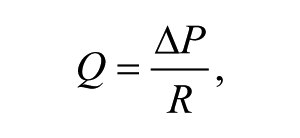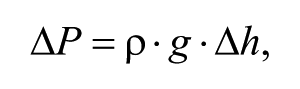Abstract
In this article, we present a microfluidic platform, compatible with conventional 96-well formats, that enables facile and parallelized culturing and testing of spherical microtissues in a standard incubator. The platform can accommodate multiple microtissues (up to 66) of different cell types, formed externally by using the hanging-drop method, and enables microtissue interconnection through microfluidic channels for continuous media perfusion or dosage of substances. The platform contains 11 separate channels, and each channel has six tissue compartments. Primary rat liver tissues were cultured over 8 days, and multiple tumor tissues (HCT116) were exposed to various concentrations of 5-fluorouracil for platform characterization.
Introduction
Multicellular spheroids have emerged as a promising developmental 3D tissue model system in response to the observed limitations of 2D culture systems in mimicking in vivo–like environments.1,2 Spherical microtissues (MTs) are scaffold-free cell clusters that have proven to feature tissue-like phenotype and functionality.3,4 They can be reliably produced in well-controlled sizes at high throughput in an automated way.5–7
Owing to their compact constitution that makes them easy to handle with conventional cell culture equipment, microtissues have become a more and more popular choice for use with microfluidic culturing setups. Microfluidic systems allow for precisely controlled perfusion and can be engineered in a way that microtissue cultures experience realistic physiological conditions with regard to liquid-to-cell ratios, fluid residence times, and the respective dynamic mechanical environment. 8 Furthermore, miniaturization allows for a substantial reduction of required reagents and enables precise liquid volume dosage of drug compounds and offers various sampling options for media and tissue analysis.
Recently, more complex microfluidic systems have been designed, in which even different organ models can be hosted and can be fluidically interconnected. The goal is to develop multiorgan models, or so-called body-on-a-chip experimental setups,9–13 which potentially can provide a more systemic view on the effects of drug compounds in the human organism.
Also for multiorgan models, spheroids are highly interesting, because they can be formed from different cell types while using the same fabrication method—the hanging-drop technique. This advantage can substantially reduce system complexity and improve reliability, a prerequisite for reproducible experimentation.
Spheroids have been formed and characterized inside microfluidic systems14–17 or transferred to those after external MT formation. 18 These experiments reveal that perfusion can have an important impact, which has to be taken into account when designing 3D cell-based assays.
The currently available culturing platforms are all relatively difficult to operate and rely on rather complex fabrication processes. Typically, they are not designed to host multiple spheroids of different cell types while providing a robust fluidic interconnection for continuous nutrient supply and compound exposure experiments. Finally, the microfluidic setups are mostly very specific and not compatible with SLAS standards and commonly used tissue culture incubators. This compatibility, however, is critical for commercialization and automation of the developed microfluidic system with existing techniques. There have been efforts in developing 96-well format-based cell culturing or drug testing systems that can be operated through pipetting.19–21
