Abstract
Handling and dosing of cells comprise the most critical step in the microfabrication of cell-based assay systems for screening and toxicity testing. Therefore, the immediate drop-on-demand technology (I-DOT) was developed to provide a flexible noncontact liquid handling system enabling dispensing of cells and liquid without the risk of cross-contamination down to a precise volume in the nanoliter range. Liquid is dispensed from a source plate within nozzles at the bottom by a short compressed air pulse that is given through a quick release valve into the well, thus exceeding the capillary pressure in the nozzle. Droplets of a defined volume can be spotted directly onto microplates or other cell culture devices. We present a study on the performance and biological impact of this technology by applying the cell line MCF-7, human fibroblasts, and human mesenchymal stem cells (hMSCs). For all cell types tested, viability after dispensing is comparable to the control and exhibits similar proliferation rates in the absence of apoptotic cells, and the differentiation potential of hMSCs is not impaired. The immediate drop-on-demand technology enables accurate cell dosage and offers promising potential for single-cell applications.
Introduction
Although the use of robotic workstations for liquid handling has increased enormously throughout recent decades to meet the requirements for high accuracy and high throughput of current bioanalytics, 1 automated and integrable dispensing systems for cell applications are limited. Various techniques are applied to dispense volumes in the nanoliter range. 1 Most of them use drop-on-demand (DoD) technology for ejecting single liquid droplets. 2 However, dispensing small volumes of cell suspensions (e.g., for single-cell analysis or diagnostics) remains a great challenge. Many bioprinting devices such as inkjet printers are still in the research stage. Cell dispensing differs from printing of bioactive molecules such as DNA or proteins. The latter are deposited onto dry substrates and dry out quickly after deposition: cells require suitable materials or technical process modifications to prevent the drying of the cells. 3
Plenty of groups work with inkjet printers, modifying them for cell application and for 3D fabrication. Boland et al. report the successful use of a modified inkjet printer to print cell suspensions. 4 They further investigated whether fragile mammalian cells (primary neurons and the QCE-6 cell line) could be used as bio-ink. 5 DoD printers using all three forms of actuation (thermal, piezoelectric, and electrostatic) have been reported as suitable for dispensing living cells. 6 Human fibroblast cell line (HT 1080) has been printed with a piezoelectric-actuated, DoD inkjet printing system (Microjet MJ-AB-01; Microfab, Plano, TX). 6 Lorber et al. demonstrated printing of adult rat retinal ganglion cells and glia by a Microfab dispensing device. 7 The physiological properties of mammalian cells strongly depend on environmental conditions, being much more sensitive to heat and mechanical stress. Hence, there is concern that the cells could be damaged or lysed by the conditions present during thermal printing. However, previous studies demonstrate cell survival after thermal inkjet printing. 3 Parameters such as printing pressure and needle shape are crucial factors for cell viability. Thus cells can be encapsulated in hydrogels to achieve higher viability.8,9
Besides, inkjet printers using laser-based techniques [biological laser printing (BioLP), e.g., laser-induced forward transfer (LIFT)] are developed to displace cells. 10
The Gyger Fluidics CERTUS nanodispenser (Gyger, Thun, Switzerland), based on the solenoid valve principle, could be a tool for dispensing cells in small volumes. 11 Biofluidix (Freiburg, Germany) provides the piezoelectric-actuated PipeJet dispenser for aliquoting and distributing biological fluids and cells. 12
With regard to automated high-throughput use, most of the devices have drawbacks in sterile charging of the reservoir and handling of patient-specific cells without cross-contamination, or they lack the flexibility for automated handling.
In the present study, we report on our in-house developed immediate drop-on-demand technology (I-DOT). 13 This nanodispensing technology belongs to the class of air displacement systems that applies pressure to a liquid reservoir for liquid dispensing. The ejected volume is controlled by the pressure height and the applied valve-opening time, which allows liquid dispensing down to the nanoliter range. Droplets with a defined volume can be spotted directly into microplates [customized plates as well as plates that meet the standards of American National Standards Institute/Society for Laboratory Automation and Screening (ANSI/SLAS) 1-2004], assay carriers, cell culture devices, or other reaction tubes. Furthermore, the I-DOT provides an alternative for the creation of microarrays with densities up to three spots per mm. The parallel processing of up to eight wells enables the rapid filling of large multiwell plates (up to 1536 wells) by rapid moving of the dosing head over the entire plate. The system has a high viscosity range, enabling both aqueous solutions and highly viscous media up to approximately 800 mPas to be dispensed by a single device. I-DOT control software enables the fully automatic dispensing of arrays and diluent series, and reformatting of SBS plates. Also, easy mixing of liquids by using the target plate in a further process step as a source plate is possible.
