Abstract
The open-source release of self-replicating rapid prototypers (RepRaps) has created a rich opportunity for low-cost distributed digital fabrication of complex 3-D objects such as scientific equipment. For example, 3-D printable reactionware devices offer the opportunity to combine open hardware microfluidic handling with lab-on-a-chip reactionware to radically reduce costs and increase the number and complexity of microfluidic applications. To further drive down the cost while improving the performance of lab-on-a-chip paper-based microfluidic prototyping, this study reports on the development of a RepRap upgrade capable of converting a Prusa Mendel RepRap into a wax 3-D printer for paper-based microfluidic applications. An open-source hardware approach is used to demonstrate a 3-D printable upgrade for the 3-D printer, which combines a heated syringe pump with the RepRap/Arduino 3-D control. The bill of materials, designs, basic assembly, and use instructions are provided, along with a completely free and open-source software tool chain. The open-source hardware device described here accelerates the potential of the nascent field of electrochemical detection combined with paper-based microfluidics by dropping the marginal cost of prototyping to nearly zero while accelerating the turnover between paper-based microfluidic designs.
Introduction
Bowyer’s open-source release of self-replicating rapid prototypers (RepRaps)1–3 has created a rich opportunity for low-cost distributed digital fabrication of complex 3-D objects. There has been a surge of interest among scientists from a wide swath of fields in using RepRap 3-D printers to design, manufacture, and share the open-source digital designs of scientific equipment.4–6 As these open-source digitally replicable scientific tools are freely distributed and thus widely accessible for the cost of materials, 3-D printing components, or entire devices, the result is substantial economic savings—with costs that are generally 90%–99% less than those of proprietary commercial equipment. 5 Preliminary analysis of the economic value of an open hardware approach 7 indicates that scientific funders can obtain a return on such investments in the hundreds or thousands of percent. 8 This has resulted in an explosion of high-quality 3-D printable lab tools for biology labs 9 and biotechnological and chemical labware,10–12 optics and optical system components, 13 colorimetery, 14 nephelometers 15 and turbidimeters, 16 nitrate testing, 17 liquid autosampling 18 with robot arms, 19 automated sensing arrays, 20 and compatible components for medical applications. 21 Not only are relatively inexpensive 3-D printed parts being used to replace conventional scientific tools, but they are also being used to create new scientific opportunities for investigation. For example, utilizing a 3-D printer in the lab makes it easier for researchers to design and build laboratory tools faster and more closely aligned to specific research needs. For example, a highly configurable reaction vessel made with a 3-D printer can integrate reagents, catalysts, and a purification apparatus into a single reactionware,22,23 which can significantly simplify multistep reactions by integrating equipment into a single component. These 3-D printable reactionware devices offer the opportunity to combine open hardware microfluidic handling 24 with simple “lab-on-a-chip” reactionware. 25
There is a large interest in creating such inexpensive lab-on-a-chip devices for rapid detection of health-related biomarkers,26,27 explosives,28,29 and gas components. 30 The goal of such applications is to save lives by detecting molecules in short periods of time, typically less than 1 h. While the field of microdevices is currently exploding, cost is a constant concern with these devices. In 2008, the Whitesides group pioneered paper microfluidics. 31 This discovery was quickly followed by other complex 3-D structures made in paper that controlled the capillary flow of biological liquids by creating channels bordered with hydrophobic wax.32–34 Such paper microfluidic devices have reduced or eliminated the need for pumping of fluids, and this reduces both the footprint and cost of the overall device. The paper base is also inexpensive and readily available throughout the world, making it an ideal point-of-care platform. Originally, groups were creating wax channels using lithography, 32 but this is time-consuming and expensive. Recently, wax printing has been developed that provides more flexibility in design and reduces the cost of device creation. 33
