Abstract
Today's biomedical research requires instrumentation that is both functional and versatile. While high throughput screening (HTS) and drug discovery laboratories require instrumentation that can be automated, pilot assay laboratories may not necessarily need total automation. Towards that end, Bio-Tek has developed the μFill, a 96-/384-well microplate reagent dispenser capable of running stand-alone or computer-controlled as part of a robotics system. The μFill is compatible with both 96- and 384-well microplates, and using a specially designed adaptor can also dispense to deep-well microplates. It is capable of dispensing from 10–3000 μl for 96-well plates and 5–1500 μl for 384-well plates in 1-μl increments. The μFill can dispense 20 μl into a 96-well plate in four seconds and into a 384-well microplate in 12 seconds. The microprocessor-controlled syringe pump is based on a tested, low-maintenance design that requires no calibration, yet provides a high degree of accuracy and precision. The accuracy with an 80 μl dispense is within 1 μl with a percentage CV of less than 2%, and with a dispense volume of 20 μl, the percentage CV is still less than 5%. For those needing to dispense organic solvents or sterile aqueous solutions, a model that is autoclavable and has increased solvent resistance is also available. The programming allows for the control of flow rates from 225 μl/well/sec, for dispensing to cell cultures, and to 1000 μl/well/sec for rapid and vigorous reagent dispensing. The flexible software provides complete programming capabilities from the keypad. For more complete automation, robotics interfaces can be developed using ActiveX® software commands. The μFill's size, with a 14 times 14-inch footprint and a height of seven inches, allows it to be used almost anywhere.
Introduction
Biomedical research often requires the rapid and accurate dispensing of liquid reagents to microplates. The use of microplates has allowed for routine assays to be automated. Because of the small volumes associated with the wells of microplates, both 96- and 384-well, the ability to dispense small volumes accurately, precisely and in a timely fashion is necessary to perform most assays. Towards that end, Bio-Tek has developed the μFill 96-/384-Well Reagent Dispenser (Figure 1). The μFill has the capability of dispensing solutions to 96- or 384-well microplates without any hardware changes. Volume selection, dispense rate, strip count, and plate type are all software selectable from the instrument keypad. Removal of the plate holder allows the μFill to fill deep-well microplates as well. This reagent dispenser has a small footprint, a robot-friendly plate carrier, and ActiveX® software for easy robotics integrations. In this monograph we will discuss the μFill's speed, accuracy, and precision at fluid volumes that span its reported range.

μFill 96-/384-Well Microplate Dispenser with aerosol lid.
Materials and Methods
Dispense accuracy and precision were determined using either a gravimetric method or the dilution of dye. Determinations using the gravimetric method were performed by weighing 8-well strips or plates using a Sartorius A 120S analytical balance. After dispensing fluid to the plate using the μFill, the plate and/or strip was quickly re-weighed. The resultant weight change, when divided by the number of plate or strip wells, returned an average per-well dispense-volume. When calculating the accuracy of dispense using the dye method, distilled water containing FD&C yellow number 5 dye was dispensed into plates with the μFill reagent dispenser. In order to adequately measure the absorbance of the colored solution at low dispense volumes, it was often necessary to add distilled water to each well. To insure thorough mixing prior to measurement, the plate was mixed on an orbital shaker (Bellco Technology, Vineland, NY) at a speed setting of seven with a 1 mm orbit for 30 seconds, after which the absorbance at 450 nm (630 reference) was measured using a μQuant microplate spectrophotometer (BioTek, Winooski, VT). The dye solutions were made fresh daily from powder (Warner-Jenkinson, St Louis, MO). The resultant absorbance values were compared to a pre-existing calibration curve and the dispense volume interpolated. The data from both experiments was then exported to Microsoft® Excel and the data reduction was performed.
Results
The time necessary for routine dispensing by the μFill was examined for various volumes at both the slowest and fastest dispense rates. The μFill can dispense 10 μl into each well of a 96-well microplate in less than eight seconds and 5 μl into a 384-well microplate in approximately 10.5 seconds. This time includes the time required to move the plate carrier from the home position to the manifold at the beginning of the dispense cycle as well as returning to the home position at the completion of the cycle. As one might imagine, with larger per-well volumes, the time required to fill a plate increases. Dispense-times for deep-well plates were also measured at volumes of 500, 1000, and 2000 μl per well. As demonstrated in Table 1, the time necessary for 300 μl to be dispensed into all 96 wells is 44.5 seconds at rate 1. However, at larger per-well volumes, faster fluid rates are available, which can cut the dispense time in half. The degree of increase is dependent on the volume being dispensed, with larger volumes showing the greatest increase. Similar results were seen when 384-well plates were used. The standard model of the μFill Reagent Dispenser has five different dispense rates that affect the time required to fill a plate.
