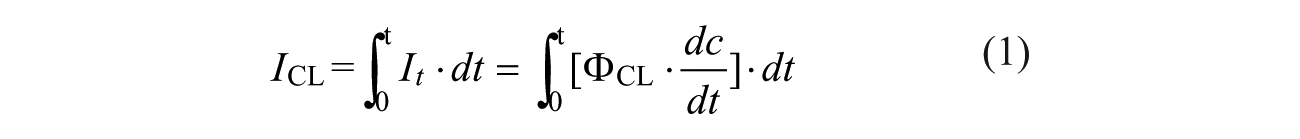Abstract
A fully automated microfluidic-based detection system for the rapid determination of insulin concentration through a chemiluminescence immunoassay has been developed. The microfluidic chip used in the system is a double-layered polydimethylsiloxane device embedded with interconnecting micropumps, microvalves, and a micromixer. At a high injection rate of the developing solution, the chemiluminescence signal can be excited and measured within a short period of time. The integral value of the chemiluminescence light signal is used to determine the insulin concentration of the samples, and the results indicate that the measurement is accurate in the range from 1.5 pM to 391 pM. The entire chemiluminescence assay can be completed in less than 10 min. The fully automated microfluidic-based insulin detection system provides a useful platform for rapid determination of insulin in clinical diagnostics for diabetes, which is expected to become increasingly important for future point-of-care applications.
Introduction
Diabetes is an epidemic metabolic disease, and its increasing prevalence affects all aspects of society. 1 Globally, an estimated 371 million people had diabetes in 2012, with type 2 diabetes making up about 90% of all cases. 2 Diabetes is a worldwide disease, but the largest increase in recent years takes place in Asia and Africa, where the majority of diabetic patients are expected to be found by 2030. 3 At present, diabetes diagnosis usually uses the blood glucose test, 4 but irregular blood glucose level, unfortunately, is not a good indicator for early diagnosis. Previous studies have shown that the levels of insulin, connecting peptide, and other immune indicators are better suited for the early diagnosis of diabetes. 5 Insulin, a peptide hormone produced by the beta cells of the pancreas, is central to regulating glucose and fat metabolism in the body. Any abnormal behavior in the insulin level is an effective auspice to diagnose diabetes at the early stage, which is critically important to diabetes prevention. 6
The current clinical methods for insulin detection typically require complex procedures, time-consuming assays, and skilled labor, which make them unsuitable for future demands when a large increase in the number of diabetic patients is expected. An improved insulin detection system must have the advantages of simple analytical platforms, inexpensive supplies, accurate results, rapid procedures, and convenient operation. 7 Microfluidic systems meet many of these requirements, including low reagent consumption, short analysis time, high reliability and sensitivity, and multiprocess integration.8,9 More importantly, microfluidic systems’ portability arising from their compact forms can be a key factor in point-of-care-based medical service in addition to offering a practical solution for rapid detection of insulin. Furthermore, magnetic microparticles, used in many established biomedical assays, can also be effectively applied in the microfluidic systems to enhance the enrichment process and increase the surface area for connecting antigen to antibody in a microfluidic environment.10,11
The chemiluminescence immunoassay combines chemiluminescence detection and a specific immunosorbent assay to become one of the most sensitive immunodetection systems in clinical diagnostics. 12 Chemiluminescence immunosensors have been used widely in environmental,13,14 medical, 15 food-processing, 16 and other related biological and biomedical areas. 17 For example, Tanaka and Matsunaga developed a sensitive chemiluminescence immunoassay for insulin, 18 in which the detection limit was shown to be 2 µU/mL and the linear correlation between the light signal and insulin concentration was apparent over the range of 19–254 µU/mL. However, their detection system was not equipped with a rapid detection platform, and the chemiluminescence immunoassay was therefore time consuming, requiring about 60 min to complete the detection process.
In this article, we present a fully automated insulin detection system based on an integrated microfluidic platform. The microfluidic chip in the platform, fabricated from polydimethylsiloxane (PDMS), is composed of multiple individually controlled pneumatic micropumps and microvalves, and one micromixer, all of which enable the automated chemiluminescence immunoassay and detection process. At a high injection rate of the developing solution, the chemiluminescence signal is excited and recorded by a chemiluminescent light detector over the entire light emission process. The integral value of the light signal is then used to determine the insulin concentration. The microfluidic-based insulin detection system has been demonstrated to have a low detection limit and high specificity, which makes it highly suitable for rapid determination of insulin in clinical diagnostics of diabetes and future point-of-care applications.
