Abstract
We demonstrate direct nuclear delivery of DNA into live mammalian cells using the photothermal nanoblade. Pulsed laser-triggered cavitation bubbles on a titanium-coated micropipette tip punctured both cellular plasma and nuclear membranes, which was followed by pressure-controlled delivery of DNA into the nucleus. High-level and efficient plasmid expression in different cell types with maintained cell viability was achieved.
Introduction
Methods for intracellular gene delivery have been instrumental in biomedical research and clinical applications. Viral expression vectors and packaging systems have been an important method of choice for achieving stable, efficient, and high-level expression of delivered DNA sequences. However, this and other related methods have significant constraints that include often high cellular cytotoxicity and limited DNA size packaging capacity that may be restricted to a few kilo-base pairs (kpbs). 1 Nonviral gene delivery approaches, such as chemical and biophysical methods, have demonstrated efficient delivery of DNA mainly into the cytoplasm of cells. However, several significant barriers exist before the delivered DNA can enter the cell nucleus to be expressed. First, the DNA is often rapidly degraded by cytosolic nucleases, yielding a half-life of ~50–90 min. 2 Also, the diffusion of introduced DNA molecules larger than ~2 kbp is severely impeded within the gel–sol composition of the cytoplasm, resulting in inefficient DNA trafficking into the nucleus. 3 Finally, the nuclear envelope is impermeable to passive diffusion of molecules larger than 40 kDa (or 60 bp DNA), requiring inefficient and poorly defined active transport processes to move introduced DNA into the nucleus for expression. 4 As a result, except for lentiviral infection, 5 successful DNA transfection is limited to actively dividing cells in which the transgene can enter the cell nucleus during cell division when the nuclear envelope temporarily breaks down. Few methods exist that can overcome the barriers to direct DNA delivery into the cell nucleus to avoid these impediments for successful gene delivery and expression. Nucleofection uses electroporation and proprietary cell type–specific buffers to transfer DNA into the cell nucleus. 6 Also, direct nuclear injection using a glass micropipette <0.5 µm in tip diameter can achieve ~50–100% gene expression efficiency in mouse LMTK− cells. 7
Our group previously reported the delivery of a wide range of differently sized, shaped, and composed cargo into live cells using a photothermal nanoblade.8,9 Briefly, the nanoblade uses a pulsed laser illumination to trigger a localized vapor bubble on a capillary pipette tip coated with a heat-conducting, thermally stable metal that is in light contact with a cell plasma membrane. Fast bubble expansion and collapse by the “lightning rod effect” 10 transiently disrupt the plasma membrane through the generation of localized shear forces, enabling active, pressurized cargo transfer from the pipette bore into the cytoplasm without advancing the pipette into the cell interior. Here, we demonstrate a new enabling application in gene transfer technology by direct DNA delivery into the cell nucleus using the photothermal nanoblade. By simply positioning the nanoblade pipette directly above the cell nucleus, the rapid bubble expansion and collapse that are characteristic of this approach efficiently open both the plasma and nuclear membranes simultaneously for DNA delivery with maintained cell viability.