The I-DOT has been used to dispense the human breast cancer cell line MCF-7, human fibroblasts, and human mesenchymal stem cells (hMSCs) to investigate the response of the cells to the mechanical and fluid stresses associated with the dispensing process. In comparison to other available systems, the I-DOT is a promising technology for the automated handling of viable and functional cells.
Materials and Methods
Immediate Drop-on-Demand Technology
The nanodispensing device used in this study applies the I-DOT and has been described in detail in patent specification (US8759113 B2).
In brief, liquid is dispensed from a special source plate with a nozzle at the bottom. Application of pressure to the liquid reservoir leads to droplet ejection. Droplet volume is thereby determined by liquid properties, pressure height, application time, and nozzle diameter.
The I-DOT benchtop instrument is composed of two extendable drawers for the target and source plate and an above-stored dosing head. The distance between the source plate and the target is optimally adjusted so that drop distraction is minimal. Further deionization during the retraction of the lower drawer is performed to discharge electrostatically charged target plates. A special source plate with nozzles at the bottom (Ø 100 µm) is used for dispensing the liquid to the target. The small diameter of the nozzles keeps the liquid inside the well due to capillary forces and prevents leakage under ambient pressure.
To start the dispensing process, the filled source plate is inserted in the upper drawer. In the lower drawer, a target plate or any other carrier (e.g., glass slides) is placed. The eightfold dosing head is positioned on the selected source wells, and a short compressed air pulse is applied through a quick-release solenoid valve into the wells, exceeding the capillary pressure in the nozzle and leading to a defined release of liquid ( Fig. 1 ). Linear spindle actuators (RCP5; IAI, Schwalbach, Germany) and servo-motors (PD2-N41; Nanotec Electronic GmbH & Co. KG, Feldkirchen, Germany) enable the positioning needed to dispense from any source well to any target position. Droplets with a defined volume can be spotted directly into different microplates.
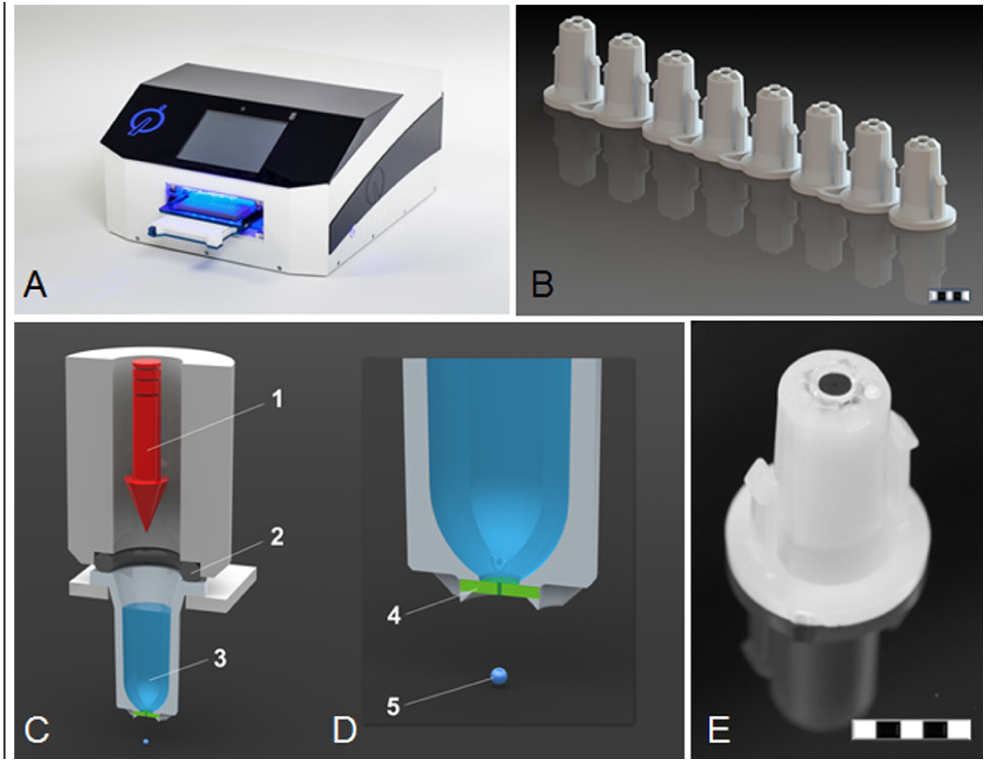
(
The special quick-acting solenoid valve emits extremely short compressed air pulses that generate single droplets with volumes as low as 1 nl. The duration of the pressure pulse is less than 1 ms. This enables the generation of up to 400 droplets per second, and thus also larger overall volumes can be dispensed within an extremely short time.