To further drive down the cost while improving the performance of lab-on-a-chip paper microfluidic prototyping, this study reports on the development of a RepRap upgrade capable of converting a Prusa Mendell RepRap into a wax 3-D printer for paper-based microfluidic applications. An open-source hardware approach is used to demonstrate a 3-D printable upgrade for the 3-D printer, which combines a heated syringe pump with the RepRap/Arduino 3-D control. The bill of materials, designs, basic assembly, and use instructions are provided along with a completely free and open-source software tool chain. The results of this approach are discussed.
Materials and Methods
The mechanical and electrical design of the wax 3-D printer upgrade followed standard RepRap design methods for scientific equipment outlined in detail elsewhere, 5 thus relying on only open-source electronics, RepRap printable custom parts, off-the-shelf globally available nonprinted parts (referred to as vitamins in the RepRap literature), and a completely open-source software tool chain. The designs are released under a CC-BY-SA license. In addition, no expensive tools, specialty equipment, or advanced skills are needed for assembly.
Both the custom 3-D printed parts and the wax print designs are created in OpenSCAD. 35 Open-source and freely available OpenSCAD is a script-based, parametric CAD program possessing powerful 3-D modeling capabilities. Three-dimensional models are created by adding and subtracting primitive objects created by code to produce the desired complex shapes. The script language is based on C++. As only a small number of methods are required to produce very complex designs, the learning curve is short for students with programming experience even if they are not familiar with conventional CAD. The scripts are written such that designs are parametric; thus, by changing the values of a single variable, the design can easily be altered. Models rendered in OpenSCAD are saved as a (.scad) file and are also exported as a stereolithography (.stl) file for the slicing process. Slicing converts an stl file into layers of equal thickness in the z direction in G-code, a human-readable file format specifying the path the print head must follow to produce a physical object. Cura, an open-source and freely available package, was used to slice the stl models. 36
The mass of the printed parts was calculated with Cura LulzBot Edition 14.09-1.18 using a normal quality setting for polylactic acid (PLA) for the syringe carriage, and the remainder of the parts were printed in acrylonitrile butadiene styrene (ABS) for its relatively higher-temperature resistance.
Results and Discussion
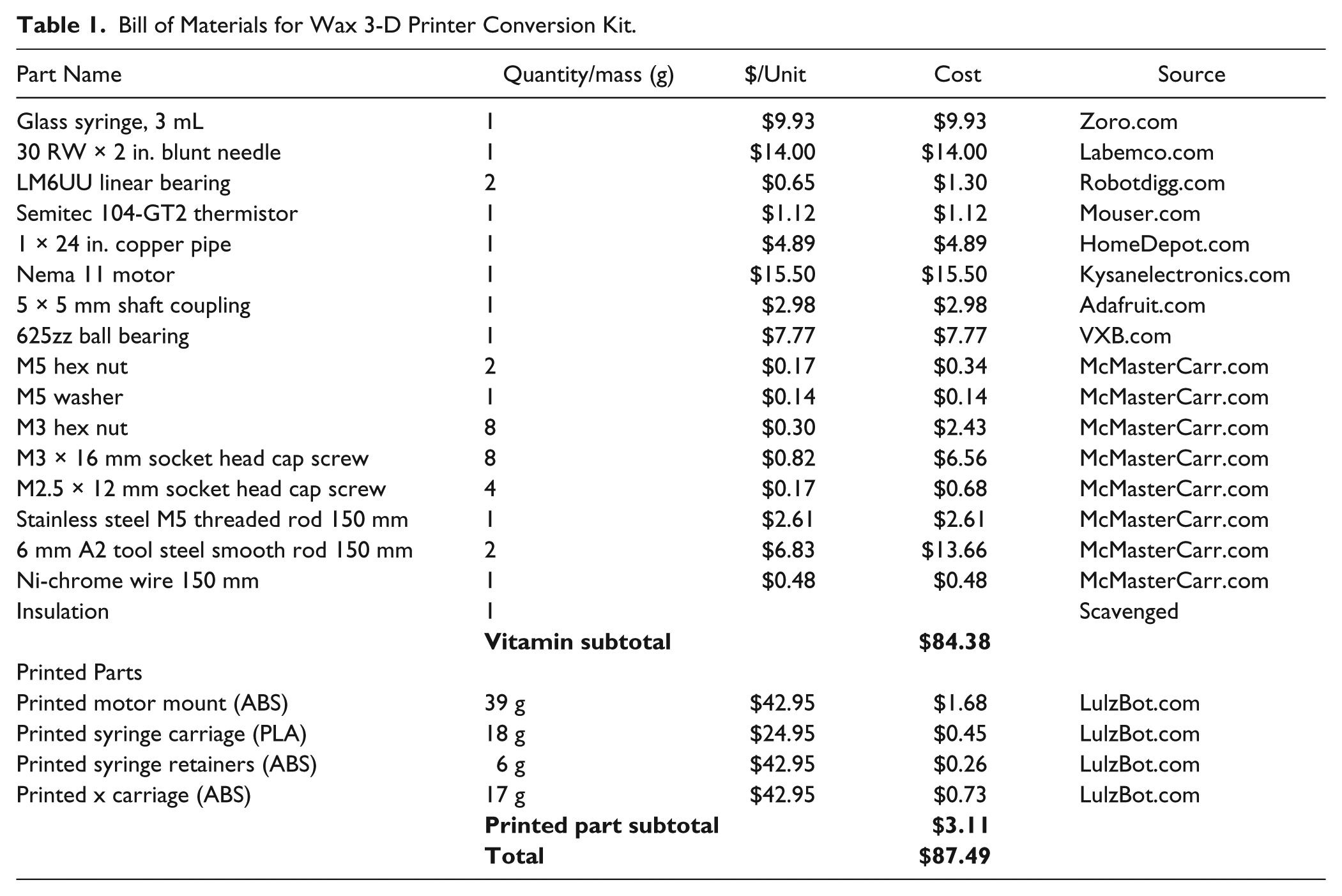
The syringe design was derived from the Wijnen et al. open-source 3-D printable syringe pump library. 37 There are five 3-D printed custom parts needed for the conversion, including a motor mount ( Fig. 1 ), syringe carriage ( Fig. 2 ), x carriage ( Fig. 3 ), and two syringe retainers ( Fig. 4 ). The 3-D printed parts can be printed in about 5 h on any standard RepRap 3-D printer, consuming $3.11 of commercial PLA filament. The entire bill of materials, including part, count, cost, and source, totals $87.49, as seen in Table 1 . This bill of materials assumes that only a single conversion kit is purchased, and it should be pointed out that there are discounts for most of the components when purchased in bulk (e.g., fasteners). The conversion kit assembly follows the basic steps outlined by Wijnen et al. 37 for syringe pumps and is shown completed without the syringe in Figure 5 .

