Abstract
Well-designed microfluidic platforms can be excellent tools to eliminate bottleneck problems or issues that have arisen in biological fields by providing unprecedented high-resolution control of mechanical and chemical microenvironments for cell culture. Among such microtechnologies, the precise generation of biochemical concentration gradients has been highly regarded in the biorelated scientific fields; even today, the principles and mechanisms for gradient generation continue to be refined, and the number of applications for this technique is growing. Here, we review the current status of the concentration gradient generation technologies achieved in various microplatforms and how they have been and will be applied to biological issues, particularly those that have arisen from cancer research, stem cell research, and tissue engineering. We also provide information about the advances and future challenges in the technological aspects of microscale concentration gradient generation.
Introduction
Soon after the arrival of the new field of micro-electro-mechanical systems (MEMS) in the 1980s, a wide variety of MEMS devices were fabricated for chemical, biological, and biomedical applications, resulting in a new domain, bioMEMS, in the 1990s.1–3 The combination of MEMS technology and fluidic channels created microfluidic systems that are useful for cell biology, with features including the collection of quantitative data, high-throughput analysis, faster experiments with lower costs, the ability to mimic an in vivo environment, supplying chemical factors, and applying physiological mechanical stresses to the cells.4–8 Engineers and scientists have proven that the growth of cell cultures on such microplatforms is efficient for creating a cell-friendly microenvironment and analyzing detailed characteristics of cells and tissues.9,10
Among the various types of bioMEMS devices, there has been particular interest in microfluidic systems for generating concentration gradients, which have been used in the areas of chemotaxis, 11 toxicity, 12 drug screening, 13 and even cell sorting. 14 The concept of such gradient chips is applicable not only for reagent concentrations but also other properties that can stimulate cells such as electromagnetic fields, 14 temperature, 15 materials, 16 and gaseous molecules. 17
Earlier devices for gradient generation used pipette tips, 18 gel reservoirs, 19 and diffusion chambers, 20 which were unable to maintain spatiotemporal control of concentration gradient profiles at single-cell scale resolutions (10–100 µm) or maintain the gradient fields for a long duration (days) because the main mechanism of gradient generation relied only on diffusion, and thus continuous changes in gradient profiles were unavoidable. To overcome such problems, the microfluidic gradient generator was developed in 2000 ( Fig. 1 ) 21 ; a Christmas tree–like microchannel geometry was adopted to generate and control the concentration gradient field, and then the same microsystem was applied to cells. 22 Since then, various attempts have been made to create unique microplatforms using flow-based gradient generation for their own biomedical applications.
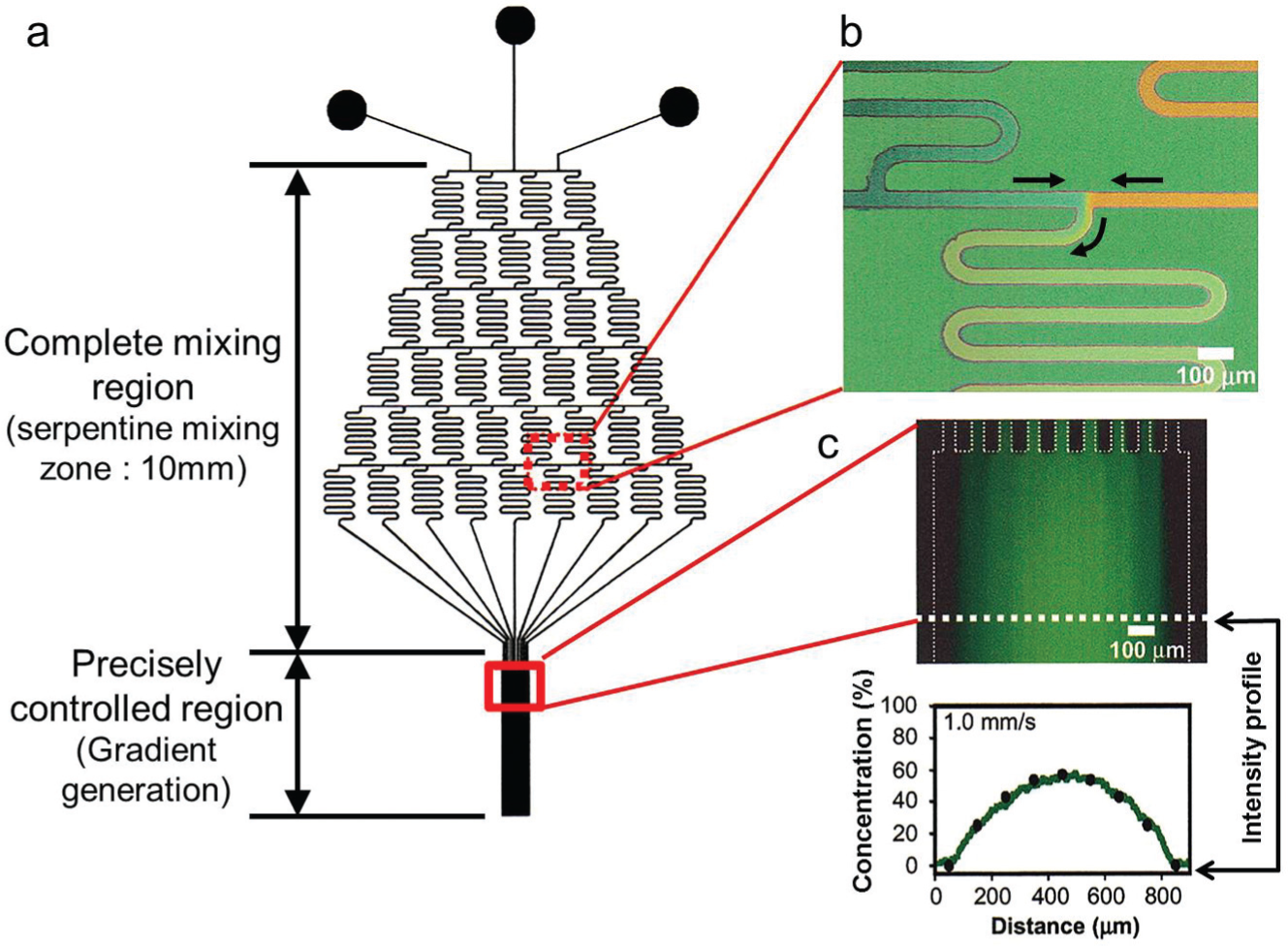
(
This review covers the current status of gradient generation microtechnology by categorizing the microsystems in terms of gradient types (chemical/physical properties) and biological applications (cell biology and drug screening). Through this review, we hope to demonstrate the possibilities for further innovations in gradient generation microsystems to meet further requirements proposed by biologists, tissue engineers, clinicians, and biomedical engineers.