In this article, we present a microfluidic platform, which is compatible with 96-well automation tools and allows for parallel culturing of spherical MTs of different cell types. Its basic concept relies on our previous work 22 and includes the reliable formation of spheroids outside the platform and their subsequent direct transfer into the microfluidic platform. Gravitational forces are used for MT positioning in the microfluidic channel as well as for subsequent flow control. The new system combines this concept with a substantial increase in throughput. Together with the compatibility with standard SLAS formats, it significantly improves usability and makes handling simple and robust enough to be used by other laboratories without microtechnology experience. The device was fabricated by casting poly(dimethylsiloxane) (PDMS) between two molds aligned to each other, which is similar to an injection molding process. Therefore, the device design can be translated to an injection molding process with minor modifications and due regard to process-specific details. Fabrication through injection molding would enable routine use of our platform in an automated fashion. At the same time, the format is still complex enough to interconnect different microtissue types. With the presented system, over 100 different multitissue experimental conditions can be conducted in parallel in a conventional incubator at moderate operation complexity. Without tubing and active pumps, liquid handling is kept as simple as possible and enables robust continuous nutrient supply and various compound exposure protocols.
Materials and Methods
Layout of the Microfluidic Platform
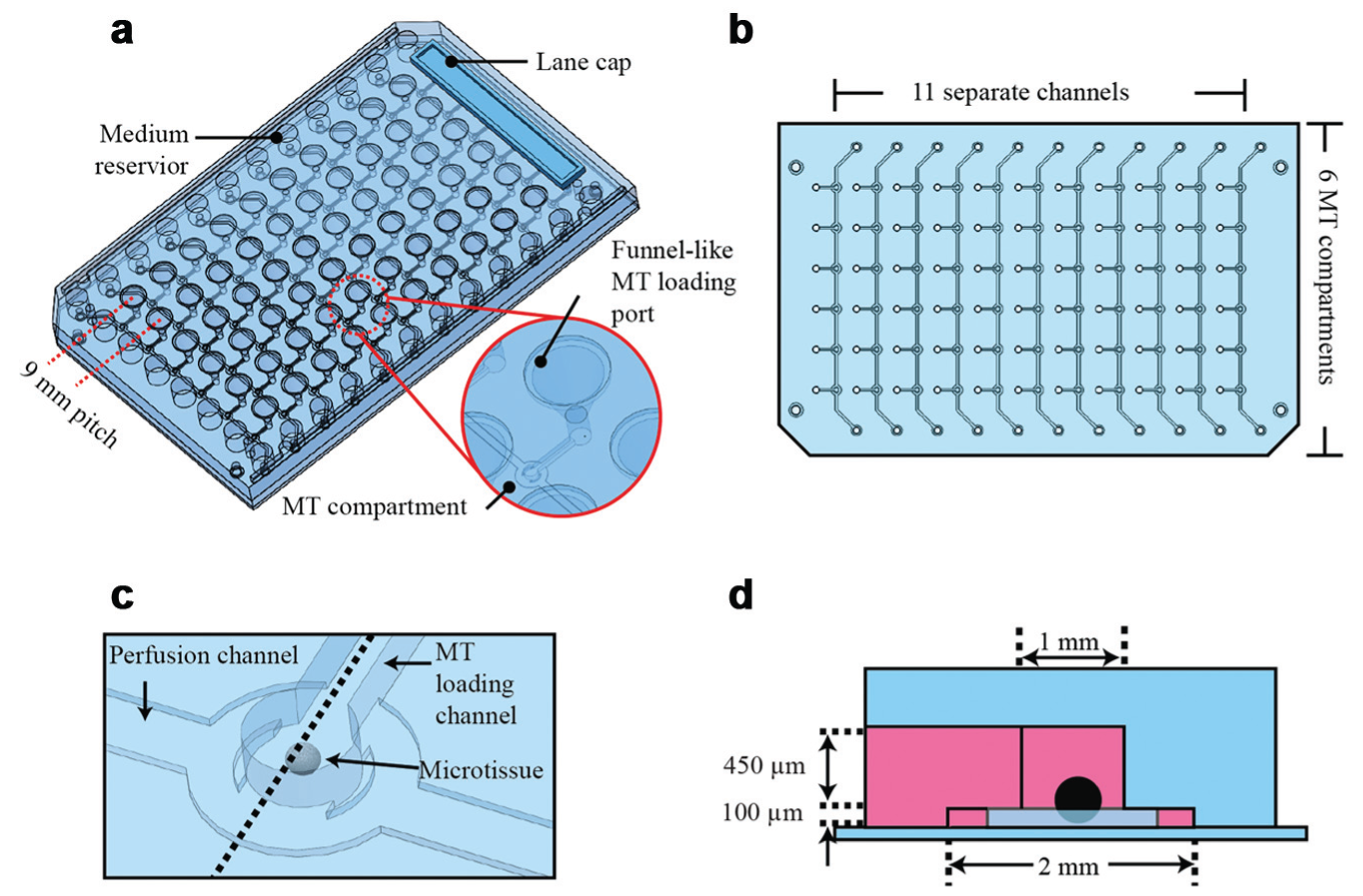
The microfluidic platform is presented in
Figure 1
. It comprises 11 separate straight perfusion channels or lanes, and each of these lanes features six serially aligned MT compartments. Open medium reservoirs are located at both ends of each channel, which can host up to 150 µL medium. The MTs are introduced into the platform through funnel-like loading ports. Both medium reservoirs and loading ports are arranged at a 9-mm pitch and match the positions of conventional 96-well plates, which enables easy loading of multiple MTs and convenient media exchange using typical multichannel pipettes and conventional 96-well equipment (see “Gravity-Controlled MT Loading and Perfusion for Cultivation” for more details). All MT compartments are shifted between two loading ports of neighboring channels. This arrangement enables the gravity-based reliable loading and good optical accessibility for transmitted light microscopy. The distance of the MT compartments to the loading ports is 4.5 mm and therefore matches the positions of the 384-well format for standard readout and analysis equipment. Specifically designed lane caps are used to close the MT loading ports. Up to 66 MTs can be accommodated in a single platform. For parallelized experiments, it is possible to simultaneously culture six identical or different MTs under 11 identical or different conditions (
Figure 1b
). In other platform versions, we arranged seven channels with eight compartments or 14 channels with four compartments on the platform (
Layout of the microfluidic platform. (
The MT compartments are located at the intersection of the loading channels and the perfusion channel ( Fig. 1c ). Figure 1d shows a cross section through the MT compartment. Its diameter is 1 mm, and the height is 550 µm. The loading channel has a length of 4.5 mm, a height of 450 µm, and a width of 500 µm. The perfusion channel has a length of 64 mm, a width of 600 µm, and a height of only 100 µm.
Platform Fabrication
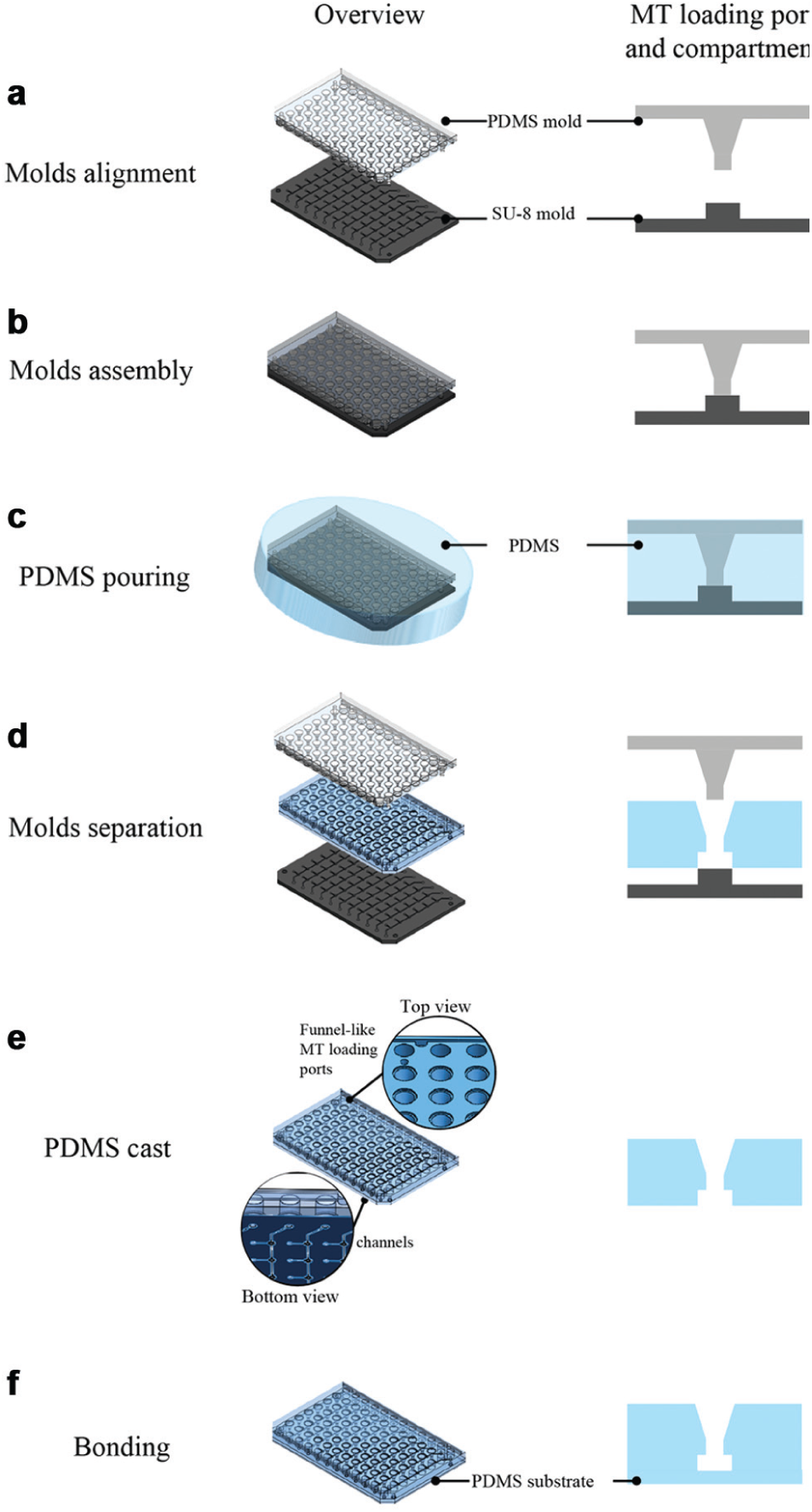
