the ECM through bioconjugation (between the streptavidin-coated beads and collagen fibers) in order to create a pretension in the ECM when under the influence of an external magnetic field. The advantage of using this magneto-microfluidic system is that the resulting change in the local deformability of the collagen fibers is only apparent to a cell at the pericellular level near the site of an embedded bead, while the global intrinsic material properties of the ECM remain unchanged. The results demonstrate that this system represents an effective tool for inducing noninvasively an external force on cells through the ECM, and suggest the possibility of creating desired stiffness gradients in the ECM for manipulating cell behavior in vitro.
Keywords
Introduction
The microenvironment of cells influences their behavior in migration, proliferation, and differentiation. Two groups of factors, namely, the soluble cues and the mechanical cues, exert such influence. Soluble cues consist of chemical growth factors, such as the vascular endothelial growth factor (VEGF); metabolites; and dissolved gases, while mechanical cues involve composition, architecture, and mechanical properties of the extracellular matrix (ECM). It has been shown that ECM stiffness, an insoluble cue, influences many types of cell behavior. Migration of endothelial cells (ECs) during angiogenesis in particular is significantly influenced by the stiffness of the ECM.
For angiogenesis, changes in endothelial capillary cell shape and function can be produced by changing ECM elasticity, adhesivity, or topography; by applying mechanical stresses; or by altering cell-generated traction forces.1–6 There has been evidence that stress-induced distortion of the capillary cell cytoskeleton regulates Rho activity by controlling its upstream inhibitor, p190RhoGAP, which binds to the transcription factor TFII-I.7,8 TFII-I binds to the initiator (Inr) region of the VEGFR2 gene promoter, which is the main receptor for the growth factor VEGF and essential for angiogenesis. It has been shown that an appropriate level of ECM stiffness may be required for optimal VEGFR2 expression and vascular development in vitro and in vivo. Furthermore, sudden changes in the elasticity of the ECM will cause a change in the active growth and differentiation of functional vascular networks in vivo. 9
Methods are available for altering the ECM stiffness in order to observe in vitro cell behavior. The three main methods are changing collagen concentration, varying the pH value of the ECM, and varying the temperature of the ECM. It has been reported that changing the collagen concentration to 0.66, 1.5, 3, and 5 mg/mL yielded a stiffness of 13.1, 49.9, 290, and 189 Pa, respectively. 10 In another approach, the ECM pH value is varied, as the solution acidity during fibrillogenesis affects collagen stiffness. It has been shown that the Young’s modulus of an ECM sample changed from 0.5 to 5.5 MPa when the pH value was reduced from 8.5 to 6 at 37 °C. 11 Changing the temperature of an ECM sample can also lead to a change in its stiffness. The Young’s modulus of an ECM sample has been shown to increase with polymerization temperature, for example, from 0.3 Pa at 4 °C to 22.7 Pa at 37 °C. 12 These reported methods mainly manipulate soluble cues to vary the initial ECM stiffness. The ECM stiffness after fibrillogenesis is then assumed to constant. However, most ECMs in vivo have a stiffness that varies with time. Moreover, changes in the stiffness of ECM effected by methods based on soluble cues are irreversible.
Manipulating ECM stiffness by mechanical means offers an attractive alternative. Recent research has demonstrated the possibility of changing the mechanical and rheological properties of highly elastic polymer embedded with magnetic particles in the matrices by the application of an external magnetic field. The magnetic field induces a magnetic moment on each of the micromagnetic particles embedded in the polymer matrices, resulting in a buildup of forces within the polymer matrices. It had been reported that these materials show significant changes in their mechanical properties under the influence of magnetic fields.13,14 Similarly, the ECM is also a polymer matrix made up of many carbohydrate polymers attached to glycoproteins to form proteoglycans. 15
The technique of active manipulation of ECM stiffness involves embedding in the ECM magnetic beads (that bind onto these polymer chains) and then applying an external magnetic field to manipulate the stiffness of the ECM. 16
Varying the strength of the magnetic field results in change in the stiffness of the ECM. In particular, a stronger magnetic field will increase the magnetic forces acting on the magnetic beads, resulting in greater resistance of the ECM to deformation in the direction opposing these magnetic forces. This technique of active manipulation of ECM stiffness enables investigation of how cells respond to such resistance to ECM deformation. Specifically, when applying this technique in the context of chemotaxis, if the magnetically induced force gradient is set to oppose the chemical gradient, cells (as they form focal adhesions in order to cleave through the ECM) will experience a stronger resistance to deformation in the ECM, which in turn may affect their behavior. It has been shown that cells in general, and ECs in particular, are highly mechanosensitive and react to changes in external forces and in the compliance of their microenvironment.17,18
The emergence of microfluidic devices has enabled the creation of chemical gradients in vitro in order to observe cell migration during chemotaxis. 19 This is typically achieved by fabricating devices having a gel region, for cell seeding, flanked by two media channels that can be filled with chemical cue (such as VEGF) of different concentrations to create a chemical gradient across the gel region.20,21 Recently, mechanical gradients have also been incorporated in such devices to study mechanotransduction of various cell types by introducing a shear flow or by changing the stiffness of the gel region.22–24
In the work reported in this article, we studied the effect of ECM stiffness on angiogenic sprouting using a magneto-microfluidic system. We applied the technique of active manipulation of ECM stiffness and studied its effect on in vitro angiogenic sprouting of HMVECs under the influence of a VEGF gradient. Magnetic beads were embedded in an ECM through bioconjugation (between the streptavidin-coated beads and the collagen fibers) in order to alter the local stiffness of the ECM when under the influence of an external magnetic field. We refer to such bead-embedded ECM samples as bECM. HMVECs were allowed to form sprouts in the bECM with a VEGF gradient, and their sprouting behavior was observed during a period of 4 days from the time the HMVECs were seeded in the microfluidic device.
We quantified the sprouting behavior in terms of two categories of indicators. The first concerns the number and height of sprouts, which reflect the extent of sprouting at the cellular level. The second concerns the number and size of focal adhesions associated with the sprouts, which reflect the activities of HMVECs in sprout formation at the molecular level. Focal adhesions function as cell–ECM mechanical linkages in the form of biological signaling complexes. These complexes concentrate and direct various signaling proteins through integrin binding and clustering. 25 Because of their dynamic nature, focal adhesions may vary in size due to their constituent proteins that respond to changes in the mechanical properties of the ECM. One such property is stiffness. Focal adhesions are thus able to sense ECM stiffness and react accordingly. 26 A stiffer ECM leads to more and larger stable focal adhesions. 27 Vinculin is known to be a key regulator of focal adhesions; 28 its level of expression reflects the extent of focal adhesion activities. Cells depleted of vinculin have fewer and smaller focal adhesions than wild-type cells; 29 conversely, higher levels of vinculin recruitment have been shown to increase the number and size of focal adhesions. 30
Our experimental results about these two categories of indicators show that the introduction of a magnetically induced force gradient markedly promotes sprouting and formation of focal adhesions. We observed in statistically meaningful samples an increase of 56% in sprout height for sprouts whose heights were above 100 µm, and an increase of 49% in vinculin recruitment in focal adhesions when an external magnetic field was applied on the bECM. These results demonstrate that under the influence of an external magnetic field, the bECM (compared with pure ECM) provided a preferential sprouting environment.