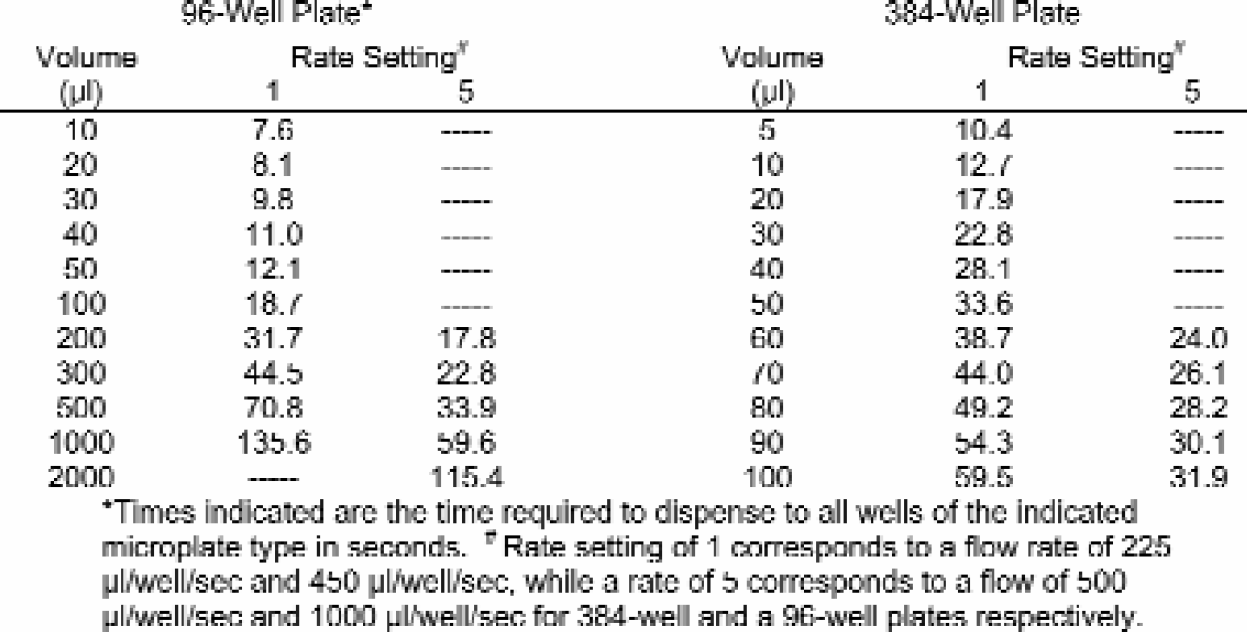
Times indicated are the time required to dispense to all wells of the indicated microplate type in seconds.
Rate setting of 1 corresponds to a flow rate of 225 μl/well/sec and 450 μl/well/sec. while a rate of 5 corresponds to a flow of 500 μl/well/sec and 1000 μl/well/sec for 384-well and a 96-well plates respectively.
The accuracy and precision of the μFill were determined using a number of different techniques. As demonstrated in Table 2, the μFill is accurate across the entire range of its volume settings for 96-well plates. When the minimum setting for 96-well plates (10 μl) was selected, the dispense-volume, determined gravimetrically, was found to deviate from the expected rate by 11.7% (8.8 μl vs. 10.0 μl). The deviation rapidly diminished with larger volumes to less than 1%. In all cases, the dispense volume was quite precise. As demonstrated in Figure 2, the coefficient of variance (percentage CV) was found to be less than 7% at 10 μl per well. The percentage CV also decreased rapidly with larger volumes to less than 1% at volumes above 50 μl per well. When the dispense accuracy into 384-well microplates was examined, similar results were found. When dispensing 5 μl into each well of a 384-well plate, the average deviation was calculated to be 12.4% (Table 3). Again, as the per-well volume was increased, the percentage deviation from the expected quickly diminished to less than 1% by 20 μl per well. The precision when dispensing into 384-well plates was also quite good, with percentage CVs being less than 4% at the lowest volume setting (Figure 3). Because the μFill enables the selection of different fluid dispense-rates, the accuracy of pipetting different volumes at different rates was examined. As demonstrated in Figure 4, the ability of the μFill to accurately dispense fluids is not compromised at higher dispense speeds. Using the rate settings of 1, 3, and 5, which represent fluid dispense rates of 450, 750, and 1000 μl/well/sec, for 96-well plates respectively, the determined volumes were virtually identical. However, in order to maintain accuracy and precision, the higher dispense rates are not available at lower dispense volumes.
Dispense accuracy into 96-well plates. Individual 8-well microplate strips were weighed. After the μFill Dispenser had dispensed the indicated volume of fluid into each well of 12 strips (96 wells), the strips were re-weighed. The volume of each well was calculated by dividing the change of weight by the number of wells (8) in a strip. Note that the data represent the average of 12 different 8-well strip determinations. The percent deviation is the ratio of the difference between the calculated and expected values to the expected values.
