Abstract
Angiogenesis is an essential part of bone regeneration, as neovascularisation supports the supply of necessary oxygen, nutrients, and cellular transport. Bioactive glasses (BGs) have shown promise in enhancing both angiogenesis and bone regeneration. For the evaluation of the angiogenic potential of BGs, the chorioallantoic membrane (CAM) assay constitutes an attractive experimental model and has already gained increasing attention in BG-focused research. However, there is conflicting evidence as to whether the addition of cells, such as bone-marrow-derived mesenchymal stromal cells (BMSCs) to the CAM is necessary to facilitate the evaluation of the angiogenic potency of BGs. Therefore, in this study, the angiogenic potential of mesoporous bioactive glass nanoparticles (MBGNs; molar composition = 70% silica (SiO2) and 30% calcium oxide (CaO)) was assessed by using the in ovo CAM assay, both in the presence and the absence of exogenous BMSCs. Compared to the BMSC-free and MBGN-free control groups, both BMSCs alone and MBGNs without the addition of BMSCs were able to induce an equally strong, significantly enhanced angiogenic response on the CAM. The combination of MBGNs with BMSCs did not yield a more pronounced angiogenic response, as compared to MBGNs without the addition of cells. Thus, MBGNs exhibit a strong intrinsic angiogenic potential that is not dependent on the presence of exogenous BMSCs. Therefore, the CAM assay without added cells can be used as a simplified, reproducible and cost-effective method for accurate preclinical testing of the angiogenic potential of BGs.
Introduction
Angiogenesis is a key factor in tissue regeneration. The regeneration of bone tissue depends, among other things, on sufficient local blood flow, which allows the bone defect to be adequately supplied with oxygen and nutrients.1,2 Blood flow also enables the removal of waste products during regeneration, and the transport of progenitor cells to the damaged site, without which bone healing would not be possible.2–4 It has been demonstrated that insufficient vascularisation of the bone defect can limit its regeneration, highlighting its importance in the healing process. 5 Given the importance of angiogenesis, ways in which the process can be stimulated to enhance tissue regeneration have been widely investigated — for example, by using angiogenesis-promoting drugs or bioactive materials.6–8
Among synthetic bone substitute materials, bioactive glasses (BGs) have shown promise in promoting angiogenesis.7,9 BGs are a group of bioactive silica-based materials that, after contact with body fluids, eventually form a hydroxyapatite layer on their surface through several intermediate chemical steps.10–13 Through this mechanism, BGs not only bond firmly to their surrounding tissue and bone, but also release therapeutically active ions into their surroundings.14,15 Besides their osteogenic potential, their pro-angiogenic abilities make them an attractive biomaterial to facilitate bone defect regeneration. The promotion of neovascularisation by BGs is caused by the release of therapeutic ions that are incorporated into the silica-based material.9,14 The ions incorporated into the BGs are interchangeable, with certain ions like boron or copper increasing the material’s angiogenic potential.9,16–21
In the search for reliable models to evaluate the angiogenic potential of biomaterials, the chorioallantoic membrane (CAM) assay has been increasingly used alongside conventional in vitro and in vivo models.22–25 The in ovo CAM assay makes use of the CAM, a highly vascularised membrane in the fertilised chicken egg that is responsible for the exchange of gas and electrolytes in the embryo. The assay serves as a bridge between in vitro and in vivo testing, offering a more ethically responsible model that preserves many of the complexities of in vivo angiogenesis. Furthermore, it is easier to reduce variability between experimental groups in the CAM assay, as the environment and experiments can be standardised and controlled. 26 The CAM assay was first developed for use in tumour research, to provide a suitable environment for cell and tumour transplantation for further studies.27–31 It was later realised that the CAM’s properties, in particular its dense network of blood vessels and rapid development, also make it a promising model for studying angiogenesis and provide researchers with a simple assay to observe the angiogenic response of biomaterials like BGs in ovo.23,32
When it comes to evaluating the angiogenic potential of biomaterials in the CAM assay, two different approaches can be employed, namely testing in the presence of exogenous bone marrow-derived mesenchymal stromal cells (BMSCs) or without the addition of exogenous cells. In the approach without exogenous cells, only the biomaterials are transplanted onto the CAM — this permits the evaluation of the intrinsic angiogenic potential of the biomaterial and of its components.22,23,33,34 In CAM assays performed with exogenous cells, the tested biomaterials are coated or seeded with cells and then transferred onto the CAM.21,35 In this approach, the biomaterial might stimulate the cells toward an angiogenic involvement. Therefore, the result of the CAM assay might either be related to the actions of the exogenous cells, to the biomaterial itself, or to stimulation of the exogenous cells by the biomaterial. Thus, the reliance on the addition of exogenous cells further complicates the interpretation of the results obtained, raising the question of whether the angiogenic effects of bioactive materials can be effectively evaluated in ovo, without the need for exogenous cell support.