Types of Gradient Generation
Among the various chemical and physical properties that can be used for gradient generation in microplatforms, the most frequently used and successful is the soluble biochemical gradient. 23 In this section, the microplatforms for biochemical gradient generation will be reviewed first, followed by a description of the gradient generation of temperature, substrate stiffness, and other properties that are also important physiological stimuli for cells. Table 1 includes a list of previously reported example microplatforms for various types of gradient generation and their biological applications.
Various Microplatforms for Biological Applications.
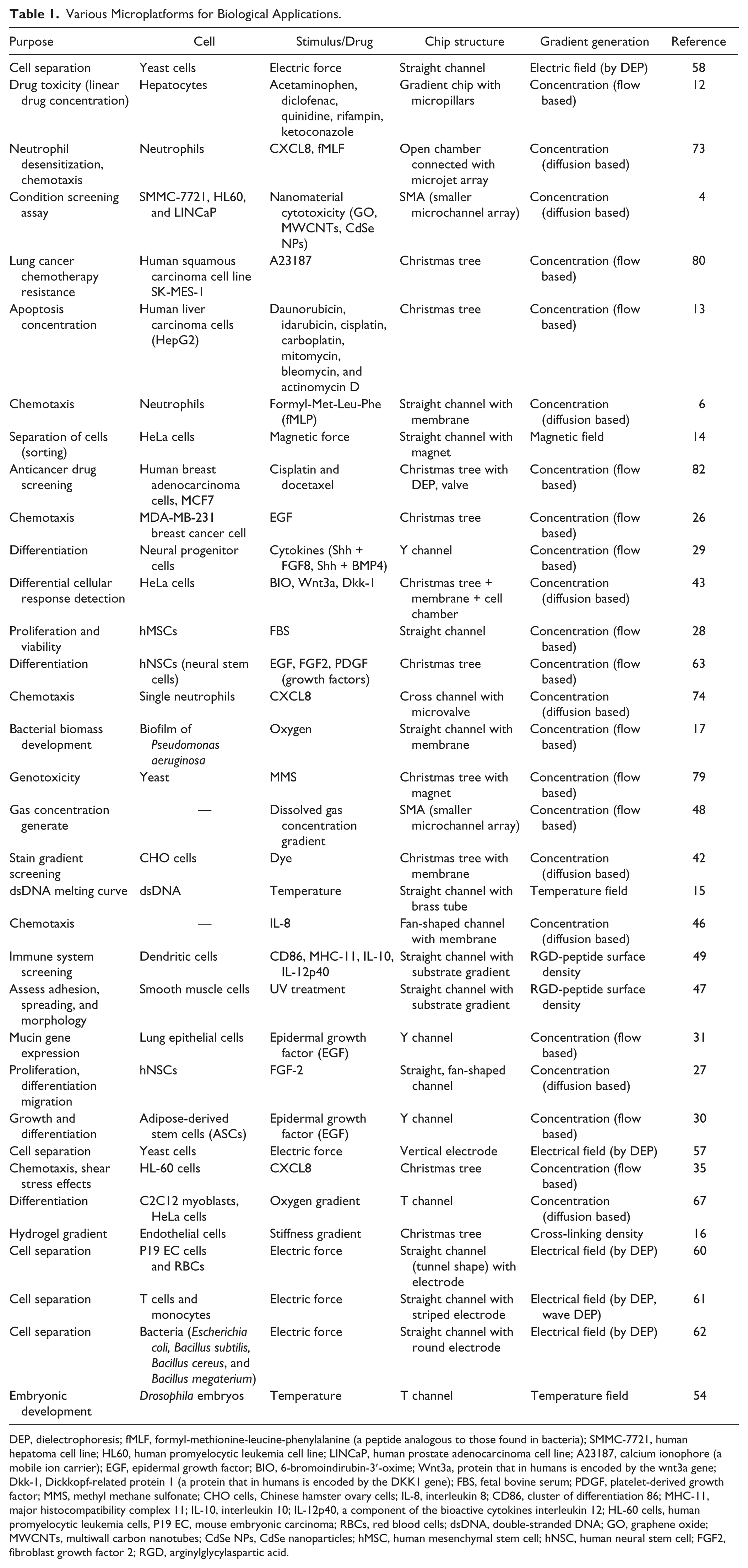
DEP, dielectrophoresis; fMLF, formyl-methionine-leucine-phenylalanine (a peptide analogous to those found in bacteria); SMMC-7721, human hepatoma cell line; HL60, human promyelocytic leukemia cell line; LINCaP, human prostate adenocarcinoma cell line; A23187, calcium ionophore (a mobile ion carrier); EGF, epidermal growth factor; BIO, 6-bromoindirubin-3′-oxime; Wnt3a, protein that in humans is encoded by the wnt3a gene; Dkk-1, Dickkopf-related protein 1 (a protein that in humans is encoded by the DKK1 gene); FBS, fetal bovine serum; PDGF, platelet-derived growth factor; MMS, methyl methane sulfonate; CHO cells, Chinese hamster ovary cells; IL-8, interleukin 8; CD86, cluster of differentiation 86; MHC-11, major histocompatibility complex 11; IL-10, interleukin 10; IL-12p40, a component of the bioactive cytokines interleukin 12; HL-60 cells, human promyelocytic leukemia cells, P19 EC, mouse embryonic carcinoma; RBCs, red blood cells; dsDNA, double-stranded DNA; GO, graphene oxide; MWCNTs, multiwall carbon nanotubes; CdSe NPs, CdSe nanoparticles; hMSC, human mesenchymal stem cell; hNSC, human neural stem cell; FGF2, fibroblast growth factor 2; RGD, arginylglycylaspartic acid.
Concentration Gradient of Soluble Biochemicals
Laminar flow is a flow of undisturbed paralleled layers where no cross-currents exist. Meanwhile, turbulent flow is characterized by the chaotic and random motion of fluid particles, which effectively enhances the convective mixing. These two different flow regimes are usually determined by the Reynolds number, a dimensionless number that is defined as the ratio of inertial forces to viscous forces:
Such benefits in the precise control of concentration gradients have been well used in cell biology studies.
25
It is physiologically normal for in vivo cells to be exposed to similar concentration gradients. Thus, culturing cells on the microchannel or microchamber where a concentration gradient is achieved and maintained allows the observation of more physiological cellular reactions in the in vitro microplatforms compared with the cells being bulk cultured in Petri dishes. Studies of cells in various gradient generation microplatforms were successful in a range of biological applications such as drug screening,
12
cancer metastasis,
26
and stem cell development.