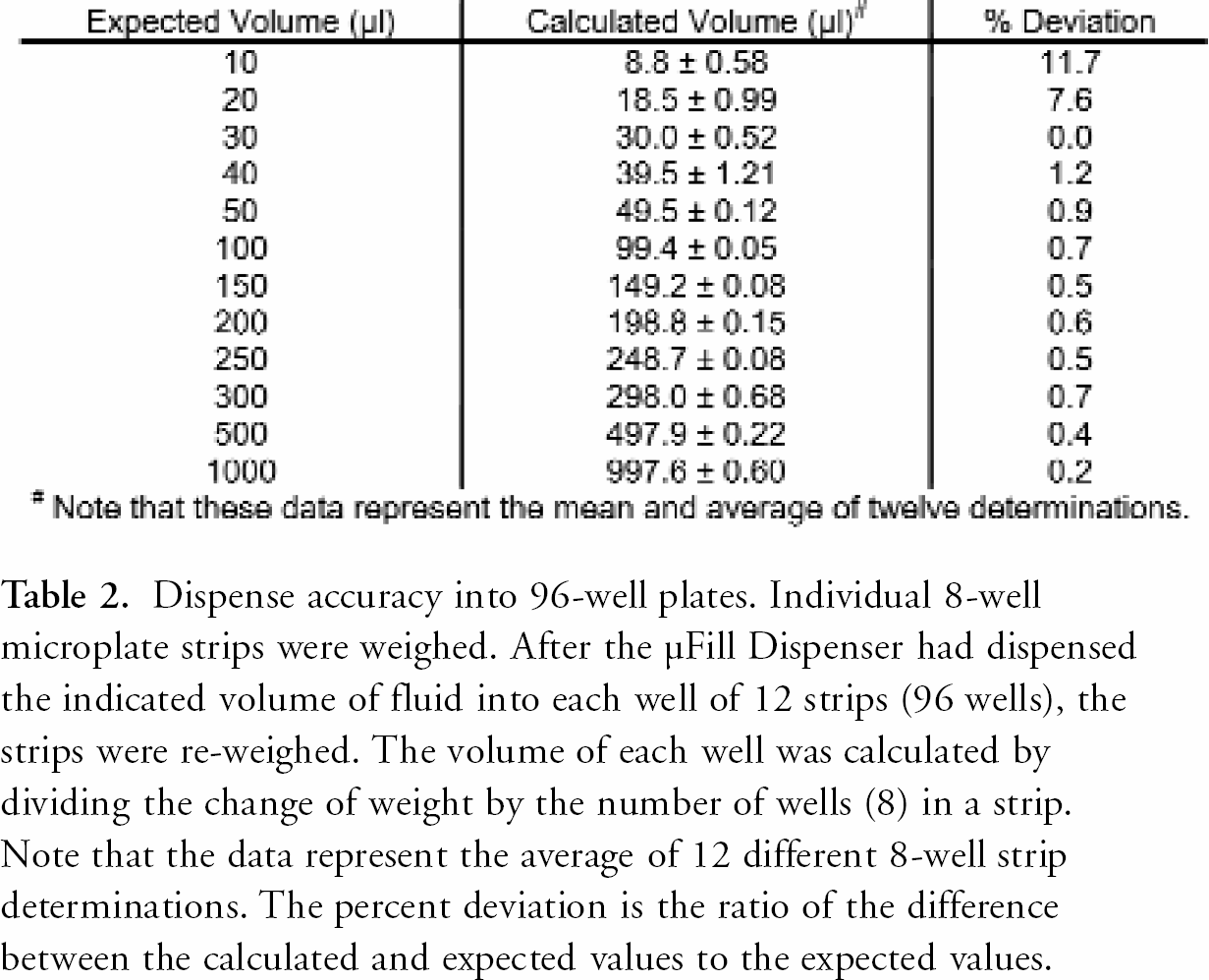
Note that these data represent the mean and average of twelve determinations.
Dispense accuracy into 384-well plates. Nunc flat-bottomed 384-well microplates were weighed. After the μFill Dispenser had dispensed the indicated volume of fluid into each well of the 384-well plate, the plates were re-weighed. The volume of each well was calculated by dividing the change of weight caused by the number of wells (384) in a plate. The percent deviation is the ratio of the difference between the calculated and expected values to the expected values. Each data point represents the mean and standard deviation of eight different 384-well plates.