Materials and Methods
Overview of the Magneto-Microfluidic System
Figure 1 illustrates the key features of the magneto-microfluidic system. Streptavidin-coated magnetic beads are embedded in the ECM (i.e., the bECM), with the beads forming covalent bonds with the collagen fibers. The bECM is contained in a microfludic device. Seeded on one side of the bECM is a monolayer of HMVECs. In close proximity to the monolayer is a permanent magnet for inducing a force gradient in the bECM. A VEGF gradient in the bECM spans across the channel width, with its low concentration located at the monolayer. The details of this setting and the experiment procedure are described in the following subsections.
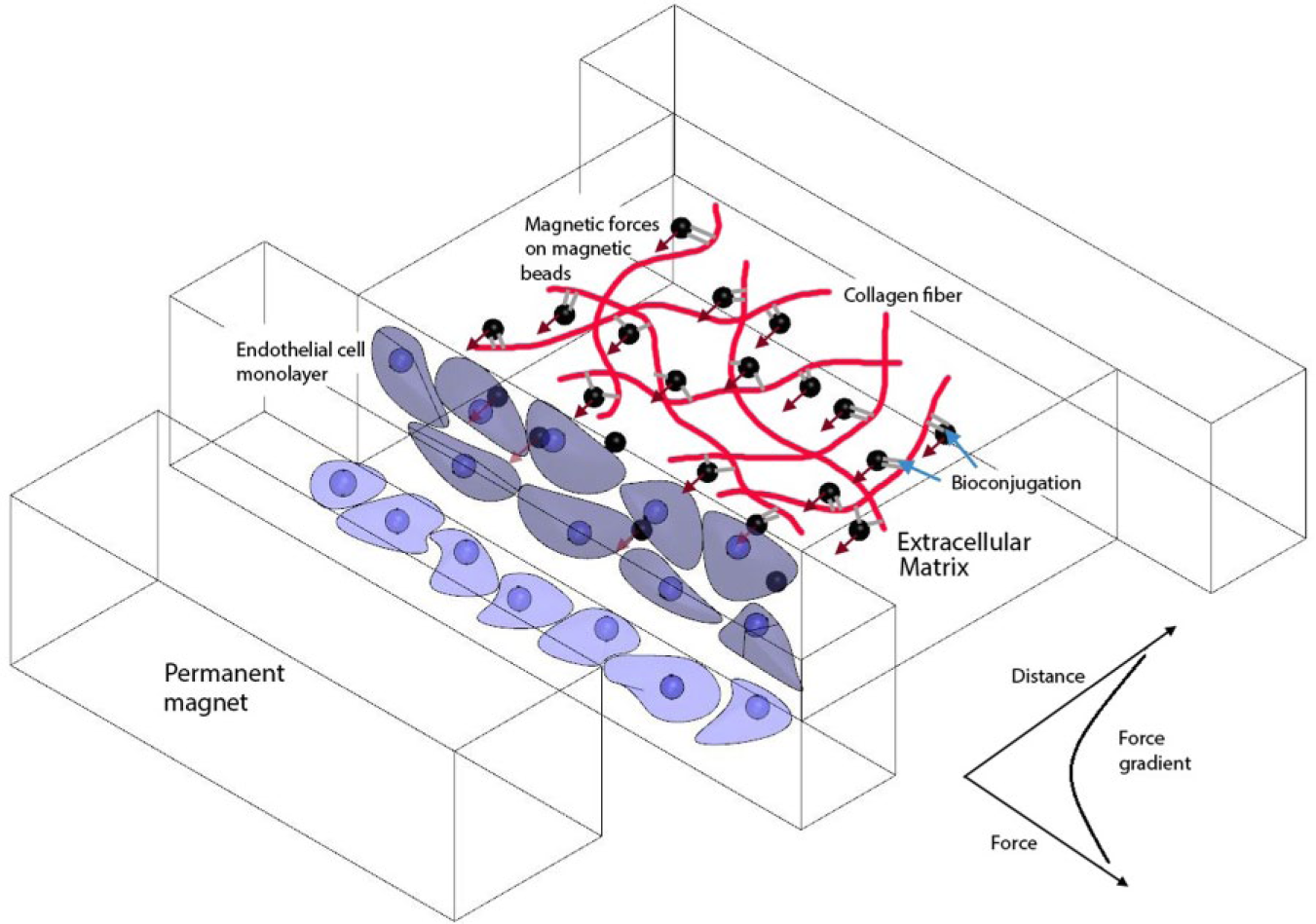
Schematic illustration of the proposed approach. The magnetic beads are embedded in the ECM via bioconjugation between the beads and the ECM fibers. Applying an external magnetic field on the ECM produces forces on the beads and collectively produces a force gradient on the beads to resist the dislocation of the ECM fibers due to the migration of ECs.
Microfluidic Device Design and Replacement of Magnets
The microfluidic device (shown in Fig. 2 ) was fabricated based on the design by Farahat et al. 31 Purposely patterned features were made on the surface of a polydimethylsiloxane (PDMS) substrate by imprinting with a silicon wafer mold using negative photoresist (SU-8). The PDMS layer is cut into individual units, and through-holes are punched in each unit for the delivery of collagen, HMVECs, and media. A single unit was then plasma bonded onto a glass slide to create one microfluidic device. Such a device has three channels, a–c, and 37 slots. Each slot spans the region between two posts of trapezoidal cross section, where HMVECs undergo angiogenic sprouting in response to a VEGF gradient.