27
In a simple single T or Y channel (two inlets, one outlet) with controlled flow rates, the proper concentration can be generated in the desired position28–30; the mixing starts at the convergence point of the two solutions from each inlet, and the concentration gradient profile spreads downstream of the flow. Diffusion between the paralleled flow streams is the main mechanism for gradient generation, so the flow velocity should be controlled at a reasonably slow rate.
31
However, the effect of the shear stress of fluid flow on the cells has been overlooked in most previously reported gradient generation microsystems that used laminar flow for gradient generation. Cells are exposed not only to the concentration gradient but also to fluidic forces (shear stress and pressure) simultaneously. For some cells, shear stress does not affect phenotype; however, other cells have mechanisms to sense and react to shear stress.
32
It was reported that a very low shear stress level (10−4–10−2 dyne/cm2) can regulate endothelial cell function,
33
a response that occurs independently from the biochemical concentration effect. The typical shear stress level of general microfluidic channels ranges in the order of magnitude 1 dyne/cm2.
34
Such shear stress level has an influence on neutrophil cell movements.
35
Therefore, it is important to check the shear stress level of the fluid flow while designing a microsystem. When a cell type is selected to be tested in the microsystem, the shear stress level should be controlled under the threshold value triggering specific cellular responses due to shear stress. The amount of shear stress that the cells experience can be estimated as the wall shear stress. This case assumes that the cells attach to the microchannel wall, spreading widely and thinly on the wall. In laminar flow, wall shear stress is calculated from Newton’s law, τ = −µ(d
Thus far, we have discussed laminar flow–based gradient generation in microfluidic systems. To generate and maintain concentration gradient profiles, utilization of fluid flows is essential. Although some devices created a concentration gradient solely depending on diffusion without fluid flow, 38 continuous changes in their gradient profiles are unavoidable. Therefore, shear stress cannot be separated from stable gradient generation ( Fig. 2a ). 39 Some gradient generation microsystems have been proposed to minimize the shear stress effect while maintaining stable gradient generation using extremely slow flows ( Fig. 2b ) 29 ; however, some types of cells can respond to such extremely slow flows.33,40 Depending on the research purposes and target cell types, shear stress effects need to be carefully controlled or removed completely. With this perspective, the gradient generation microsystems for cell culture and analysis can be categorized into two types: first, convection-based systems in which cells are exposed to both concentration gradients and shear stresses and, second, diffusion-based systems in which cells are located remotely from the main flow channel and are thus free from the shear effects.
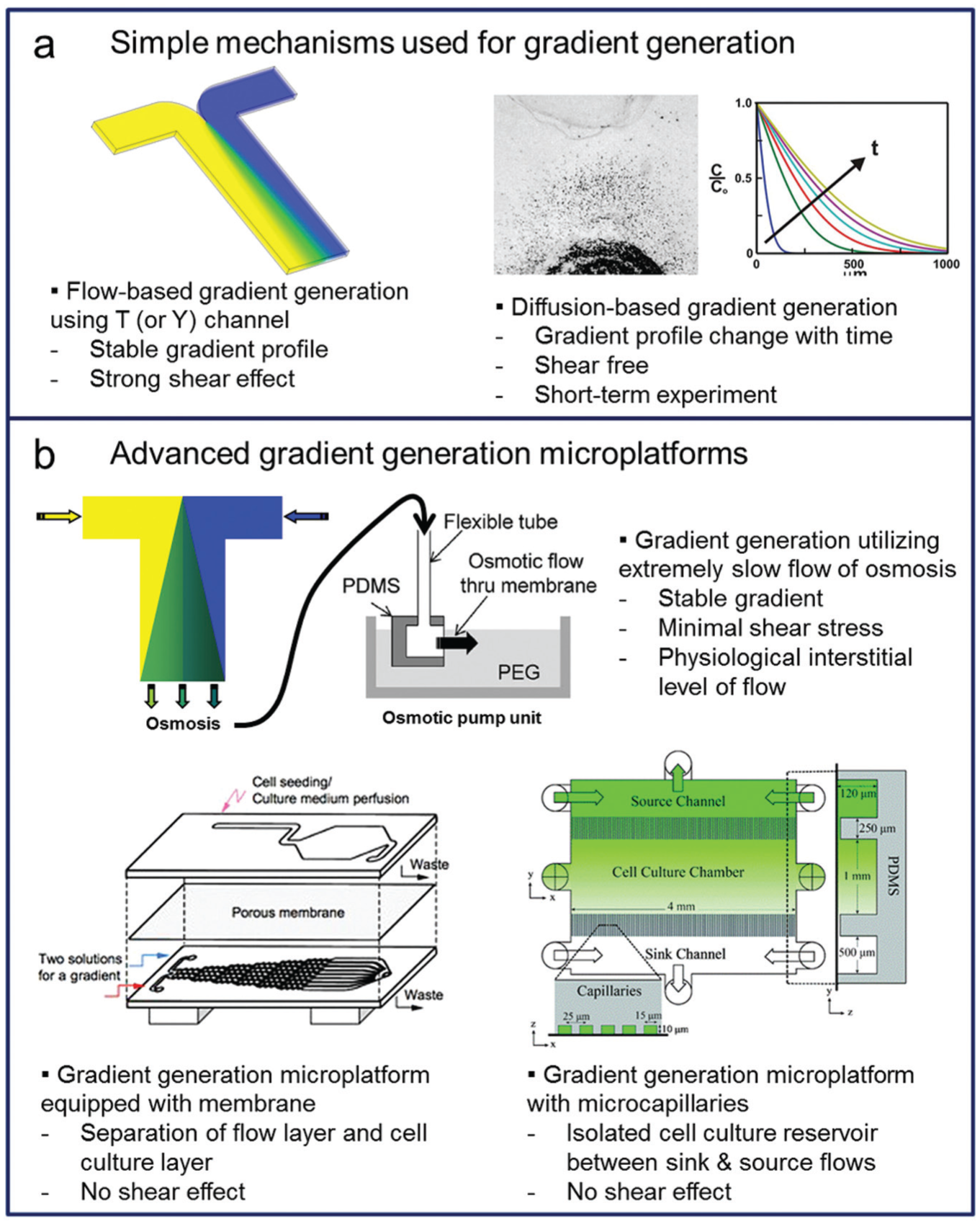
Microplatforms for the gradient generation of soluble molecules have evolved from (
A convection-based system is more simple in terms of the fabrication and operation schemes compared with a diffusion-based system; chemicals are carried by constant fluid flows and directly supplied to cells with shear effect from the flow. 8 Due to the simplicity, most gradient generation microsystems are convection-based systems ( Table 1 ). However, unwanted side effects can frequently be found due to the shear stress in certain cell types; therefore, the results should be carefully examined, and well-defined control tests are necessary to prevent unwanted effects from interfering with data analysis. 41 It is important to note that most cells are located in tissues, where shear stresses are far lower than those found in convection-based systems and cells rely mainly on diffusion for survival. 4
Diffusion-based systems, on the other hand, can be more complex in terms of fabrication, design, and operation, because they require additional components to block shear stress from the main flows. As we discussed above, main flows are required to create a stable concentration gradient. To culture cells in a shear-free environment, there should be supplementary components such as small channels,4,7 membranes,42,43 or gels 44 to block the main flows and their shear effects. One example of the diffusion-based systems are microcapillaries, which prevent shear effects of the main flows ( Fig. 2b ); unlike in conventional gradient generation by two-phase flows, the cell culture chamber is exposed only to diffusion and is free of shear stress. 45 Another example is a double-layered microchannel structure, where the flow generates a gradient in the bottom microchannel while cells are cultured on the top microchannel, which is separated from the bottom by the membrane that blocks the shear effect of the flow ( Fig. 2b ).42,43 A combination of membranes and ladder-type channels has also been reported; the concentration gradient was created in a fan-shaped cell chamber to study the chemotaxis of cells, while vertical membranes were installed to keep the cells in the chamber and also to prevent the flows from entering the chamber. 46
From the above-mentioned examples, it is clear that there is a need for additional components to prevent cells from being exposed to shear stresses in diffusion-based systems. Various kinds of microsystems can create concentration gradients of soluble biochemicals for cell biology studies. Various classifications of microplatforms for gradient generation are listed in Table 1 .
