Abstract
The significance of microfluidics-based and microelectromechanical systems–based biosensors has been widely acknowledged, and many reviews have explored their potential applications in clinical diagnostics, personalized medicine, global health, drug discovery, food safety, and forensics. Because health care costs are increasing, there is an increasing need to remotely monitor the health condition of patients by point-of-care-testing. The demand for biosensors for detection of biological warfare agents has increased, and research is focused on ways of producing small portable devices that would allow fast, accurate, and on-site detection. In the past decade, the demand for rapid and accurate on-site detection of plant disease diagnosis has increased due to emerging pathogens with resistance to pesticides, increased human mobility, and regulations limiting the application of toxic chemicals to prevent spread of diseases. The portability of biosensors for on-site diagnosis is limited due to various issues, including sample preparation techniques, fluid-handling techniques, the limited lifetime of biological reagents, device packaging, integrating electronics for data collection/analysis, and the requirement of external accessories and power. Many microfluidic, electronic, and biological design strategies, such as handling liquids in biosensors without pumps/valves, the application of droplet-based microfluidics, paper-based microfluidic devices, and wireless networking capabilities for data transmission, are being explored.
Introduction
Laboratory testing has been undergoing a major paradigm shift in recent years to adapt to the new requirements that are driven mainly by ever-changing health care scenarios, technology, regulations, and various market forces. According to a report published by the Milken Institute,
1
data from US Centers for Disease Control and Prevention (CDC) indicate that chronic illnesses affect one of every two adults in the United States, and are responsible for 75% of health care costs. In 2008, health care costs in the United States were 16.8% of GDP, and by 2022 they are projected to be approximately 20%.
1
Even though the urbanization trend around the globe has been increasing, 55% of inhabitants in the developing world still live in rural areas. There is a demand for affordable technologies that can enable local communities in developing regions to improve health care, environmental safety, animal health, and food safety to accomplish the agendas of the Millennium Development Goals
2
beyond 2015. According to the World Health Organization (WHO), after cardiovascular disease, infectious diseases are the second leading cause of mortality around the world.
3
This problem is greater in resource-limited settings in developing countries with poor hygiene. The burden of disease is typically measured in terms of disability-adjusted life years (DALYs),
4
a unit that accounts for the years of life lost due to both mortality and disability caused by the disease. More than 95% of deaths due to major infectious diseases that include acute respiratory infections (ARIs), malaria, HIV, and tuberculosis (TB) occur in developing countries, with by far the largest burden on Africa.
5
A large reduction of DALYs resulting from the major causes of disease in low-resource settings can be achieved by developing improved and appropriate diagnostics. In developed countries, in spite of many advances in health care, there are many challenges related to pathogen outbreaks, sexually transmitted diseases, and food safety that need to be addressed. In the past decade, the demand for rapid and accurate on-site detection of plant disease diagnosis has increased due to emerging pathogens with resistance to pesticides, increased human mobility, and regulations limiting the application of toxic chemicals to prevent spread of diseases. Many portable devices have been developed for plant pathogen detection, but current technologies are limited to detecting known pathogens with limited detection accuracy.
6
A biosensor for plant pathogen detection should be portable, simple enough to be used on-site by growers and producers, and capable of detecting multiple plant pathogens and their associated variants. Due to the ongoing war on terrorism and rising security concerns, the need for rapid-detection biosensors against bio-warfare agents for military and civil defense applications has increased. Diagnosis is the first step to treat a condition or take preventive steps. However, one of the major hurdles in monitoring and controlling diseases and contaminations is the availability of easy-to-use, low-cost, and robust diagnostic tests. Diagnostics for limited-resource settings does not get the required attention from large companies due to its low profit margins, whereas the limited research contribution from smaller companies and academia is not sufficient to make a major impact in developing diagnostic products. In many low-resource settings without diagnostic tests, disease is often treated based on clinical symptoms and local prevalence of disease. Such syndromic disease management can be cost-effective and has been recommended by the WHO for certain diseases, such as malaria and sexually transmitted infections. However, this approach sometimes unnecessarily treats patients who do not require treatment, and thus may accelerate drug resistance. The WHO Sexually Transmitted Diseases Diagnostics Initiative has developed a set of generic guidelines for the development of diagnostic tests appropriate for the developing world that can be summarized under the acronym ASSURED:
7
This review article will focus on the integration of different techniques that led to the development of portable biosensors. This review will also outline the significance, pros and cons, and challenges in the development of portable biosensors for various applications, with selected examples of point-of-care testing (POCT) for patients, detection of biological warfare agents (BWAs), and on-site detection and diagnosis of plant disease. An overview of various microfluidics/electronics/biological design strategies for biomarkers, measurement techniques, sample preparation, fluid-handling techniques, data collection and analysis challenges, and quality specifications for the development of portable biosensors will be provided.
Biosensor
A biosensor is a sensing device comprising a bio-recognition element and a transducer. A bio-recognition element specifically identifies and interacts with an analyte, and the changes in its physicochemical properties (optical, thermal, electrical, and thermodynamic properties) are usually converted into an electrical signal by a transducer. Figure 1 illustrates the schematic of a biosensor. The bio-recognition element is a sensing material and may include enzyme, antibodies, microorganisms, tissues, organelles, DNA, and RNA. The biosensors can be classified based on the type of bio-recognition element or the transducing method used. Based on the bio-recognition element, the biosensors can be classified as enzyme sensors, immunosensors, nucleic acid probe sensors, or cell-, tissue-, or organelle-based sensors. Based on the transducing method, biosensors can be classified as piezoelectric, optical, or electrochemical biosensors.
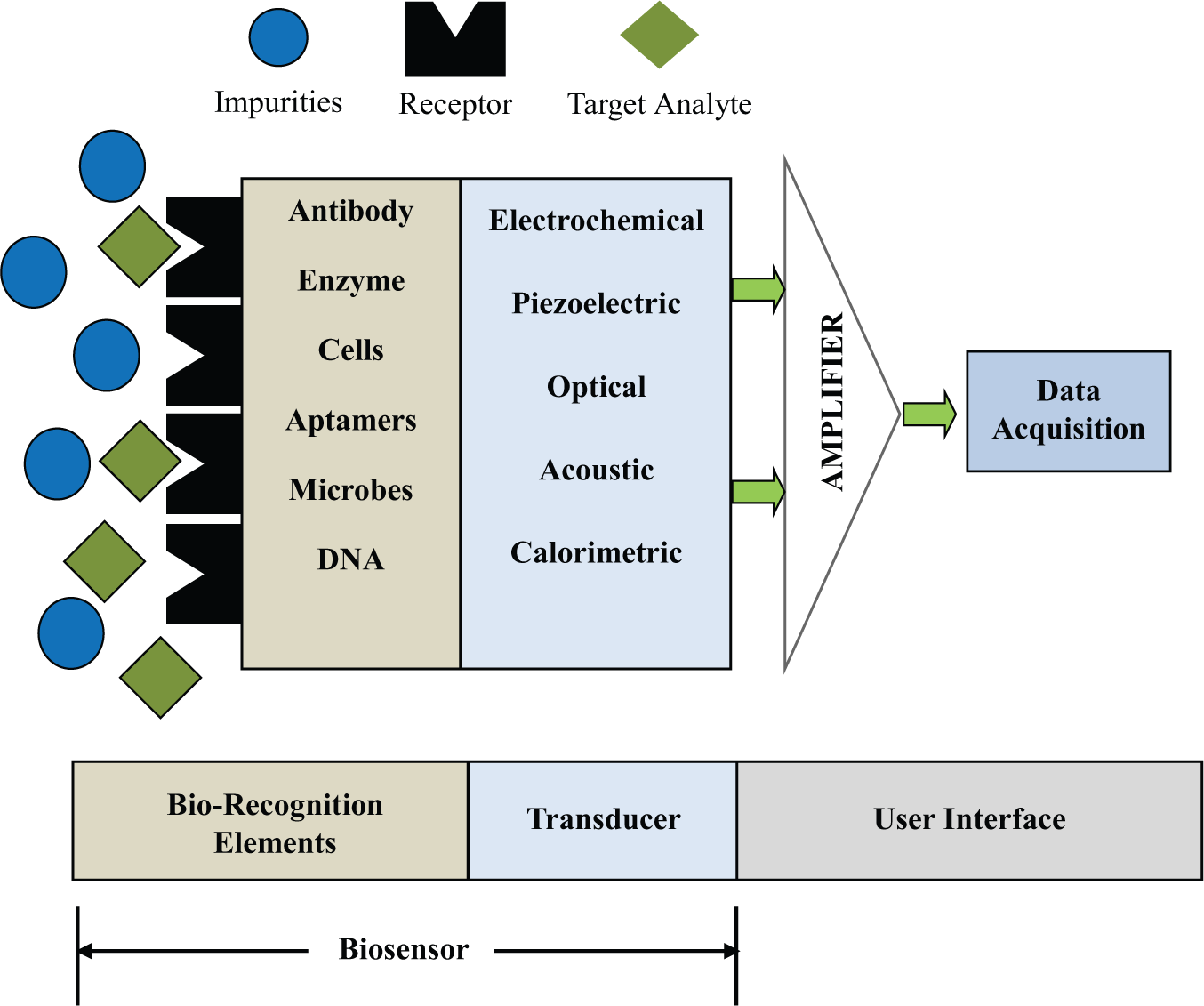
Schematic of a biosensor.
Bio-recognition Element
Antibody-based sensors, also known as immunosensors, are based on the specificity of an antibody–antigen reaction to produce a change in the transducer signal. Both polyclonal and monoclonal antibodies can be used, and the suitability depends mainly on their specificity. Antibodies can be directly immobilized on the surface of the transducer by covalent bonding through amino, carboxyl, or aldehyde groups. Antibodies can also be attached to the surface of magnetic beads to perform immunomagnetic separation and detection. Antibody-based biosensors with various transducing methods have been reported.8–11 Enzyme-based biosensors12–14 are based on using enzymes that are specific to the biomolecules under detection to catalyze the generation of a product that can be quantified by a transducer. Most of the enzymes used in biosensors are oxidases that react with dissolved oxygen to produce hydrogen peroxide. Nucleic acid–based biosensors 15 integrate a natural and biomimetic form of oligo- and polynucleotides, and rely on the very strong base pair affinity between complementary sections of nucleotide strands for their high sensitivity and selectivity. In recent years, the use of synthetic oligodeoxyribonucleotides (ODNs) is gaining popularity. End labels, such as thiols, disulfides, amines, or biotin, are incorporated to immobilize ODNs 16 to the surface of a transducer element. Electrochemical DNA biosensors, 17 in which the base-pair recognition event is converted into a measurable electrical signal, are being used for the diagnosis of genetic diseases, pathogens, biomolecules of clinical interest, and so on. Cell-based biosensors (CBBs) are based on viable whole cells that are integrated onto the biosensor platform and function as the sensing unit to indicate the presence of a particular analyte or a group of analytes. CBBs can use bacteria, yeast, or higher eukaryotic cells, including vertebrate or mammalian cells. 18 CBBs are designed to record the deviation from normal cellular activities of the “sensing cells” due to their exposure to an analyte. Unlike the nucleic acid– or antibody-based biosensors, CBBs respond to the toxins in a physiologically relevant manner to provide additional information on the mechanism of action and toxicological outcome of exposures.