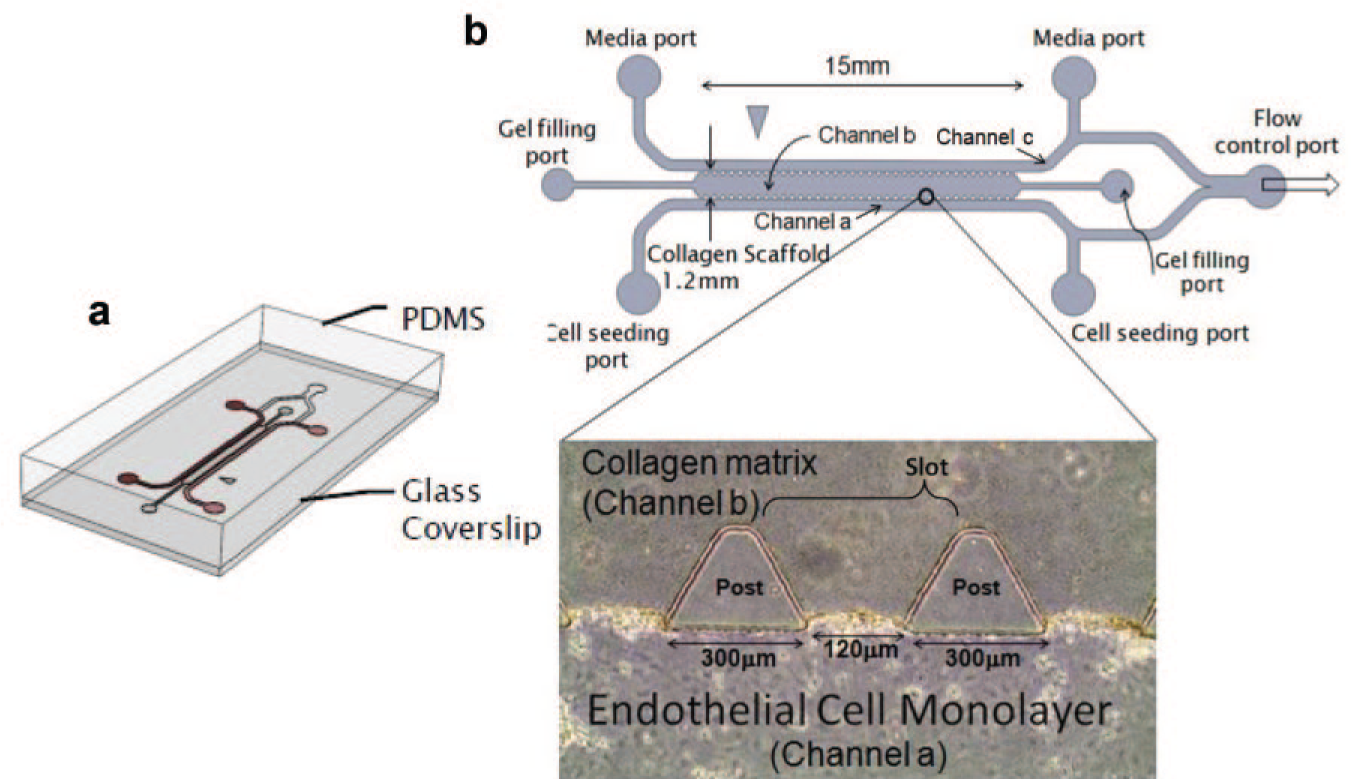
Microfluidic device for observation of HMVEC sprouting during angiogenesis. (
Four permanent magnets, made of an alloy of neodymium, iron, and boron (NdFeB), each sized 5 × 4 × 3 mm and capable of producing a magnetic field of 0.65 T at the surface, were placed on the cell seeding side of the microfluidic device. The magnets were located approximately 1.4 mm away from the EC sprouting region, as shown in Figure 3b .

Experiment setup. (
Human Microvascular Endothelial Cell Culture
Adult HMVECs (Lonza, Walkersville, MD) were cultured in Endothelial Growth Medium-2MV (EGM-2MV, 2% serum, Lonza). Media was changed every 2 days, and cells were kept in a humidified, 5% CO2 environment at 37 °C. HMVECs were passaged once from passages 6–7 using EDTA solution (Gibco, Grand Island, NY). The HMVECs were delivered into channel a (i.e., the cell seeding channel, as shown in
Fig. 2
) in the microfluidic device at
Embedding Beads in ECM
To embed beads in the ECM so that they are bioconjugated with the collagen fibers, the beads were coated with streptavidin, which contains an Arg-Tyr-Asp (RYD) amino acid sequence that mimics the Arg-Gly-Asp (RGD) receptor domain of fibronectin. 32 The complementary shapes, charges, polarity, and hydrophobicity of the streptavidin and collagen fibers permit multiple weak interactions, which in combination produce a tight binding.33,34 The streptavidin-coated magnetic beads BM551 were procured from Bangs Laboratory (Fishers, IN).
Once fibrillogenesis occurred, the magnetic beads and collagen fibers were observed by reflectance microscopy. Figure 4a , b shows the reflectance microscopy image of the beads. It can be observed from the images that after fibrillogenesis, the ECM consists of a network of magnetic beads (bright spots) and collagen fibers (bright lines).
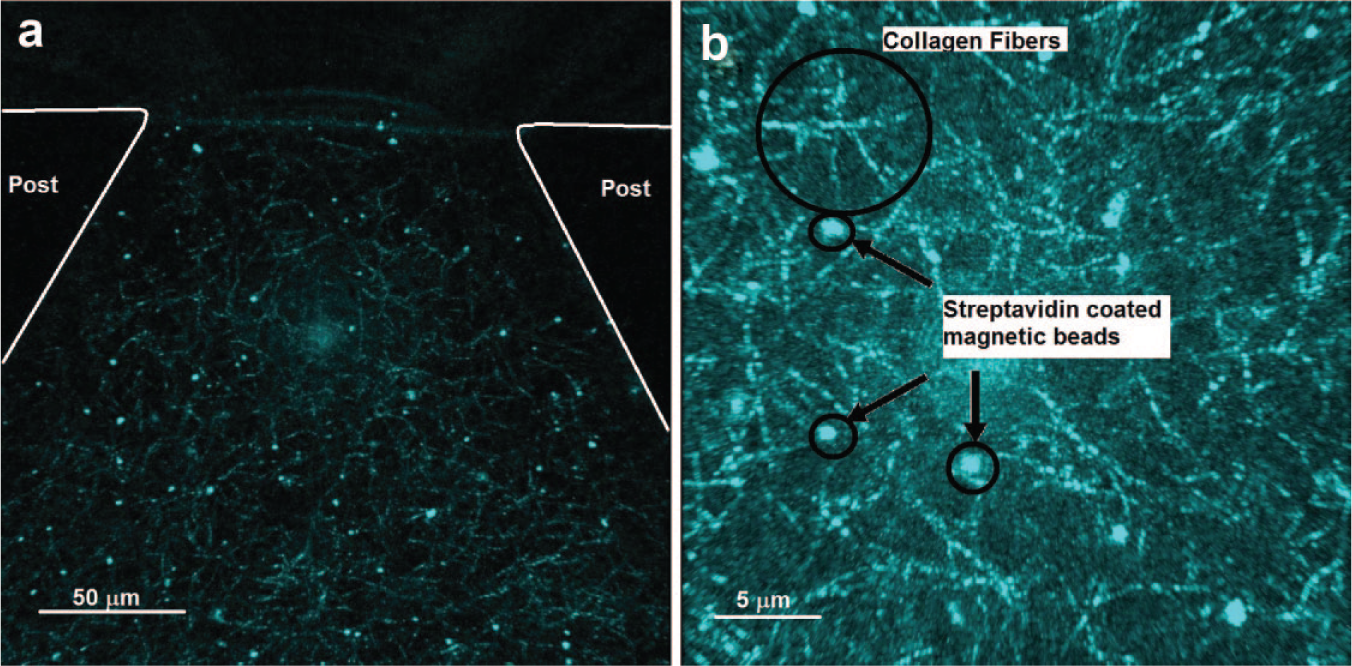
Reflectance image showing the location of superparamagentic beads embedded in collagen. (
Tissue Culture in Microfluidic Device
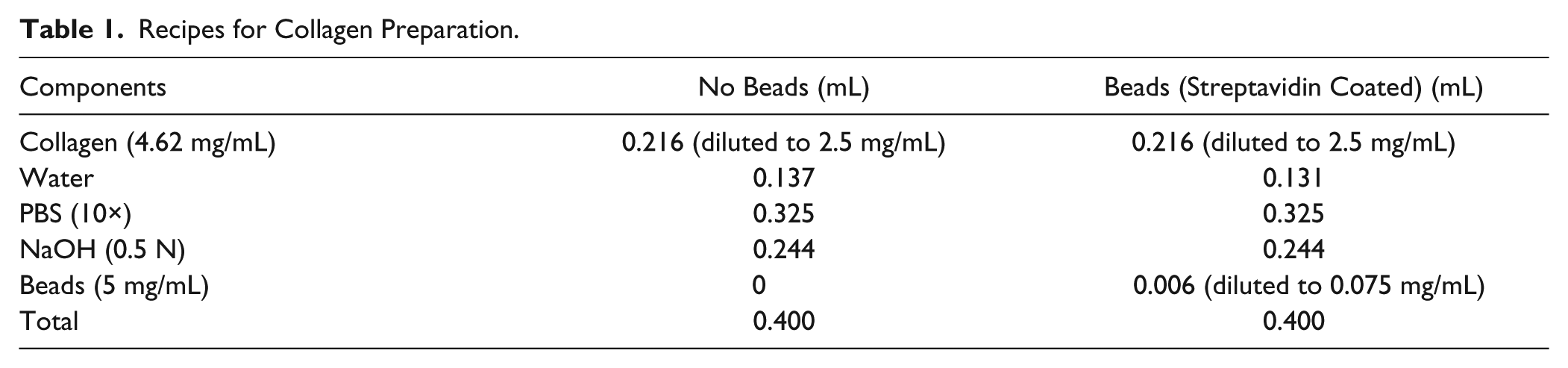
The collagen was prepared according to the recipes listed in Table 1 . All the samples contain 2.5 mg/mL of Rat Tail Collagen Type I (obtained from BD Biosciences, Franklin Lakes, NJ). The streptavidin-coated magnetic beads were mixed into the collagen mixture. The collagen with a bead concentration of 0.075 mg/mL was prepared according to the Table 1 . Another collagen mixture without magnetic beads was also prepared as shown in the second column of Table 1 to serve as a controlled experiment.
Recipes for Collagen Preparation.