Gradients of Substrate Stiffness, Gas, and Temperature
Even though gradients of biochemical components such as growth factors, nutrients, and cell-secreted cytokines play important roles in the biological applications of gradient generation microsystems, many other kinds of gradient generation are necessary, each with its own purpose. It is frequently necessary to create gradients of properties other than molecular concentrations in liquids; examples include substrate gradients, 47 liquid-gas mixture gradients, 48 and temperature gradients ( Fig. 3a ). 15
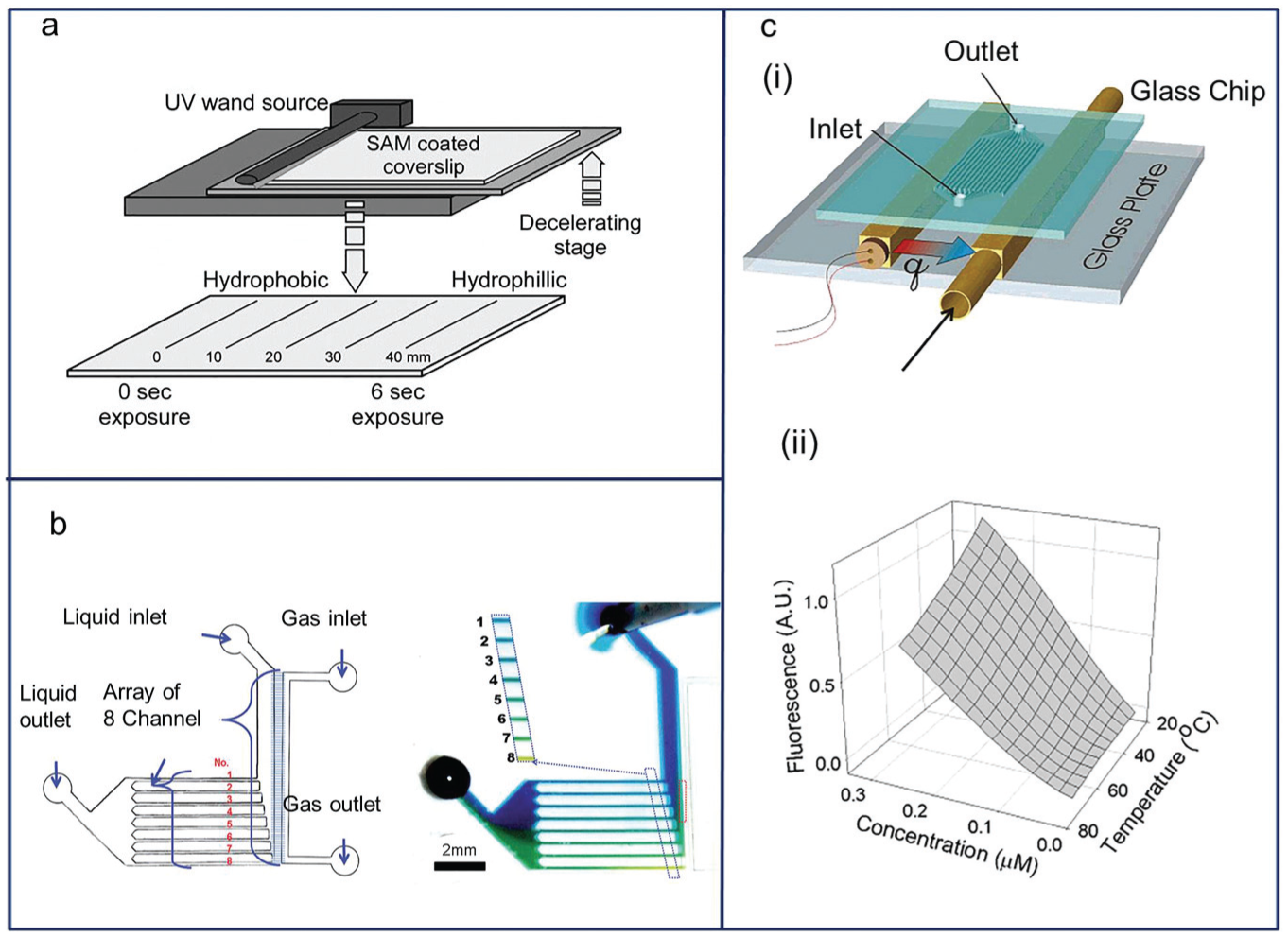
(
To elicit a desired cell response, controllable substrate treatment was studied to observe biofunctionalization by cell-material interactions. The rise in UV exposure time (0–6 s) led to increasing amounts of ozone-derived oxidation on the substrate, gradually converting the hydrophobic (CH3-terminated) layer to hydrophilic (OH- and COOH-terminated) species, which could be used as initiation sites for further functionalization ( Fig. 3a ). 47 Using the same method, immunogenic or tolerogenic responses of dendritic cells were measured by danger signals on a simple straight arginylglycylaspartic acid (RGD)–peptide gradient channel. 49
The stiffness of materials is another target for gradient generation. Hydrogel-based microplatforms have been reported that incorporated a controlled photopolymerization process for the generation of a stiffness gradient.16,50 A prepolymer gradient was subsequently UV-polymerized into a water-swollen hydrogel in a Christmas tree–like microplatform similar to that of Figure 1a to produce both a cross-linking density (stiffness) gradient and an adhesive ligand gradient, which were tested with endothelial cells. 16 Another example of stiffness gradient generation was accomplished by changing the UV exposure time of a hydrogel. A time-dependent photopolymerization process can establish a stiffness gradient in a hydrogel functionalized with fibronectin. 50 This study revealed that hydrogel stiffness and the corresponding changes in Young’s modulus can control cell spreading.