ARTEL MVS
The Alternative Solution Preparation with Multichannel Verification System (ARTEL MVS) was applied to validate printed volume coefficients of variation. After application with the I-DOT, the volumes of dispensed water were verified with the ARTEL MVS according to the manufacturer’s instructions.
Biological Experiments
Cell Line and Culture Conditions
The human breast cancer cell line MCF-7 was acquired from the Leibniz Institute DSMZ—German Collection of Microorganisms and Cell Cultures (DMSZ-No. ACC115). Primary human fibroblasts were isolated by the Fraunhofer Institute for Interfacial Engineering and Biotechnology (IGB). Mesenchymal stem cells, isolated from bone marrow, were provided from the Medical Department of the University of Tübingen.
All these cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) with 4.5 g/l glucose and supplemented with 10% fetal bovine serum (FBS), 100 U/ml penicillin, and 100 µg/ml streptomycin (Gibco, Darmstadt, Germany), this medium hereafter being called DMEMr. Frozen stored cells were thawed and routinely cultured at 37 °C, 5% CO2, and 95% relative humidity. At a confluency of 80%, the cells were detached and either used for experiments or subcultured. For the detachment of cells, the culture medium was removed and the flask rinsed once with Dulbecco’s Phosphate-Buffered Saline without magnesium and calcium (DPBS; Gibco). The cells were detached using 3.5 ml StemPro Accutase Cell Dissociation Reagent (Life Technologies, Darmstadt, Germany) per T-flask. After an incubation time of 20 min at 37 °C and 5% CO2, the detached cells were centrifuged at 20 °C for 5 min at 323 g. Afterward, the cell concentration and the cell viability were measured using the Countess Automated Cell Counter (Life Technologies).
Cell Viability
Different methods were applied to determine the viability of the cells dispensed with the I-DOT: the Trypan Blue Viability Test (Life Technologies), LIVE/DEAD Viability/Cytotoxicity Kit (Invitrogen, Karlsruhe, Germany), and In Situ Cell Death Detection TMR Red Kit (Roche Diagnostics GmbH, Mannheim, Germany). For each method, a manual control was performed, replacing the application with the I-DOT by a transferring step of the same volume with a 10–200 µl pipette (Eppendorf, Hamburg, Germany).
Trypan Blue Viability Test
Ten microliters of a defined concentration of cells per milliliter were applied with the I-DOT into four-well chamber slides (Lab-Tek Chamber Slides, Permanox, Nunc, VWR, Darmstadt, Germany). Experiments for each cell type were replicated fivefold in triplicates. The cell viability was determined immediately after the application.
LIVE/DEAD Viability and Cytotoxicity Kit
Viable cells are discriminated from dead ones by staining with green-fluorescent calcein-AM to indicate intracellular esterase activity and with red-fluorescent ethidium homodimer-1 (EthD-1) to indicate loss of plasma membrane integrity. 14 For the detection of cells, which lost their ability to adhere, the test kit was adapted to analyze the content of adherent cells and cells that are in suspension.
Adherent cells
Twenty microliters of a defined cell concentration of cells were dispensed with the I-DOT into 180 µl propounded cell culture medium in Permanox chamber slides. After overnight incubation (37 °C and 5% CO2), the test kit was applied as described in the manual. Immediately after staining, the samples were evaluated using fluorescence microscopy [fluorescence illumination system (MT10D), Olympus, Hamburg, Germany].
Cells in Suspension
Ten microliters of a cell suspension with a defined concentration were dispensed with the I-DOT into a fluorescence 96-well plate (Greiner, Frickenhausen, Germany) and stained immediately after the dispensing according to the instructions of the LIVE/DEAD manual.
In Situ Cell Death Detection, TMR Red Kit (TUNEL Assay)
The applied kit is based on the detection of single- and double-stranded DNA breaks that occur at the early stages of apoptosis. 15 This method is termed TUNEL [terminal deoxynucleotidyl transferase (TdT)-mediated dUTP-X nick end labeling]. 16 Performing the TUNEL assay, apoptotic cells are fixed and permeabilized. Subsequently, the cells are incubated with the TUNEL reaction mixture. After washing, the label incorporated at the damaged sites of the DNA is visualized by fluorescence microscopy.