Transducer Element
Optical biosensors include surface plasma resonance (SPR) biosensors, adsorption and reflection biosensors, luminescence biosensors, fluorescence biosensors, and optical fiber biosensors. SPR and fluorescence biosensors are the most popular optical biosensors. Fluorescence biosensors use fluorescent labels for signal generation, and the intensity of the fluorescent signal is used to determine the amount of the antigen being detected. 19 SPR is a direct optical-sensing technique that measures the refractive index change due to bio-specific interactions occurring in the vicinity of a thin metal film surface. 20 SPR biosensors for detecting influenza virus 21 and plant virus 22 have been reported. Piezoelectric biosensors consist of mass-sensitive piezoelectric crystals with excitation electrodes. The piezoelectric transducers allow a binding event to be converted into a measurable signal, such as resonant frequency changes. 23 The most popular transducer design is based on the quartz crystal microbalance (QCM), adapted to the liquid medium, which gives a direct response signal to characterize the binding event between a sensitive layer grafted onto the surface transducer and an analyte to be detected. 23 QCM has been used for the detection of DNA, protein–ligand interactions, virus capsids, bacteria, and mammalian cells. 24 A microcantilever is another type of mass-sensitive biosensor. The principle of this detection is based on the change in the mechanical response of a cantilever due to molecular adsorption on the functionalized cantilever surface. The sensitivity of a microcantilever is high enough to detect a single virus particle. 25 Electrochemical biosensors can be classified into amperometric, potentiometric, and impedance biosensors based on the electrical parameters they measure. Potentiometric biosensors use ion-selective electrodes (ISEs) and ion-sensitive field-effect transistors (ISFETs) to measure the change in electric potential due to the accumulation of ions resulting from an enzyme reaction. 26 Amperometric biosensors measure the change in electrical current (typically, in the nanoampere to microampere range) due to the oxidation/reduction process of an electroactive species. The working electrodes are usually a noble metal or a screen-printed layer covered by a bio-recognition element. 27 The amperometric biosensors are often used for analytes such as glucose, lactate, 28 and sialic acid. 29 In addition, the detection of biological agents such as Bacillus cereus and Mycobacterium smegmatis, 30 the serological diagnosis of Francisella tularensis, 31 pesticides, and nerve agents 32 has been reported. Potentiometric biosensors measure the potential difference generated across an ion-selective membrane separating two solutions at virtually zero current flow. A potentiometric sensor array 33 for investigating the cytotoxicity of hydroquinone to cultured mammalian V79 cells has been reported. Impedance biosensors are based on the combined measurement of the resistive and capacitive properties of the targets in response to a small-amplitude sinusoidal excitation signal.34–35 Impedance detection involves measuring the change in impedance caused by the binding of analytes to receptors (antibodies, DNA, proteins, etc.) immobilized on the surface of electrodes, 36 change in the conductivity of the medium caused by the growth of bacteria, 37 bacterial cells captured on an electrode surface using dielectrophoresis, 38 and change in the ionic concentration of a medium caused by activity of enzyme used as labels. 39 Calorimetric biosensors are based on measuring the heat of a biochemical reaction at the sensing element. These biosensors consist of temperature sensors with immobilized biomolecules. Once the analyte comes into contact with the biomolecules, the reaction temperature is measured. The total heat produced or absorbed is proportional to the molar enthalpy and the total number of molecules in the reaction. The change in temperature is proportional to the analyte concentration. Calorimetric biosensors have been used for food, cosmetics, pharmaceutical, and other analyses. For example, a microcalorimetric sensor has been used for L-malic acid determination 40 in various types of food.
Point-of-Care Testing
POCT can be defined 41 as “provision of a test when the result will be used to make a decision and to take appropriate action, which will lead to an improved health outcome.” Another definition 42 of POCT is “patient specimens assayed at or near the patient with the assumption that test results will be available instantly or in a very short timeframe to assist caregivers with immediate diagnosis and/or clinical intervention.” POCT platforms include lab-on-a-chip platforms, micro total analysis systems, and fluidic cartridges/lateral flow (LF) strips. These platforms may have an integrated or dedicated readout system. The footprint of POCT platforms can range from a small chip to a tabletop system that might be placed in doctors’ offices, hospitals, or ambulances and ideally is capable of being operated by minimally trained users. POCT systems should essentially require minimal operator intervention, have required reagents, and preferably have automated processing steps that are integrated within the system.
Even though there are many other definitions of POCT, the most critical elements 43 of POCT are rapid turnaround, communication of results to guide clinical decisions, completion of testing, and follow-up action in the same clinical encounter. A rapid turnaround of results is important for the test results to influence clinical decisions such as triage, referral, and decision to discharge the patient. Rapid can refer to seconds, minutes, or a few hours while the patient is still present at the site. Figure 2 illustrates the rapid turnaround of results with POCT when compared to a conventional testing procedure. A rapid test result that does not also have a mechanism for rapid reporting of the result to the care providers and its translation to a treatment plan is highly unlikely to have any significant impact on public health outcomes. 44 For example, in some countries with limited resources, a rapid diagnostic test (RDT) may be performed, but the test results may not reach the care provider until after a few days. In conventional laboratory testing, which typically takes at least a day, the patient has to go to a remote testing facility and may not return to continue the treatment process. POCT can overcome this problem by making it convenient for both patients and care providers by completing the diagnostic process “in the same clinical encounter” and allowing a treatment plan to be implemented on the same day. POCT can occur at various settings. POCT can provide considerable savings in health care costs by reducing the number of patients visiting health centers. POCT can improve the quality of life for patients by reducing their number of visits to health care facilities. An early diagnosis based on POCT can also enable the doctors to start the treatment process earlier and thereby increase chances of preventing or curing the disease. POCT can also reduce human errors leading to mix-ups of patient samples sent to off-site laboratories. In future, a lowering of medical costs due to POCT might benefit both insurance companies and consumers.
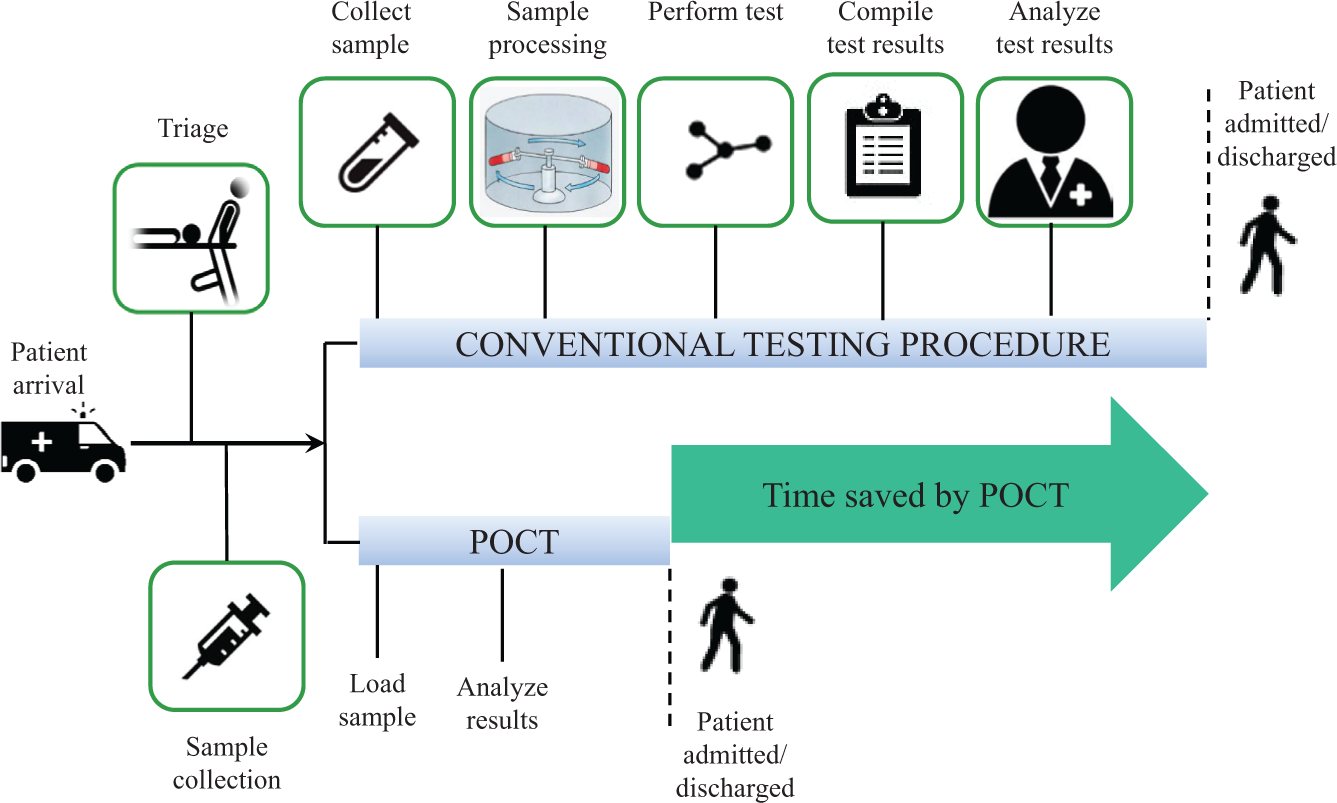
Rapid turnaround of results with point-of-care testing (POCT) compared to a conventional testing procedure.
POCT Users and Settings
A detailed description of various POCT users and settings has been published. 43 The simplest POCT with the largest commercial market is self-testing by patients in a home setting for self-assessment and referral. Self-monitoring for blood glucose is one of the oldest applications, with the earliest patents for glucose strips being filed in 1963. 45 Some other examples of self-testing include testing for pregnancy and HIV. In some countries such as the United Kingdom, patients are allowed to monitor their own international normalized ratio (INR) to adjust their oral anticoagulation therapy. 45 Community POCT usually refers to testing conducted for triage and referral by trained health workers such as pharmacists 45 in pharmacies, village workers, and paramedics 43 in a community such as aged care facilities. For example, pharmacies in the United Kingdom and Switzerland have diabetes screening programs that include blood tests and lifestyle advice for an early and cost-effective identification of patients with diabetes and their referral to a general practitioner. Some other examples of testing in the community include testing for malaria, HIV, and dengue. POCT for outpatients in clinics by health care providers (doctors and nurses) is performed for diagnosis and treatment. POCT devices can include a combination of RDTs and handheld instruments. POCT in clinical setting can include testing for HIV, malaria, syphilis, dengue, and Strep A. POCT in peripheral labs is performed by lab technicians for diagnosis, treatment, and monitoring. POCT devices can include not only RDTs but also molecular tests, enzyme-linked immunosorbent assay (ELISA), and microscopy. POCT in peripheral labs can include testing for TB, HIV, malaria, hepatitis B virus, Clostridium difficile, CD4 cell counts, methicillin-resistant Staphylococcus aureus (MRSA), flu, urinary tract infections (UTIs), and viral loads. POCT can also occur in hospital settings 46 for diagnosis, treatment, and monitoring. Tests are performed by hospital staff in emergency rooms, operating rooms, and intensive care units to eliminate the need to perform the testing in laboratories. For example, rapid HIV testing can be performed by hospital staff in labor wards 47 to reduce mother-to-child transmission.
The cost of POCT devices can make them unaffordable in many countries, and health care providers may receive incentives from private laboratories for ordering tests instead of performing a POCT. The lack of proper guidelines and policies based on strong evidence of the validity and advantages of POCT can also be a barrier to the implementation of POCT. The validation of a POCT device requires strong regulations to ensure that substandard and poor-quality RDTs do not reach the market. A good infrastructure that includes power supply, refrigeration, and waste disposal units at the care centers is necessary for implementation of certain POCTs. A shortage of staff with the required knowledge and training to perform POCT with quality assurance can hinder the implementation of POCT. It may not be possible to claim POCT expenses from insurance providers when these tests are performed in community or home settings. A lack of awareness among health workers and care providers regarding the latest and advanced POCT options available may lead to patients being sent to laboratories for testing. Stigma and a lack of confidentiality associated with tests such as those for HIV may discourage the social acceptance of POCT in community settings.
Microfluidic Techniques
The application of microfluidics for the development of biosensors has proven to play a significant role in analytical investigations of biological and chemical samples. A generic analytical procedure can be broken down into three broad categories, 48 as shown in Figure 3 : the analytical principle on which the measurement is based, the analytical method that is the concept for optimizing the conditions of the analytical principle chosen, and the analytical procedure that includes all considerations from the analyte to the analytical result.