The platform was fabricated using a conventional soft lithography process ( Fig. 2 ). The first part was an SU-8 mold that was fabricated on a 6-inch (100) silicon wafer by using standard photolithography. Then, 100-µm-thick SU-8 100 photoresist (Microchem Corp., Newton, MA) was spin-coated onto the wafer and exposed to UV through a transparency mask to define the perfusion channels. Next, a 450-µm-thick SU-8 100 layer was coated on top. A second transparency mask was used to UV-pattern the MT compartments and loading channel structures. The second part was a PDMS (Dow Corning, Midland, MI) mold, used to define the MT loading ports and medium reservoirs. The PDMS mold was casted from a plastic mold fabricated in a 3D printing process. The SU-8 mold and PDMS mold were coated with an antiadhesive layer of trichloro(1H, 1H, 2H, 2H-perfluoroctyl)silane (product no. 448931; Sigma-Aldrich Chemie GmbH, Buchs, Switzerland) in a vapor silanization procedure. All molds can be used several times to produce many identical devices.
Fabrication procedure of the 96-well format-based platform. (
The platform body was then produced in a PDMS casting process using the two fabricated molds aligned and pressed onto each other in a sandwich-like arrangement ( Fig. 2a , b ). The applied pressure and the soft nature of the PDMS material ensured a tight contact between the two molds so that no PDMS could flow into regions, where, for example, the loading ports were in contact with the loading channels. The two PDMS components, monomers and a curing agent, were mixed at a ratio of 10:1 and poured in the space between the molds ( Fig. 2c ). After curing at 65 °C for 4 h, the molds were separated from the cast ( Fig. 2d , e ). The obtained PDMS cast was bonded to a 1-mm-thick PDMS substrate after O2-plasma treatment ( Fig. 2f ; Diener Electronic GmbH & Co., Ebhausen, Germany) and placed on a rectangular cell culture dish (Nunc OmniTray; Milian SA, Geneva, Switzerland). Finally, the PDMS channel surface was coated with Biolipidure 502 (2-methacryloyloxyethyl phosphorylcholine [MPC] polymers; NOF Corporation, Tokyo, Japan) to reduce compound adsorption and cell adhesion. 23
Microtissue Fabrication
All microtissues were fabricated off-chip by using the hanging-drop method. Then, 40 µL of the cell suspension with defined cell concentration was applied to each well of the GravityPLUS plates (InSphero AG, Schlieren, Switzerland). After spheroids were formed in a standard incubator, they were transferred to GravityTRAP plates (InSphero AG). The MTs were then incubated again in the plate for 1 more day before they were loaded into the microfluidic devices.
Rat liver microtissues (rLiMTs) were obtained from InSphero AG and delivered in rat liver maintenance medium. Their initial diameter was 250 µm. Tumor MTs were formed by seeding 250 HCT116 eGFP cells (human colon carcinoma cell lines; Sirion Biotech, Martinsried, Germany) into each hanging drop, resulting in MTs with approximately 350 µm diameter at day 6 after seeding. RPMI 1640 (product no. E15-885; Chemie Brunschwig AG, Basel, Switzerland) containing 10% (v/v) of fetal bovine serum (FBS; product no. F2442; Sigma-Aldrich), 100 µg/mL penicillin (product no. P11-010; Sigma-Aldrich), 10 µg/mL streptomycin (product no. P11-010; Sigma-Aldrich), and 0.3 µg/mL puromycin (product no. P8833; Sigma-Aldrich) was used for HCT116 MTs.
Gravity-Controlled MT Loading and Perfusion for Cultivation
Three important factors ensure simple loading and cultivation of the MTs in the platform. First, the MT loading ports have a 9-mm pitch so that multiple MTs can be easily and rapidly transferred into the microfluidic network using a conventional multichannel pipette. Second, the funnel-like MT loading port guides the pipette tips so that the MTs reliably fall into the respective loading channels. Third, MT positioning in the compartment and subsequent media perfusion are both driven by gravity and actuated through simple tilting of the platform. No tubing connections or additional pumping systems are required during the experiments. Therefore, facile and automated operation of the device is possible, also for nonexpert users.