Adherent cells
Twenty-microliter cells of a determined cell concentration were dispensed with the I–DOT into 180 µl propounded cell culture medium in Permanox chamber slides. A control slide for dead cells was cooled down for 20 min at −80 °C. The kit was applied in accordance with the manual. Twenty microliters of Flouromount G (Biozol Diagnostica GmbH, Eching, Germany) per chamber were applied to cover the slides. The slides were analyzed by fluorescence microscopy.
Cells in suspension
The cells in suspension were treated as described in the manual: 190 µl of buffer was prepared, and 10 µl of cell suspension was added manually or dispensed with the I-DOT. A dead control was performed in a separated well plate, and 10 µl of cell suspension was pipetted into the wells and cooled down in a −80 °C freezer for 20 min. Finally, the absorption of TMR red was measured at 580 nm.
Stem Cell Differentiation
For the differentiation assays, hMSCs in passages ≤5 were used. For the manual control, 1.00E+05 cells were seeded per well and cultured until they reached confluence. For the application with the I-DOT, 20 µl cells (2.00E+06 cells/ml) were dispensed with the I-DOT into 180 µl cell culture medium and cultured until they reached 100% confluency as well. To induce adipogenesis in confluent monolayer cultures, DMEMr supplemented with 100 µM insulin (Sigma-Aldrich, Steinheim, Germany), 500 µM IBMX (Sigma-Aldrich, Steinheim), 1 µM dexamethasone (Sigma-Aldrich, Steinheim), and 100 µM indomethacin (Sigma-Aldrich, Steinheim) were used. 17 All differentiation assays were performed threefold using duplicates for each lineage.
Osteogenesis was induced by the usage of a differentiation medium consisting of DMEMr with 50 µg/ml L-ascorbic acid 2-phosphate (Sigma-Aldrich, Steinheim), 10 mM glycerol 2-phosphate (AppliChem, Darmstadt, Germany), and 100 nM dexamethasone (Sigma-Aldrich, Steinheim). 18
The differentiation to the adipogenic lineage was performed for 14 days with adipogenic differentiation medium. For the differentiation to the osteogenic lineage, the cells were cultured for 28 days using the osteogenic differentiation medium. The negative control assays were cultured using the standard cell culture medium, DMEMr. At the end of culture, a specific staining was performed for each differentiation lineage.
Confirmation of adipogenic differentiation
Cultures were washed with DPBS once and afterward fixated using 4% paraformaldehyde for 10 min. Then, the slides were washed with distilled water and incubated for 5 min in 60% 2-Propanol. Next, the 0.3% Oil Red O working solution (Sigma-Aldrich, Taufkirchen, Germany) was applied and incubated for 10 min. Subsequently, the slides were again washed with 60% 2-Propanol, and then in distilled water. After that, the hemalaun solution was applied and incubated for 5 min, and the slides were rinsed for at least 1 min in water. The chamber slides were put in distilled water and thereafter mounted with 20 µl Aquatex (Merck, Darmstadt, Germany) and a cover slip. The slides were placed underneath a fuming hood on a level surface and dried overnight. The following day, the slides were examined and evaluated with a bright-field microscope. Oil Red O stain dyes fat droplets red, whereas hemalaun dyes cell nucleus blue.
Confirmation of osteogenic differentiation
Cultures were washed with DPBS once and afterward fixated using icy cold methanol for 10 min. Next, the slides were washed with distilled water. Then, the 1% alizarin red S staining solution was applied for 2 min. Thereafter, the slides were washed three times with distilled water, and afterward mounted using the mounting agent Aquatex and a cover slide. The slides were placed underneath a fume hood on a level surface and dried overnight. The following day, the slides were examined and evaluated with a bright-field microscope. Alizarin red S dyes potassium phosphate pinkish red.
Results
Volume-Dispensing ARTEL MVS
With the ARTEL MVS volume variation, coefficients for different drop volumes could be assessed. The calculated volumes thereby increased according to drop numbers ( Fig. 2 ).