Materials and Methods
Materials and Procedure
Figure 1
shows an illustration of the biological protocol for insulin detection using a chemiluminescence immunoassay. Step 1 is taking advantage of the specific interaction between insulin antibodies (monoclonal mouse) and insulin antigens (recombinant human insulin standard); super-paramagnetic microparticles (~1.0 µm) coated with insulin antibodies (~30 µg/mL, 40 μL) are first used to capture the target insulin antigens in the sample (20 μL), as shown in
Figure 1A
Biological protocol for insulin detection using a chemiluminescence immunoassay: (
Microfluidic System
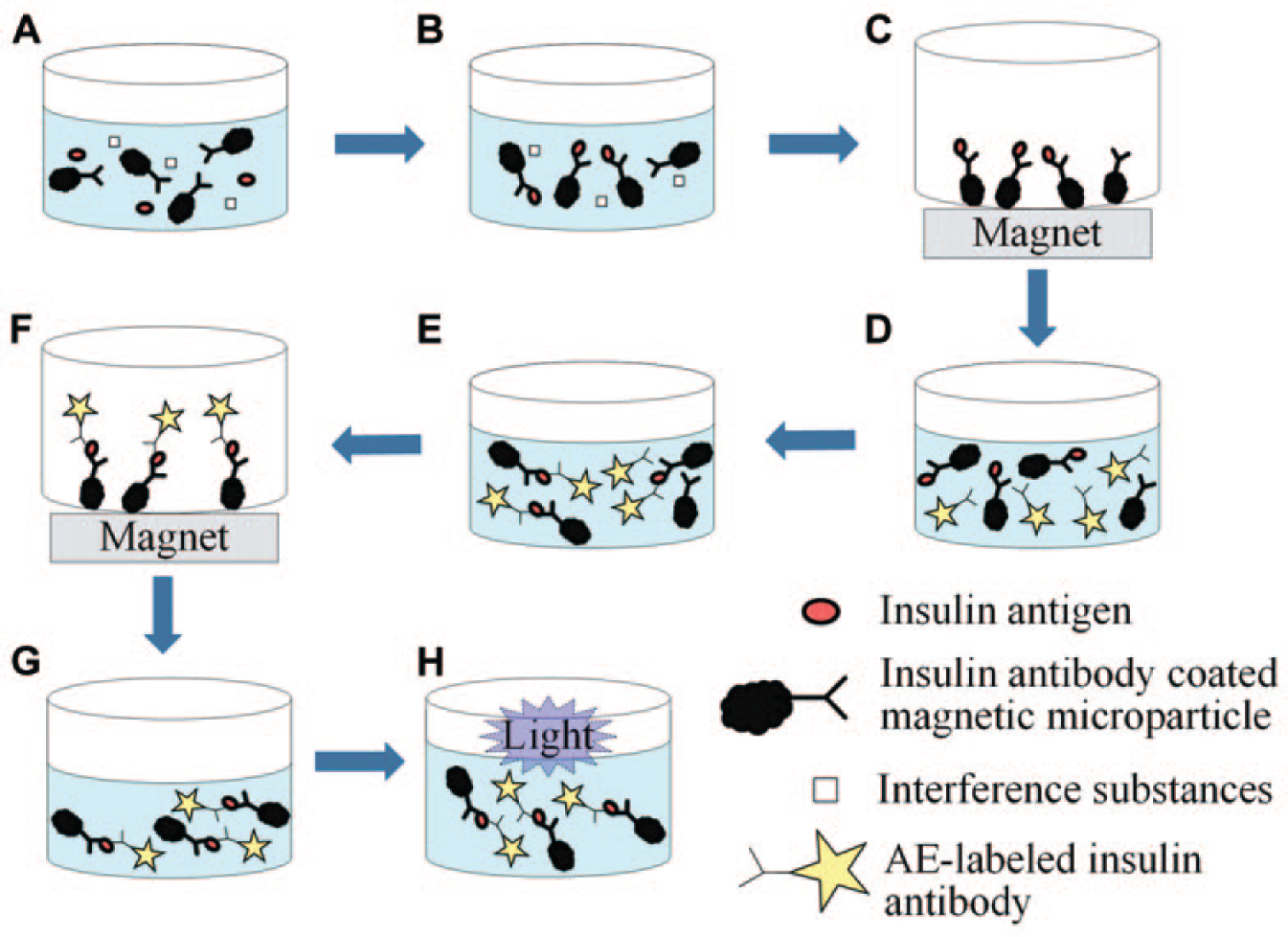
An enlarged picture of the microfluidic chip is shown in Figure 2 . The overall dimensions of the chip are 38 mm by 40 mm. It is fabricated from two PDMS layers and one glass layer using micromolding and oxygen plasma bonding. 19 The chip consists of five individually controlled pneumatic micropumps, five microvalves, and one micromixer. The insulin detection procedure begins with preloading the reagents and samples into their corresponding inlet chambers. By using the micromixer, the incubation between insulin antibodies and insulin antigens takes 3 min (Step 1 and Step 3). A wash buffer is flushed through the mixing