Before usage, the platform was first degassed for approximately 1 h and sterilized under UV for at least 30 min. Then, reservoirs and channels were filled with culture medium. Bubbles trapped during liquid filling were taken up by the previously degassed PDMS. Six MTs were picked up into 200-µL pipette tips from the GravityTRAP plate in parallel with 10 or 20 µL of media; the six MTs were then gently released into the loading ports of one lane ( Fig. 3a ). The whole platform was then tilted, to simultaneously transfer all MTs into the compartments under the influence of gravity ( Fig. 3c ). At the same time, excess medium flew into the lateral medium reservoirs located slightly lower than the loading ports. Capillary forces retained the liquid, which prevented that the loading ports completely drained. This procedure was repeated for all lanes. Types and numbers of tissues can be easily varied with respect to experimental setups and requirements. The loading ports were then closed with lane caps. Spillover, which may cause cross-contamination between the different lanes, was not observed due to the low medium levels in the loading ports.
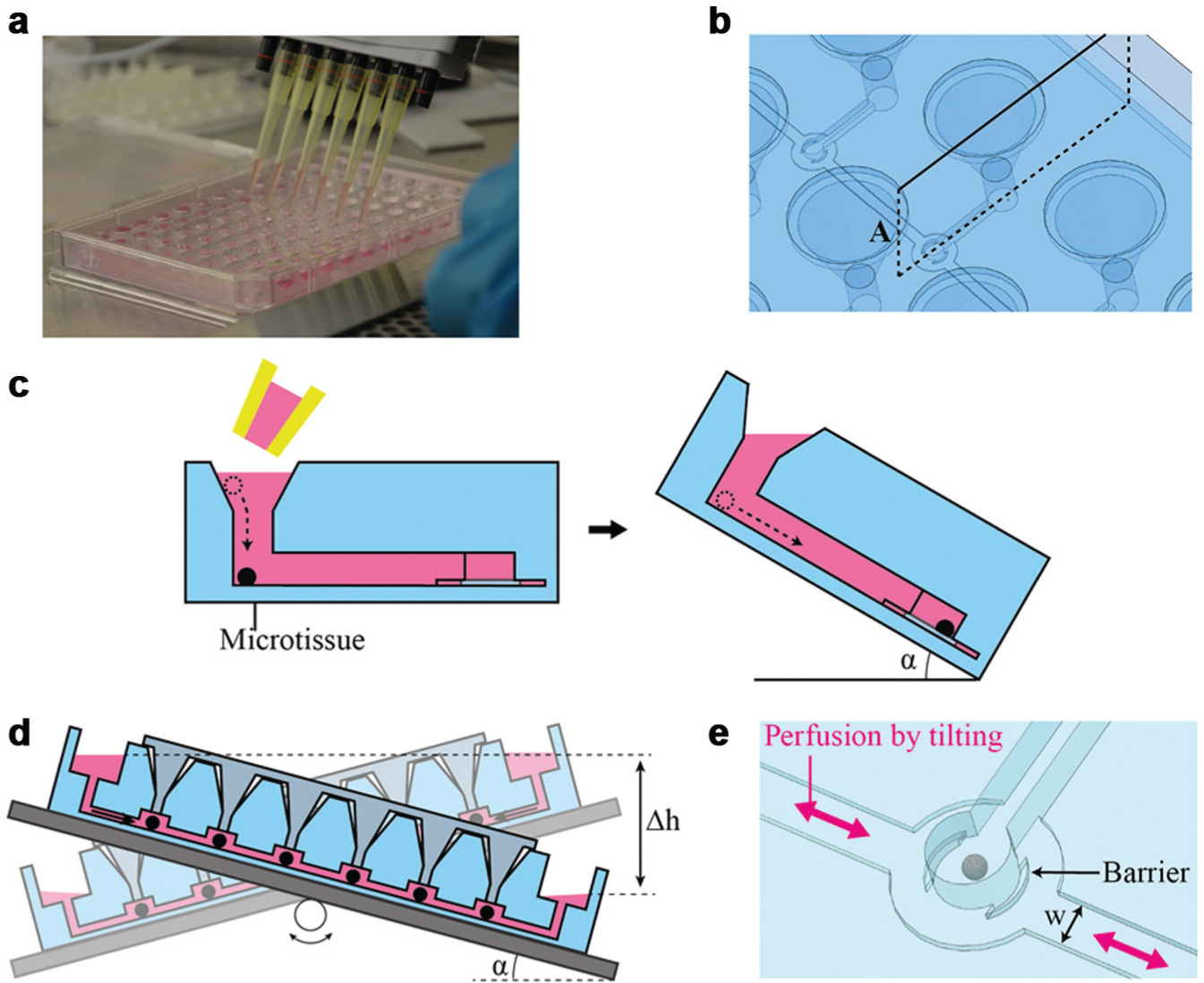
Tilting schemes for loading microtissues (MTs) and media perfusion. (
The loaded platform was afterward placed on an automated tilting plate, of which the tilting frequency, tilting angle, and tilting speed can be programmed. The whole setup was operated in a 5% CO2, 37 °C humidified incubator. When the platform is tilted by a specific angle, the two open reservoirs are located at different heights, which results in flow of medium from the higher reservoir through the 100-µm-high perfusion channel to the lower reservoir ( Fig. 3d ). In this way, media are shuttled back and forth, imitating circular flow and continuous fluidic intertissue communication. The low channel height of 100 µm prevents MTs from leaving the respective compartment during media perfusion ( Fig. 3e ). Moreover, integrated side barriers in the compartment exclude direct flow onto the tissue to minimize shear forces.
The gravity-driven flow rate (Q) can be theoretically estimated by
where ΔP in this case is the hydrostatic pressure difference and R is the flow resistance of the rectangular microchannel. ΔP is given by
where ρ is the fluid density, g is the standard gravity, and Δh is the height difference of the media in the two reservoirs (
Fig. 3d
). The gravity-controlled perfusion system does not require any tubing to be connected. The open reservoir concept offers several benefits, such as bubble-free perfusion, easy sampling, and media exchange. Furthermore, it enables highly parallelized experiments because multiple platforms can be operated in parallel by simply stacking several of them on the tilting plate (see
For cultivation of rLiMTs, 120 µL of rat liver maintenance medium (InSphero AG) was introduced in each channel, and two different numbers of MTs (three and six MTs) were loaded into the channels to investigate the effects of different media volumes per MT, which included 20 µL/MT and 40 µL/MT, respectively. The volume of the compartment is approximately 0.43 µL, which is about 300 times smaller than the total volume of perfused media (120 µL) but 20 to 50 times larger than the cell volume of rLiMTs (approximately 0.008 µL) and HCT116 MTs (approximately 0.022 µL). Sufficient media exchange and nutrition supply are therefore ensured. The whole medium volume in the reservoir (the channels were not emptied) was collected for albumin quantification, and media were exchanged every 2 days during 8 days of culturing.