Cura rendering of motor mount.

Cura rendering of syringe carriage.

Cura rendering of x carriage.

Cura rendering of the small and large syringe retainers.
Bill of Materials for Wax 3-D Printer Conversion Kit.

Assembled conversion kit without syringe.
The Prusa Mendell RepRap, 38 which can be fabricated for less than $600 and assembled in 24 h, 39 is itself capable of printing all of the conversion components ( Figs. 1 – 4 ). After printing the ABS and PLA components with the standard direct drive head, the mechanical components of the syringe depressor are assembled as shown in Figure 5 and the heater made up of the copper pipe and nichrome heating element wrapped in Kapton polyimide tape for the syringe is installed ( Fig. 6 ). Then the syringe is placed into position along with the temperature feedback provided by the thermistor and the assembly is insulated as shown in Figure 7 . Finally, the extruder driver and standard hot end is removed to be replaced by the assembled syringe pump ( Fig. 8 ). It should be noted that similar conversions can be made for all other RepRap designs using a direct drive extruder, although further work is needed to convert delta-style 3-D printers with Bowden sheaths.

Assembled conversion kit with heater and copper pipe installed.

Fully assembled conversion kit with syringe, thermistor wiring, and insulation.

Prusa Mendel open-source RepRap converted wax 3-D printer.
The wax printer can then be fully assembled utilizing a Prusa Mendel open-source RepRap 3-D printer for the frame and electronics as shown in Figure 8 . The resultant wax 3-D printer has a horizontal print area of 150 mm width by 200 mm length, and about 30 mm in height. The printer uses an open-source electronics prototyping platform of the Arduino Atmega 2560, 40 with a RepRap Arduino Mega Pololu Shield (RAMPS) to control the motors and heating element. 41 The glass syringe is then filled with a wax and heated to melting, and the wax is printed on a paper substrate as shown in Figure 9 at 5 mm/s.