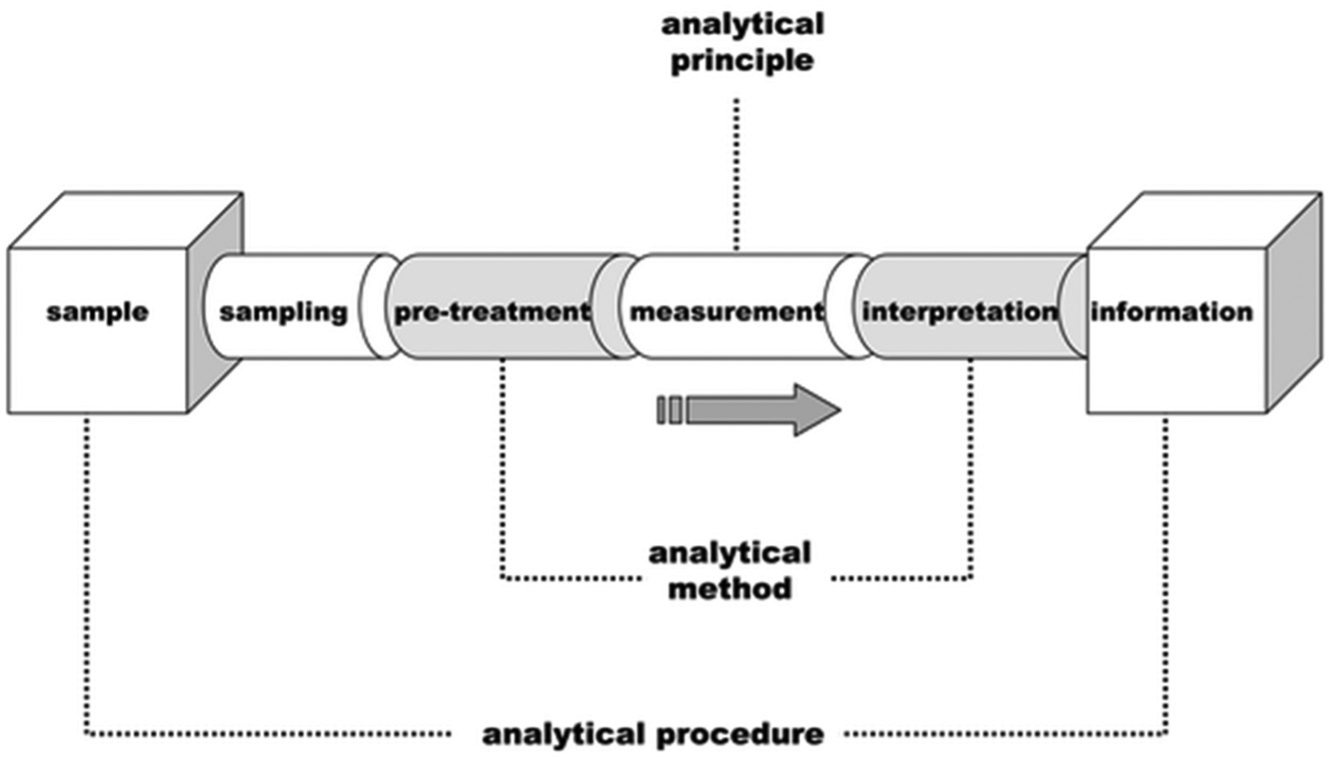
Schematic of a generic analytical process. Reproduced with permission from Ref. 48
Microfluidic devices inherently possess characteristics that make them advantageous for POCT applications: smaller quantities of test samples and reagents required for analysis that can also be precisely handled, drastic reductions in test times due to reductions in diffusion path lengths in microfluidic devices, performing multiple test types simultaneously (multiplexing), and the capability to integrate and automate various process steps on the same platform. A sample pretreatment and enrichment step is necessary for detecting biological samples at very low concentrations.
Antibodies, Antibody Alternatives, and DNA-Based Assays
Among the various biorecognition elements, antibody- and DNA-based approaches have been used widely. Antibody-based detection is one of the main analytical techniques and has proven to be successful in specific and high-affinity detection of biological samples. In recent years, engineered antibody fragments, recombinant antibody fragments (rAbs), single-chain-variable fragments (scFvs), and monovalent antibody fragments (Fabs) have evolved from antibody-based detection and are now emerging as credible alternatives. 49 These fragments not only retain the targeting specificity of conventional antibodies but also can be produced more economically. Antibody-based probes can be used for detection of specific proteins, toxins, and also whole cells. For example, antibody-based probes have been used for detection of cholera toxin subunit B (CTB), 50 a surrogate biotoxin (ovalbumin) in a raw-milk sample, 51 and microarray immunoassays for simultaneous detection of proteins and bacteria. 52 Antibody-based whole-cell detection in microfluidic systems has been demonstrated in combination with the chemiluminescence technique, 53 the impedance technique, 54 and various other techniques. Antibodies are relatively easy to use and are widely available. However, they are costly, have poor chemical and physical stability, require animals for their production, and are limited in options for some analytes. In recent years, alternatives to antibody-based approaches such as enzyme–substrate reactions, 55 molecularly imprinted polymers, 56 aptamers, 57 and antimicrobial peptides 58 have been developed.
DNA hybridization assays 59 provide more sensitive, specific, and rapid detection of target nucleic acids when compared to conventional antibody-based assays. Many microfluidic DNA-based probes coupled with techniques such as fluorescence resonance energy transfer (FRET), 60 conductance impedance, 61 and surface plasmon resonance imaging (SPRi) 62 have been reported. Peptide nucleic acid (PNA) is a DNA mimic and has a peptide backbone instead of a sugar phosphate backbone. PNAs have good chemical and thermal stability, 63 resistance to enzymatic degradation, faster hybridization kinetics, and the ability to hybridize at lower salt concentrations. A PNA beacon 64 has been designed for detection of 16S rRNA of Escherichia coli in a droplet-based microfluidic device. PNAs, however, are more expensive than DNA probes.
Amplification
PCR 65 is an enzymatic assay that plays a very important role in genetic analysis and biochemical research. PCR enables amplification of a specific DNA fragment from a complex pool of DNA by cycling through three temperature steps and creating several million copies of target DNA within a few hours. Microfluidic devices, when combined with the PCR technique, 66 could achieve significant reductions in reaction times. One of the main challenges with PCR in microfluidic systems is prevention of sample evaporation. A design approach 67 using nonmiscible mineral oil to cover sample was reported to achieve evaporation loss of less than 5% and simultaneously detect multiple pathogens using an oscillatory-flow multiplex PCR. In recent years, the isothermal nucleic acid amplification 68 of DNA and RNA has become popular because it does not require as much energy and thermal momentum for temperature cycles when compared to PCR-based systems. Some of the methods for isothermal amplification include loop-mediated isothermal amplification (LAMP), 69 helicase-dependent amplification (HDA), 70 nucleic acid sequence–based amplification (NASBA), 71 recombinase polymerase amplification (RPA), 72 and rolling circle amplification (RCA). 72
Sample Preparation
Sample preparation steps are crucial to achieve high sensitivity and specificity in any detection technique. The two important approaches are enrichment of target analyte and/or removal of inhibitors. Sample preparation can be complicated with biological fluids such as saliva, blood, and environmental samples. Some of the popular sample preparation techniques in microfluidic devices include dielectrophoresis (DEP), microparticle- and nanoparticle-based approaches, and filters.
DEP is a term that was first introduced by Pohl 73 to describe the translational motion of particles when subjected to nonuniform electrical fields. The DEP force depends on several parameters, such as the dielectric properties, the shape and size of the particle, the frequency of the applied field, and the electrical properties of the suspension medium. A microfluidic device has been developed for continuous DEP fractionation and purification of sample suspensions of biological cells, 74 in which the sorted cells are then delivered to two sample collection ports. A variation of DEP using a combination of both positive and negative DEP has been reported to continuously separate and concentrate bacteria 75 from cerebrospinal fluid and blood. Insulator-based DEP (iDEP) in a microchannel containing an array of cylindrical insulating structures, 76 in which nonuniform fields are produced by using arrays of insulating structures, has been reported for the concentration and fractionation of a mixture of bacteria and yeast cells. Different electrode structures also have been implemented in a three-dimensional (3D) DEP 77 to control the electrical field distribution for continuously sorting and concentrating different types of bacteria.
Micro- and nanobeads have high surface-to-volume ratios with low diffusion times and have been applied to capture and separate target analytes for obtaining higher sensitivity and selectivity in detection. Micro- and nanobeads have been widely used for various applications such as nucleic acid enrichment78–79 and whole-cell enrichment.54,80–81 A magnetic nanobead amplification-based QCM immunosensor, 82 with magnetic nanobeads coated with anti-H5 antibodies for amplification of the binding reaction between antibody and the H5N1 virus, has been developed. Magnetic micro- and nanobeads provide a means for better particle manipulation when compared to the nonmagnetic micro- and nanobeads. A typical protocol for applying magnetic micro- and nanobeads is based on mixing the functionalized beads with the target sample, 83 followed by application of magnetic field to capture the beads and a wash step to remove any unbound target antigen and analyte. For example, an impedance biosensor system based on a combination of magnetic nanobeads coated with avian influenza virus subtype-specific antibody for capture (separation and concentration) of a target virus, followed by impedance measurement of the nanobeads–virus complexes, has been developed. 84 A magnetoresistive immunosensor 80 for detection of E. coli, which can detect small variations in the magnetic field caused by the conjugation of magnetic beads to previously immobilized antigens on the surface, has been reported.
Microfluidic filters based on physical filtration provide an alternative to magnetic separation for the rapid separation and enrichment of target samples. A microfluidic filter chip 53 with a stepped microchannel configuration has been reported to capture and enable the formation of a sandwich complex of a monolayer of beads, bacteria, and peroxidase-labeled antibodies inside a microchamber. Polyacrylamide membranes with nanopores 85 for simultaneous concentration of viral particles and separation of virus–fluorescent antibody complexes was observed to improve the sensitivity and detection time when compared to the use of an electrophoretic immunoassay alone. Microfabricated pillars that were functionalized with an affinity for bacterial cells inside a PCR chip have been applied to detect E. coli in blood samples.
Microfluidic Platforms
At the micrometer scale with increased surface area–to-volume ratio, surface forces become dominant and influence the operation of microfluidic devices. It becomes challenging to mix fluids in microfluidic devices due to limited convection and reliance on diffusion. In recent times, various design approaches have been developed to enable microfluidic devices to be more suitable for POCT. Typically, POCT devices are expected to have low cost, portability, user-friendliness, reduced power requirements, and reduced accessories required for their operation. The most power-consuming and expensive components that also increase the footprint of a POCT device are usually the transducer and the pumping components with valves. Any size reduction in the pumping system or even eliminating a need for pumps and valves would be of great advantage in the development of portable POCT devices. Microfluidic platforms can be classified 86 based on liquid propulsion principles such as pressure-driven flow.
Capillary Flow Platforms
Capillary flow platforms are commonly known as test strips in which the sample liquids are driven by capillary forces without any applied pressure. The liquid flow is controlled by the wettability and feature size of the microstructured substrate. All of the required chemicals are pre-stored within the test strip. For example, capillary flow platforms such as the LF immunoassay or blood glucose test strips are among the most commercially successful microfluidic POCT devices today. Figure 4 (reproduced from Mark et al. 87 ) shows a schematic design of a lateral flow test. Typically, the test results are optical signals and are usually implemented as a color change in the detection area that is visible to the naked eye. Capillary flow platforms are widely implemented and are generally inexpensive. However, capillary flow platforms are difficult to adapt to complicated assays, requiring steps such as mixing, dilutions, washing, and so on. In recent years, numerous efforts have been made to overcome these limitations and enable multiplexing on capillary platforms.