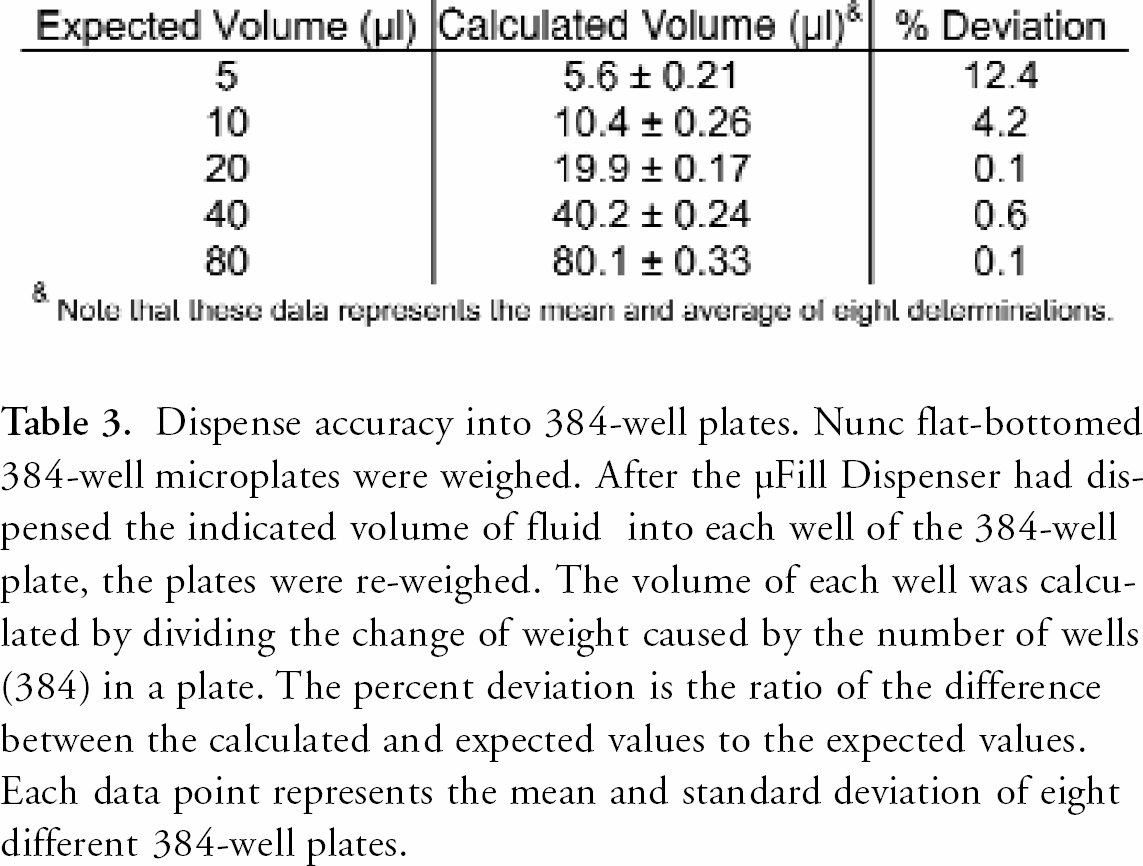
Note that these data represents the mean and average of eight determinations.
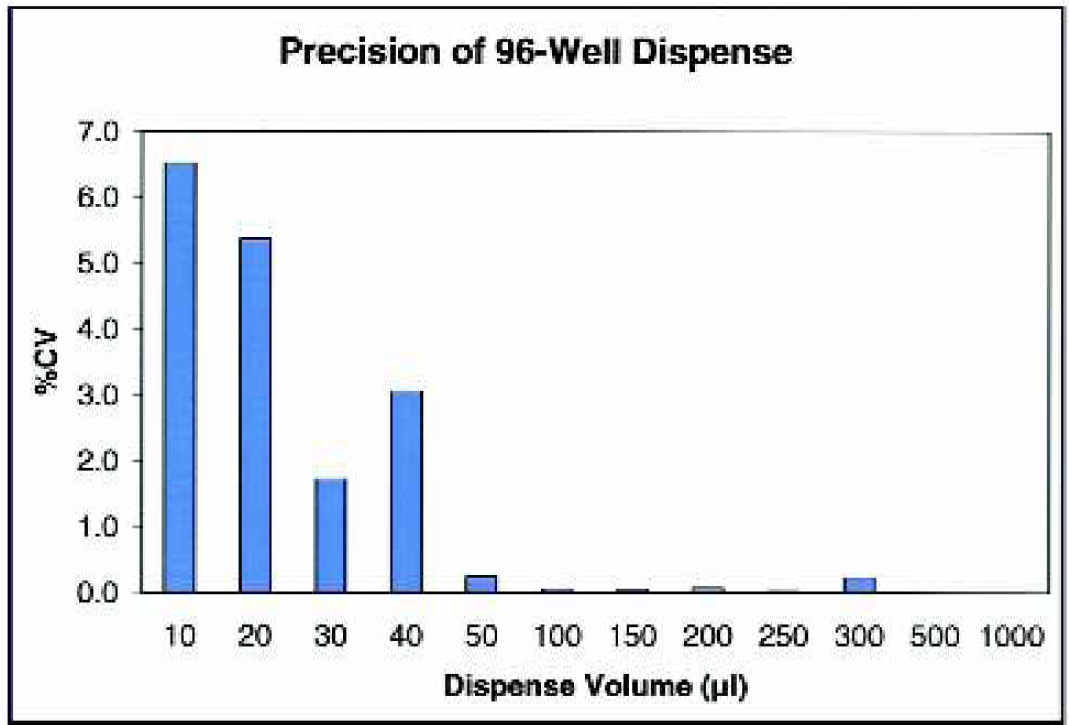
Dispense precision into 96-well plates using the μFill 96-/384-Well Reagent Dispenser at various dispense volumes. Each of the indicated volumes of deionized water was dispensed to 12 8-well strips using the μFill. For each strip of 8 wells, an average per-well dispense-volume was calculated gravimetrically. Note that each data bar represents the percentage CV for the average of the 12 strips.