Since BGs have demonstrated potent pro-angiogenic properties in different experimental settings, the CAM assay is most certainly an attractive method with which to evaluate their angiogenic potential in physiological surroundings. However, while there is evidence that BGs act in a pro-angiogenic manner on the CAM when exogenous cells are involved, there are also studies reporting that BGs alone are not able to provoke angiogenic reactions on the CAM.21,35–37
This study therefore aims to evaluate whether the addition of exogenous cells to MBGNs is necessary to promote angiogenesis on the CAM. The MBGNs used in this study have previously demonstrated angiogenic potential in in vitro and in vivo settings, 34 and the ions released by them have been shown to exhibit pro-angiogenic activity.9,38–43
Materials and methods
Bone marrow-derived mesenchymal stromal cell isolation and culture
To obtain bone marrow-derived mesenchymal stromal cells (BMSCs), density gradient centrifugation of bone marrow aspirates from patients undergoing total hip arthroplasty was used as previously described.44,45 After centrifugation, the cells were cultured in T175 cell culture flasks (Sarstedt, Nümbrecht, Germany) coated with 0.1% w/v gelatin (Sigma-Aldrich, Steinheim, Germany) and grown in expansion medium (EM) consisting of Dulbecco’s modified Eagle’s medium (DMEM) high glucose, 12.5% v/v fetal calf serum (FCS), 2 mM
Ethical approval
Patients undergoing total hip arthroplasty were approached prior to surgery, and gave written consent for collection of the bone marrow aspirates. The use of the cells for the purpose of this study was approved by the ethics committee of the Medical Faculty of the University of Heidelberg (S-340/2018).
In Germany, the use of avian embryos for experimental research is regulated by §4c of the Tierschutzgesetz, which permits the use and euthanasia of chicken embryos for scientific purposes. 47 Furthermore, §14 of the Tierschutz-Versuchstierverordnung requires ethical approval for vertebrate embryos prior to hatching only if they are expected to survive and suffer post-hatching. 48 As our study involved chicken embryos terminated before hatching, no ethical approval was required under current German law. This is also in line with EU Directive 2010/63/EU, which provides a general framework for animal experimentation within the EU, but does not explicitly regulate the use of chicken embryos at any stage of development. 49
Synthesis and characterisation of MBGNs
MBGNs with a nominal molar composition of 70% silica (SiO2) and 30% calcium oxide (CaO) were synthesised by using a micro emulsion-assisted sol-gel method under basic conditions (pH 9.5–10.5). Cetyltrimethylammonium bromide (CTAB; Merck, Germany) served as a surfactant to form micelles, while ethyl acetate (Merck) created microemulsion droplets, acting as a template for particle formation. An ammonium hydroxide solution (VWR International, Germany) was used to maintain a basic environment for the hydrolysis of tetraethyl orthosilicate (TEOS; Sigma-Aldrich), which served as the silica precursor. Calcium nitrate tetrahydrate (Sigma-Aldrich) was later introduced to incorporate calcium ions into the silica network.
To synthesise the MBGNs, 1.12 g of CTAB were dissolved in 52 ml of Milli-Q water at 37°C under magnetic stirring for 30 minutes. Subsequently, 16 ml of ethyl acetate was added dropwise, and the solution was stirred for another 30 minutes to form a stable microemulsion. The pH was adjusted by adding ammonium hydroxide, followed by the dropwise addition of 6 ml of TEOS under continuous stirring. Silica nanoparticles began to form after 30 minutes and continued growing over time. Then, 2.8 g of calcium nitrate tetrahydrate were introduced, and the reaction mixture was left at room temperature for four hours to facilitate silica condensation and calcium incorporation.
The resulting whitish suspension was centrifuged at approximately 6800g (Centrifuge 5430R, Eppendorf, Germany) for 10 minutes to separate the nanoparticles. The collected particles were washed twice with water and once with ethanol (VWR International). The precipitate obtained was dried overnight at 60°C, ground with a mortar and pestle, and then calcined at 700°C for three hours at a heating rate of 2°C/min to remove organic residues.