On-Chip Drug Testing
For parallelized on-chip drug exposure experiments, 5-fluorouracil (5-FU; product no. F6627; Sigma-Aldrich) was chosen as drug compound, and its effect on HCT116 MTs was tested. Five different concentrations (0, 1, 3.16, 10, 31.6, and 100 µM) were prepared with 0.1% DMSO in RPMI 1640 media from a stock solution (100 mM 5-FU in DMSO; product no. D8418; Sigma-Aldrich). HCT116 MTs were exposed to 5-FU under continuous perfusion condition in the platform for 3 days. Five replications for each concentration were run in parallel.
Biochemical Assays
The amount of albumin secreted by the rLiMTs was measured by analyzing the collected supernatants with an enzyme-linked immunosorbent assay (ELISA) (product no. E110-125; LuBioScience GmbH, Luzern, Switzerland). Albumin was chosen because it is a protein synthesized by the liver and a good biomarker for the metabolic activity of liver. At the end of the experiments, MTs were collected, and a luminescence-based adenosine triphosphate (ATP) assay (product no. G7570; Promega AG, Dübendorf, Switzerland) was used to determine cell viability. The unloading procedure comprised tilting the platform so that the MTs could be transferred back through the loading channel to the loading ports, removal of the lane caps, and subsequent withdrawal of the MTs by using a conventional pipette.
Imaging
The size of MTs was measured optically every 2 days using an inverted microscope (Axiovert 25; Carl Zeiss AG, Jena, Germany) with a 10× objective in bright-field mode.
For the determination of viability, a dead staining was performed using propidium iodide (PI; ex/em 545 nm/610 nm; product no. P3566; LuBioScience GmbH, Luzern, Switzerland) directly inside the platform on the last day of the experiment. Then, 10 µL of PI stock solution (1 mg/mL in water) was added to 1 mL of cell culture medium. For staining of the MTs, 120 µL of the medium containing the dyes was added to the one of the reservoirs after removal of the old media. The dyes were introduced to the MTs through the perfusion channel by repeated tilting and incubated for 30 min inside the incubator. The platform was transported to an inverted fluorescence microscope (Leica DMI6000; Leica, Heerbrugg, Switzerland) with a 10× objective for fluorescence imaging. Staining and imaging are, therefore, straightforward and can be done without unloading the MTs from the platform.
Results and Discussion
Flow Rate Characterization
Platform tilting induces a flow between the two reservoirs. The flow rate is determined by two parameters, the height difference of the reservoirs, which generates a hydrostatic pressure, and the hydrodynamic resistance of the perfusion channel. The flow rates in the presented platform were examined for three different tilting angles: 10°, 20°, and 30°. While 150 µL of the media was flushed from the upper to the lower reservoir, the transferred volume of the flowed media was measured with time until complete drainage. In Figure 4 , the obtained flow rates as a function of time are displayed and compared with model calculations. The calculations show a linear decrease over time as a result of the change of the liquid levels in both reservoirs. The experimental results show higher flow rates at the beginning and lower flow rates toward the end. This effect gets more pronounced for increasing tilting angles and may be caused by capillary effects in the reservoir bottom, which is almost empty. Variations of the flow rates between different channels in the chip are quite high using 30° tilting, but of reasonable reproducibility at tilting angles below 20°. Furthermore, the estimated values are in good agreement with our measurements for angles lower than 20°. All following studies were therefore carried out with a tilting angle of 10° and a tilting interval of 9 min. The low flow rate in the range of 10 µL/min exerts low shear forces, and sufficient volume of medium (approximately 100 µL) is exchanged during one cycle. Moving the overall medium volume through tilting is not recommended, because in some cases, the reservoirs are completely emptied, which introduces bubbles at the media inlets, which then will disturb media perfusion over extended times.
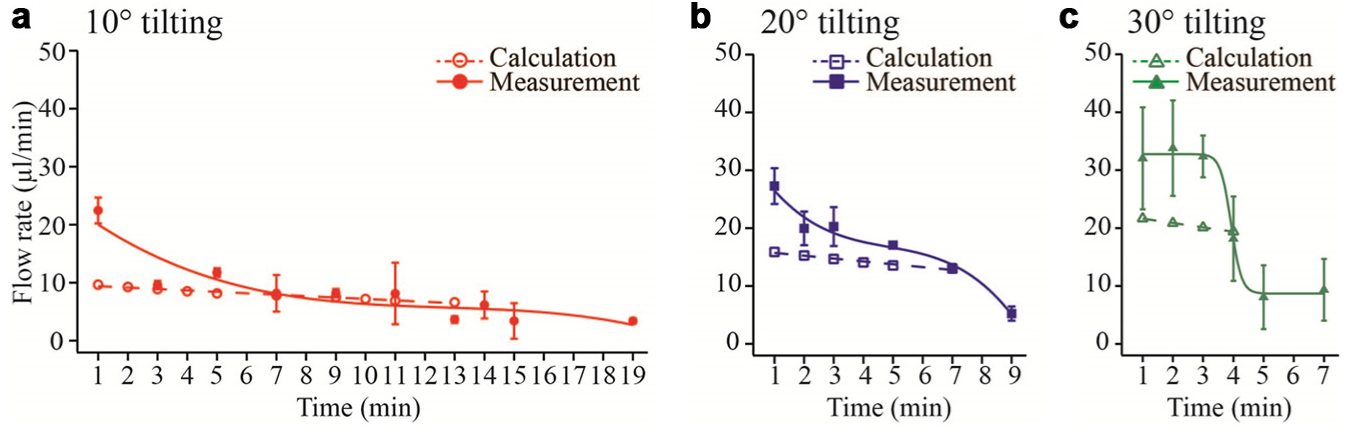
Flow rates as a function of time until complete reservoir drainage measured and calculated for platform tilting angles of (
Gravity-driven perfusion between two reservoirs has the advantage that is compatible with any other material the platform will be fabricated of in the future. Furthermore, open reservoirs will provide sufficient gas exchange. Regarding antiadhesion and antiadsorption coatings, care has to be taken when replacing tested and established materials in an injection molding process for mass production.