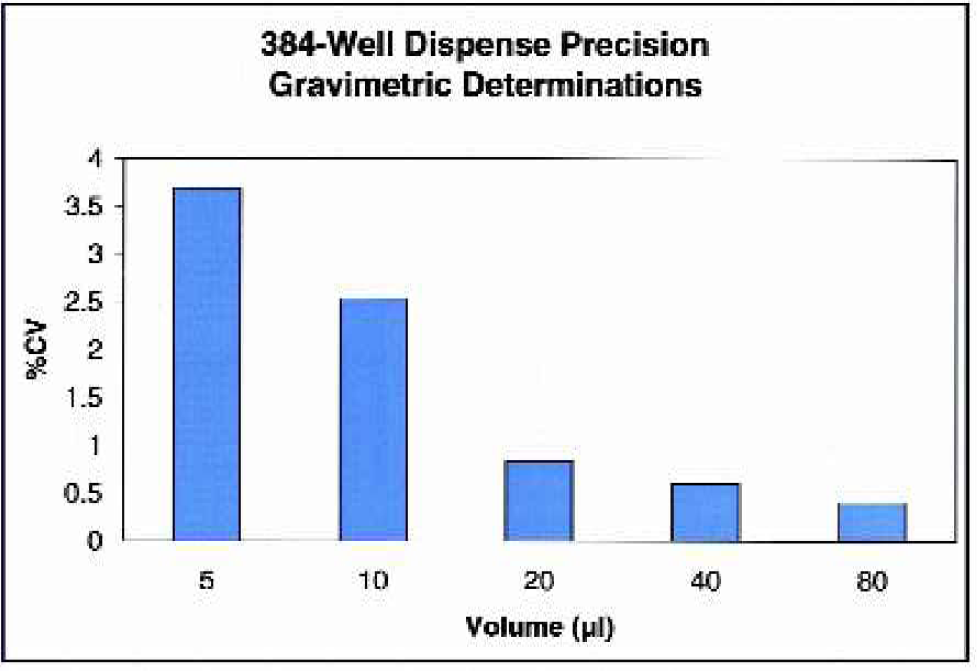
Dispense precision into 384-well plates using the μFill 96-/384-Well Reagent Dispenser at various dispense volumes. Each of the indicated volumes of deionized water was dispensed to eight 384-well microplates using the μFill. For each plate, an average per-well dispense-volume was calculated gravimetrically. Note that each data bar represents the percentage CV for the average of the eight plates.
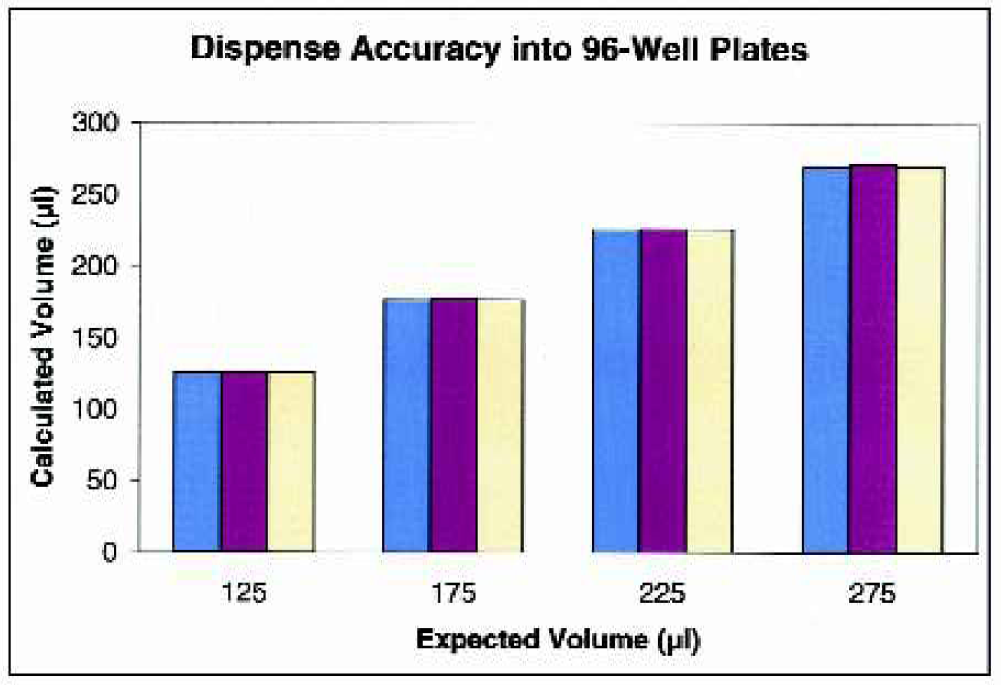
Comparison of Dispense Accuracy with Dispense Rate. Each of the indicated volumes of yellow dye solution was dispensed into three strips of a Costar 96-stripwell plate at three different dispense-rate settings. After dispensing, the absorbance was measured and the values interpolated against a previously prepared standard curve.