Gas concentration gradient chips have also been reported. Since gas plays an important role in cell biology, a microplatform was proposed to create a gas concentration gradient using the microchannel array connected to a gas reservoir ( Fig. 3b ). 48 In this system, the gas-liquid interface required for gas exchange was maintained by the large capillary force of the fluid in microchannels; with proper design and flow velocity control, linear, parabolic, and even logarithmic gas concentration profiles were achieved. Among human organs, the liver is the most multifunctional, as it is involved in protein/macromolecule synthesis, urea synthesis, protein/blood storage, and immune/hormonal system modulation, among other functions. Seventy to 80% of the liver consists of cells called hepatocytes, which have a unique feature of high oxygen consumption. To study drug metabolism by hepatocytes as well as a wide variety of other physiologically important processes, the development of an in vitro system in which a microscale oxygen gradient can be achieved is necessary. Control of oxygen level in microfluidic bioreactors has been previously reported; it uses the cellular uptake of oxygen to generate the oxygen gradient in a microchannel.51,52 These are physiologically relevant systems where the upstream cells consume oxygen to generate a gradual decrease of oxygen concentration downstream.
Some reports have shown the usefulness of temperature gradients on microplatforms. To construct the melting curve for double-stranded DNA (dsDNA), a linear temperature gradient chip was designed (ranging from 16–101 °C over a 20-mm distance;
Fig. 3c
).
15
Similarly, by setting different temperature tubes on a gradient chip, we can obtain two distributions simultaneously for temperatures and drug concentrations.
53
A temperature gradient allowed the spatial and temporal regulation of embryonic development. At the contact point of two different temperature flows from the T channel,
Electromagnetic Gradient
Dielectrophoresis (DEP) is a phenomenon in which force is exerted on a dielectric particle in a nonuniform electric field. Since most cells are polarizable particles,
55
DEP is used for the separation of cells or cell sorting in biological applications.14,56 The particles (or cells) can move toward regions with higher or lower electric field strength depending on the permittivity of particles relative to the medium.57,58 Methods for generating an electric field gradient can be categorized as follows: microfabrication techniques to create electrodes of different shapes, inducing a change in the electric field magnitude across the particles; a traveling wave DEP that changes the phase of the applied electric field; or an isolating DEP that uses a nonhomogeneous dielectric medium between the electrodes.
59
First, different electrode shapes induce a change in electric field magnitude (
Fig. 4a
)
60
; a 3D-asymmetric electrode increased the discriminated electric force to endothelial cells and red blood cells, resulting in cell separation. The second method involves changing the phase of the applied electric field (
Fig. 4b
)
61
; DEP force and traveling wave DEP force were applied to cells (T cells and monocytes), and the amount of force depended on the frequency and cell volume. The last method of electric field gradient is generated by a nonhomogeneous dielectric medium (
Fig. 4c
)
62
; using DC nonuniform electric fields, four types of bacteria (
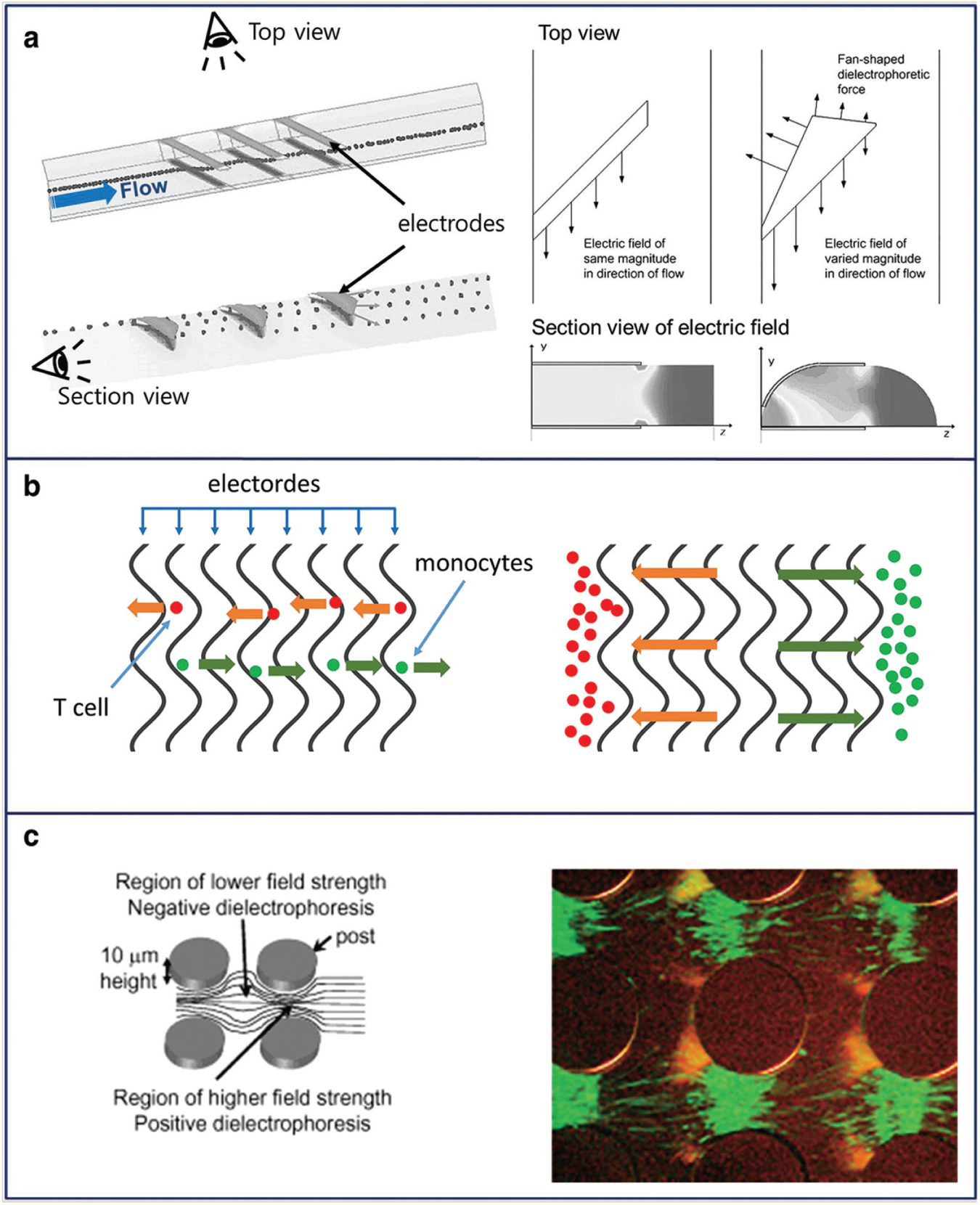
(
Biological Applications
As the above-mentioned novel microplatforms have been developed, their successful applications in characterizing cells and pathologic/physiologic analysis have been published. Table 1 shows the representative microplatforms regarded as successful in the field of cell biology. In this section, we present detailed introductions on such good examples of microplatforms used in cell biology.