The morphology of the MBGNs was analysed by scanning electron microscopy (SEM) (FESEM, LEO 435VP, Carl Zeiss AG, Germany) at 2 kV. Energy-dispersive X-ray (EDX) spectroscopy at 20 kV was performed for qualitative compositional analysis. The amorphous or crystalline nature of the MBGNs was examined using an X-ray diffractometer (Rigaku Miniflex 600, Rigaku, Germany) operated at 40 kV and 15 mA. Before SEM analysis, the samples were sputter-coated with gold by using a sputter coater (Q150/S, Quorum Technologies, UK).
Overview of the general experimental design
The MBGNs were transplanted onto the CAM, either with or without the addition of exogenous cells (i.e. BMSCs). The BMSCs were added to the CAM of fertilised eggs as a suspension. All transplants were applied in a 1:1 mixture of cell culture medium (CCM) and Cultrex Basement Membrane Extract Type 3 (Cultrex BME Type 3; Bio-Techne, Wiesbaden, Germany), which is an extracellular matrix hydrogel that mimics the in vivo environment. The angiogenic response on the CAM was analysed seven days after transplantation. The following experimental groups were used: — Control group: A mixture of 20 μl CCM and 20 μl Cultrex BME Type 3, without the addition of either BMSCs or MBGNs, was applied to the CAM. — MBGNs group: In this group, 307 μg MBGNs in 20 μl CMM/20 μl Cultrex BME Type 3, were applied to the CAM. — BMSCs: A suspension of 7.5 × 105 BMSCs in 20 μl CMM/20 μl Cultrex BME Type 3, was applied to the CAM. — MBGNs/BMSCs group: In this combined group, 307 μg MBGNs and 7.5 × 105 BMSCs, in 20 μl CCM/20 μl Cultrex BME Type 3, were applied to the CAM.
The CAM assay
The day of arrival of the fertilised chicken eggs in the lab was defined as embryonic development day (EDD) 0. Eggs were obtained from a local organic hatchery (Geflügelzucht Hockenberger, Eppingen, Germany). On EDD0, eggs were cleaned with sterile water and incubated with the wider side facing upwards at a temperature of 37.5°C and a humidity of 53% in a hatching incubator (HEKA Favorit-Olymp 192; HEKA Brutgeräte, Rietberg, Germany). On EDD3, a hole was made on the wider side of the egg using a 20-gauge needle (Becton Dickinson, Heidelberg, Germany) and 3 ml of albumin were extracted to separate the CAM from the eggshell. The puncture site was then covered with non-sterile medical tape (Leukosilk; BSN Medical, Hamburg, Germany) and the eggs were put back into the incubator laying on their side. Six days later, on EDD9, the CAM was exposed by cutting a window into the upper side of the egg. A sterile silicone ring (Greiner Bio-One, Frickenhausen, Germany) with an inner diameter of 9 mm was then placed on the exposed CAM, to highlight the location of the transplant and to define an area of interest (AOI) for quantification. The transplants were applied inside the ring after scratching the surface of the CAM three times from one side of the ring to another with a 20-gauge needle. The purpose of the scratching is to break the outermost layer of the CAM open, to allow the transplants to get in contact with the inner layers as well as in direct contact with the vasculature. The window in the eggshell was then closed with medical tape, and the eggs were further incubated at an increased humidity of 65% to prevent drying of the eggs. On EDD16, the CAM was imaged in ovo by using a Zeiss Axioplan 2 microscope (Carl Zeiss, Oberkochen, Germany). Immediately after the image was captured, the chick embryos were euthanised with an intravascular injection of 100 μl Narcoren (Boehringer Ingelheim Vetmedica, Ingelheim, Germany).
Macroscopic evaluation of angiogenic response
To quantify the angiogenic response of the CAM in the presence of the transplants, the vessel density within the AOI was measured, as well as the vessel ingress. For the evaluation of the vessel density, the number of vessels inside the AOI was counted manually and then divided by the surface area of the AOI (254.5 mm2). The vessel ingress is characterised by the number of vessels entering the AOI from outside the defined area. To measure the number of vessels entering the AOI, a modified version of the ‘vascular index’ by Barnhill et al. 50 was used, in which every vessel entering the AOI at an angle of at least 45 degrees was counted.