chamber to remove the nonreactive substances and transport them to the waste chamber (Step 2 and Step 4). In the development process, developing solution 1 is mixed with the microparticles in the mixing chamber for 30 s (Step 5), and developing solution 2 is then injected into the mixing chamber, and chemiluminescence on the microparticles is emitted (Step 6). The entire insulin detection assay in the microfluidic system requires less than 10 min to complete.
An enlarged picture of the integrated microfluidic chip.
Automation System
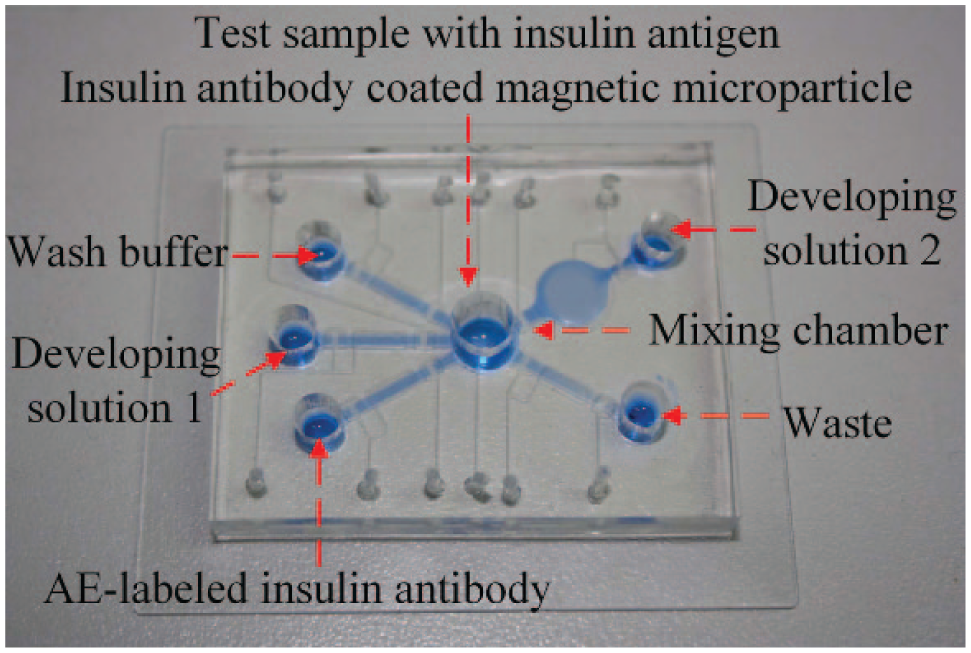
The insulin detection process is automatically performed by a single microfluidics chip with custom-designed control systems and optical detection components. Figure 3 is a schematic diagram of the fully automated insulin detection system. A personal computer controls the entire detection process and also records the detection result, using the software to set the parameters and read the signal from the photomultiplier tube (PMT; CR105-01, Hamamatsu, Hamamatsu City, Japan). The PMT is used to quantify the chemiluminescence signal, and it surpasses other sensitive photoelectric-detecting apparatus with the advantages of fast response, low cost, a large photocathode area, and high magnification.