The pure collagen mixture or the collagen–bead mixture was thoroughly vortexed for 2 min in an ice bath until a homogeneous solution was formed. A volume of 20 μL of the mixture was then pipetted into the central 3D collagen gel port (channel b) such that no visible air cavities were formed. For gelation, the devices were placed in an external incubator at 37 °C and 5% CO2. To achieve self-assembly of collagen molecules into fibers and binding of beads to the collagen fibers, the samples were placed in the incubator for at least 22 h to ensure that gelation occurred throughout the entire collagen strip.
Once gelation had occurred, HMVECs were delivered into channel a of the microfluidic device, and 80 μL of media (EGM-2MV) was added with 20 and 40 ng/mL concentrations of VEGF (VEGF-A isoform VEGF(165), R&D Systems, Minneapolis, MN) to channels a and c, respectively, to form a VEGF gradient spanning across channel b. The media in channels a and c were changed every day, with the 20–40 ng/mL gradient maintained for a period of 4 days.
Magnetic Force Gradient and Induced Stress in ECM
Computing the forces experienced by superparamagnetic particles requires the applied magnetic field and its gradient, together with the magnetic moment of the beads
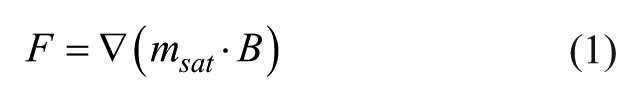
The magnetic field equation along the x axis for a cuboid permanent magnet is given by 36
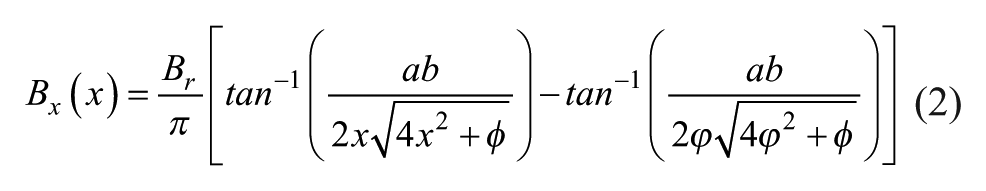
where
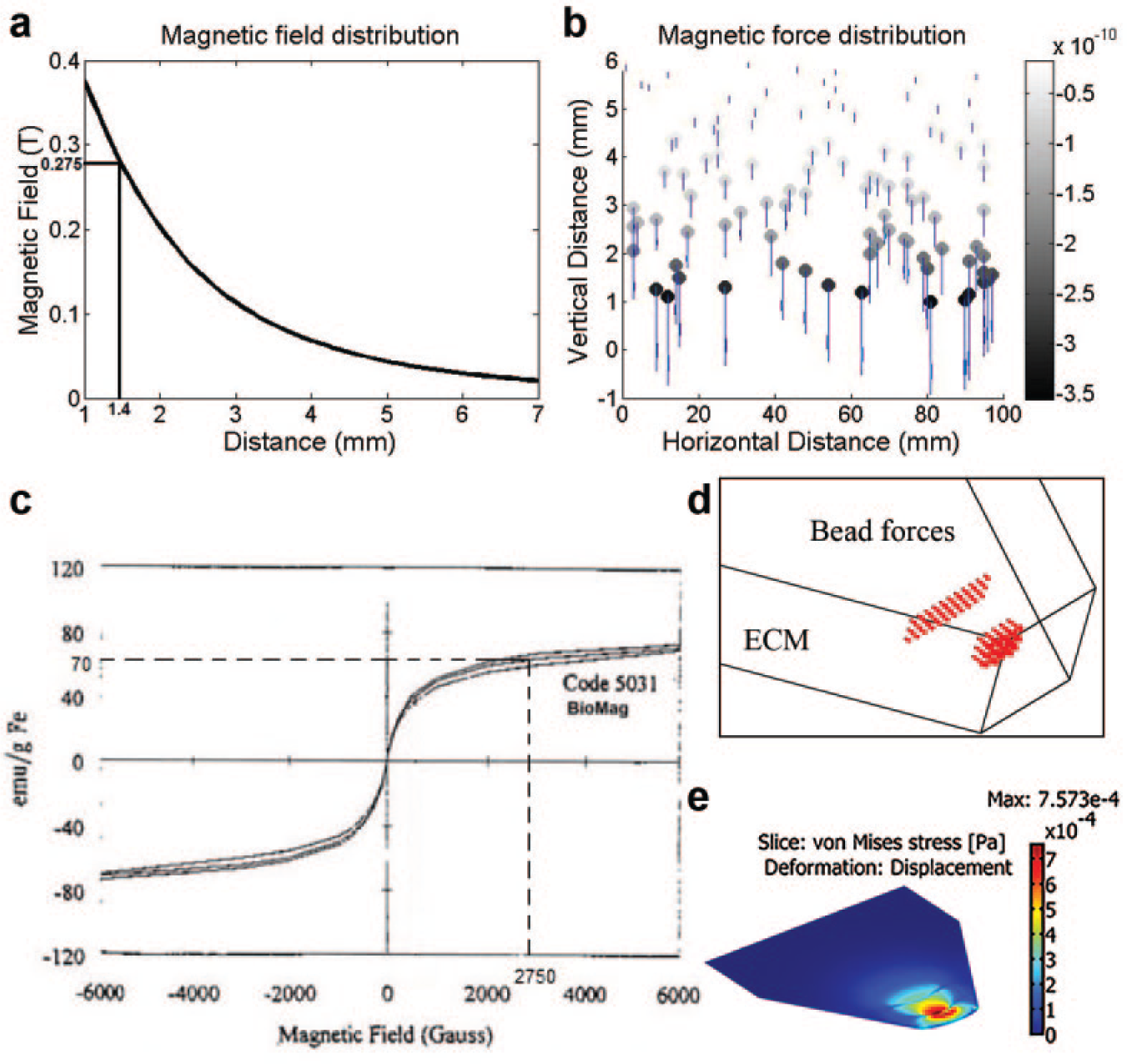
Magnetic field and force analysis of BioMag superparamagnetic particles in bECM. (
The effect of this force gradient on the ECM was analytically evaluated by a numerical simulation (using COMSOL) to estimate the stresses exerted by these magnetically induced forces on the ECM. Specifically, the von Mises stress produced by two layers of magnetic beads in the microfluidic device was investigated. The first layer consists of 15 beads and is situated 50 μm away from the monolayer, while the second layer has 33 beads and is at 100 μm, as shown in Figure 5d . The result of the simulation is shown in Figure 5e . It can be seen that the slot region near the monolayer (where sprouting is expected to occur) exhibits the highest stress.
Experiment Design and Quantification of Sprouts and Focal Adhesions
A set of experiments was conducted under four scenarios as specified in
Table 2
. Scenarios 3 and 4 served as the controlled experiments. Each scenario involved five devices and was completed twice, yielding a total of
Experiment Scenarios.
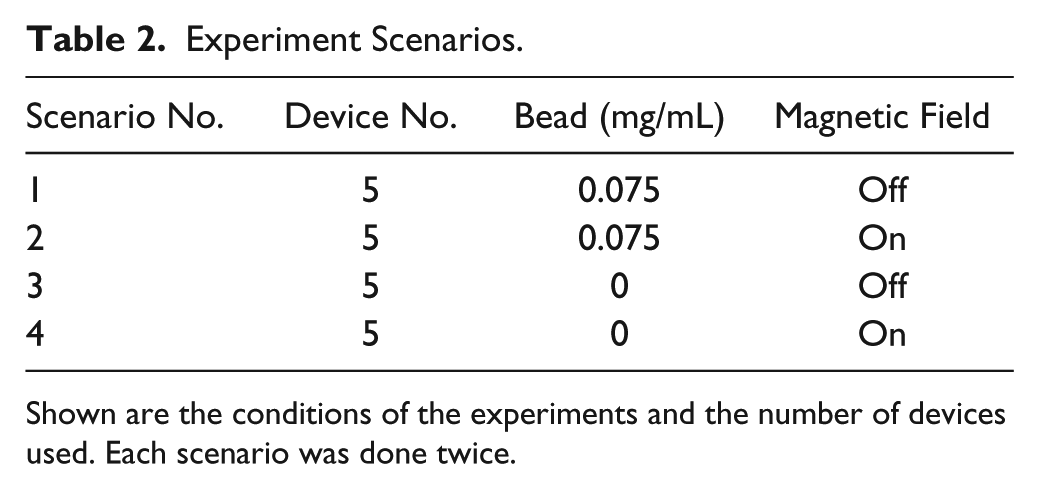
Shown are the conditions of the experiments and the number of devices used. Each scenario was done twice.
In quantifying the sprouts, analysis of the images obtained from 32 devices (out of the total of 40) consisted of counting the number of sprouts per device and measuring the height of each sprout. The average number of sprouts with heights above 100 µm per device (together with their standard deviations) was calculated.