Statistics
Statistical analyses were performed by using IBM SPSS Statistics (Version 29; IBM, Armonk, NY, USA). Significant differences between groups were identified by using the Mann–Whitney U test. The level of significance was set at p < 0.05. Where appropriate, values are shown as rounded means. All results are shown as x-fold of the control group. For each measurement, between nine and thirteen biological replicates were included (i.e. n = 9 to n = 13). Graphs were designed by using Microsoft Excel and Microsoft PowerPoint (both Microsoft, Redmond, WA, USA).
Results
MBGNs characterisation
MBGNs were synthesised using the micro-emulsion-assisted sol-gel method. The morphology and structural characteristics of the obtained nanoparticles were analysed by using SEM, EDX and XRD techniques. Figure 1(a) and (b) presents the SEM images of the synthesised nanoparticles at two different magnifications. The images reveal that the particles exhibit a uniform, monodispersed distribution with a spherical morphology. The average particle size is approximately 140 nm (measured using ImageJ software), indicating successful control over the synthesis process to achieve nanoscale dimensions.
51
Characterisation of the MBGNs. (a) and (b) Scanning electron microscopy images of the MBGNs at two different magnifications, showing monodispersed, spherical particles. (c) Dispersive X-ray (EDX) spectrum, confirming the presence of Si and Ca; and (d) X-ray diffraction (XRD) pattern displaying the amorphous nature of the produced MBGNs.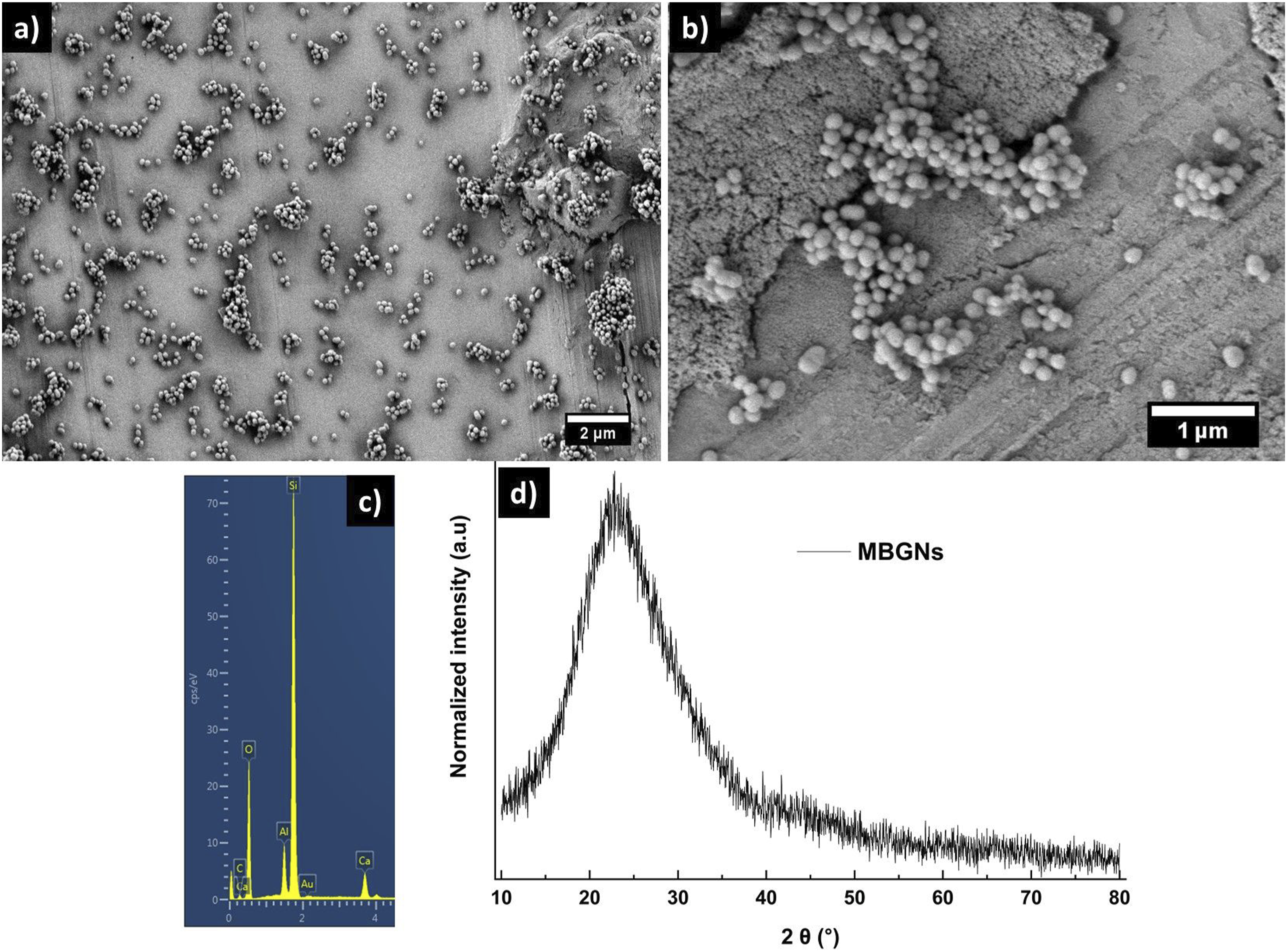
The energy-dispersive X-ray (EDX) spectra further validate the elemental composition of the synthesised nanoparticles. The silicon (Si) and calcium (Ca) peaks confirms the presence of SiO2 and CaO in the synthesised MBGNs, as expected based on the precursor materials used during synthesis. The absence of unwanted impurities suggests the high purity of the obtained nanoparticles.