Schematic diagram of a fully automated insulin detection system. A personal computer controls the detection process in the microfluidic chip and records the results of chemiluminescence from the photomultiplier tube.
The control system consists of a mini–air compressor (D730A-23-01, Hargraves Technology Mooresville, NC, USA), a pressure regulator (IR2000-02, SNS, Zhengjiang, China), a pulse generator (S3C2440, Samsung, Seoul, South Korea), 12 electromagnetic valves (EMVs; S070M-5BG-32, SMC Inc., Tokyo, Japan), an electromagnet (TAU-0730, Kendrion, Villingen-Schwenningen, Germany), and a stepper motor (N20, Arduino, Ivrea, Italy). Using an RS232 serial port for data communication with a computer, the control parameters can be set by the user, including driving frequency, duration time, electromagnet position, and the stepper motor motion. A series of pulse air pressure generated from the EMVs are connected to the microfluidic chip. Both air pressure and EMV driving frequency are important parameters that can influence the working performance of the micropump and micromixer. The characteristics of pneumatic micropumps have been researched in our previous works. 20
To automatically capture and separate the magnetic microparticles from the fluid, the electromagnet controls the vertical position of the permanent magnet, which is a programmable control and provides a stronger magnetic field than electromagnets only. During the incubation process (Step 1 and Step 3), the permanent magnet is in a lower position, and there is no magnet field in the mixing chamber. During the washing process (Step 2 and Step 4), the permanent magnet is pushed up by the electromagnet. The immunoassay is carried out with the microfluidic chip positioned above the permanent magnet, and then the chip is horizontally moved by a stepper motor to the optical detection zone to record the chemiluminescent light (Step 5 and Step 6).
To maximize detection sensitivity, the microfluidic chip is placed inside a custom-designed black box, and 12 Teflon tubes supply the pulse air pressure for the microfluidic chip. The signal output of the PMT is expressed in relative light units (RLUs). Figure 4 demonstrates the experimental setup. The computer screen in the figure shows a typical light intensity versus the time trace of an experimental run.

The experimental setup of the automated insulin detection system.
Results and Discussion
Flow Rate of Developing Solution
The flow rate used to inject developing solution 2 into the microfluidic chip plays an important role in determining the sensitivity of the insulin assay. Figure 5 demonstrates the effect of the speed of the injection flow rate on the chemiluminescence intensity emitted from the mixing chamber for an insulin concentration of 522 pM. At a low flow rate of ~40 µL/min, the peak and integral value of the chemiluminescence signal are much smaller than those at the high flow rate of 600 µL/min. As can be observed in Figure 5 , at a low flow rate, the chemiluminescence signal declines slowly after reaching the peak. Conversely, at a high flow rate, the chemiluminescence signal rises quickly to a much larger peak value and then falls back to the baseline value after a short delay. This result proves that a more efficient chemiluminescence process takes place at a high injection flow rate of developing solution 2.
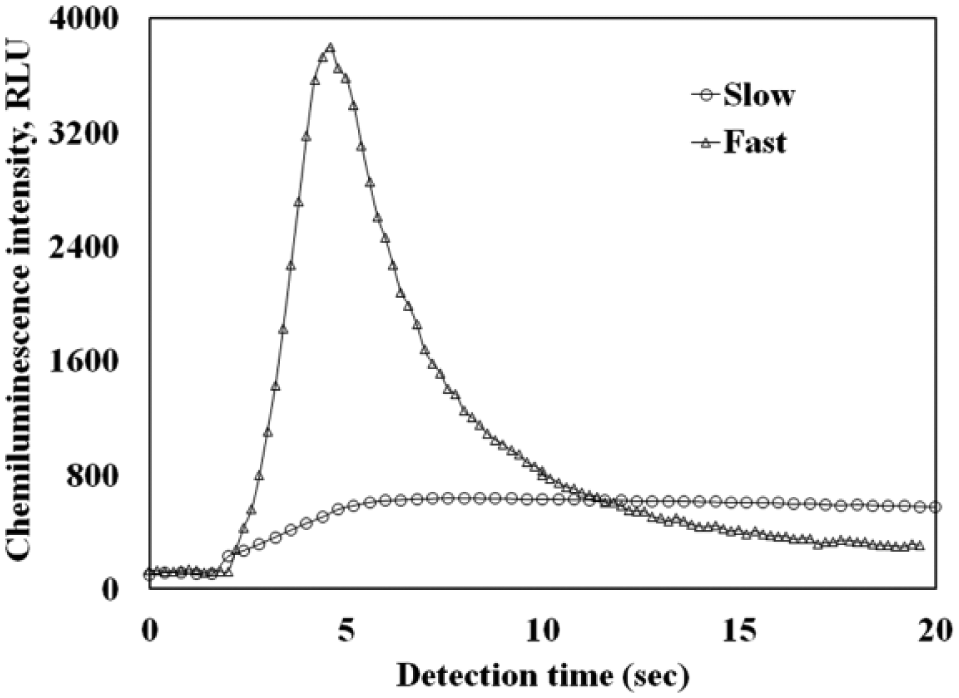
Comparison of the chemiluminescence signals obtained at two different flow rates of the developing solution. Both chemiluminescence curves are for the same insulin concentrations of 522 pM.