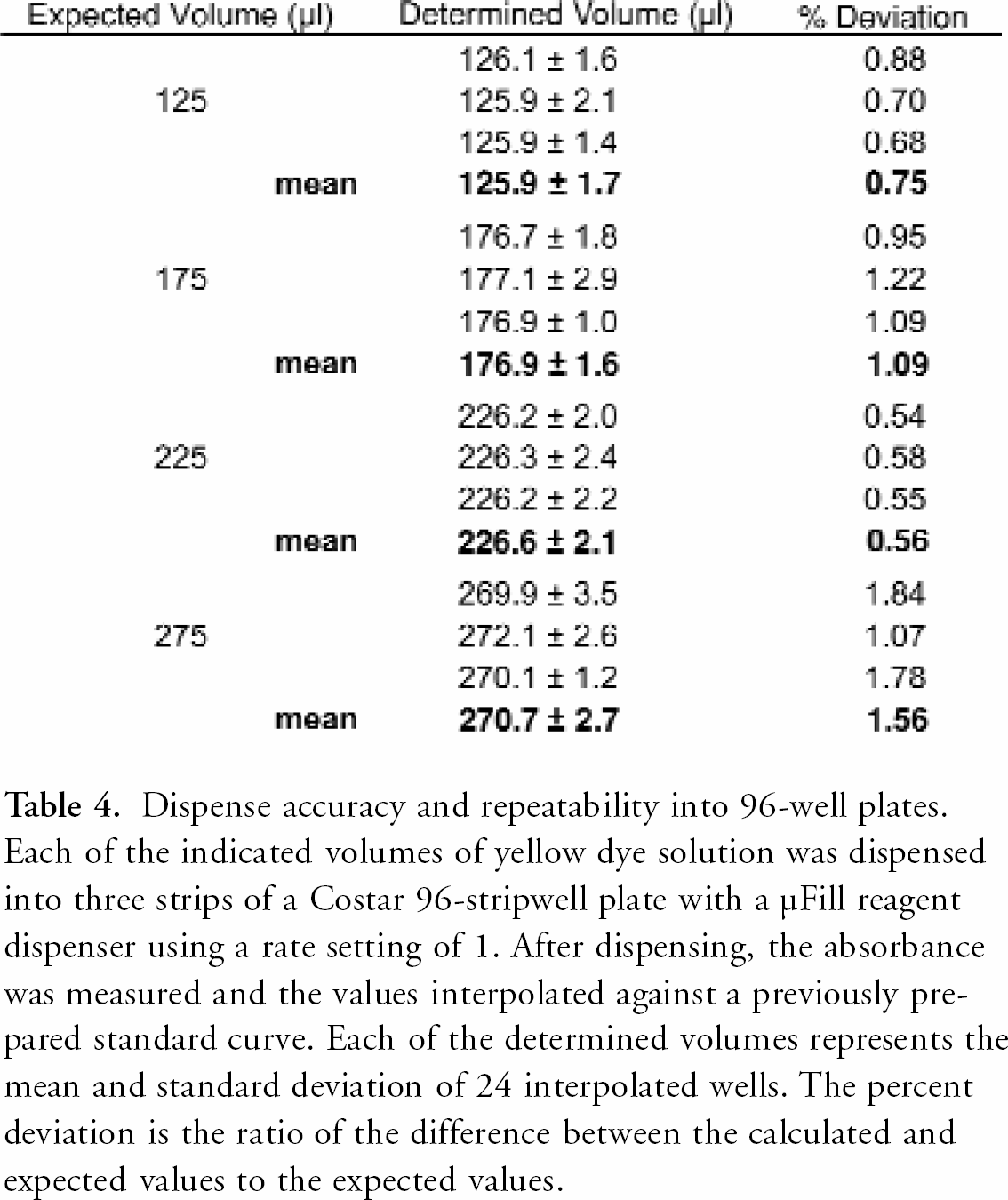
Using a colored dye solution, the accuracy and precision of the μFill can be determined colorimetrically. In these experiments, the absorbance of dye solution dispensed into the wells of microplates can be compared to previously prepared standard curves and the volume calculated by interpolation of the curve. As demonstrated in Table 4, the determined volume when dispensing various volumes of solution into 96-well plates agrees closely with the expected value. In addition, the data demonstrate the repeatability of the μFill, as three separate experiments returned similar volumes. Figure 5 demonstrates the dispense precision into 384-well plates. While the coefficient of variance (percentage CV) using absorbance is slightly higher than what was recorded using the gravimetric methods, the overall pattern of higher CVs at the lowest volumes remains the same. The increase in absolute values most likely reflects the natural variation of the meniscus of the solution in microplate wells. Note that in order to obtain adequate coverage of the wells with solution, it was necessary to add deionized water to all wells such that the final volume was 100 μl.
Dispense accuracy and repeatability into 96-well plates. Each of the indicated volumes of yellow dye solution was dispensed into three strips of a Costar 96-stripwell plate with a μFill reagent dispenser using a rate setting of 1. After dispensing, the absorbance was measured and the values interpolated against a previously prepared standard curve. Each of the determined volumes represents the mean and standard deviation of 24 interpolated wells. The percent deviation is the ratio of the difference between the calculated and expected values to the expected values.