Additionally, the X-ray diffraction (XRD) analysis provides insight into the structural nature of the synthesised particles. The broad, diffused peaks observed in the XRD spectra indicate the amorphous nature of the MBGNs. This lack of long-range crystalline order is a characteristic feature of bioactive glass materials, which enhances their bioactivity and dissolution properties, making them suitable for biomedical applications.
The characteristics obtained from SEM, EDX, and XRD analyses are consistent with previously reported studies, including our earlier work,34,52 further confirming the reproducibility and reliability of the synthesis method.
Survival rate
Survival rate of fertilised eggs for each experimental group on embryonic development day 16.

Angiogenic response
Figure 2(a) shows representative pictures of the different groups on the CAM that have been used to quantify vascularisation by evaluation of the vessel density within the AOI (Figure 2(b)) and the vessel ingress (Figure 2(c)). Both the vessel density and the number of vessels entering the AOI were significantly higher in the MBGNs group, as compared to the control group (Figure 2(b); Figure 2(c)). In comparison to the control group, the presence of BMSCs alone also resulted in an enhanced angiogenic response of the CAM with significantly more vessels entering the AOI (Figure 2(c)). In the MBGNs/BMSCs group, there was also a significant increase in vessel ingress compared to the control group (Figure 2(c)). Still, the extent of vessel ingress in the MBGNs/BMSCs group was no greater than in the MBGNs group or in the BMSCs group. While the vessel density inside the AOI was not significantly altered by the presence of BMSCs, it was significantly increased by the co-stimulation of BMSCs and MBGNs (Figure 2(b)). However, there were no significant differences in angiogenic response detectable between the MBGNs, BMSCs or the MBGNs/BMSCs groups. Evaluation of vascularisation on the CAM. (a) Representative macroscopic images of vascular formation on the CAM. The brightness and contrast of the images was adjusted for better visualisation of the vascular structures. (b) Vessel density and (c) vessel ingress into the area of interest (AOI) on EDD16 (embryonic development day 16). Values are normalised to the control group (x-fold of control). Asterisks (*) mark significant differences compared to the control group. The level of significance was determined at p < 0.05. MBGNS = mesoporous bioactive glass nanoparticles; BMSCs = bone marrow mesenchymal stem cells.