In quantifying the focal adhesions, vinculin staining was done on day 4 on the other eight devices. The sprouts were fixed at day 4 with 4% parafomaldehyde into both the HMVEC channel and the opposite media channel and incubated at room temperature for 20 min. The devices were washed thrice by delivering 1× phosphate-buffered saline (PBS) into both channels. The cells were permeabilized with 0.5% Triton X-100 for 10 min in room temperature. Focal adhesions were stained with primary antibody anti-vinculin (Sigma V9131) overnight in 4 °C and washed five times with wash buffer (0.5% bovine serum albumen (BSA) in PBS) and secondary antibody goat anti-mouse (Invitrogen A21123, Waltham, MA) for 2 h. After washing with wash buffer three times, cell nuclei and the actin filament were stained with 405 Hoechst (Invitrogen H1399) and 488 phaloidin (Invitrogen A12379) at 4 °C overnight.
Images were obtained using a phase contrast microscope, Olympus CKX 41, and a confocal microscope, Olympus IX 81. Fluorescent image processing was done using IMARIS software to extract the different immunofluorescence channels from the image data. A two-tailed, nonpaired Student t test was used to compare the number of focal adhesions in each scenario.
Results
The Induced Force Gradient in the ECM Created a Preferential Environment for Sprout Formation and Growth
Figure 6 shows the growth of sprouts under two experimental conditions on the same day, that is, adding only micron-sized magnetic beads and both the same beads and a magnetic field to the ECM. The acquired phase images a and b collectively indicate that with such a magnetic field, ECs involved were more active and exhibited a higher level of angiogenic activities, as reflected by a greater number of sprouts formed, combined with an apparent increase in sprout size, than that occurring when the magnetic field was absent. The appearance of widened sprouts suggested the promoted migration ability of the ECs and their spreading behavior. Further evidence regarding the associated change in focal adhesion formation would be discussed later. The observations made with images a and b were basically consistent with the result of our investigation into the expression of actin filament (stained with 488 phalloidin green) under the two conditions. Using the magnetic field could lead to more pronounced development of actin stress fibers, as shown by stronger green signals in image d than those appearing in image c for the control, as well as an elevated protrusion activity. There was not much difference in the cell densities of the monolayer between images c and d, manifesting that no toxicities were inflicted by the magnetic beads on the HMVEC. The further examination of ECM remodeling revealed that the addition of the magnetic field (shown by image f) produced an enhanced cellular contractility, as shown by a higher level of fiber compaction in the sprout regions.