Cellular Differentiation under Chemical Gradients
Stem cells can differentiate into more specialized cells (pluripotency) and can also divide to produce more stem cells (self-renewal). Recent trends in microtechnology have led to a better understanding of the importance of controlling multiple stimulations in evaluating higher-order cellular responses in stem cells. Differentiation occurs numerous times during the development of complex systems and is influenced by various signaling molecules. Spatial gradients of diffusible signaling molecules play important roles in controlling the differentiation of cells, thus determining their developmental fate. Therefore, it is important to find ways to observe and understand the effects of the concentration gradient of signaling molecules controlling and optimizing the differentiation cell types. 29 Microtechnology-based platforms can meet such needs because chemical gradients can be easily achieved, controlled, and maintained in microfluidic systems. Below are several example studies of microfluidic gradient generation and its effects on stem cells.
Numerous studies using microsystems for neural stem cell culture have been conducted to find cell-based therapies for human neural disorders.63–65 The relationships of growth factors with cell proliferation, differentiation, and even migration have been verified with many types of stem cells.27,30 For example, some growth factors could induce opposing effects on the proliferation and astrocyte differentiation of human neural stem cells (hNSCs). 63 By generating suitable concentration gradients, these differentiation phenomena can be measured easily. Human embryonic stem cells were differentiated into neural progenitors with gradients of the cytokines and growth factors, including sonic hedgehog (Shh), bone morphogenetic protein (BMP4), and fibroblast growth factor 8 (FGF8), to evaluate the oppositional effects of an agonist (Shh) and antagonist (BMP4) ( Fig. 5a ). 29 Fibroblast growth factor 2 (FGF2) was also found to trigger hNSC differentiation ( Fig. 5b ). 27 In addition, microfluidic platforms were found useful for investigations of Alzheimer disease. Amyloid-β oligomeric assemblies were re-created, and their neurotoxicity level was measured in a microfluidic channel in which primary neural progenitor cells were cultured and a biochemical gradient profile was developed under an interstitial level of slow flow. 66
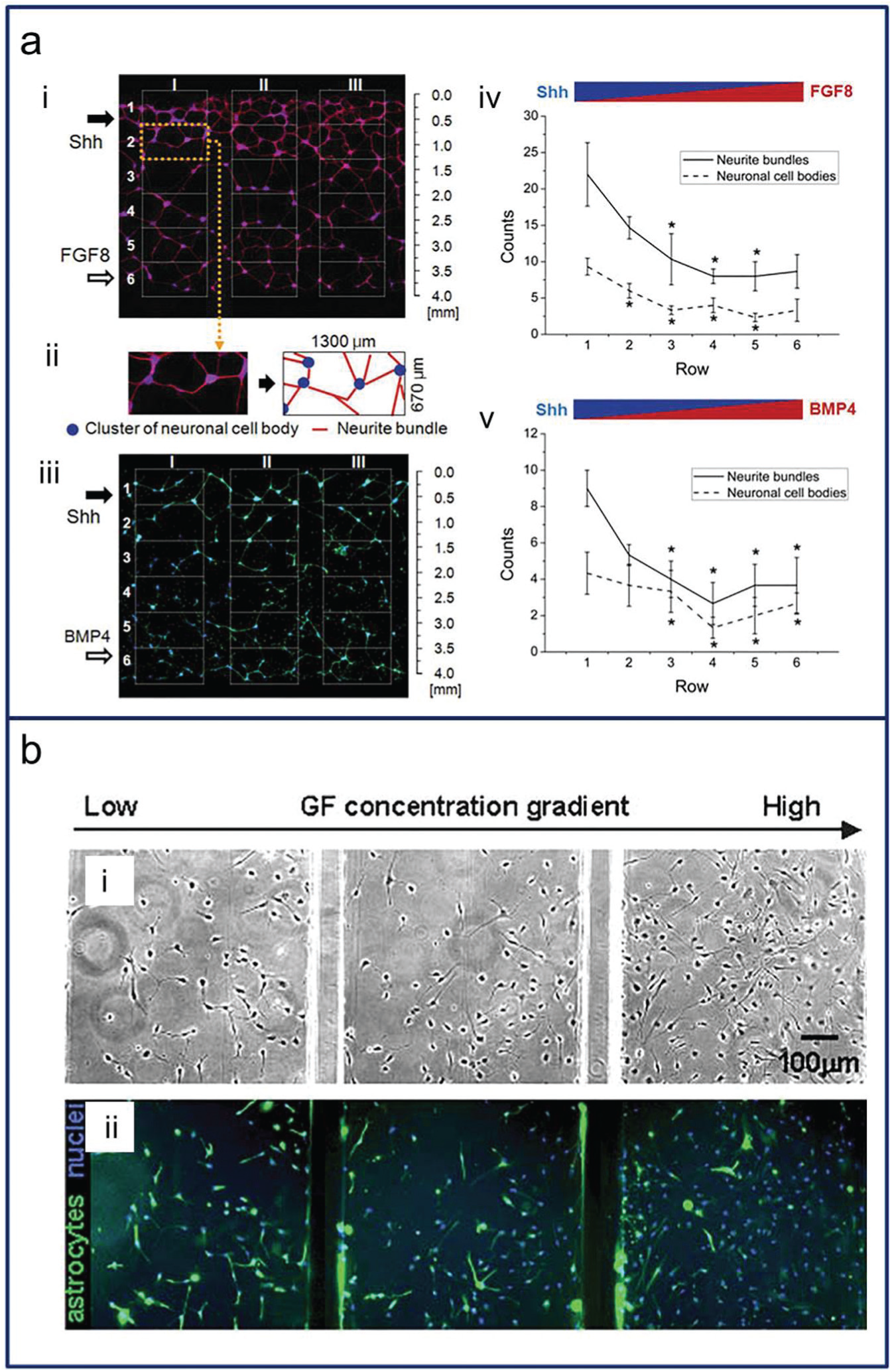
(
Other types of stem cells have also been studied on microplatforms. The oxygen gradient effect on C2C12 myoblast differentiation has been observed 67 ; the cell chamber had a closed end and open entrance contacting oxygen-rich media to generate an oxygen gradient in the chamber. Near the entrance, cells were alive, attached, and differentiated with differentiation medium because they were exposed to the oxygen-rich media. Another study used agarose gel to allow the diffusion of oxygen and nutrients while preventing convective mixing. 68 This system provided useful tools for investigating the effects of the metabolic microenvironment on cells in culture.