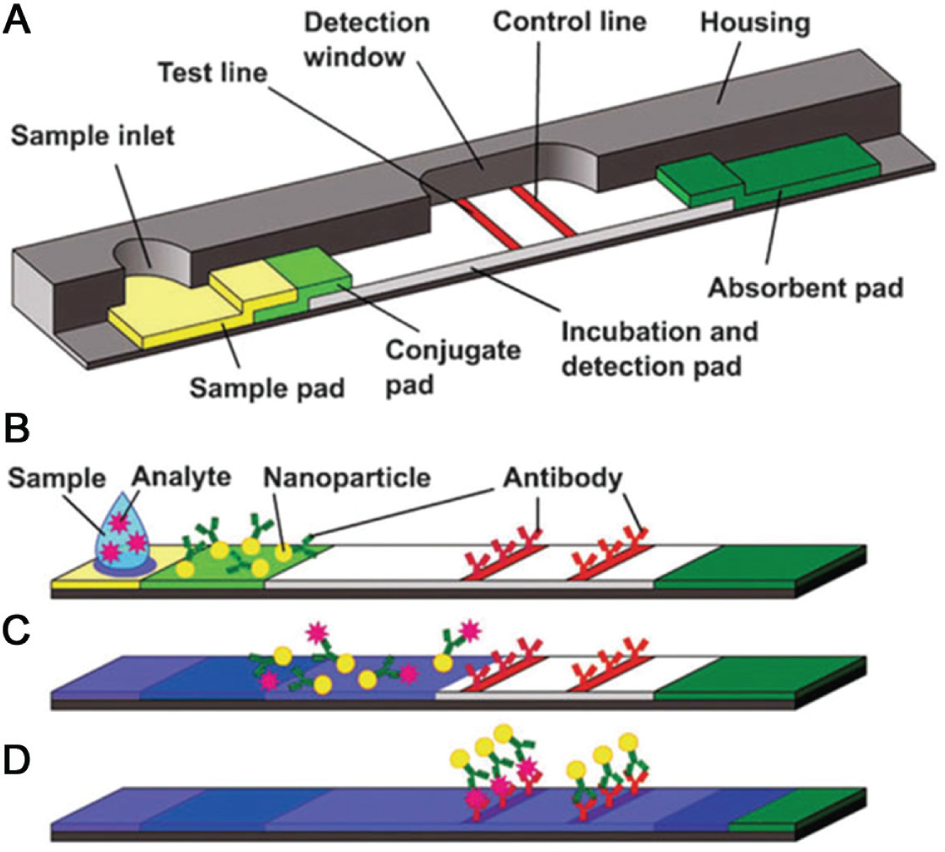
Schematic design of a lateral flow test: (
Applied Lateral Pressure Platforms
Applied lateral pressure platforms are pressure-driven devices in which external pumps or built-in micropumps are applied to drive fluids through the platform. This platform allows a variety of fluidic operations 88 such as mixing, valving, metering, and separation. However, the presence of pumps with fluidic connections to the pump leads to large dead volumes.
Applied Transverse Pressure Platforms
Applied transverse platforms are based on transverse pressure to propel or stop the fluid flow. The microchannels and reservoirs are made of soft elastomers that can be squeezed or pinched off by external pressure exerted by fluid such as compressed air flowing in adjacent channels. Figure 5 shows an example of a transverse pressure-driven flow. The material for these platforms is limited to soft elastomers such as polydimethylsiloxane (PDMS). 89 It is possible to design large fluidic networks 90 on these platforms, and they are suitable for high-throughput applications.
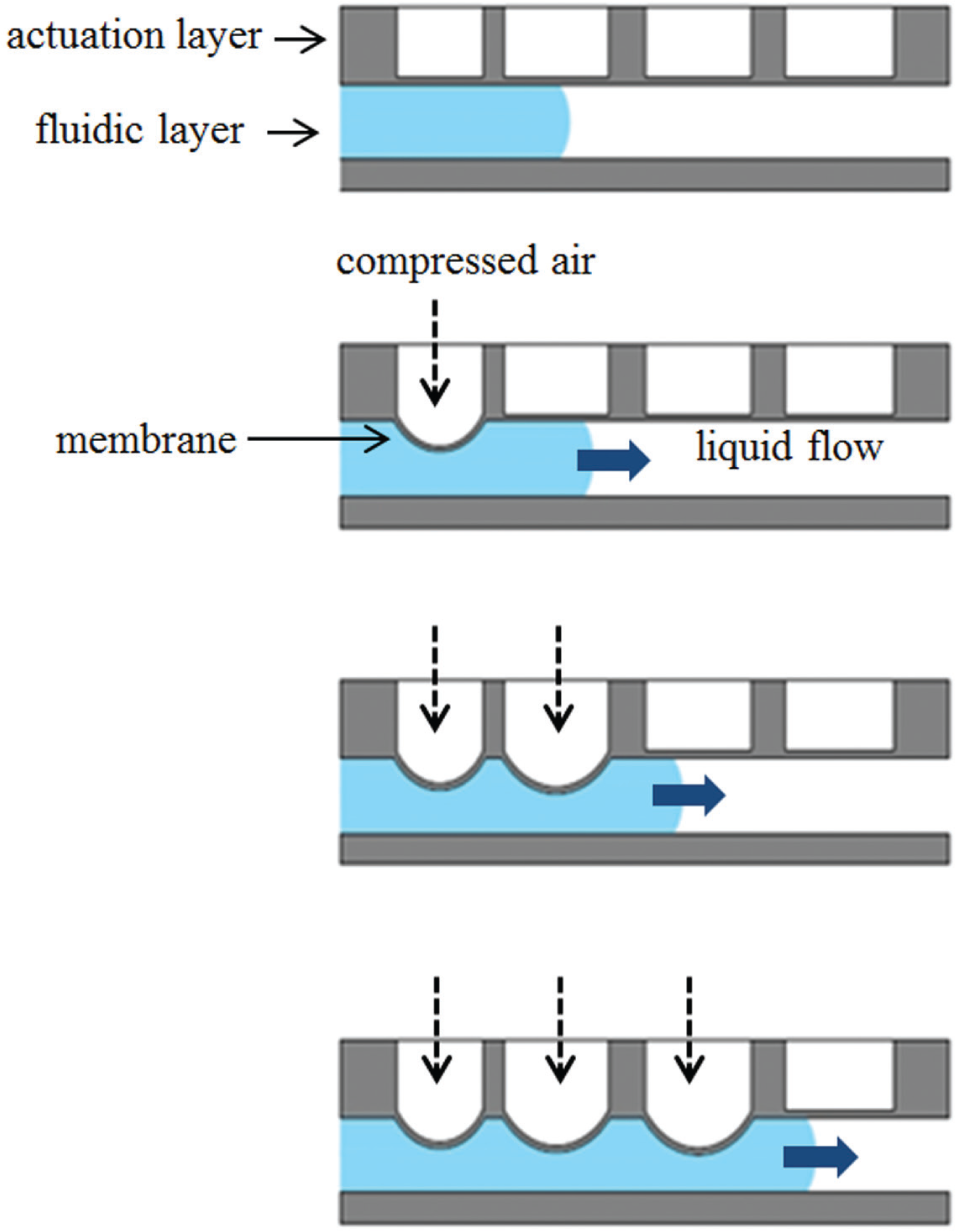
Applied transverse pressure platform. A sequential deflection of polydimethylsiloxane (PDMS) membrane due to pneumatic actuation displaces the liquid underneath to generate flow.
Droplet Microfluidics
Droplet microfluidics, 91 a subset of microfluidics that involves the generation and manipulation of micrometer-sized emulsion droplets on a microfabricated platform, has started to attract greater attention in recent years. Droplet microfluidics makes the need for conventional micropumps redundant in a microfluidic platform. A controlled and rapid mixing of fluids in the droplet reactors 92 results in decreased reaction times when compared to continuous-flow microfluidic devices. Droplet microfluidics can also store all the reagents in droplets and eliminate the need for fluidic coupling to external reagent reservoirs. Any scaling up of droplet microfluidic device does not increase device size or complexity, thereby making these devices very suitable for high-throughput analysis for POCT applications. In digital microfluidic devices, unlike the droplet microfluidic devices, it is possible to address each droplet separately in an array of electrodes without any microchannels, and the droplets can be moved based on the electrowetting-on-dielectric principle, making the device suitable for high-throughput assays. A fully integrated and reconfigurable droplet-based “digital” microfluidic lab-on-a-chip for performing a colorimetric enzymatic glucose assay on human physiological fluids has been developed. 93
Centrifugal Platforms and Lab-on-a-CD
Another approach that can eliminate the need for external tubing and pumping systems is a “lab-on-a-CD,” a technique based on fluid flow driven by centrifugal force achieved on a rotating CD platform. 94 Based on a rotating disc, centrifugal pumping can generate a wide range of flow rates 94 (from less than 10 nl/s to greater than 100 µl/s) depending on the disc geometry, rotational rate (rotations per minute), and fluid properties. Also, the pumping is not very sensitive to the physicochemical properties such as pH, ionic strength, or chemical composition. Centrifugal pumping of a variety of liquids, including aqueous solutions, solvents, surfactants, and biological fluids (blood, milk, urine, etc.), has been demonstrated. The valving in these systems is achieved either by “capillary” valves in which capillary forces can stop fluids until rotationally induced pressure is sufficient to overcome capillary pressure at the burst frequency or by hydrophobic methods. A wide range of fluidic functions such as valving, decanting, calibration, mixing, metering, sample splitting, and separation can be implemented on a lab-on-a-CD. All of these characteristics of a lab-on-a-CD make it suitable for multiplexing and POCT. A fully integrated lab-on-a-CD that can perform both multiple biochemical analysis and sandwich-type immunoassay simultaneously has been developed 95 and is shown in Figure 6 (reproduced with permission from Lee et al. 96 ). This platform is suitable for POCT because the required sample volume of blood is small (350 µL vs. 3 mL), takes less time (22 min vs. several days), and does not require specially trained operators or expensive instruments to run the biochemical analysis and immunoassay separately. Researchers at Sandia National Laboratories 97 have developed SpinDx, a centrifugal platform for conducting simultaneous multiplexed immunoassays and white blood cell counts from a single finger prick of whole blood with a total sample-to-answer time of less than 15 minutes. A centrifugal microfluidic platform for rapid and accurate determination of the concentration of hemoglobin in human whole blood has been reported. 98
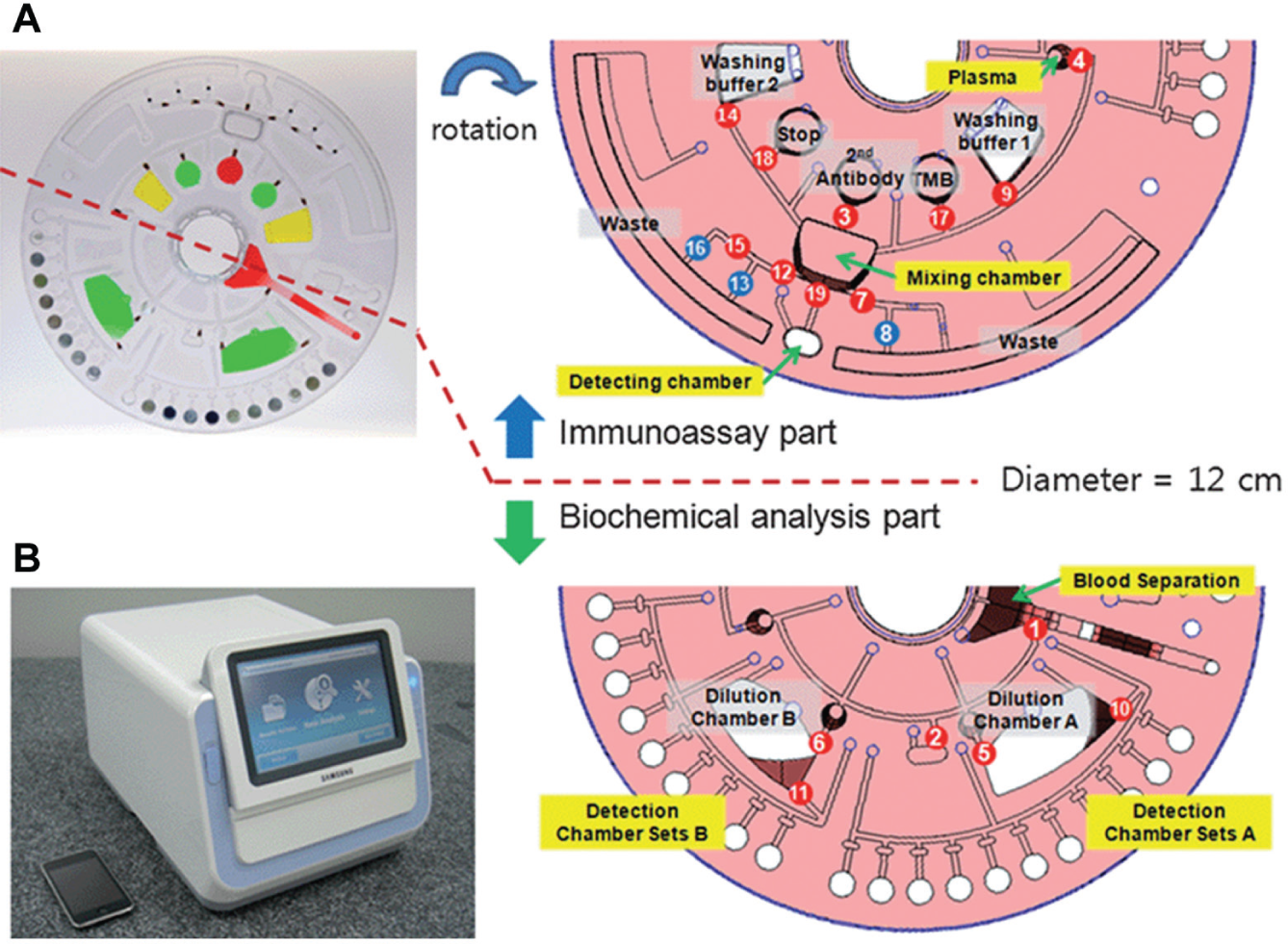
(
Acoustically Driven Flows
Surface acoustic waves (SAWs) traveling over the surface of substrate to propel a liquid droplet in the wave propagation direction have been applied to generate, propel, mix, and break up liquid droplets. 99 SAW platforms enable enhanced mixing and centrifugation within individual droplets on a microscale. 100 However, due to the open configuration of SAWs, for application such as PCR that involve a heating step, sample evaporation might be an issue.