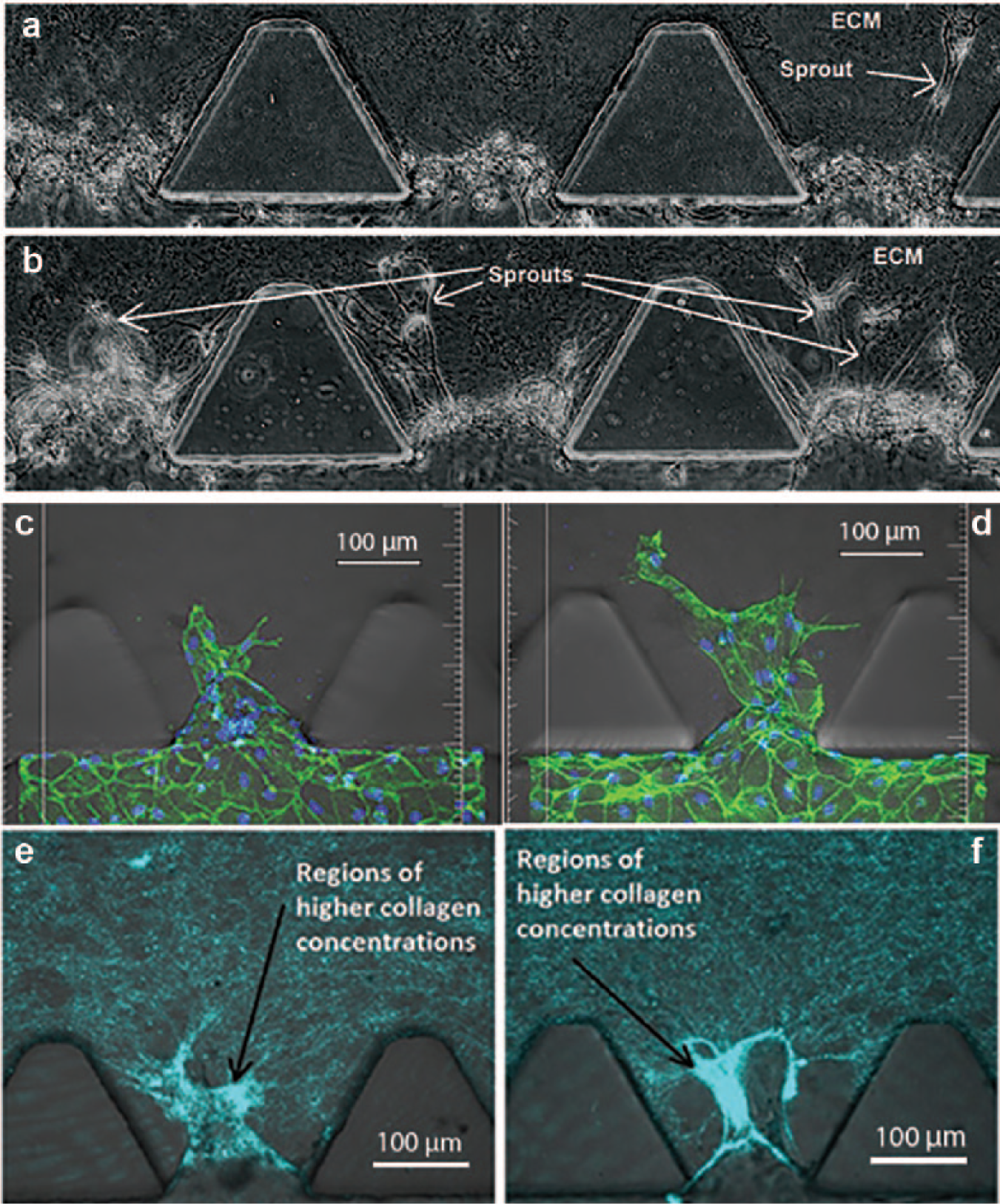
Sprout invasion into the ECM. Once activated by the VEGF (with a low concentration of VEGF in the cell channel and a high concentration in its opposite channel), sprouts appeared in the regions of cell clusters and were elongated forward in the ECM with beads in the absence (
The effect of the induced force gradient on sprout growth was further investigated by quantitatively examining the potential difference of sprout height from that observed in the control. Figure 7a illustrates our measurement of sprout height, that is, the vertical difference from the sprout tip to its bottom. Heights of more than 200 sprouts for both scenarios 1 and 2 were indicated in Figure 7b . By comparing the two scenarios, a bunch of sprouts in scenario 1 were less extended than the majority of those in scenario 2. The number of sprouts with a height equal to or larger than 100 μm was counted for all four scenarios, S1, S2, S3 (pure ECM), and S4 (with only a magnetic field), to investigate their promotion of stable growth of sprouts, as shown by panel c. The result shows that scenario 2 could produce much more well-developed sprouts than the other three scenarios, indicating that it was capable of elevating EC migration and angiogenic activities. We continued to examine the average height of all sprouts observed in the four scenarios, involving a total of nearly 800 sprouts. This suggested that the average height of sprouts in scenario 2 was also significantly greater than that of each of the other three scenarios (p < 0.001), that is, 126 µm versus a value of less than 112 µm. It can also be seen from the image that the exclusive use of beads and the magnetic field yielded a result similar to that of using the pure ECM.