However, in a microfluidic chip, an increase in the flow rate will also increase the static pressure within the microchannel and the likelihood of leakage. For the present study, an optimized injection rate for developing solution 2 is determined to be 600 µL/min, which is high enough for rapid insulin detection without causing any leakage. In all subsequent discussions, the results were obtained using the 600 µL/min injection flow rate.
Chemiluminescence Intensity
Two representative chemiluminescence curves for insulin concentrations of 522 pM and 784 pM are shown in Figure 6 . During the chemiluminescence reaction in the micromixer, the chemiluminescence curves show a consistent trend: The light intensity reaches its peak value rapidly, and then declines back to the baseline within 20 s. The level of the peak value is directly proportional to the level of the insulin concentration in the sample.
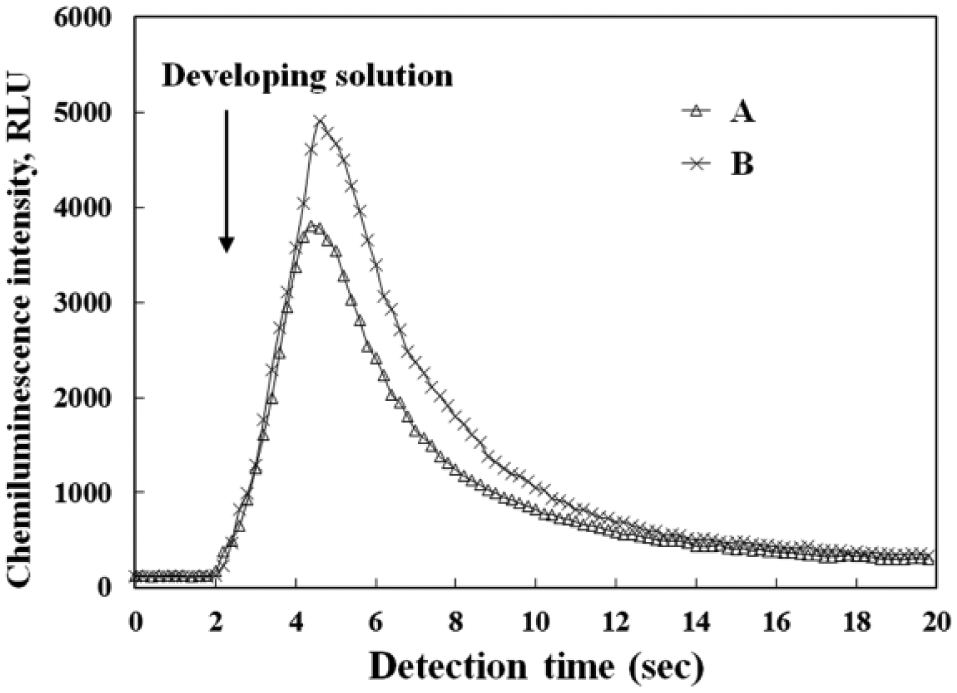
Two representative chemiluminescence traces. Traces A and B are for insulin concentrations of 522 pM and 784 pM, respectively.