POCT Platforms
Microfluidic diagnostic technologies can be separated into lab-based testing devices and POCT platforms. 86 Lab-based testing devices are not required to be completely integrated. For example, sample preparation can be done outside before introducing it into the device. Also, lab-based devices are not restricted by the requirements of readout equipment and theoverall size or weight of the platform. POCT platforms are expected to have significant integration of various assay steps of compact techniques that can be challenging during the development of compact, inexpensive, portable/desktop diagnostic platforms. POCT platforms can be broadly classified into three types 86 based on a microfluidics perspective. Depending on the complexity of the assays, some of the tests may not require steps such as washing and are suitable for adaptation to LF platforms. Handheld and desktop POCT platforms can perform multiplexing or do quantitative automated multiple tests. Molecular diagnostic POCT platforms represent the fastest-growing POCT segment, 86 and many new products based on this platform are expected in the near future.
Strip-Based/LF POCT
Strip-based/LF tests using capillary forces are the most commercially successful POCT and can be divided into two categories: qualitative tests that can identify the presence or absence of an analyte in the sample, and quantitative tests that provide concentration of the analyte using a readout device. A popular example of a qualitative strip-based/LF test is the pregnancy test. Pregnancy test strips typically are based on an immunoassay in which the presence or absence of a specific protein such as gonadotrophin (hCG) 86 is detected based on the interaction of antibodies immobilized on the substrate with antigens in the sample. A test strip for blood glucose level measurements is one of the most popular quantitative LF tests. A drop of blood is placed on a test strip, and capillary action moves the sample to a region of the strip where an enzyme such as glucose oxidase is embedded. The reaction of glucose with glucose oxidase is detected electrochemically in a handheld reader by amperometric detection of the peroxide reaction product, 86 and the current is proportional to the blood sugar level in the sample. LF testing has evolved in recent years to include more sensitive bio-recognition elements such as nucleic acid hybridization, 101 resonance-enhanced absorption, 102 chemiluminescence, 103 and silver-enhanced gold nanoparticle labels. 104 A lot of research is now focused on simplification of LF devices to their highly simplified ingredients: patterned filter paper impregnated with ingredients, the so-called bioactive paper that forms the basis of paper-based analytical devices (µPADs). Recently, paper has been functionalized as a substrate to construct microfluidic devices for use in rapid diagnostics. 105 A patterning of paper into regions of hydrophilic channels separated by hydrophobic barriers provides four basic capabilities 105 on a single µPAD: (1) the distribution of sample into spatially segregated regions to enable multiple assays simultaneously; (2) the ability to move the sample based on capillary action without the need for any pumps; (3) the capability to handle small-volume samples such as tears, saliva, urine from neonates, and drops of blood from finger sticks; and (4) easy disposal of hazardous wastes because these devices can be incinerated. The fabrication process is simple, and fabrication material for these devices is usually a cheaper material such as cellulose fiber. Figure 7 (reproduced from Mao et al. 106 ) shows a µPAD with a multilayer 3D fluidic network formed by stacking layers of patterned paper and double-sided adhesive tape that has been demonstrated for glucose and protein assays.
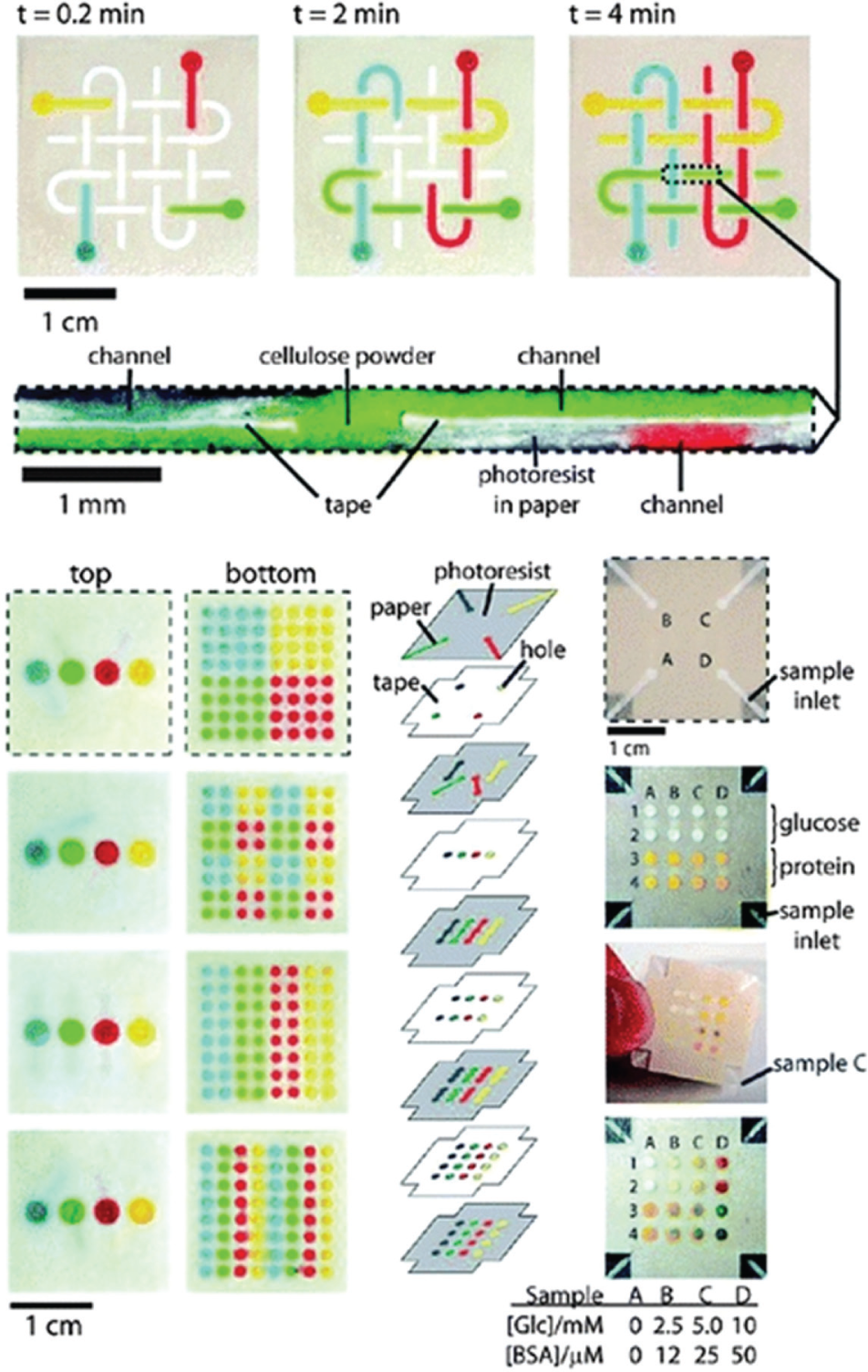
Three-dimensional (3D) paper microfluidic channel network and multiplex detection. Reproduced with permission from Ref. 106
Handheld and Desktop POCT
Handheld and desktop POCT meets the requirements of multistep tests with automated sample processing, assay steps, and detection. Handheld and desktop POCT platforms use microfluidic technologies ranging from centrifugal fluidics to electrokinetic flows. Typically, desktop platforms use a set of microfluidic cartridges for various types of tests. The popular handheld analyzer i-STAT 107 (Abbott, Abbott Park, IL), as shown in Figure 8A (reproduced with permission from Chin et al. 107 ), uses separate fluidic cartridges for blood gases and electrolytes, lactate, coagulation, hematology, and cardiac biomarkers. The operation of i-STAT 108 begins with introducing sample in the form of whole blood into the cartridge, which is then sealed and inserted into the analyzer. The various processing steps in i-STAT are completely automated to perform amperometric or potentiometric detection on the thin-film biosensor array. Epocal (acquired by Alere in 2010; Alere, Waltham, MA) has developed a portable blood chemistry analyzer, as shown in Figure 8B (reproduced with permission from Chin et al. 107 ). Fluid manipulation and acutation are achieved using a combination of electroosmotic and pneumatic pumps and capillary flow. Epocal uses self-contained cards called Flexcards with fluidic circuits.