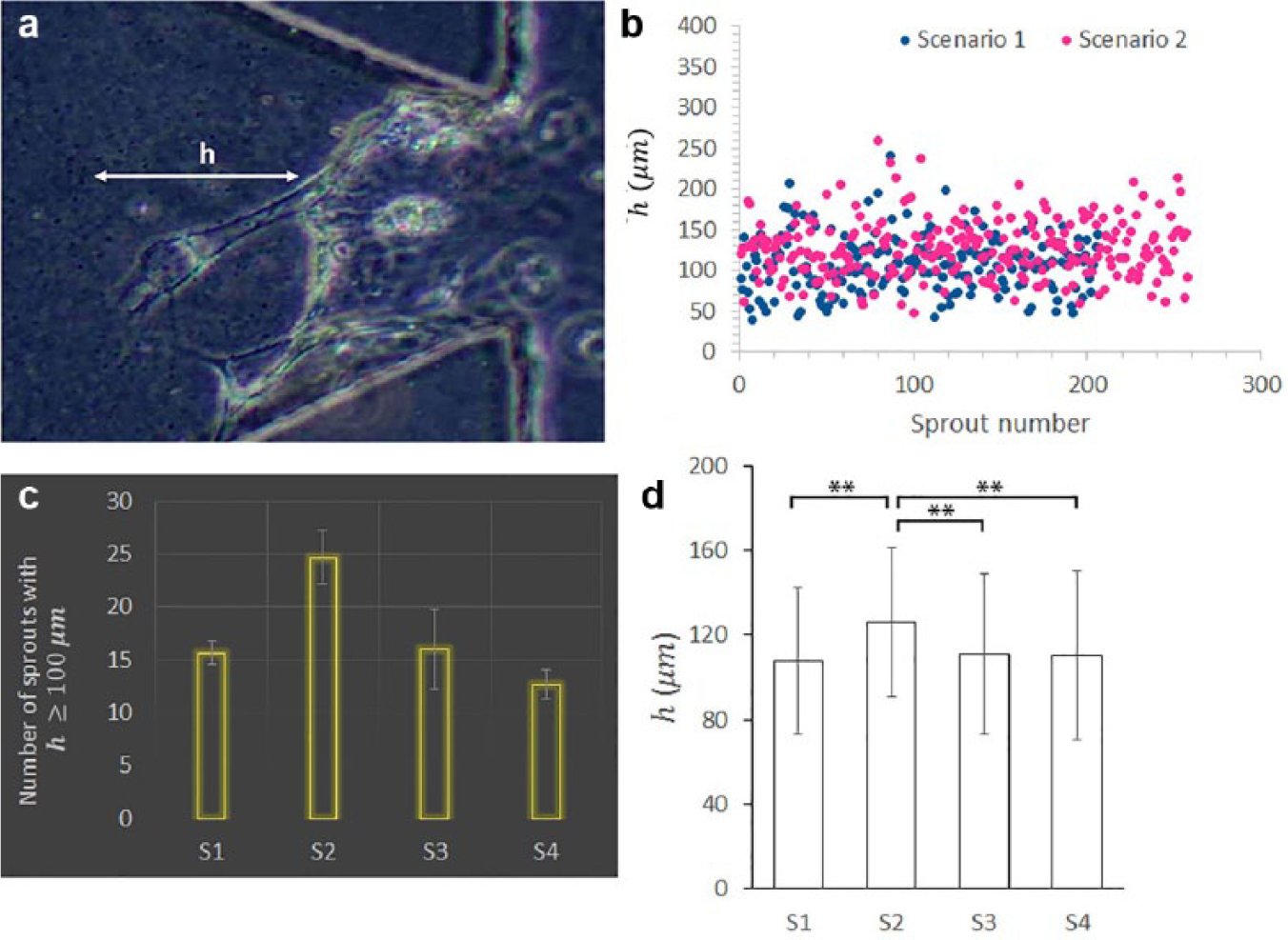
Quantification of sprout height in different scenarios. (
The Elevated Level of Focal Adhesion Formation Promoted Sprout Growth
As previously discussed, the addition of a magnetic field to the ECM containing magnetic beads had an effect of promoting sprout growth and widening sprouts. It was necessary to further verify whether such changes were linked to the elevation of EC migration ability. To do so, the expression of vinculin at the tips of sprouts was examined. Figure 8a–d shows four samples of red fluorescent vinculin-stained spots for scenarios 1–4, respectively, together with their blue fluorescent (Hoechst)–stained nuclei. A higher level of vinculin recruitment was present in scenario 2 (panel b) than that occurring in the other three scenarios, reflected by a greater number of red spots distributed along the entire periphery of the observed sprout. This suggests that the combined use of beads and the magnetic field could enhance focal adhesion formation of the ECs, and thus their migration ability.
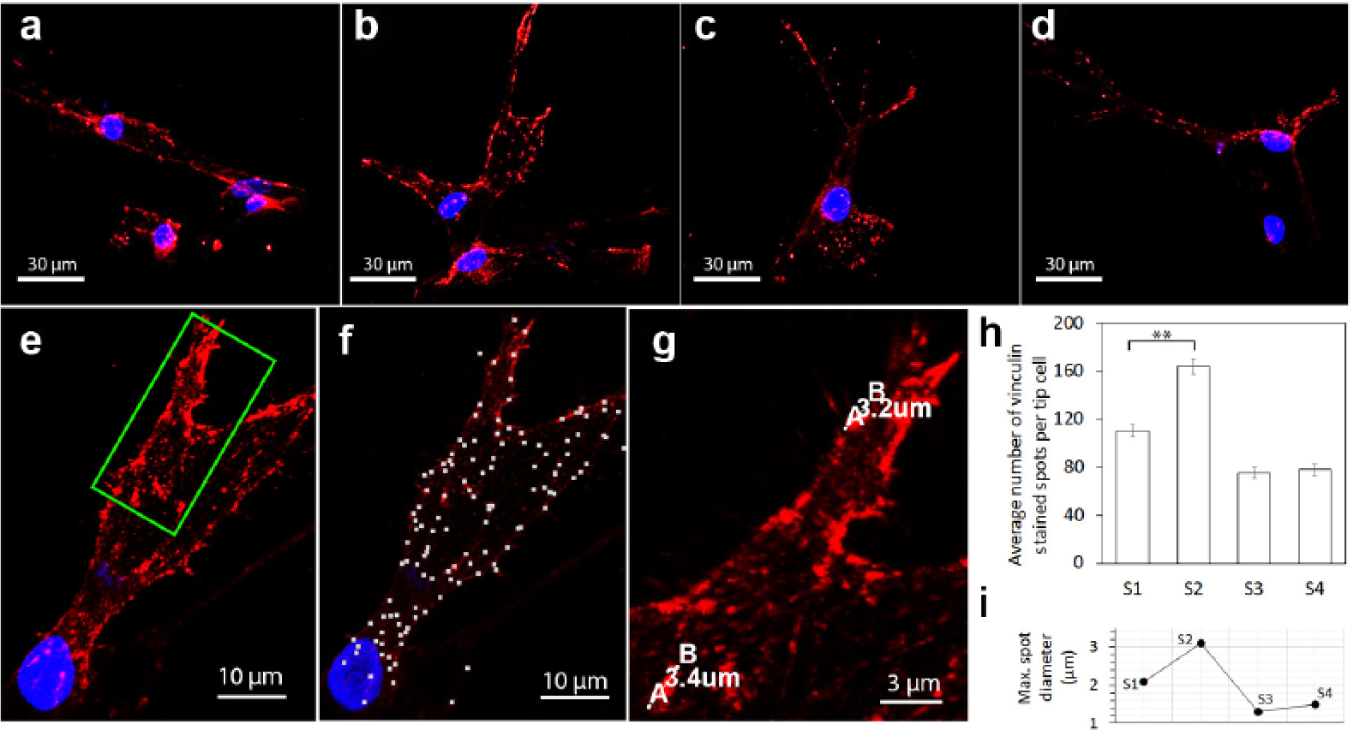
Formation of focal adhesions in different scenarios. (
To quantify vinculin expression, the number and maximum diameter of the red fluorescent vinculin-stained spots per tip cell were determined with the Spots and Measurement Points tools in IMARIS, respectively. Figure 8e–g collectively illustrates the result of identifying vinculin-stained spots, marking them with white dots in image f for a sample sprout tip shown by image e. The diameter measurement is demonstrated in image g, showing an enlargement of a highlighted (green rectangle) region in image e. The statistics produced from a total of 80 tip cells are displayed in images h and i. Scenario 2 had a significantly greater number of focal adhesions than the other three scenarios, for example, a 49% increase compared with that of scenario 1, indicating cell adhesion to the ECM was much improved by the effect of the magnetic field imposed. This was further evidenced by the enlarged focal adhesions due to the existence of the magnetic field, as shown by the result in image i.