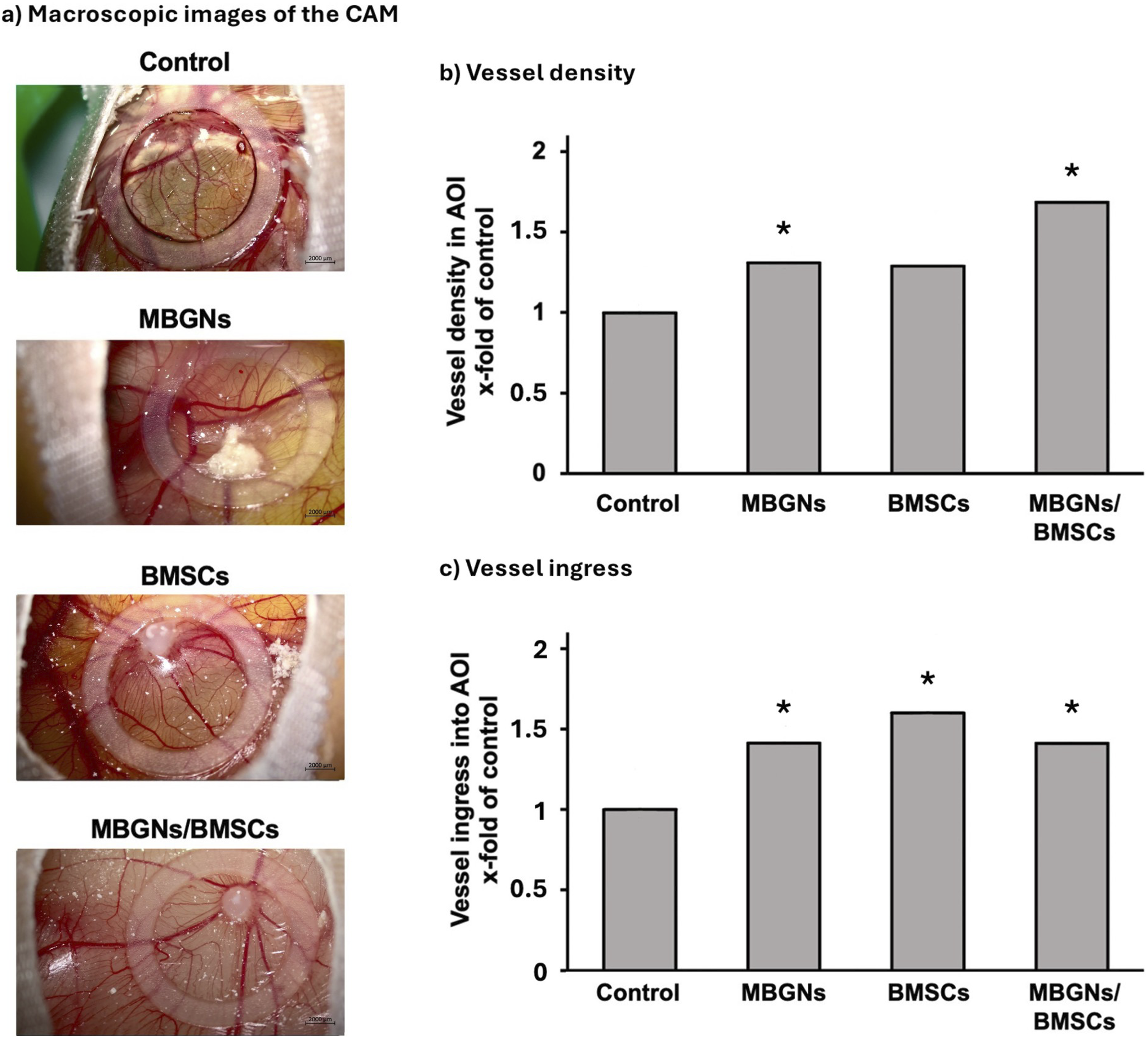
Discussion
This study addressed the following questions: (i) whether it is necessary to add cells to the CAM assay when evaluating the angiogenic potential of BGs in the form of MBGNs, and (ii) whether MBGNs exhibit an intrinsic angiogenic potential that alone is able to stimulate an angiogenic response on the CAM without exogenous cells. The rationale behind adding cells to a biomaterial-based experimental setting is to induce vessel growth by pro-angiogenic mediators released by the cells.53–55 It has been shown before that cells may respond to the BG-contact with paracrine reactions that result in increased expression of pro-angiogenic genes such as vascular endothelial growth factor (VEGF-A) or hypoxia inducible factor (HIF-1α).55–61
Traditionally, the angiogenic properties of BGs have been evaluated in the presence of cells in ovo, using different experimental designs. For example, in a previous study conducted by our group, BMSCs were transplanted after pre-stimulation with BGs in cell culture on the CAM and later their presence was confirmed histologically after explantation of the CAM. 21 Handel et al. were able to induce significant vascularisation in a different setting, using human adipose tissue-derived stem cells (hASC) seeded on 3D 45S5 BG-based scaffolds in the CAM assay, but they were unable to achieve the same effect with cell-free 45S5 BG scaffolds. 35
However, the results presented in this study indicate that MBGNs independently induce an angiogenic response in the CAM, demonstrating their intrinsic angiogenic potential. This is evidenced by increased vessel density and ingress within the AOI in response to the presence of MBGNs. Furthermore, the addition of BMSCs to the MBGNs did not significantly affect the angiogenic activity in the CAM, highlighting the ability of MBGNs to stimulate angiogenesis autonomously.