The chemiluminescence intensity of a chemical reaction is related to the reaction rate through the following equation:
where ICL is the chemiluminescence intensity; It is the chemiluminescence intensity at time t; ΦCL is the quantum yield; and dc/dt is the reaction rate. 21 In our previous studies, the peak value of the chemiluminescence signal was chosen to represent the insulin concentration because higher concentrations result in higher peak values. 22 However, because the chemiluminescence process consumes the AE immobilized on the microparticles during the entire light-emitting sequence, the integral value, instead of the peak, of the light intensity trace should be more representative of the level of the sample insulin.
Sensitivity and Detection Limit
Figure 7 shows the relationship between the normalized integral value of the chemiluminescence intensity and the corresponding insulin concentration. The concentrations of the reagents and developing solutions for the measurement are optimized for the microfluidics chip. Reagents are transported at a pumping rate of 300 µL/min. The mixing chamber is activated at a driving frequency of 5 Hz and an applied pressure of 10 psi. After the incubation process, wash buffer is flushed at a pumping rate of 380 µL/min.
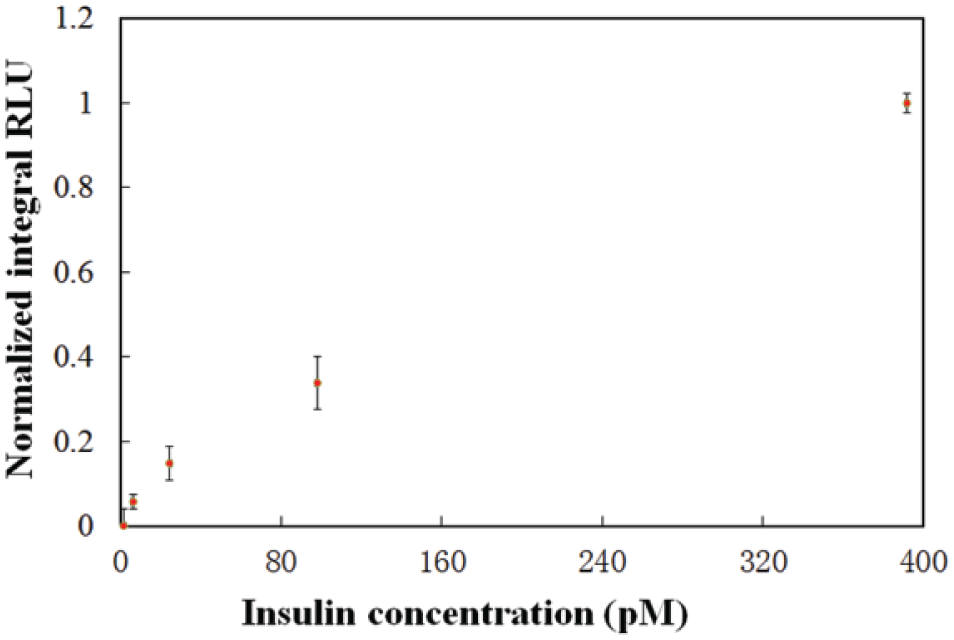
Relationship between the normalized integral value of the chemiluminescence intensity and the corresponding insulin concentration.
Based on the result shown in Figure 7 , the detection limit of the automated insulin detection system using microfluidic chips is determined to be 6.1 pM at a CV (coefficient of variance) of 6.1% and a signal-to-noise ratio of 3. A relatively linear relationship is found between the insulin concentration and the integral value of the chemiluminescence intensity in the insulin concentration. The equation of the calibration curve is y = 0.0025x + 0.0522 and R2 = 0.9899. The results indicate that the average accuracy of the microfluidic system is 14.6% within a range of insulin concentrations from 1.5 pM to 391 pM. The measured insulin concentrations are found to be reasonably repeatable because CVs are smaller than 8.2%.