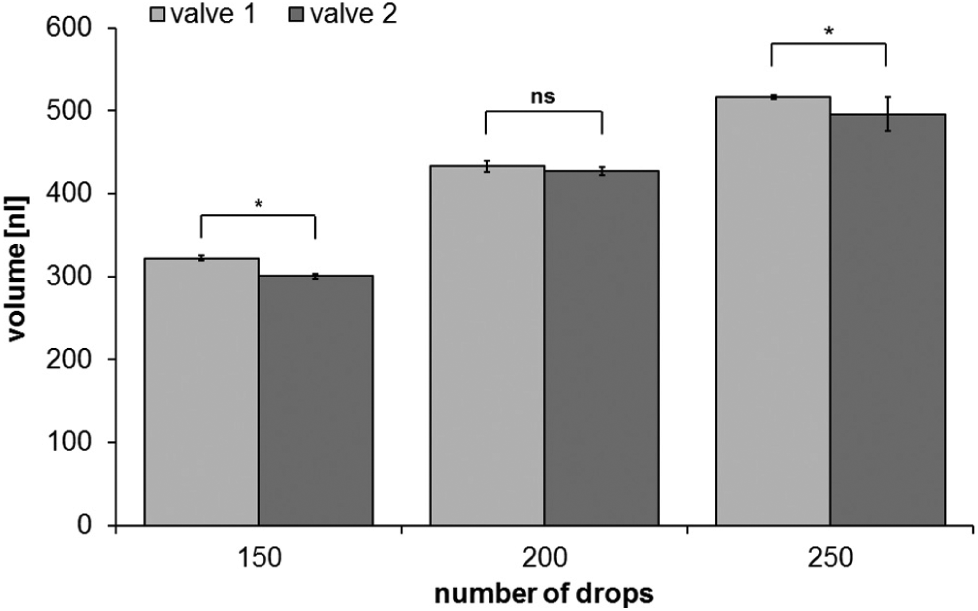
Calculated volume of different amounts of dispensed drops of water. Drop counts of 150, 200, and 250 dispensed with two different valves and each with two different source wells. Volume is calculated with the Alternative Solution Preparation with Multichannel Verification System (ARTEL-MVS).
Array Spotting
In Figure 3 (left), arrays of aqueous liquid spots with different drop volumes are shown. Liquid dispensing with I-DOT allowed for a precise spatial droplet position without satellite drop depositions at different target volumes. As expected, deposited drop size was thereby increased with higher target spot volume. Dispensing of cell suspensions at different cell concentrations from 0.1 to 5 million cells per ml in the source well resulted in reproducible cell spots. According to initial cell concentrations, liquid spots exhibited increasing cell numbers per spot ( Fig. 3 , right).
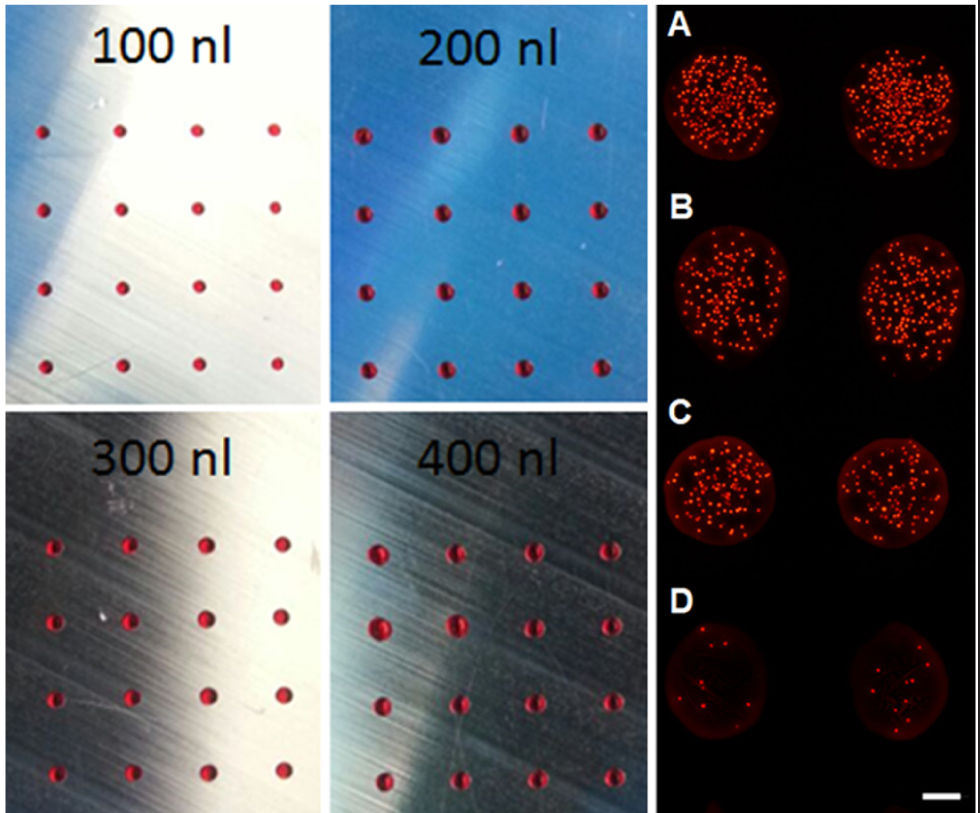
(
To analyze the possibility of applying the I-DOT device for cell application procedures, we dispensed several cell types and assessed different cell parameters, such as cell morphology, viability, and differentiation capacity.