The stiffness gradient of material mimicked an in vivo environment that regulates mesenchymal stem cell (MSC) differentiation by tissue stiffness. 69 An elasticity gradient established by creating a hydrogel crosslinking gradient in 10% acrylamide/0.3% bis-acrylamide was obtained via the selective activation of a photoinitiator. Immuno-fluorescent staining for MyoD, a myogenic regulatory factor, and phalloidin was observed as a function of culture time and hydrogel stiffness. A substrate stiffness gradient is advantageous since it permits the tuning of the material to elicit the desired cell behavior. 70 In this study, a stiffness gradient produced by a temperature gradient during curing was applied to observe osteogenic differentiation in rat MSCs.
Chemotaxis
Chemotaxis is the directional movement of cells, bacteria, and other organisms in response to certain chemicals in their environment. 46 For chemotactic cells, small concentration differences as low as 2% can influence the movement of the cells. 71 As an example, bacteria swim either to find food by directing themselves to the highest concentration of food molecules or to evade danger by moving away from high concentrations of repellants. 72 The exploration of this phenomenon, chemotaxis, was greatly influenced by Stephen Boyden with his Boyden chamber/transwell assay in 1962. 38 Two chambers that contain different factors are divided by a transwell membrane, and cells move through the membrane pores and are counted to quantify the degree of chemotaxis. However, the inability to directly visualize cells and control the biomolecular gradient makes that method unsuitable for correlating specific cell responses. 39 The next-generation chambers such as the Zigmond chamber (1977) and Dunn chamber (1991) were developed to better understand chemotaxis, but those also have time scale limitations of up to 2 h. Through recent developments, a gradient chip may be used to analyze cell movement with a stable long-term system. 26 In a gradient chip, migrating cells do not take a linear path along the gradient but rather move in an ambulatory fashion with net motion in the direction of the increasing gradient, as seen in Figure 6 .73,74 Hydrogel-based gradient platforms have been used for 3D chemotaxis studies.75,76 The platforms had one cell-injected channel and two side channels containing different media to establish a concentration gradient. These three channels were connected with an agarose gel, which allowed nutrients and chemicals to diffuse through and provided a barrier to convective flows. With this system, the chemotactic responses of murine dendritic cells to a gradient of CCL19 75 or CCL21 76 were quantified. In addition to chemotaxis, cells can respond to direct current electric fields (dcEF) by migrating to the electrodes of the field (electrotaxis). 77 Coexisting chemical gradients and dcEF were applied to evaluate T-cell chemotaxis and electrotaxis between a chemokine CCL19 gradient and applied dcEF.
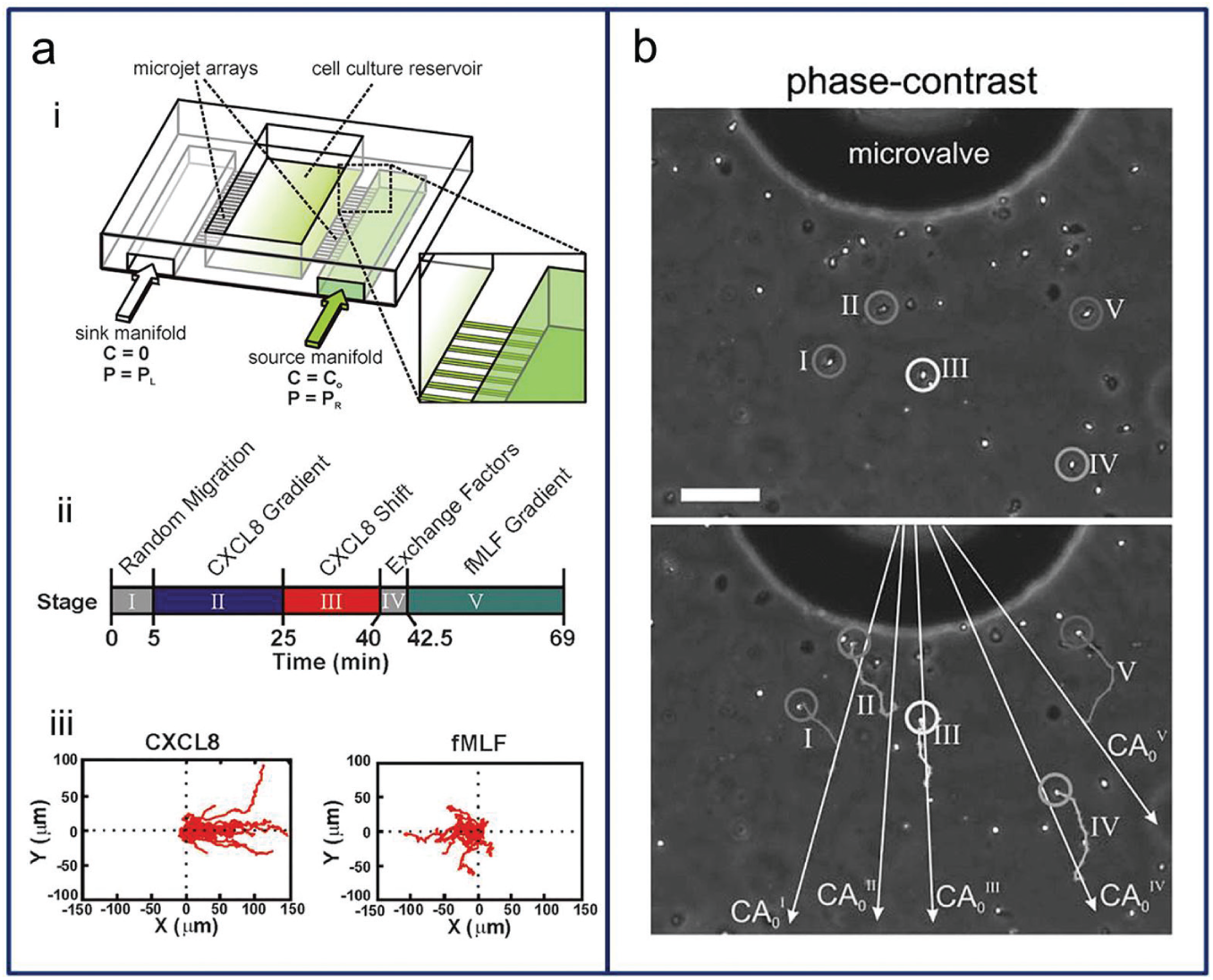
(
Drug Screening and Toxicity