Results
Figure 1
illustrates the operating principle for direct cargo delivery into the nucleus using the photothermal nanoblade. Recipient cells and their nuclei are visualized by standard phase contrast microscopy on an inverted microscope stage. The nanoblade pipette is fabricated by sputter deposition of a 100 nm thick titanium thin film on a glass microcapillary tip with a 1.5 µm tip diameter, and its hollow bore is filled with green fluorescent protein (GFP) encoding plasmid DNA. Using a standard stage-anchored micromanipulator, the nanoblade pipette tip is visually positioned on top of the cell nucleus. The pipette is then lowered until the tip comes into gentle contact with the cell’s plasma membrane without appreciable indentation. A nondamaging, nonfocused laser pulse (532 nm in wavelength, 6 ns in pulse width, and 180 mJ/cm2 in fluence, with parameters readily adjustable based on cell type) transiently irradiates the nanoblade pipette tip to induce an explosive vapor bubble that disrupts the underlying plasma and nuclear membranes simultaneously. A synchronized pressure-driven flow transfers plasmid DNA from the delivery pipette directly into the cell nucleus (
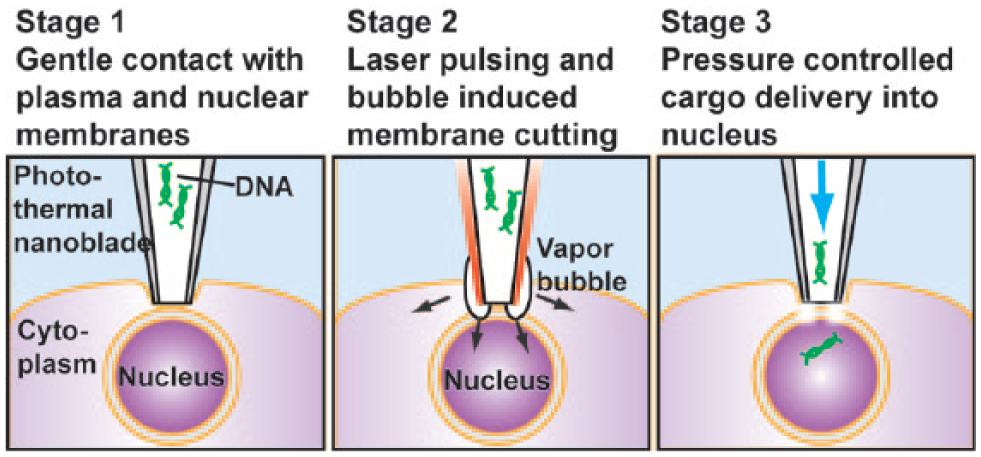
Schematic of cargo delivery directly into a cell nucleus with the photothermal nanoblade. A titanium-coated glass micropipette is positioned lightly touching the plasma membrane directly on top of a target cell nucleus. After a nanosecond laser pulse illumination, an explosive vapor bubble disrupts both the plasma and nuclear membranes in contact with the nanoblade tip. Synchronized pressure-driven flow transfers DNA into the cell nucleus before the membranes reseal.
To verify cargo delivery into the cell nucleus, a high-molecular-weight red fluorescent dextran molecule (tetramethylrhodamine dextran, 70 kDa, 1 mg/mL) was co-delivered with a GFP encoding plasmid (pmaxGFP, 3 kbp, 10 µg/mL). When the cargo was successfully delivered into the nucleus, red fluorescence could be detected only in the nucleus (
Fig. 2
, top row). In contrast, when the cargo was delivered into the cytoplasm, red fluorescence was excluded from the nucleus (
Fig. 2
, bottom row). This is due to the nuclear envelope barrier to passive diffusion for macromolecules >40 kDa in cells in which the nuclear membrane has not broken down during cell division. In some cases, cargo was delivered into both the nucleus and the cytoplasm, and was counted as a successful nuclear delivery. For each run, plasmid and dextran were delivered into ~50 cells with the photothermal nanoblade. Treated cells were cultured in a stage-top incubation chamber and imaged every hour for 24–32 h post delivery. Hourly time-lapse imaging is required because nuclear dextran could be repartitioned into the cytoplasm once the delivered cell undergoes mitosis (
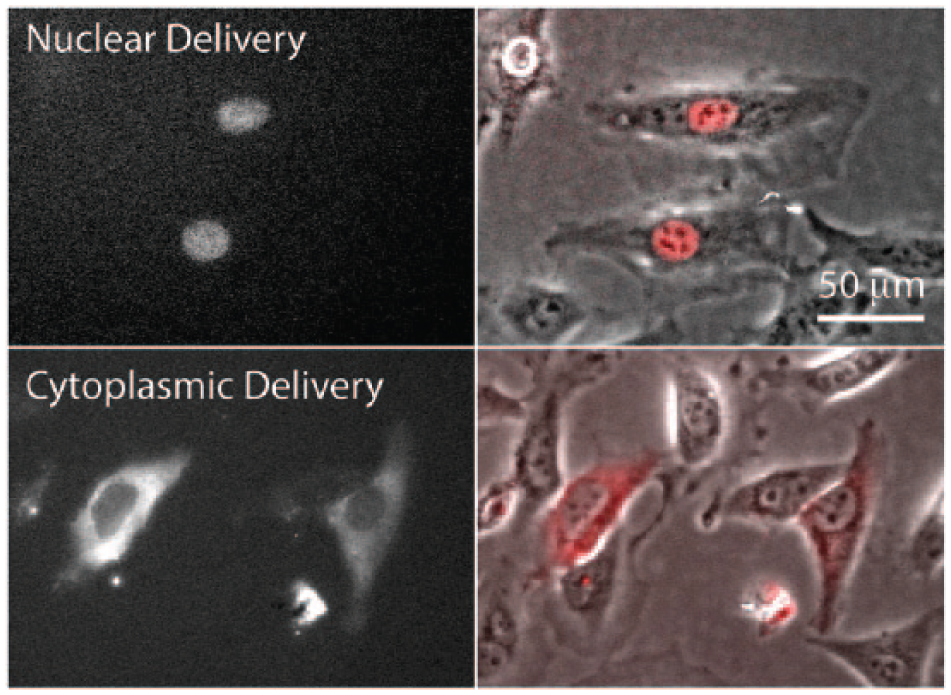
Nuclear versus cytoplasmic cargo delivery by the photothermal nanoblade. Fluorescent dextran (70 kDa molecular weight) was delivered successfully into the nucleus of HeLa cells and verified by localized fluorescent signals restricted to the nucleus (top panels) using phase contrast (right panels) and immunofluorescence (left panels) microscopy.