Dispense precision into 384-well plates using the μFill 96-/384-Well Reagent Dispenser at various dispense volumes. Each of the indicated volumes of yellow dye solution was dispensed into two strips of a Nunc 384-well plate with a μFill Reagent dispenser using a rate setting of 1. After dispensing, the absorbance was measured at 450 nm (630 nm reference) using a μQuant microplate spectrophotometer. Each of the data bars represents the percentage CV of 24 absorbance-measurements.
Figure 6 demonstrates the linearity of dispense of the μFill reagent dispenser. Using the Link feature of the μFill, several individual dispense routines can be run in succession, each with a different volume setting. Volumes of dye solution ranging from five to 60 μl, in 5-μl increments, were dispensed into a 384-well microplate (two strips at each volume). After dispensing the dye solution, deionized water was added to all wells such that each well contained 100 μl of solution and the absorbance for each well of the microplate was determined. When the mean absorbance at each volume is plotted against the volume dispensed a linear relationship is observed. When linear regression analysis of the data was performed, the coefficient of regression (r2) was calculated to be greater than 0.999. The resolution of dispense is demonstrated in Figure 7. When dispense routines in increments of 1 μl are linked together and the resultant absorbance values plotted, the mean value for each volume is linear. The coefficient of regression of dye solution was calculated to be greater than 0.999. Note that while minimum and maximum values at each volume overlap, the two parallel the same trend line as the mean.
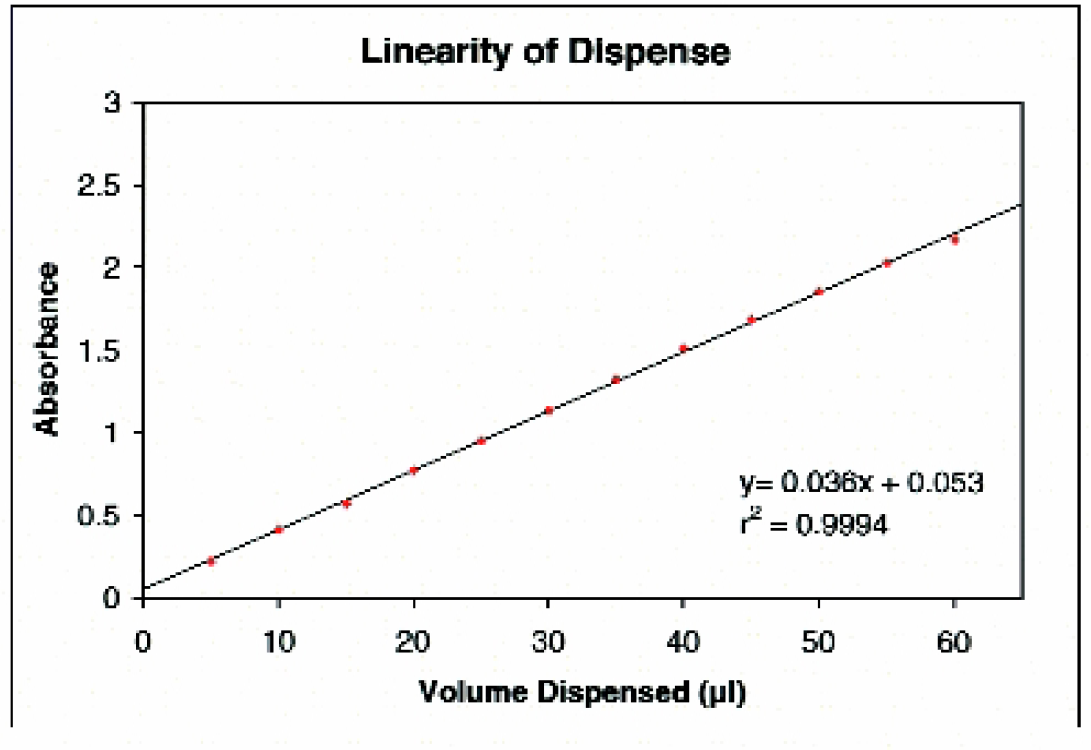
Linearity of Dispense using the μFill Reagent Dispenser. Using the “link” function of the μFill, several different dispense protocols were linked together such that various volumes, in 5-μl increments, of yellow dye solution were dispensed to two strips each of a Nunc 384-well plate. After dispensing, deionized water was added to the wells of the microplate such that all of the wells contained 100 μl of solution. The absorbance at 450 nm (630 nm reference) was then determined for each well using a μQuant microplate spectrophotometer. Data points at each volume (diamonds) were plotted along with the linear regression (line).