These reactions could be explained by the contact of the MBGNs with the cells present in the CAM. Direct contact is made with the chorion — the outermost layer of the CAM — and its cells. In addition to chorionic cells, mesenchymal cells are also found in this layer, facilitating the response to the materials — a role that is at least in part provided by the addition of cells in the cell-dependent CAM model. 62 The capillaries in this layer also allow direct contact with the vasculature, influencing endothelial and haematopoietic cells.62–64 In addition, the scratching of the CAM surface prior to transplantation causes minimal bleeding and provides another entry point for the MBGNs and their therapeutic ions into the vasculature. As there are large intercellular gaps in the chorion, MBGNs applied to the CAM can also make indirect contact with the other two layers of the membrane, particularly the mesoderm, which is the largest layer where most vessels are found.62,65 Therefore, ions released from MBGNs can reach the outermost layer via direct contact, and access more deeply-located cells through intercellular gaps in the chorion. This enables the ions released from MBGNs to influence the whole vascular network, affecting not only their immediate environment but also the CAM as a whole.
Haro Durand et al. were also able to stimulate angiogenesis in ovo by applying ionic dissolution products (IDPs) of 45S5-BG doped with borate in a Ca2+- and Mg2+-free Hanks’ Balanced Salt Solution without added cells. 66 Although they did not observe similar changes with the original 45S5-BG composition (i.e. without the incorporation of borate), their results showed that stimulation of angiogenesis on the CAM model does not require the addition of cells. Other biomaterials, such as biodegradable polymer scaffolds, have also been successfully used in the CAM assay without the addition of exogenous cells. For example, Stanzani et al. demonstrated that poly (3‐hydroxybutyrate‐co‐3‐hydroxyhexanoate)–nanocellulose composite scaffolds can intrinsically promote angiogenesis in the CAM assay, suggesting that specific material characteristics may be the cause of neo-vascularisation rather than angiogenic growth factors released by added cells. 67 These results confirm those of the present study, underscoring that specific biomaterials can modulate the CAM’s angiogenic response intrinsically.
In other studies, no changes in CAM vascularisation were observed, when in direct contact with cell-free 45S5-BG scaffolds.35–37 Gorustovich et al. and Vargas et al. tested for the first time the potential of the CAM assay as a biocompatibility assay for BGs by applying the scaffolds directly to the CAM without the addition of hydrogel, CCM or other supporting components.36,37 While the primary focus of their studies was on biocompatibility, no effect on vascularisation was observed up to five days after transplantation. When the scaffolds were explanted and examined histologically, it was found that the scaffolds were encapsulated by an epithelial layer and thus were separated from the circulation of the CAM. 37 These observations, that are in conflict with the observations made in this study, might be explained by differences in the experimental approach — i.e. 3D 45S5 BG derived scaffolds were applied without any additives (in this study, Cultrex was used as an additive) and without the scratching of the surface of the CAM prior to transplantation. Furthermore, the duration of this experiment was shorter than that of our study. In addition to this, Duan et al. described a synergistic effect of released silicon (Si) ions and VEGF co-delivered by porous Si microparticles. 68 A similar effect could occur in our study model, as Cultrex BME Type 3 contains pro-angiogenic growth factors.69–71 It is possible that the simultaneous application of MBGNs and Cultrex enhances the angiogenic effects of the MBGNs. In a study conducted by Ryan et al., 72 BG nanoparticle containing collagen scaffolds were transplanted onto the CAM in a technique that compares to that used by Gorustovich et al. 36 and Vargas et al. 37 Rather than an unstimulated group being used as the control, the study 67 employed a collagen scaffold control group. Between the two groups, no differences in angiogenic response were detected. However, when copper-doped BGs were used in the same study, angiogenesis on the CAM was significantly enhanced, demonstrating the potential of the CAM assay to detect intrinsic angiogenic potential of BGs.
Porous Si microparticles have been shown to induce pro-angiogenic effects on the CAM without the addition of cells, after only one day. 68 The biomaterial used releases Si ions in a similar pattern to the MBGNs used in our study. In addition, MBGNs are known to release ions more rapidly than 45S5-BG. 73 Therefore, MBGNs may be able to promote angiogenesis faster than 45S5-BG-based scaffolds in the CAM assay.