Discussion
We have described a magneto-microfluidic system for investigating the effect of active manipulation of ECM stiffness on angiogenic sprouting of HMVECs in vitro. Magnetic beads were embedded in the ECM through bioconjugation (between the streptavidin-coated beads and the collagen fibers) in order to create a pretension in the ECM when under the influence of an external magnetic field. The advantage of using this magneto-microfluidic system is that the resulting change (induced externally and mechanically) in the local deformability of the collagen fibers is only apparent to a cell at the pericellular level near the site of an embedded bead and is localized at that site, while the global intrinsic material properties of the ECM remain unchanged. Embedding magnetic beads in the ECM creates a cross-linking zone around the individual beads. Under influence of the external magnetic field, a bead located approximately 1.4 mm away from the permanent magnet is estimated to exert a force of 0.12 nN on its surrounding ECM. A HMVEC invading from the monolayer into the collagen gel generates (through its focal adhesion traction forces) on the ECM at a magnitude up to 57 nN. 37 When aligned to counteract these traction forces, the magnetically induced forces collectively produce a stronger resistance to deformation in the ECM as perceived by the cell at the invasion front. This induced resistance to ECM deformation has been analyzed by numerical simulation and illustrated in Figure 5e , which shows the region of higher stress to be near the monolayer where sprout formation was observed to initiate.
In an experiment using this magneto-microfluidic system, HMVECs were allowed to form sprouts in 3D in the modified ECM under a VEGF gradient for a period of 4 days, and confocal fluorescence and bright-field images were acquired to analyze sprout morphogenesis, as well as cell adhesion reflected by the level of vinculin expression. A set of experiments was conducted under four scenarios. Scenario 1 describes a situation where angiogenesis of HMVECs is carried out with a 20–40 ng/mL VEGF gradient in a magnetic bead–embedded ECM in the absence of a magnetic field; scenario 2 is identical to scenario 1 except that a magnetic field is present; scenarios 3 and 4 (as the controlled experiments) describe the situations where angiogenesis of HMVECs is carried out with a 20–40 ng/mL VEGF gradient in a pure ECM in the absence and presence of a magnetic field, respectively. A total of nearly 800 sprouts from 32 devices were quantified to yield statistically meaningful results.
Results from analyzing the confocal images obtained in this set of experiments revealed that the presence of a magnetic field affected noticeably both the size and the cell adhesiveness of the sprout. Specifically, it was observed that with the presence of the magnetic field, there was a higher level of sprout morphogenesis (as is evident in an increase of 17% in the sprout heights compared with that from the controlled experiments), and there was a significant increase in tip-cell adhesiveness, as indicated by a 49% increase in the number of tip-cell focal adhesions. The observed sprouting behavior points to the conclusion that during angiogenic sprouting, HMVECs tend to migrate preferentially in the environment in which the induced force gradient acts against the general direction of the migration. This observation is consistent with prior results reported in the literature that in areas of higher ECM stiffness, ECs exhibit increased motility and tend to spread.26,38–42
Research reported in the literature has established that a reduction in collagen density would lead to significant downgrading of vinculin expression, while an ECM with higher collagen density promotes the recruitment of vinculin in cell–matrix adhesions.43–46
Our immunofluorescent staining results indicate that the level of vinculin recruitment, in terms of the average number of vinculin stain red spots per tip cell, increased by 49% in sprouts formed in bead-embedded ECM under the influence of the magnetic field. This can be attributed to the deformation and movement of the collagen when the beads were pulled toward the permanent magnet, resulting in an area of higher collagen density near the monolayer where sprouting occurred.
To develop a full understanding of how the magnetically induced force gradient influences angiogenic sprouting, experiments and analysis are needed to clarify a number of key issues. To begin with, the effect of a number of variables on the stiffness of the bead-embedded ECM remains to be explored; these variables include bead concentration, size and placement of the magnets and their distance to the ECM sample, and the strength of the magnetic field. The use of magnetic beads in the experiments also needs further investigation. Although magnetic beads have been used in many applications (both in vivo and in vitro), their effect on the actual biological behavior of cells is not yet fully understood. This is particularly true when an ensemble of beads is embedded in the ECM via bioconjugation. It may be argued (based on available experimental results47–49) that the biocompatibility and toxicity of these magnetic beads with respect to their biological environment solely depend on the coating on the surface of the magnetic particles. It is therefore essential to verify that such an ensemble of beads does not directly interfere with the natural behavior of cells.
Finally, the mechanical interaction among the HMVECs, the collagen fibers, and the bioconjugated beads represents a rich area of study in the context of biophysics and mechanobiology. Advances in this area will have implications not only on how externally induced forces affect angiogenic sprouting in particular, but also on how such forces can be used to manipulate cell behavior in general.
Such technical challenges notwithstanding, the work reported in this article has demonstrated the effectiveness of the proposed magneto-microfluidic system for active manipulation of ECM stiffness and for investigating the effect of ECM stiffness in influencing the angiogenic sprouting of HMVECs. The results obtained from the experiments conducted using this magneto-microfluidic system suggest the possibility of creating desired stiffness gradients in the ECM for manipulating cell behavior in vitro.
Footnotes
Acknowledgements
The authors thank the A*Star Science and Engineering Research Council (under Public Sector Funding Grant Project No. 1321202077) and BioSystem and Micromechanics Interdisciplinary Research Group (under the Singapore-MIT Alliance for Research and Technology Program) for their financial support. S.C.B.H. acknowledges the financial support provided by the National University of Singapore. D.Y. acknowledges the financial support provided by the China Scholarship Council and by the Department of Mechanical Engineering at the National University of Singapore.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the A*Star Science and Engineering Research Council (under Public Sector Funding Grant Project No. 1321202077) and BioSystem and Micromechanics Interdisciplinary Research Group (under the Singapore-MIT Alliance for Research and Technology Program) for their financial support. S.C.B.H. acknowledges the financial support provided by the National University of Singapore. D.Y. acknowledges the financial support provided by the China Scholarship Council and by the Department of Mechanical Engineering at the National University of Singapore.