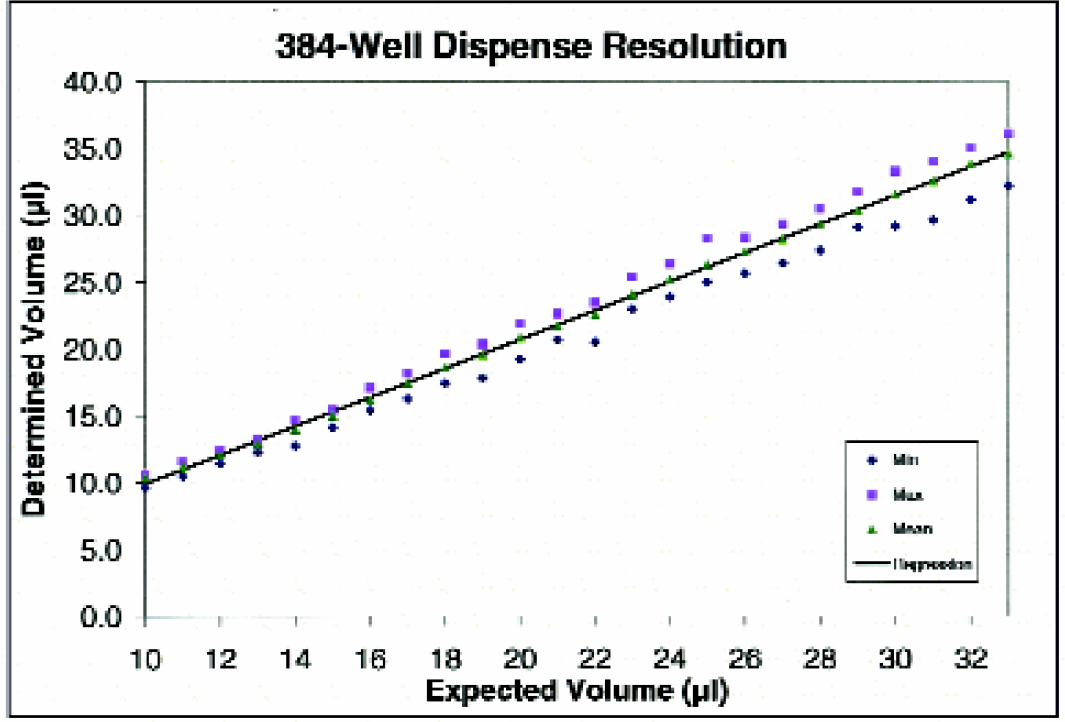
Dispense volume resolution of the μFill Reagent Dispenser. Using the “link” function of the μFill, several different dispense protocols were linked together such that various volumes, in 1 μl increments, of yellow dye solution were dispensed to strips of a Nunc 384-well plate. After dispensing, the absorbance at 450 nm (630 nm reference) was determined for each well using a μQuant microplate spectrophotometer and the values interpolated against a previously prepared standard curve. Data points representing the minimum (diamonds); maximum (squares); and mean (triangles) values at each volume were plotted along with the linear regression (line).
Discussion
The data indicate that the μFill Reagent dispenser can be used to dispense either large or small volumes precisely and accurately. The μFill routinely exceeded the accuracy and precision specification outlines for 20-μl and 80-μl dispense volumes. The μFill is accurate at any valid dispense rate, allowing the end user to decide the appropriate rate based on their application. The μFill's design is such that it can accommodate 96- and 384-well plates without changing the manifold. Deep-well microplates can be accommodated by removal of the plate holder from the carrier, while dispensing into plate types with intermediate heights can be accomplished via software control of the manifold dispense height. Multiple dispense routines can be linked together, allowing multiple per-well volumes, rows to be skipped, different dispense rates, all within the same plate.
The μFill can also be used to dispense solutions other than distilled water containing dye. Commonly used buffers such as TE (10 mM Tris, 1 mM EDTA) and phosphate buffered saline (PBS) have been dispensed with equivalent gravimetric results (data not shown). Because the μFill utilizes the same proven syringe-piston pump technology as Bio-Tek's microplate washers, solutions containing surfactants, such as Tween® 20 and Triton X-100, or proteins, such as casein and BSA, can be successfully dispensed as well. Interestingly enough, in our hands low concentrations (0.1% v/v) of Tween® 20 or Triton X-100 will often improve the percentage CVs when using the dye method (data not shown). This phenomenon is most likely the result of the surfactant providing a more uniform meniscus from well to well leading to more uniform absorbance measurements.
There are several priming features available with the μFill. The priming trough has a fitting that allows for a drain tube to be connected. Besides the prevention of overflow from the trough by removal of primed reagent, the reagent can be recollected and precious reagent can be saved. Built-in software features include an autoprime feature that allows for periodic priming of the reagent dispenser during periods of inactivity. Selection of the “New Buffer Prime” provides for a sufficient priming volume to purge the fluid path of one reagent and replace it with another.
The small footprint of the dispenser allows for easy use in biosafety cabinets, as well as incorporation into automated systems. The μFill is based on proven microplate washer technology that ensures a high degree of accuracy and precision for long periods of time. The manifold and tubing are easily accessible for cleaning or replacement.
Conclusions
The μFill exceeds the accuracy and precision specifications at 20 μl and 80 μl.
The μFill's dispense rate does not affect accuracy and precision. While not all rates are available for low-volume dispense routines, selection of a valid rate setting does not alter performance, merely the time required for dispensing.
The μFill is a versatile and capable instrument capable of dispensing into multiple plate types and formats without any hardware changes other than to remove the plate holder for deep-well microplates.
The range of dispense volumes is only limited by the capacity