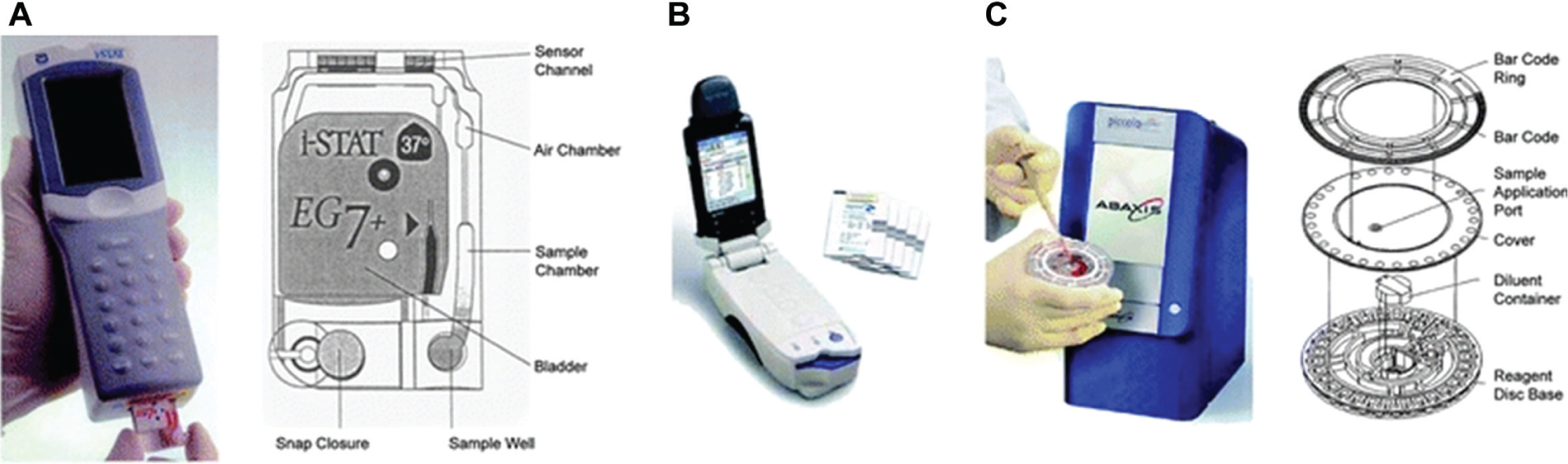
(
Molecular Diagnostic POCT
There are two major reasons 86 for a growing demand for molecular diagnostic POCT. The first reason is the seroconversion delay, which in the case of many infectious diseases has consequences such as permanent damage or death and the possibility that an undiagnosed carrier will transmit the disease to other people. The second reason is the advantage of increased sensitivity that can be achieved by amplification of nucleic acids by the PCR technique. There are numerous molecular diagnostic POCT platforms that are commercially available or under development. For example, Cepheid’s GeneXpert (Cepheid, Sunnyvale, CA) is a real-time PCR-based DNA analysis system capable of DNA extraction, real-time DNA amplification, and detection. 109 The system is capable of “sample-to-answer” results from an unprocessed sample in approximately an hour. A disposable cartridge contains all the reagents, cell lysis is carried out by ultrasonic energy, and the platform uses an integrated fluorescence detection system. A barcode system is used to store the test results of each test, and the system has a small footprint with low power consumption. Other examples of commercially available molecular diagnostic POCT include Idaho Technology’s FilmArray 110 (BioFire, Salt Lake City, UT) and the Roche cobas Liat (lab-in-a-tube) System (Roche Diagnostics, Basel, Switzerland). 111
Applications of Portable Biosensors
Infectious Diseases
Infectious diseases are one of the major causes of illnesses and mortality around the world, particularly in developing countries with limited resources. Early detection of pathogens is essential for preventing, treating, and controlling infectious diseases from gaining pandemic proportions. For example, according to the CDC, the 2014 Ebola epidemic is the largest in history, affecting multiple countries in West Africa, and is a serious concern with efforts being made to detect and prevent further spreading of the disease. A complex combination of greater global movement of people and animals, demographic shifts, ecological changes, climate changes, and changes in animal husbandry practices has led to the emergence of an increasing number of new diseases. 112 In developed countries also, there are numerous challenges with regard to pathogen outbreaks, sexually transmitted diseases, and food safety. Traditional methods of pathogen detection such as cell culture and colony-counting methods, PCR, and immunology-based methods such as immunomagnetic separation are time-consuming and laborious. Portable biosensors with the required sensitivity and specificity can overcome these limitations. A rapid detection of an antigen has a significant effect on the success of disease zoning, control, or eradication. Disease surveillance, when combined with good data collection and management, will provide better decision-making tools and early warning systems for disease emergency vigilance. One of the main requirements in developing a biosensor for infectious diseases is a low detection limit without compromising the selectivity. There are many infectious diseases that can spread very rapidly 113 before any symptoms are observed, and a biosensor with a capability to detect low levels of antigen at the onset of the disease will be a highly invaluable tool. Portable biosensors used for infectious disease also should be inexpensive and robust with high-throughput capabilities.
More than half of the biosensors applied for detection of pathogens are based on electrochemical transducers. 114 This is mainly because electrochemical biosensors have properties such as low cost, high sensitivity, and independence from solution turbidity, and they easily lend themselves to miniaturization, are adaptable to microfabrication, and have low power requirements and relatively simple instrumentation. 115 An amperometric immunosensor 116 for Newcastle disease antibody in poultry serum samples has been developed for concentrations up to 443.24 ng/ml with a detection limit of 11.1 ng/ml. A impedance biosensor for detection of the avian influenza H5N1 virus 117 at a titer higher than 103 EID50/ml (EID50: 50% egg infective dose) within 2 h has been developed. A portable amperometric biosensor for the rapid evaluation of Salmonella pullorum contamination in chicken with a detection limit of 100 colony-forming units (CFU) per milliliter in culture media and chicken samples within 10 min for each sample has been reported. 118 A novel potentiometric immunosensor 119 for the detection of hepatitis B surface antigen by means of self-assembly to immobilize hepatitis B surface antibody on a platinum disk electrode based on gold nanoparticles has been developed. A dynamic concentration range of 4–800 ng/ml and a 1.3 ng/ml detection limit were observed, and results were observed to be in agreement with the standard ELISA method. The bioelectric recognition assay 120 (BERA) is a CBB method that detects the electric response (membrane potential) of cultured cells suspended in a gel matrix to various ligands that bind to the cell and/or affect its physiology. BERA has been applied for detection of hepatitis C virus 121 in blood samples to rapidly (assay time 3–5 min) and specifically detect at a concentration lower than 100 pg/ml. Impedance spectroscopy is also one of the electrochemical methods that has been applied widely for pathogen detection. Impedance spectroscopy provides the advantage of label-free/reagentless bioaffinity sensing, 122 making it suitable for real-time monitoring. A portable impedance biosensor for detection of multiple avian influenza viruses 123 has been reported. It has been proven that impedance spectroscopy is an efficient method when analyzing antigens suspended in clean buffered solutions. 124 However, the detection of antigens in a complex medium such as blood remains challenging. It has been observed that the noise signal from nonspecific adsorption is an issue, and many electrode surface modification approaches 125 to minimize noise are being explored in this direction. Similarly, conductometric biosensors based on changes in conductance before and after a biorecognition event are also being applied for rapid detection of various foodborne pathogens 126 and achieve lower detection limits. A biosensor for detection of bovine viral diarrhea virus 127 that is sensitive at an average concentration of 103 cell culture infective doses (CCID) per milliliter in artificial blood serum samples has been developed. The application of nanomaterials, such as carbon nanotube modified electrodes 128 for electrochemical detection, has enhanced the detection capabilities of these devices for various biomolecules. In recent years, screen-printed electrodes 129 have garnered a lot of attention in DNA-, immune-, and enzyme-based biosensors. Screen-printed electrodes are a more economically feasible option for mass production of disposable electrodes.
Optical biosensors also have received a lot of attention for detection of pathogens. Numerous optical detection modes such as absorption, reflection, fluorescence, chemiluminescence, and phosphorescence have been applied for biosensors. However, SPR- and fluorescence-based biosensors are more popular for pathogen detection. The detection limit of optical techniques in combination with optical fiber 130 is comparable to that of traditional benchtop analyzers. The Sensata Spreeta SPR sensor (Sensata, Attleboro, MA), a commercially available, low-cost SPR sensor, has been demonstrated 131 for detection of E. coli O157:H7 in beef, apple juice, and ground beef extract with a detection limit of 102 CFU/ml in 30 min with good specificity (it is nonresponsive to E. coli K12 and Shigella). The Spreeta sensor has also been applied for Campylobacter jejuni in poultry meat 132 at a low detection limit of 103 CFU/ml with a low cross-reactivity with Salmonella typhimurium. A SPR-based immunosensor based on synthetic peptides has been developed for detection of hepatitis A virus 133 in human serum with sensitivity comparable to that of ELISA. A SPR biosensor was applied to study the coronavirus 134 of severe acute respiratory syndrome (SARS) with the capability to verify that the N-terminal deleted proteinase dimer adopts a state different from that of the full-length proteinase dimer. SPR provides excellent sensitivity, but the required instrumentation is complex and requires a layer of metal to be deposited on the device, thereby raising cost. Also, SPR suffers from strong temperature dependence, which can be a hindrance for portable biosensors in outdoor conditions. Fluorescence-based immunoassays are also of interest for pathogen detection due to their high sensitivity. 135 A fluorescence-based multi-analyte immunosensor 136 has been developed for simultaneous analysis of multiple samples with various microorganisms and toxins without significant cross-reactivity. A fluorescence immunosensor for detection of dengue virus 137 has been reported. One of the drawbacks of the fluorescence technique is that the required reagents are expensive and reaction times are often longer.
Piezoelectric biosensors such as QCMs and SAWs also have been developed for pathogen detection, even though their performance is not as good 138 as that of electrochemical and optical biosensors. A QCM biosensor for foot-and-mouth disease virus based on a self-assembled monolayer of alkane thiol 139 has been reported. A piezoelectric-based DNA biosensor for the detection of hepatitis B virus 140 has been developed by immobilizing a nucleic acid probe immobilized onto a gold electrode via a polyethyleneimine–glutaraldehyde cross-linking process. The sensitivity and reliability of piezoelectric biosensors for pathogen detection are comparable 141 to those of the conventional ELISA method. However, the stability of the sensor surface in biological fluids and crystal regeneration remains a challenge.
Chronic Diseases: Monitoring and Diagnostics
Glucose measurement using handheld glucometers is the world leader in commercial volume for POCT. The production volume of glucose test strips for home use has increased tremendously, approaching 1010/year, with single production lines making devices at a rate of 106/h using printing and laminating technology. 142 The science and technology of these devices have been extensively described elsewhere. 143 Also available are POCT systems for continuous monitoring of glucose, which can replace the invasive, intravenous electrode with the minimally invasive location of a microdialysis catheter in subcutaneous tissue. 144 Currently available commercial systems include the Guardian RT from Medtronic (Medtronic, Dublin, Ireland), the Abbott Navigator, and the GlucoDay from A. Menarini Diagnostics (Florence, Italy). For diabetes care, subcutaneous devices monitor glucose continuously and wirelessly communicate with an insulin pump in real time, which allows the insulin dose to be adjusted based on preprogrammed patient-specific algorithms.
Blood gas analyzers (BGAs) for pH, pO2, and pCO2 are typically based on electrochemical or optical sensors. Some of the commercially available BGAs include the IRMA TruPoint by ITC Medical (Edison, NJ), the Critical Care Xpress series of instruments from Nova Biomedical (Waltham, MA), the cobas b 221 from Roche Diagnostics, and the RAPIDPoint 400/405 from Siemens Medical Solutions (Erlangen, Germany). An optional configuration of a BGA is the inclusion of a CO-oximetry unit. The CO-oximetry unit is a multiwavelength spectrophotometer that measures the typical absorption spectra of the various hemoglobin (Hb) species to distinguish O2–Hb from other Hb species and to determine O2–Hb saturation. 144 Viscoelastic coagulation testing 145 is the combined analysis of plasma clotting, thrombocyte function, and fibrinolysis. Some of the commercially available instruments include the ROTEM from TEM International (Munich, Germany) and the Sonoclot series from Sienco Inc. (Boulder CO). Optical aggregometry 146 can be applied to analyze platelet function in terms of in vitro bleeding time. Examples of commercially available analyzers include the PFA-100 System from Siemens Healthcare Diagnostics (Erlangen, Germany) and the VerifyNow System from Accumetrics (San Diego, CA). The significance of plasma lipid levels to assess cardiovascular risks is very well recognized. Some of the small POCT cholesterol systems include Accutrend Plus (Roche Diagnostics), CardioChek PA (PTS Diagnostics, Indianapolis, IN), and MultiCare-in (BSI, Arezzo, Italy). 147 MultiCare systems are pocket-sized reflectance photometers in which the color intensity from a chromogen reaction is proportional to the concentration of cholesterol or triglycerides in blood. A drop of capillary blood is applied to the test strip to obtain results for cholesterol and triglyceride assays. Desktop POCT cholesterol analyzers, such as the Abaxis Piccolo Xpress (shown in Fig. 8C and reproduced with permission from Chin et al. 107 ) and the Alere Cholestech LDX, 148 are also available. The Biosite Triage MeterPlus 149 (Biosite, San Diego, CA) has immunoassay test panels for cardiac markers. In women, breast cancer is the most commonly diagnosed cancer and is second to lung cancer as a cause of cancer death. A rapid point-of-care breath test for the detection of breath biomarkers of breast cancer has been evaluated. The BreathLink system 150 (Menssana Research, Newark, NJ) collects, concentrates, and assays breath volatile organic compounds (VOCs) in approximately 6 min. The BreathLink system also has been reported to identify breath biomarkers of active pulmonary TB. 151 An ultrasensitive diagnostic platform called NanoMonitor 152 (Accu-Chek, Indianapolis, IN) has been developed to enable the rapid label-free analysis of a highly promising class of biomarkers called glycans based on the principle of electrochemical impedance spectroscopy.