Furthermore, the angiogenic response of the CAM to cell-free 45S5-BG-based scaffolds has been previously assessed after only three or five days of incubation on the CAM instead of seven days, as in our study.35–37 This may be an important difference in the response of the CAM to BGs. While mesenchymal cells are found in the chorion as early as EDD8, the recruitment of mesenchymal cells into this layer is accelerated at later stages, reaching a peak at EDD11. 62 During this time, the proliferation of cells in the chorion also increases, peaking at EDD14. 62 It can be argued that the greater presence of these cells and their proliferative dynamics are predominant for the incorporation of the bioactive materials into the membrane, allowing them to fully influence angiogenesis only later, during embryonic development. This explanation is also supported by the increase in the expression of angiogenic marker genes during the later development stages of the CAM. Makanya et al. found a peak in most pro-angiogenic genes measured, namely vascular endothelial growth factor receptor 2 (VEGFR2), VEGFA, HIF-1α and stromal derived factor-1 (SDF-1), around EDD11, suggesting that the most pronounced effect of BGs could be found at this time point. 62 It should also be noted that the full effect of gene expression itself on vessel formation may only become apparent after this time. While this behaviour would explain the more pronounced angiogenic response in our study compared to that of Handel et al., the study of Haro Durand et al. showed a significant influence of the IDPs of BGs on angiogenesis in ovo at both two and five days after transplantation. 66 This early angiogenic response could be explained by the use of IDPs on the CAM and therefore a direct effect of the ions on the cells, rather than a gradual release of the ions from the BG scaffolds (or MBGNs) causing a delayed effect. A previous study characterised the release profile of the ions from the MBGNs used in this experiment, showing a steady release over a period of 7 days, supporting the notion that longer incubation times are required to stimulate angiogenesis in a CAM assay without added cells. 34 Similarly, the 45S5-BG composition has also been shown to release therapeutic ions for up to 17 days in in vitro studies. 74 Future studies should evaluate the role of the ions released from the MBGNs on angiogenesis — for example, by application of the ions onto the CAM, the role of each ion in inducing angiogenesis on the CAM could be assessed, allowing an even more detailed understanding of the role of ions in modulating the angiogenic properties induced by MBGNs. The ions could either be extracted from the glasses by incubation, representing the whole ionic composition of the respective BG composition, or the ions could be added separately in their biologically active form, e.g. adding salts or oxides. 21
It could be argued that without the addition of cells, subtle angiogenic effects may be missed, e.g. those initiated by the activity of cells (e.g. the release of angiogenic proteins) after exposure to MBGNs. This could limit the use of CAM assays without added cells. While this is a valid concern, there is already evidence available that subtle changes in the BG composition, such as the addition of molybdenum to MBGNs, can elicit distinct angiogenic responses in a CAM assay without the addition of cells. 34 Additionally, it should be noted that no potentiating effect of BMSCs on the angiogenic response in the CAM was observed in this study.
The intrinsic angiogenic potential of MBGNs, as demonstrated in our study, may contribute to the overall regeneration of bone defects, in addition to supporting vascularisation. This hypothesis is in agreement with the results of in vitro and in vivo studies, in which various BG compositions were shown to improve osteogenesis and bone regeneration.56,75,76 Hence, the intrinsic pro-angiogenic activity of MBGNs observed in the CAM assay may reflect an overall enhanced regenerative mechanism.
Conclusions
The data presented in this study support the feasibility of a CAM model without the addition of exogenous cells for evaluating the angiogenic properties of BG-based biomaterials. The CAM model can therefore be used to bridge the gap between in vitro and in vivo testing in the early stages of translational research, preserving the full complexity of angiogenesis while reducing reliance on animal models. The MBGNs used in this study exhibit intrinsic angiogenic potential independent of the addition of exogenous cells. Therefore, given an appropriate incubation time on the CAM, the CAM assay without added cells appears to be a valid method for assessing the angiogenic potential of BG-based biomaterials.
Footnotes
Acknowledgements
We gratefully acknowledge the financial support of the Deutsche Forschungsgemeinschaft (DFG). This study contains parts of Maximilian Moll’s and Tim Ahren’s doctoral theses.
Ethical considerations
The study protocol strictly followed the contents of the declaration of Helsinki in its present form. The use of the cells for the purpose of this study was approved by the ethics committee of the Medical Faculty of the University of Heidelberg (S-340/2018).
Consent to participate
Patients were informed prior to material collection and gave written consent for its collection.
Funding
This study was funded by the DFG, grant reference BO1191/26-1 and WE6654/2-1.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest in relation to this work.