Cell Count and Viability after Dispensing
The results of the cell count and trypan blue viability test directly after dispensing are shown in Figure 4 . After dispensing, 8.4E5 ± 1.8E5 c/ml of 1.0E6 MCF-7 were counted. The cell count of 1.0E6 c/ml human fibroblasts after dispensing was 8.9E5 ± 1.2E5 c/ml, and of 2.0E6 c/ml hMSCs 2.0E6 ± 7.9E5 c/ml. For all three cell types tested, no differences in the cell counts before and after dispensing could be assessed. MCF-7 cells had an average viability of 90 ± 5% before dispensing; after the application with the I-DOT, the average viability was 84 ± 4%. The average viability of human fibroblasts before dispensing was 93 ± 4%; after using the I-DOT, the average viability was 96 ± 1%. hMSCs had an average viability of 86 ± 5% before dispensing; after the dispensing with the I-DOT, the average viability was 83 ± 6%. The trypan blue viability test indicates that the viability of cells is similar before and after dispensing.
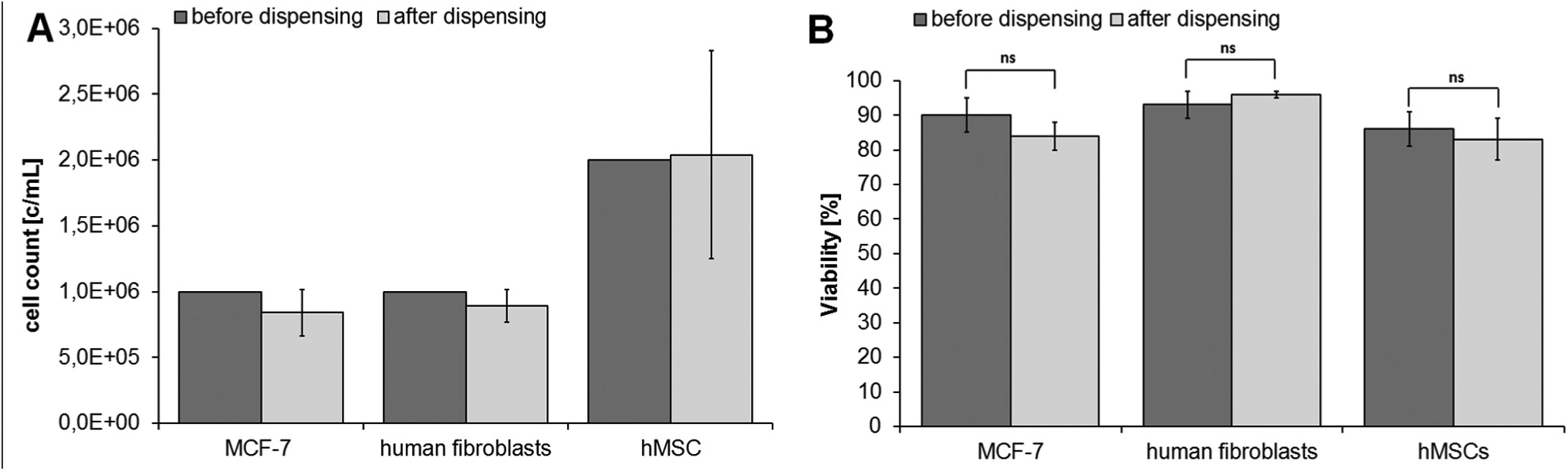
(
LIVE/DEAD Viability/Cytotoxicity Kit
Adherent cells
Cells dispensed with I-DOT as well as the control were adherent after overnight incubation. The typical cell morphology of the cells was observed for both applications. After staining, most of the dispensed cells (>95%) emitted a green signal, indicating viable cells, and cell morphology also appeared normal ( Fig. 5 , left). Controls showed the same results.