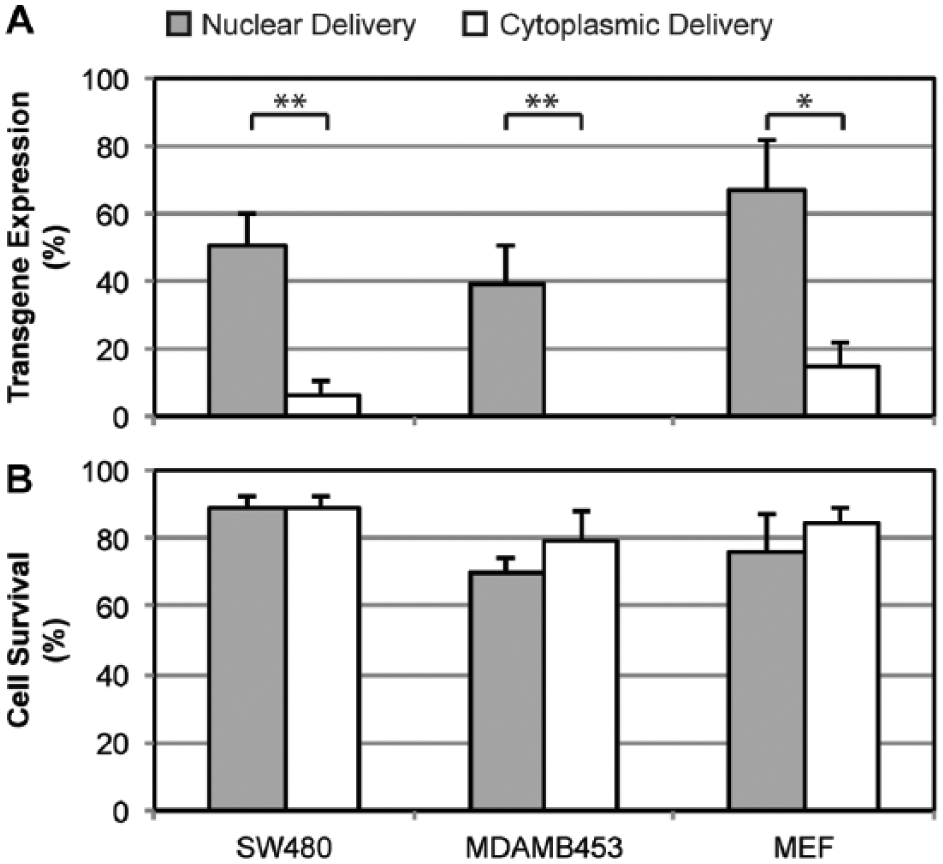
Comparison of green fluorescent protein (GFP) transgene expression and cell viability for nuclear versus cytoplasmic delivery by the nanoblade into human cancer cell lines SW480 and MDA-MB-453 and primary mouse embryonic fibroblasts (MEFs). Nuclear delivery of DNA yielded significantly higher GFP transgene expression compared to cytoplasmic delivery in all three cell types, and cell viabilities were maintained relatively constant. The symbols * and ** represent a significant difference between nuclear and cytoplasmic delivery for
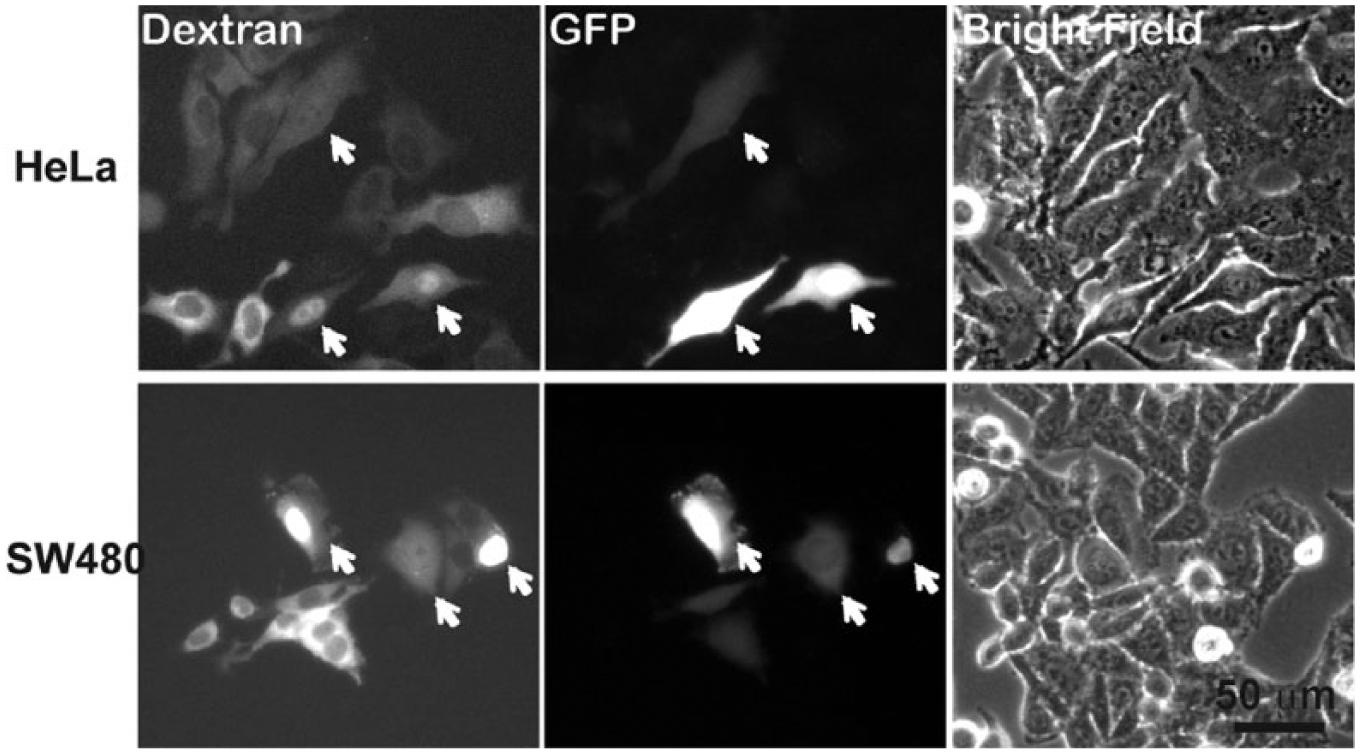
Green fluorescent protein (GFP) plasmid expression in HeLa and SW480 cells for nuclear delivery versus cytoplasmic delivery. Nuclear-delivered cells were marked by white arrows. GFP transgene expression was significantly elevated for nuclear- compared to cytoplasmic-delivered cells.
Discussion and Conclusion
To exclude the possibility of mechanical penetration through the cell membrane by the micropipette tip, “mock deliveries” were performed before each nanoblade-enabled delivery. First, the nanoblade pipette was positioned on top of either the cell nucleus or cytoplasm, then fluid pumping was initiated without laser pulsing. No cell membrane opening was confirmed before carrying out actual nanoblade-enabled deliveries with both laser pulsing and fluid pumping. The frequency of observing successful cargo delivery during “mock deliveries” was <5%, and, therefore, the data were considered as background and discarded. For conventional nuclear microinjection using a submicron glass micropipette, a zigzag motion of the sharp needle tip is needed to efficiently penetrate the cell and then nuclear membranes. This requires highly skilled