rLiMT Loading and Cultivation in the Platform
Parallel microtissue loading revealed to be very robust using the cultivation platform. Figure 5 illustrates one example, in which 36 rLiMTs were loaded into the compartments of the platform to assess microtissue viability over 8 days under gravity-controlled perfusion conditions. Six MTs were loaded in parallel into four of the first five channels and three MTs in the next four channels by using a conventional multichannel pipette. It is common to transfer MTs cultured in a conventional well plate by using pipetting. Significant damage of MTs was not observed during the MT loading by pipetting or during tilting of the device. This arrangement was used to investigate the effect of available medium volume per microtissue on the behavior of the MTs. With a total medium volume of 120 µL, we thus obtained 20 µL/MT and 40 µL/MT.
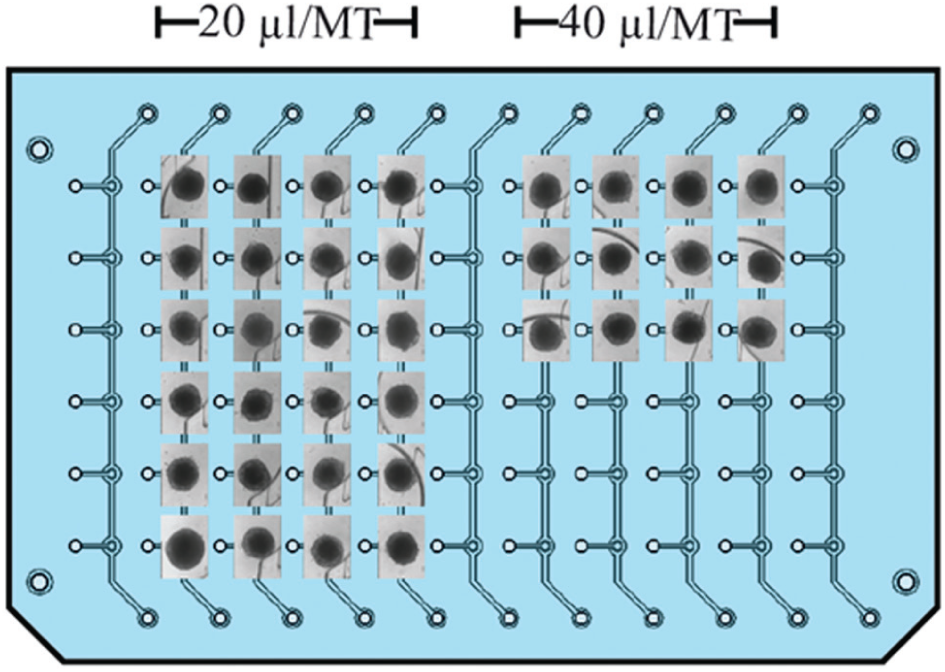
Bright-field images of rat liver microtissues (rLiMTs) loaded with a multichannel pipette and cultured in two different medium volumes per microtissue (MT), 20 µL/MT and 40 µL/MT.
In addition, rLiMTs were also cultured under static conditions in parallel to have a comparison to perfusion conditions. Figure 6a shows micrographs of rLiMTs cultured under static conditions with 40 µL/MT and perfusion conditions with 20 µL/MT and 40 µL/MT of available medium. Under all conditions, MTs preserved their morphology and their diameter of approximately 260 µm (variation of less than 10%) over 8 days due to low or absent proliferation of primary liver cells. It is important to monitor MT consistence as they shrink and disintegrate in case their viability decreases. No cell adhesion was observed in the PDMS compartments, which is an indication of the stability of the antiadhesive surface coating.
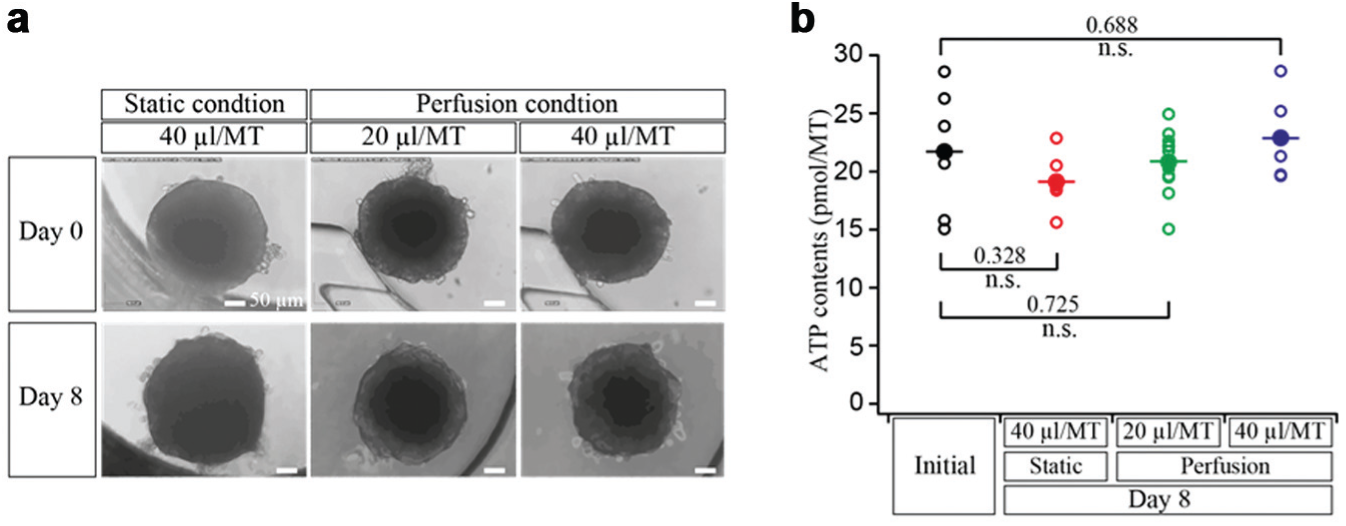
(
Figure 6b shows the viability of all rLiMTs indicated by the ATP content at day 8 for all three conditions compared with the initial values measured at day 0 (6 days after cell seeding). rLiMTs showed comparable ATP contents after 8 days of culturing under all conditions. Importantly, no significant decrease was observed with respect to the initial conditions.