Plant Pathogen Detection
Plant pathogen detection is important for detecting bacteria, viruses, and fungi in various settings such as natural landscapes, country borders, greenhouses, and mass production facilities. About 20–30% of the field crops are known to be lost annually due to various diseases. 153 It is also important to detect various plant diseases in plantations within forests and other natural environments, and more important for plants that are part of the habitats for wildlife. Among the top 10 bacterial plant pathogens 154 based on their economic and scientific importance, Pseudomonas syringae and Ralstonia solanacearum are the top two economically important pathogens that cause diseases that infect a range of crops, including potatoes and bananas. A rapid detection of plant pathogens has gained importance due to factors 155 such as developments in international trade, emerging pathogens with increased resistance to pesticides, increased human mobility, and various new regulations that impose restrictions on the application of toxic chemicals to control the spread of new plant diseases. Early on-site detection of plant pathogens with portable biosensors will enable the design of strategies to control the spread of diseases and will also help the study of disease epidemiology. 156 The agricultural sector today requires biosensors for detection of multiple potential pathogens or newly emerged pathogens 157 without necessarily knowing the plant diseases affecting that particular crop group. The development of portable and rapid plant pathogen biosensors would be of value to users from various backgrounds, including individual growers, regulatory agencies, exporters, and researchers. 158
The traditional plant pathogen detection technique 156 involves interpreting visual symptoms of disease, which is followed by pathogen diagnosis using microscopy techniques to confirm the data. However, this approach cannot be applied for pathogen diagnosis before the symptoms start appearing. Also, this approach requires analysis from plant pathologists and taxonomists, and is not suitable for on-site diagnosis of plant pathogens. There is a strong demand for developing new techniques for biosensors that would allow rapid and on-site diagnosis of plant pathogens by even unskilled users. The current techniques for plant pathogen detection using portable biosensors can be classified into direct and indirect techniques.
Direct techniques detect the pathogen itself and include immunological techniques using antibody–antibody alternatives and molecular techniques using nucleic acid probes. Among the direct techniques, immunoassays constitute a majority of commercial biosensors for plant disease diagnosis. Molecular techniques mainly include ELISA 159 and PCR-based methods. Cepheid SmartCycler, 160 a portable, real-time PCR platform, has been applied for the detection of Phytophthora ramoru, which is known to cause sudden oak death.
Indirect techniques rely on detecting only the effects of the pathogen on the physiological response of the plant. Indirect techniques 161 include spectroscopic/imaging methods and VOC detection methods. Spectroscopic/imaging methods include fluorescence spectroscopy, visible-infrared spectroscopy, and hyperspectral imaging. Fluorescence spectroscopy 162 relies on the spectral signature to monitor physiological stress levels in plants by analyzing fluorescence at various wavelengths. The spectral signature of a diseased plant 163 is influenced by the effect of the disease on plant processes such as chlorophyll degradation, photosynthesis, and so on. Thermal properties of diseased plants change, and this aspect has been applied in infrared thermography 164 to detect local temperature changes due to plant defense mechanisms in tobacco leaves. X-ray imaging 165 has been applied for detection of fungal disease in wheat. The presence of pathogens in a plant changes the VOC produced by the plant and can be sensed using electronic nose devices with integrated gas sensors and a pattern identification system. Electronic nose–based biosensors have been applied for diagnosing diseases in wheat 166 and pear 167 plants. Indirect techniques rely on comparison between healthy and unhealthy plants, and this frequently requires a recalibration of the biosensor for each plant. Also, indirect methods provide only an indication of whether a disease is present without detecting the disease-causing pathogen.
DNA sequencing is another approach that can be applied for pathogen diagnosis. Genome analysis of plant pathogens 6 provides information about the processes and genes involved in the host colonization and pathogenicity, thereby enabling the identification of unknown plant pathogens. One of the challenges for on-site application of sequencing techniques in portable biosensors for plant pathogens is the data analysis. Data analysis 168 might be difficult to perform in places where internet and communication networks are not available for sending data from a sequencing device to a central database for further analysis. A detailed review of application of DNA sequencing technologies to future portable biosensors for plant pathogen diagnosis has been published. 6
Biological Warfare Agents
The detection of toxic compounds is of great significance not only for human health but also for homeland security against bioterrorism. Many biological and chemical agents such as bacteria, viruses, fungi, and toxins are capable of causing serious damage to both animals and humans. It is also possible for the food and water supplies to be contaminated 169 during terrorist attacks or military actions with BWAs. The biosensor platforms for BWAs, apart from being sensitive and specific, must also be capable of accurately detecting a wide variety of pathogens, including even modified or previously uncharacterized agents, directly from complex sample matrices. 170 To meet the requirements of continuous monitoring and rapid detection of various BWAs with high sensitivity and specificity, cost-effective and portable biosensors need to be developed. The majority of the biosensors for BWAs have been developed based on electrochemical transducers, 171 but optical transducers 172 also have gained importance in recent years. The emergence of nanotechnology with new materials such as nanotubes, nanowires, and nanoparticles has opened new avenues for biosensors for pathogen detection. 173 Some of the most common BWAs are mycotoxins, exotoxins from E. coli O157:H7, S. aureus, Bacillus anthracis, botulinum, Listeria monocytogenes, Vibrio cholerae, and Yersinia enterocolitica.
Anthrax disease is caused by B. anthracis, and the need for rapid detection methods 174 has become critical since 2001, when letters containing the spores were mailed in the United States. A label-free detection of B. anthracis based on DNA probe functionalized QCM biosensors 175 has been reported with a detection limit of 3.5 × 102 CFU/ml of vegetative cells. A portable, label-free optical biosensor 176 for quantitative detection of a minimum of 34 spores of B. anthracis within 35 min has been developed. A portable biosensor based on oligonucleotide sandwich hybridization 177 capable of B. anthracis detection within 15 min and no cross-reactivity for 11 tested organisms has been developed. A technology called CANARY 178 (cellular analysis and notification of antigen risks and yields) has been developed based on genetically engineered B lymphocytes that emit photons within seconds of exposure to pathogens of interest. Within a few seconds, CANARY can rapidly detect as few as 1000 CFU of B. anthracis spores extracted from seeded nasal swabs, 178 demonstrating the potential to rapidly screen patients for inhalation anthrax exposure.
Botulinum toxin is an acutely lethal toxin produced by the Gram-positive bacterium species Clostridium botulinum, Clostridium barati, and Clostridium butyrium. 179 The first immunoassay for botulinum toxin 180 was based on an optical fiber with capture antibodies immobilized on the surface, and it is capable of a detection limit of 30 pM botulinum toxin A within 20 min. A FRET fluorescence-based biosensor platform 181 for detection of botulin toxin light-chain cleavage has been reported with a detection limit of 0.5 nM of toxin light chain for eight samples simultaneously in 2 h. A fluorescence detection portable platform using fluorescent electroluminescent excitation 182 has been reported for botulinum neurotoxin A detection between 31 and 62 ng/mL after 2 h of incubation. A SPR biosensor 183 has been developed for detection of three botulinum neurotoxin serotypes (A, B, and F) with detection limits between 0.5 and 1 ng/mL in less than an hour. CBBs are emerging as an alternative 18 to ELISA and immuno-PCR methods by eliminating shortcomings such as the inability of ELISA to differentiate between active or inactive toxins or holotoxin and reduced toxin. Human-induced pluripotent stem cells (hiPSCs) have been applied for detecting botulinum neurotoxin and achieving the quantitative determination of different serotypes. 184 A novel cell-based potency assay 185 coupled with sandwich ELISA has been reported for botulinum neurotoxin A detection. A comprehensive review of the trend of CBBs in botulinum neurotoxin detection has been published. 186
S. aureus is a Gram-positive coccal bacterium that can cause diseases such as pneumonia, mastitis, food poisoning, and toxic shock syndrome to humans and animals. A biosensor based on carboxyl-modified CdSe–ZnS quantum dots and oligonucleotide probe complexes 187 has been developed to detect toxic shock syndrome toxin-1–producing S. aureus with a detection limit of 0.2 µM for the target DNA. A biosensor based on diagnostic magnetic resonance (DMR-3) 188 in combination with functionalized magnetic nanoparticles has been developed to detect S. aureus in unprocessed biological samples with a detection limit of 10 CFU in less than 30 min. Moreover, the DMR-3-based biosensor has an interface to facilitate system control and data sharing over wireless networks, which in combination with built-in temperature drift compensation makes it suitable for POCT. A piezoelectric cantilever-based biosensor 189 for sensitive detection of S. aureus enterotoxin B (SEB) has been reported within a concentration range of 2.5–25 fg/mL in apple juice and milk.
Detection of multiple analytes in a single assay is a challenging task. In addition, rapid detection and differentiation of pathogenic versus nonpathogenic species, viable versus nonviable cells, and active versus nonactive toxins are also required. A sensitive detection platform for multiple pathogens, 190 including Y. pestis, B. anthracis, and SEB, has been developed; it is capable of detecting SEB and inactivated Y. pestis separately at lower concentrations of 5 pg/mL and106 CFU/mL, respectively. A multiple phage-based magnetoelastic 191 biosensor has been developed for simultaneous detection of B. anthracis spores and S. typhimurium with lower detection limits of 5 × 103 CFU/mL for both. A multiplexed high-density DNA array has been developed with probe-functionalized microspheres 192 functionalized with 18 different 50-mer oligonucleotide probes and optical encoding indicators. The DNA array included probe sequences designed for B. anthracis, Y. pestis, F. tularensis, Brucella melitensis, C. botulinum, Vaccinia virus, and Bacillus thuringiensis. The DNA array biosensor was demonstrated to achieve a detection limit of 10 fM within 30 min of hybridization for B. anthracis, Y. pestis, Vaccinia virus, and B. thuringiensis. Research in the development of biosensors for BWAs is continually evolving, and future biosensors will be designed to be portable and allow rapid, accurate, and sensitive detection.
Cell Phone Biosensors
Modern cell phones have the potential to be an important component of biosensors due to their sophisticated user interfaces, high-resolution imaging capabilities, and advanced communication and data-processing capabilities. Cell phones are one of the most widely used consumer electronic mobile devices and have become an intrinsic part of a worldwide communication network. The central challenge to exploit this ubiquitous resource for biosensing is to craft smart interfaces between the biosensors and the cell phones. 193 At present, there are two strategies for combining cell phones with biosensors: auxiliary reusable devices (ARDs) and auxiliary disposable devices (ADDs). ARDs are specifically designed for certain cell phone brands and models, and they can be of varied complexity and sophistication. ADDs have generic designs that are compatible with diverse phone brands and models in which both the biosensing part and coupling system are disposable. An ARD system configured for a Samsung Galaxy SII smart phone has been developed that can accommodate commercial immunochromatographic assays in which contrast is proportional to the analyte concentration in blood samples. 194 This system was applied to detect tuberculosis, HIV, and malaria, and the ARD provided mechanical fitting to the phone, a three-LED light source for reflection and transmission configurations, two AAA batteries, and a test cartridge securing the assay alignment within the ARD. 194 An ADD system with a SPR-based biosensor 195 has been developed for studying biomolecular interactions, in which both biosensing and coupling elements were integrated into a single disposable component. The ADD–SPR system consisted of a disposable optical coupling element with embedded fluidics in which the biosensing assay was implemented. 196 The coupler provided mechanical and optical fitting, conditioned the illumination provided by the phone screen, and guided the reflection to the front camera of the cell phone. The acquisition software was a time-lapsed image capture program. This ADD–SPR system 196 was tested on Nokia, Android, and Apple devices with a commercial β2 microglobulin (β2M) assay, an established biomarker for cancer, inflammatory disorders, and kidney disease. Cell phones have the potential to complement POCT performance, and future devices will take advantage of this aspect. However, there are obstacles to integrating biosensors with cell phones, and a future positive scenario would involve active collaboration between phone manufacturers and ARD/ADD developers to develop hardware and software for POCT applications. For a more comprehensive review on biosensing with cell phones, readers are encouraged to refer to an article by Preechaburana et al. 193
Biosensor Data Modeling
One of the important features of a biosensor is the highly selective detection of a target analyte. The selective interaction of a biosensor element with a target analyte is expected to produce a well-defined signal proportional to the concentration of the target analyte. A mathematical relationship typically based on a simple linear model is established between accurately known concentrations of the target analyte and the resulting signal. Biosensor performance is evaluated with samples containing unknown concentrations of analyte, and the accuracy of the biosensor is analytically evaluated by the coefficient of determination (R 2 ). This procedure is widely applied and is suitable for single-target analyte detection under controlled laboratory conditions. This approach can, however, be a limitation when a multi-analyte-based approach with sensor arrays is required for applications such as medical diagnosis and continuous environmental monitoring.