operators, and the fine needle tip can be broken easily during the process. In contrast, the photothermal nanoblade requires only gentle contact of the comparatively broad nanoblade tip with the cell membrane. By using a pulsed-laser triggered cavitation bubble, the nanoblade opens both the plasma and nuclear membranes simultaneously without advancing the capillary tip into the cell, minimizing cell trauma. Active fluid pumping provides efficient delivery for large molecules or objects in contrast with other noncontact laser approaches, which rely on passive cargo diffusion across transient membrane pores opened by laser pulses. To conclude, we demonstrate direct nuclear delivery of genetic material into live mammalian cells using the photothermal nanoblade. The average frequency of successfully targeting the nucleus ranged from 39% to 52% in the three cell types tested. Higher transgene expression was observed for nuclear delivery, and >70% cell viability was maintained post DNA delivery.
Footnotes
Author Contributions
T.-H.W. and P.-Y.C. had the idea for direct nuclear delivery. T.-H.W. built the experimental setup. T.-H.W. and Y.-C.W. performed the experiments and analyzed the data. E.S. fabricated the nanoblade pipettes and maintained cell cultures. P.-Y.C. and M.A.T. advised on experiments, data analysis, and paper writing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by a University of California Discovery Biotechnology Award (178517); by National Institute of Health grants AI065359, GM114188, and EB014456; and by the Broad Stem Cell Research Center at the University of California, Los Angeles, and NanoCav, LLC. This work is also funded by the National Science Foundation (CBET-1404080).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