In parallel, albumin secretion was measured to monitor the metabolic activity of the rLiMTs. Figure 7 shows relative albumin secretion measured every 2 days from medium samples. Under static conditions, we observed a slight decrease in albumin concentration over time, while it increased during the 8 days of culture under perfusion conditions. It reflects that the liver tissues were fully functional in the device, since albumin is produced only in liver and constitutes a specific biomarker to assess metabolic activity of hepatocytes in liver tissue. This increase was seen with media volume per microtissue ratios. These results indicate that perfusion conditions significantly increased secretion of the functional marker protein albumin compared with static conditions, a finding that has to be accounted for in biological and pharmaceutical research with MTs.
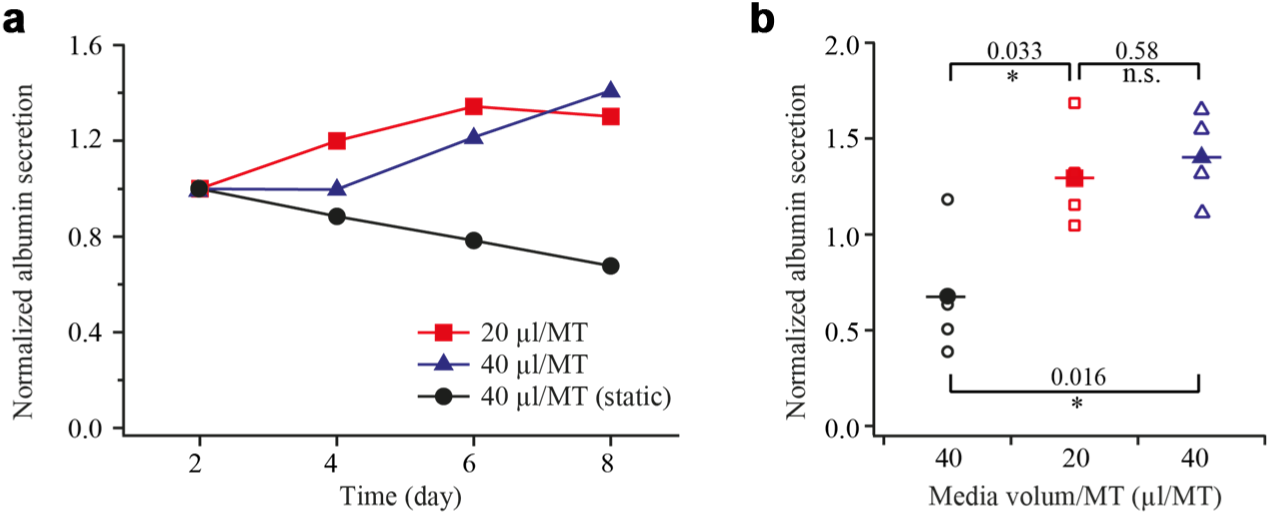
Metabolic activity monitoring of rat liver microtissues (rLiMTs). (
Compound adsorption and absorption are severe and well-known problem in PDMS devices. 24 Albumin levels from the surface-coated PDMS channels were comparable to those obtained by using a GravityTRAP plate. Absence of tubing, syringes, and pumps contributes to reducing molecule adsorption or absorption. The effects of small-molecule absorption through PDMS will have to be investigated. However, it is clear that PDMS is only suitable for prototyping and will then be replaced with an injection molding–compatible plastic material.
On-Chip Compound Testing
Growth of HCT116 MTs treated with 5-FU was monitored over 3 days. Figure 8a shows the MTs’ relative diameters with respect to their diameters on day 0. When the MTs were exposed to 0 and 1 µM of 5-FU, they grew in size over time. In contrast, the growth was suppressed upon dosage of 3.16 µM, which is evidenced by the constant MT sizes over time. Higher concentrations of 10, 31.6, and 100 µM resulted in slight reduction of the MT size. Figure 8b shows HCT116 MT images on days 0 and 3.