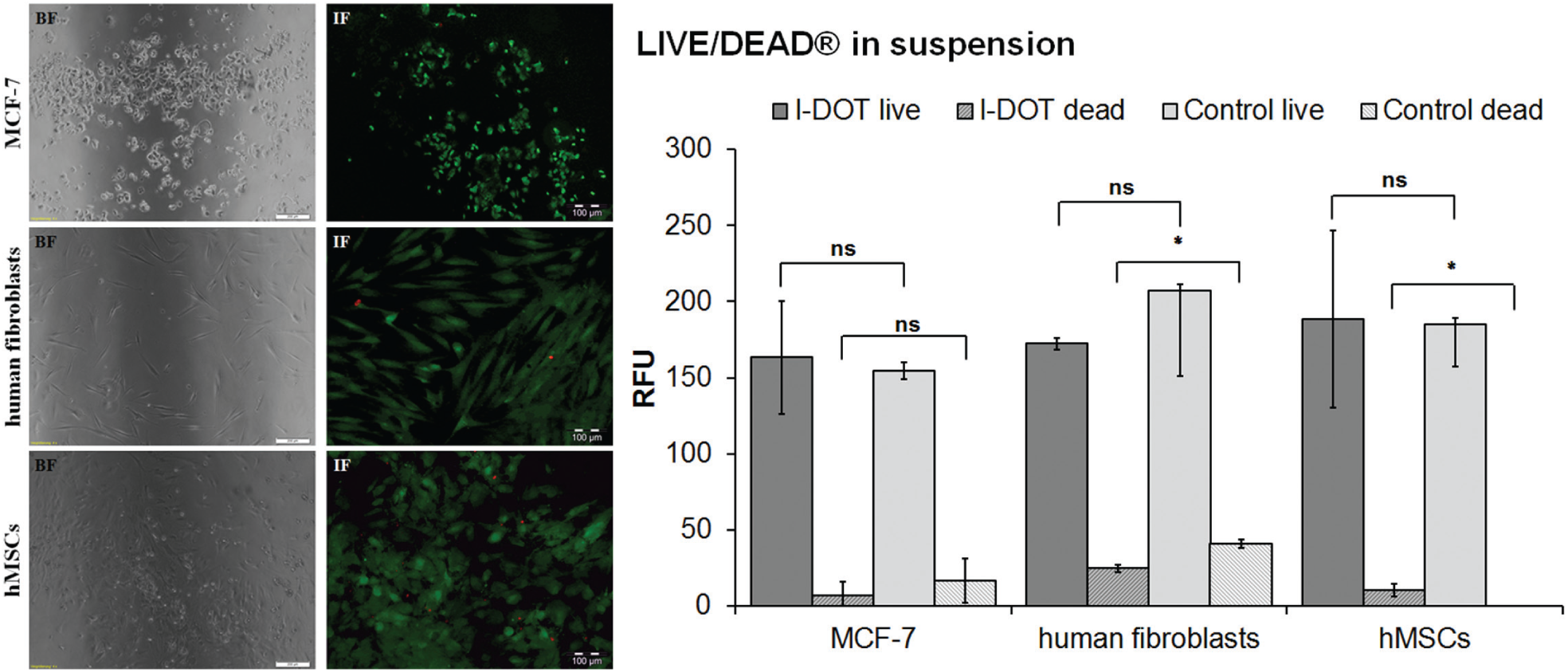
(
Cells in Suspension
The relative fluorescence unit (RFU) of cells dispensed with the I-DOT and the control (pipetted manually) are shown in Figure 5 (right). Living cells are indicated by the calcein-AM signal, and dead cells by EthD-1. Dispensed MCF-7, fibroblasts, and hMSCs have similar RFUs of calcein-AM compared to the control. RFUs of EthD-1 signals are low for dispensed and manually pipetted cells. That indicates that the viability of each cell type is not impaired by the I-DOT. T-test of data from I-DOT compared with manual controls showed no significant difference for living cells. RFUs of dead human fibroblasts and dead hMSCs differ significant from in the control group.
In Situ Cell Death Detection, TMR Red Kit (TUNEL Assay)
To determine whether dispensed cells die due to apoptotic events after application with the I-DOT, the In Situ Cell Death Detection, TMR Red Kit (TUNEL assay) was applied for adherent cells as well as for cells in suspension.
Adherent cells
Cells dispensed with the I-DOT and seeded manually were adherent after 1 day of incubation. After staining, all cells’ nuclei emitted a blue signal with DAPI excitation; however, no signal was detected using TRITC excitation, as seen in the overlay pictures ( Fig. 6 ). Manual controls showed the same staining results.