The main function of a chemical model is to establish a relationship between a set of measurements and a set of target analyte concentrations. 197 This modeling approach has undergone drastic changes with the development of complex equipment along with advances in high-speed computing and has resulted in the generation of a large amount of data. An urgent need to analyze higher-order information with proper models to obtain meaningful information led to the development of the chemistry discipline known as chemometrics. Chemometrics is dedicated to extracting relevant information from the measured chemical data. 197 Among the several linear and nonlinear models developed by chemometrics, artificial neural networks 198 (ANNs) are of great interest as modeling and calibration tools. ANNs are mathematical models attempting to mimic biological neural networks functioning in a simplified way. 199 The two main ANN architectures found in the chemical literature are multilayer perceptron (MLP) and radial basis function (RBF). Preprocessing of biosensor signals is required for some biosensors to extract meaningful features to feed the ANN model. For example, when techniques such as cyclic voltammetry are applied, signal preprocessing is necessary because of the high information order of data. 200 Examples of preprocessing techniques include fast Fourier transform (FFT), baseline correction, and canonical correlation analysis (CCA). The training of the model involves selection of an appropriate set of well-known samples from the concentration range to be modeled. 201 The accuracy of the model is estimated based on some analytical figures of merit such as the sum of square errors (SSE), the coefficient of correlation (R), and R 2 . The growth of chemometrics has been aided by the availability of software such as Matlab, Python, and Stuttgart Neural Network Stimulator. 202 A potentiometric bioelectronics tongue 203 has been developed for quantification of urea and interfering alkaline ions (ammonium, potassium, and sodium) in urine samples. The biosensor array comprised all-solid state potentiometric chemosensors and biosensors modified with urease enzyme. The data set generated by the biosensor array was designed to a fractional factorial design with three levels and four factors. 199 Calibration was performed by partial least squares (PLS1) and ANN tools. An optimal network architecture of 12 input neurons (for 12 sensors in the array), 5 neurons in the hidden layer with tansig function, and 4 neurons in the output layer was selected. A novel ANN system capable of detecting and classifying pesticide residues has been reported, 204 in which the ANN is coupled to a cellular biosensor operation based on BERA to simultaneously assay eight samples in 3 min. For a more detailed discussion on ANN, readers are encouraged to refer to the book chapter 199 on biosensors coupled with artificial intelligence.
Information Management in POCT
The true success of numerous POCT devices developed for various applications depends on whether the collected data and results are communicated and are accessible for guiding clinical decisions. A majority of the patient data generated by POCT are recorded manually in the patient chart. The decentralization of POCT has challenged the traditional laboratory computing approaches, and only 10% of POCT results are collected electronically and transmitted to appropriate patient management systems. 205 Moreover, billing and management functions for POCT are being handled by manual systems. There can be substantial loss of revenue in POCT services due to inadequate information technology support. Also, new regulatory requirements for POCT do not allow manual monitoring of comprehensive POCT programs. 206 The vendors of POCT devices are also expected to provide modern information management solutions along with their devices, and this is an important decision-making factor in purchasing POCT systems.
Regulatory requirements and hospital laboratory standards typically require access to and management of not only the test results but also information regarding patient demographics, quality control, and the instrument performance data. Many POCT devices have instrument-specific data management systems, but the challenge lies in integrating them with the laboratory information system (LIS) and hospital information system (HIS). There are various instrument- and site-specific scripted interfaces 207 that can be applied to provide links between different systems (e.g., glucometers and electrolyte systems) and the LIS/HIS. One of the options is to use wireless networks with appropriate software and hardware. Another alternative is to use dedicated docking stations that are connected to the institutional network. Also, data can be routed to the LIS via a central workstation by applying a scripted interface. One of the main problems with POCT is the lack of a formal test request in the LIS/HIS, and therefore the deposition of the test results within the LIS/HIS and the patient record is compromised. 208 This problem can be overcome by either manual entry or a terminal emulator program capable of simulating the required data entry process. A better solution is to implement an electronic order generator in the context of the POCT–LIS/HIS interface. 208 One of the main factors for meeting the information needs of POCT implementations is connectivity, which is the ability to transfer POCT data, including patient data and quality control results, into the LIS/HIS. Connectivity should be bidirectional and provide access to complete laboratory information with full data-handling capability. The American Association for Clinical Chemistry has been a driving force behind the establishment of a connectivity consortium. 209 The connectivity consortium is represented from both industry and health care providers, and will be mandated to develop industry-wide POCT connectivity standards. The focus is on “open” standards that will provide the ability to mix and match instruments from various suppliers and enable POCT results to be easily integrated into the LIS/HIS. The end goal is to establish automated data acquisition techniques by applying POCT coupled with telecommunications links to provide integrated 210 and electronic health records (EHRs). This approach will enable the enhancement of computerized decision support tools that make use of structured and integrated records systems to support improvements in health care.
Wireless Biosensors
Wireless networks can provide secure and instant access to both medical and administration records. This can help in reducing errors and enable quick decision making with improved quality of care. A wide range of commercially available components-off-the-shelf (COTS) sensors, such as electrocardiography (ECG), electromyography (EMG), electroencephalography (EEG) electrodes, pulse oximetry, carbon dioxide, blood pressure, blood sugar, and temperature sensors, have been deployed to monitor vital signals. The four major functional blocks of a wireless sensor module 211 are the power supply, radio system, processing unit, and sensing module. The sensor senses the physiological data, which are preconditioned to levels suitable for digitization and transmitted to a data acquisition device. A processor is programmed to control and coordinate the activities of the sensor node for internal data acquisition and communicating with data servers. The processor is also involved in power management by controlling the sleep and wakeup modes of the sensor nodes to save battery power when not in use. Power management is critical in wireless sensor nodes for medical applications, and lowering power consumption can prolong the operating lifetime of sensors. Also, it can be challenging to the digital signal processor (DSP) to support radiofrequency (RF) transmission between severely power-limited nodes. 211 Implanted wireless sensors are required to be self-powering because, once they are deployed, accessing the sensor to recharge the battery can be challenging. Advancements in microfabrication techniques have made smaller implantable and wearable sensors more feasible. A novel wireless glucose biosensing system that can be used as a subcutaneously implantable system for continuous glucose monitoring has been reported. 212 An implantable wireless biosensor for the immediate detection of upper gastrointestinal bleeding 213 has been developed. A high-sensitivity wireless mass-loading SAW-based DNA biosensor 214 has been developed. Continuous monitoring and evaluation of the prognostics of human health require biosensor networks to be deployed both in the environment and on patients. The applications, standards, design features, and evolution of wireless sensor technologies have been reviewed. 215 The transmission of measured data in POCT systems needs to be performed for communicating data from the biosensors to the central node and for sending the collected data from the central node to a remote medical database. Wireless body area network (WBAN) is a radiofrequency-based health-monitoring networking technology that interconnects small sensor nodes that are implanted or worn by a person called the host. 211 The sensor nodes in the WBAN have the capability to measure multiple stimuli from the host’s body, and each consists of a processor, memory, a transceiver, sensors and actuators, and a power unit. Sensor nodes are designed to withstand variations in temperature and humidity, and are powered using batteries or various energy-harvesting mechanisms. These biosensors sense physiological information, process it, transmit it to the control node, receive external signals, and trigger actions such as drug delivery. The control node functions as both the data collection and routing point. The control node also periodically sends the collected data to an external base station, where it is stored for further processing. CareNet is an integrated wireless sensor network for remote health care developed by the Vanderbilt Advanced NETwork Systems Lab in conjunction with the University of California, Berkeley and Cornell University. For further information on wireless biosensor architectures, readers are encouraged to refer to a more comprehensive review by Vasan et al. 211
Quality Specifications
Analytical methods typically can be described in terms of their two performance characteristics, practicability performance and reliability performance. Practicability performance includes skills required, speed of analysis, and volume and type of sample required. Reliability performance includes precision, bias, limit of detection, and measurement range. It is believed that speed of analysis, expressed as total turn-around time (TAT), is one of the most important drivers for establishing POCT as an additional approach in laboratory medicine. 216 The quality of medical care is the degree to which health care systems, services, and supplies for individuals and populations increase the likelihood of a positive health outcome and are consistent with current professional knowledge. 217 Apart from quality, shorter TAT is also important and is achieved by POCT platforms, especially for decisions associated with emergencies and intensive care settings. However, the quality assurance and control that have been implemented in centralized laboratory testing have not been applied much in POCT. This may be because end users of POCT might consider quality control and assurance as burdensome and not realize its effect on the quality of data produced. 218 This discrepancy of focusing on shorter TATs at the cost of test quality can lead to more errors in POCT; in fact, there are reports of twice as many analytical errors in POCT when compared to the error rate in centralized testing. 219 For example, it was observed that there was a difference in test accuracy achieved by end users when compared to what was specified by the manufacturer. 220 Because POCT results are expected to be equivalent to those of central laboratory testing, the quality requirements should be enforced regardless of the testing site, process, or procedure. 220 Quality assurance programs based on ISO 22870:2006 221 POCT requirements for quality and competence have been defined for implementation in POCT. The requirements include internal and external quality control, training programs for end users, certification and recertification of end users, evaluation of new or alternative POCT instruments and systems by professionals in laboratory medicine, evaluation and approval of proposals and protocols that are strongly directed toward the end user, and maintenance of consumable supplies and reagents.
Conclusions
Microfluidic-based diagnostics for POCT has the potential to reduce global health care costs. The general trend in POCT platforms seems to be toward miniaturization, multiplexing, and networking for data transfer and storage. The highest demand at present is for developing POCT platforms for resource-limited settings. One of the major challenges is to translate research in the POCT area into affordable products that are available at the periphery of the health care system. Intensive efforts would be required to convert the prototypes into highly reproducible POCT platforms with better performance and cost-effectiveness after scaling up. Many POCT platforms are PDMS based due to its quick prototyping and excellent properties. However, PDMS-based platforms may not be suitable for mass production. As these platforms move closer to their commercial versions, there will be emphasis on applying conventional mass fabrication processes and materials such as injection molding of thermoplastics. The development of a POCT platform that can be applied in resource-limited settings would require technology developers to closely interact with end users during the early stages of product development, especially in resource-limited settings. At present, the LF platform is the most widely accepted POCT platform for simple, mostly qualitative, low-cost diagnostics with capability to also be applied in highly sensitive, fully quantified, and multiplexed assays. The performance demands from diagnostics tests are ever increasing, and low cost alone might not be sufficient to achieve market penetration in developed countries. A careful evaluation of the POCT technology and its performance with respect to reliability of the collected data in the specific clinical context is important. It would be necessary to perform cost–benefit and cost–effectiveness analyses to justify the suitability of POCT platforms for a particular end user setting. The data management system associated with the POCT platforms is equally important. The vendors of POCT platforms and the end users have to invest in developing information technology systems networking by applying off-the-shelf data management and connectivity tools. To achieve a good level of connectivity, it would be required to implement modern communication standards and databases. The data collected on POCT platforms would not be very useful if they were not made available for clinical decision making by seamlessly integrating them with modern telecommunications. Both costs and data management issues would be critical driving forces for future POCT automation. Successful implementation and acceptance of POCT platforms in appropriate volumes at the right cost would require a systems approach with inspired vision and collaboration among end users, administrators, health care professionals, technology developers, and health insurance companies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