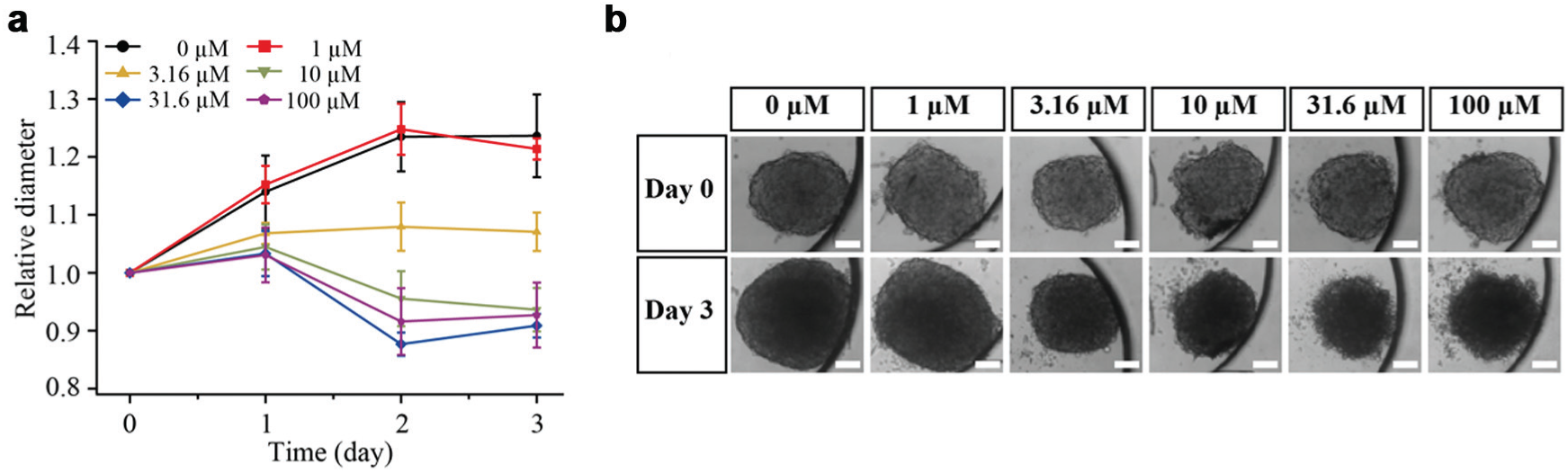
Growth of 5-fluorouracil (5-FU)–treated HCT116 microtissues (MTs). (
In Figure 9 , ATP content and fluorescence micrographs of the HCT116 MTs after exposure to different 5-FU concentrations are presented. Tissue viability was assessed with respect to the control group, which did not experience any 5-FU exposure (0 µM 5-FU in Fig. 9a ). The drug-induced effect was less than 10% in the case of MT exposure to 1 µM 5-FU. However, the viability of HCT116 MT began to significantly decrease at concentrations of 3.16 µM and went down to approximately 20% viability with respect to that of the control upon dosage of 100 µM 5-FU. The IC50 value was determined to amount to around 3 µM. These results are in accordance with the results of the growth determinations. The toxic effect can also be observed in fluorescence micrographs of treated tissues, as shown in Figure 9b .
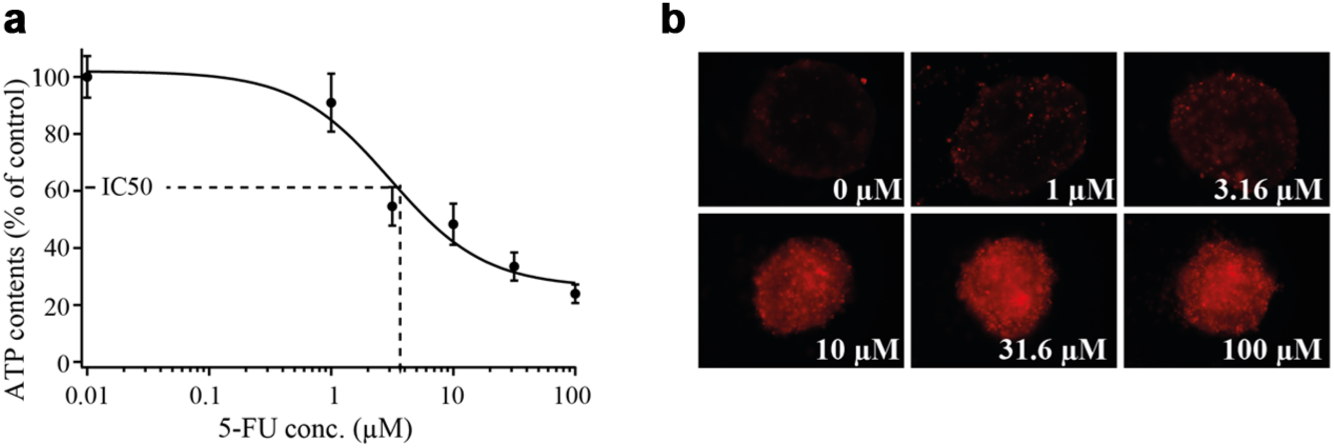
(
In conclusion, a microfluidic platform, which is compatible with conventional 96-well formats and enables parallelized 3D MT culturing and testing, has been developed. The platform can accommodate multiple MTs of the same or different cell types and fluidically interconnect those through microchannels. The platform offers various benefits compared with existing systems: (1) compatibility with existing 96-well format-based technologies, such as multichannel pipettes, automated liquid handling robots, or multiwell plate readers; (2) facile MT loading enabled by specifically designed loading ports and a gravity-controlled loading scheme; (3) robust media perfusion and operation in a conventional incubator due to gravity-driven flow and open reservoirs, which allow for bubble-free perfusion without the need for additional tubing and external pumps; (4) convenient sampling and media exchange; (5) execution of multiple experiments in parallel, as multiple channels are integrated on one platform, and as many platforms can be stacked on the tilting plate without increasing system complexity; (6) possibility of staining and fluorescence readout of MTs in the system without unloading; (7) and flexibility in designing the microfluidic connections between MTs, as various MT arrangements are possible depending on the design of the SU-8 mold. Multiple rLiMTs were successfully cultured under perfusion conditions over 8 days in the platform and showed increased metabolic cell activity under perfusion conditions in comparison to static conditions. Moreover, parallelized drug tests were conducted in the platforms using HCT116 MTs and 5-FU. The presented platform concept will help to advance current methods for 3D spheroid culturing under perfusion conditions, as it can be used with complex multitissue formats, such as “body-on-a-chip” configurations, in drug discovery and chemical safety testing. The dimension of MT compartments can be modified to accommodate different MT volumes that correspond to the volumes of different organs in a human body, for example. Furthermore, MTs displaying disease characteristics can be generated off platform and then can be introduced to the platform, where they can be further cultured in physical separation while being fluidically connected to other MTs in the platform.
Footnotes
Acknowledgements
Sirion Biotech GmbH, Germany, is acknowledged for providing the HCT116 eGFP cell line.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the FP7 of the EU through the project “Body on a Chip,” ICT-FET-296257, and an individual Ambizione Grant 142440 of the Swiss National Science Foundation for Olivier Frey.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.