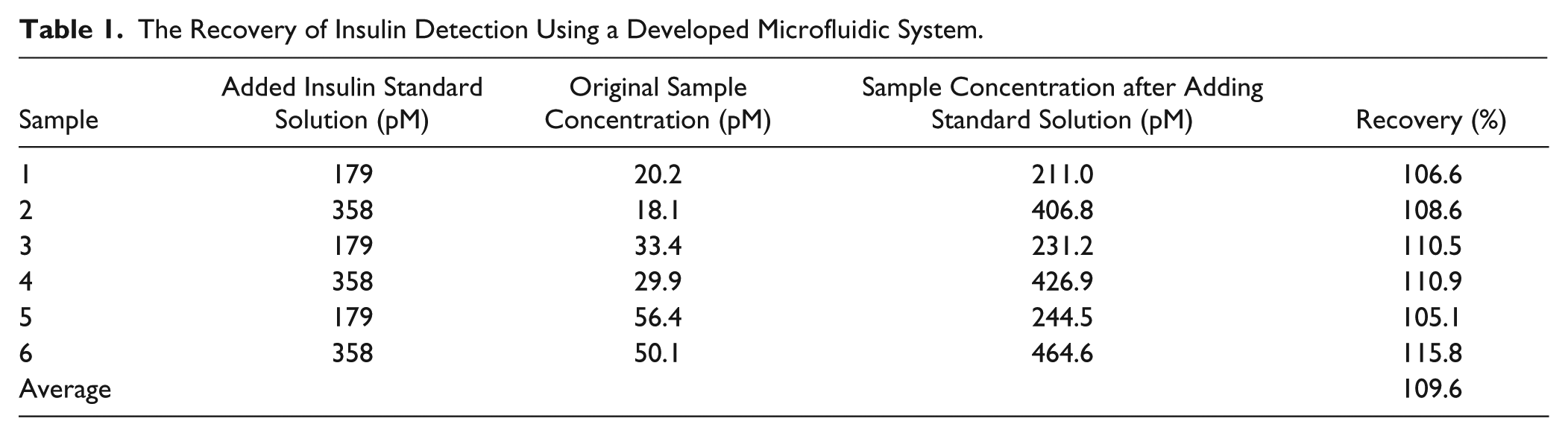
Five samples were prepared. The chemiluminescent signals at different insulin concentrations (1.5, 6.1, 24.5, 97.9, and 391 pM, respectively) have been detected by using the microfluidic system integrated with the optical detector. Each sample was measured three times. The recovery of insulin detection using a developed microfluidic system is shown in Table 1 . By adding insulin standard solution with the known concentrations of 179 pM and 358 pM into six samples, the recovery tests were performed by comparing the detection results at three insulin concentration levels (low, medium, and high). The original insulin concentration and the insulin concentration after adding standard solution of each sample were measured by the microfluidic system, and the added insulin concentration can be calculated. By comparing the measurement result of the added insulin concentration to the known value, the recovery of each sample can be determined. The average recovery of the insulin detection is 109.6%.
The Recovery of Insulin Detection Using a Developed Microfluidic System.
Insulin detection via a microfluidic system is compared with the results obtained by using a commercial immunoassay (ADVIA Centaur XP Immunoassay System, Siemens, Berlin, Germany). Clinical samples (leftover serum samples from diabetic patients at the Chinese Medical University Hospital, Shenyang, China) were used in the present study. Four serum samples at different insulin concentration levels were tested. Insulin concentrations of the same sample are measured by the developed system and conventional immunoassay separately, and the correlation between the developed microfluidic system and the conventional immunoassay is 0.978. The insulin detection sensitivity of the developed microfluidic system and the conventional immunoassay is shown in Table 2 . The insulin detection limit of the conventional immunoassay is 3.5 pM, which is similar to that of the microfluidic system.
Insulin Detection Sensitivity of a Developed Microfluidic System and a Commercial Immunoassay.

CVs, coefficients of variance.
Conclusions
This article presents a microfluidics-based automated detection system for insulin concentration using magnetic microparticles and chemiluminescence immunoassays. The microfluidics chip of the detection system is fabricated from PDMS with five individually controlled pneumatic micropumps, five microvalves, one micromixer, four inlets, and one outlet. The microfluidics chip automates the entire insulin detection process with the assistance of custom-designed control systems and optical detectors for chemiluminescence measurement. The integral value of the chemiluminescence signal is used to quantify the insulin concentration. The insulin detection time of the automated system is less than 10 min. The system has a low detection limit of 6.1 pM and a detection range from 1.5 to 391 pM. The microfluidics-based insulin detection system is expected to provide a useful platform for rapid insulin detection in clinical diagnostics of diabetes.
Footnotes
Acknowledgements
The authors thank the CAS/SAFEA International Partnership Program for Creative Research Teams.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant Nos. 61327014 and 61304251).