Wax printing on paper substrate.
Similar to the design of the conversion kit, the design of the microfluidic device takes place in OpenSCAD using parametric variables (e.g., changing the gap width variable alters the distance between the two lines of wax in a test print). The design is then exported as an stl in order to be sliced with Cura into several layers of G-code, a language that is understood and controls the movements of the 3-D printer. Thus, complex geometries for the wax print can be easily designed and printed as shown in the example in Figure 10 . The settings for Cura that are used to dictate the boundaries and how the motors move are specific to each printer. With the wax printer, Cura is set up to produce slow-moving prints, around 5 mm/s, with an extrusion rate of less than 0.5 mm/s for the wax (compared to conventional RepRap printing materials), in order to maintain print quality. The printer is meant to be operated hot enough to melt the wax, but close enough to the phase transition temperature for the wax to solidify shortly after it is deposited on the print bed. In the example shown in Figures 9 and 10 , the Cerita wax (normally used for investment casting) is printed at 70 °C. The G-code is then imported into one of several printer control software packages such as Repetier, 42 Printrun, 43 or Franklin, 44 which provides a user interface to control the printer. This software is used to load the G-code into the control board on the printer, which is then used to control the 3-D printer during the print. In this way, the marginal cost of changing a paper-based microfluidic design is reduced to the cost of the substrate paper and the wax used for the experiment. The far more time-consuming and expensive lithography-based prototyping steps, including transparency mask fabrication, SU-8 photoresisit for the channels, wafer cleaning, lithography, developing, sealing, and vacuum degassing, are eliminated. Conservatively, creating a single new microfluidic design using lithography-based methods costs more than the wax printer conversion kit capable of unlimited design options for nearly zero marginal cost. However, it should be noted that there are other lower-cost methods of microfluidic design available, such as the paper-based methods in Carrilho et al. 33 Future work is needed to compare these two methods, which would involve first optimizing the printing of the 3-D wax printer described. This is accomplished for each specific wax in a short power versus velocity study. First, an approximate extrusion temperature is found by ramping up the temperature while printing a constant velocity spiral. When this is done, a simple zigzag pattern is designed that increases the print head velocity at each corner. Then this pattern is repeatedly printed with a small x offset while varying the extrusion temperature and extrusion rate in a systematic fashion. After the optimal print head velocity and extrusion rate are found, a final set of runs are made while adjusting the temperature again by small increments to obtain optimal printing parameters. These parameters are then used in printing identical microfluidic devices using the two methods and the resultant devices are tested.

Printed wax clover leaf pattern before drive-in.
The Prusa has an x-y position accuracy of 20 microns and a minimum z accuracy of 0.4 microns. The heated bed on the Prusa can also be used to anneal the wax and/or further drive it into the paper substrate to obtain channel widths that the printer itself is not capable of obtaining. This, however, can only be achieved for a subset of geometries. Future work is needed to improve this resolution by, for example, moving to finer-pitched lead screws and belts, higher-quality stepper motors, and finer syringe needles. In addition, future improvements on the design could enable both retraction and easy removal of the plunger for wax filling, multimaterial printing with multiple heads, and indirect automated head swapping.
In addition to wax printing to replicate the work that has been accomplished in the past 33 for lower costs so that experiments are more accessible to a greater selection of scientists, 45 this 3-D printer also enables fabrication of fully integrated microdevices using a single tool and the creation of complex 3-D geometries for yet unexplored microdevice applications. For example, the most common detection method for paper microfluidic devices is colorimetric,46,47 and there are already well-established techniques for utilizing Arduino-based open-source electronics and RepRap 3-D printing to make handheld detection devices.14,15,17 However, colorimetric detection works well for yes/no detection, but is difficult to calibrate to a quantitative measure in microfluidic applications. For a microfluidic device, this is the difference between a pregnancy test and a blood glucose–level test. A more quantitative measure uses electrochemical detection. Electrochemical paper-based devices have been created to detect the oxidation of glucose with an indium-doped tin oxide electrode 47 and a more standard glassy carbon electrode. 48 Other examples include human tears being analyzed for ion detection 49 and the detection of cholesterol levels in human serum. 50 While electrochemical detection combined with paper-based microfluidics is still in its infancy, it has a high potential to become a useful and cost-effective microdevice platform for many different analytes, as well as serving to detect environmental hazards. The open-source hardware device described in this paper accelerates this potential by dropping the marginal cost of prototyping microdevices to nearly zero, while accelerating the turnover between paper-based microfluidic designs.
Footnotes
Acknowledgements
The authors would like to acknowledge the National Science Foundation for funding of this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received a grant from the National Science Foundation (CBET-1159425 and CBET-1510006).