Drug screening requires quality environmental control, different cell culture modes, and availability for use in high-throughput analysis ( Fig. 7a ) 4 ; gradient chip technology can provide a suitable platform for in vitro drug testing satisfying these requirements. 78 Drug screening using a gradient chip includes the analysis of cell responses, viability, toxicity, 79 and apoptosis. 80 For example, a hepatocyte chip can provide predictive information regarding metabolizing enzyme activities, hepatic transporters, and other differentiated functions ( Fig. 7 ).12,81 To maintain the hepatocytes’ differentiated functions, one study proposed a 3D microfluidic environment constructed with a micropillar, and five drug responses were tested (acetaminophen, diclofenac, quinidine, rifampin, and ketoconazole; Fig. 7b ). 12 Another example is an anticancer drug test that used a dielectrophoresis-based cellular microarray to reduce cell waste. Two major chemotherapeutic drugs for breast cancer, cisplatin, and docetaxel, were used for human breast adenocarcinoma cells and MCF7 (breast cancer cell line) viability drug testing. 82 An analysis of apoptosis in human cervical cancer (HeLa) cells was conducted using cisplatin, 5-fluorouracil, and cyclophosphamide as drugs 83 ; by repeating the splitting and mixing of the source solutions in a radial channel network, 65 combinatorial concentrations of drugs were evaluated.
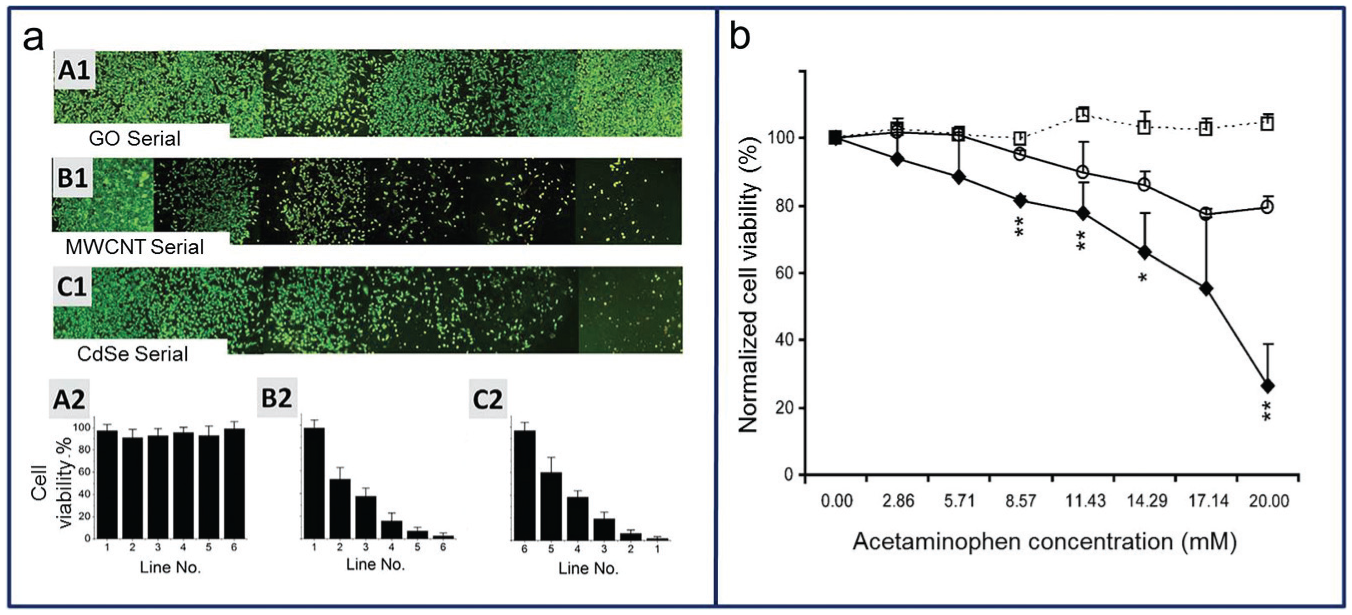
(
The 3D culture (spheroid culture) can model in vivo tissue more accurately than 2D culture, permitting interaction with surrounding cells. Therefore, the gradient drug screening of cells cultured in 3D has been highly regarded. To fabricate an in vivo screening system, this study provided a microplatform consisting of cell culture chambers and hemispherical microwells connected with a concentration gradient generator. 84 HT-29 and Hep-G2 cells were cultured as tumor spheroids, and 5-fluorouracil was used as the model anticancer drug to monitor metabolic activity.
In conclusion, microplatforms that generate gradients of various stimuli such as chemical, material, and mechanical cues have been used widely in cell biology. Such gradient generation microsystems successfully mimicked in vivo cellular environments to characterize cells. There are challenges associated with such microsystems: first, to isolate the effects from shear flows, since fluid flows are essential to create stable gradients for long-term cell culture but often have unwanted side effects; second, to control shear stress levels, because cells normally experience very low levels of shear stress created by interstitial flow; and third, to merge multiple gradient cues since multiple stimuli (including the shear stress of the flow) are the physiological norm in vivo. It is well accepted that the microplatforms generating gradient profiles of various stimuli will be extremely useful for examining higher levels of cellular mechanics. It is important to investigate and limit unwanted side effects created while applying the designed gradient cues, and thus, the results of the various gradient generation microplatforms should be carefully interpreted to avoid misleading conclusions.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Chung-Ang University research scholarship grants (2013) and by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012R1A1A1015181).