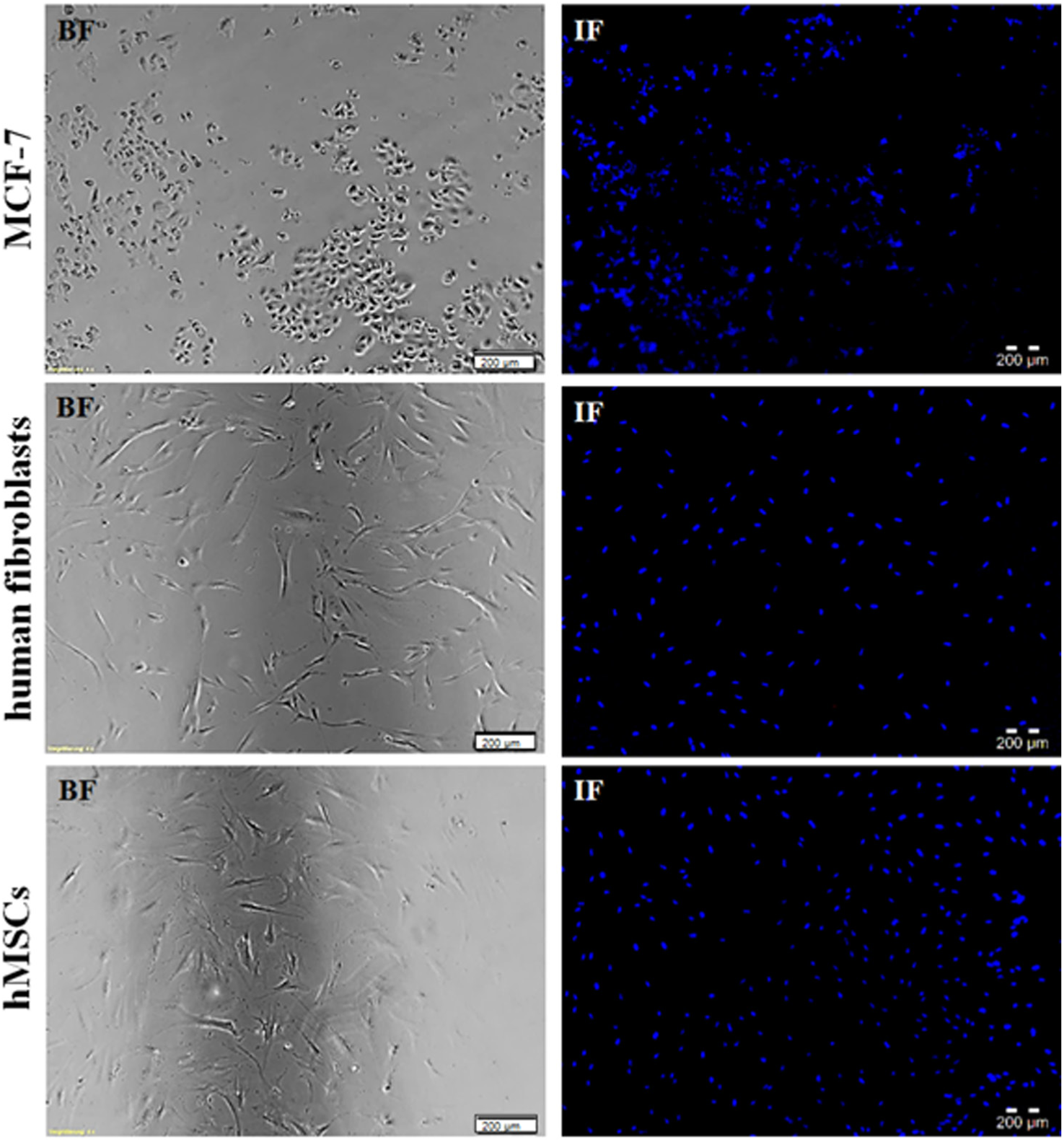
TUNEL [terminal deoxynucleotidyl transferase (TdT)-mediated dUTP-X nick end labeling] assay of adherent cells: MCF-7, human fibroblasts, and human mesenchymal stem cells (hMSCs) dispensed with the immediate drop-on-demand technology (I-DOT). (
Cells in suspension
TUNEL assay was also performed with cells in suspension. Controls verified the appropriate application of the test kit. No apoptotic signals for dispensed cells (for each cell type) could be detected.
Stem Cell Differentiation
To assess whether an application using the I-DOT affects the differentiation properties of hMSCs, dispensed and pipetted hMSCs were differentiated. The pipetted hMSCs acted as the manual control group.
Confirmation of adipogenic differentiation
One week after starting the culture with adipogenic differentiation medium, first fat droplets around the cells could be observed under the microscope. After 2 weeks of culture, large accumulations of fat droplets around the cells were seen. However, the nondifferentiated cells cultured in standard cell medium DMEMr appeared unchanged and showed no signs of differentiation during the experiment. Histological staining with Oil Red O and hemalaun stained the fat droplets around differentiated cells red and the cell nuclei blue ( Fig. 7 ).
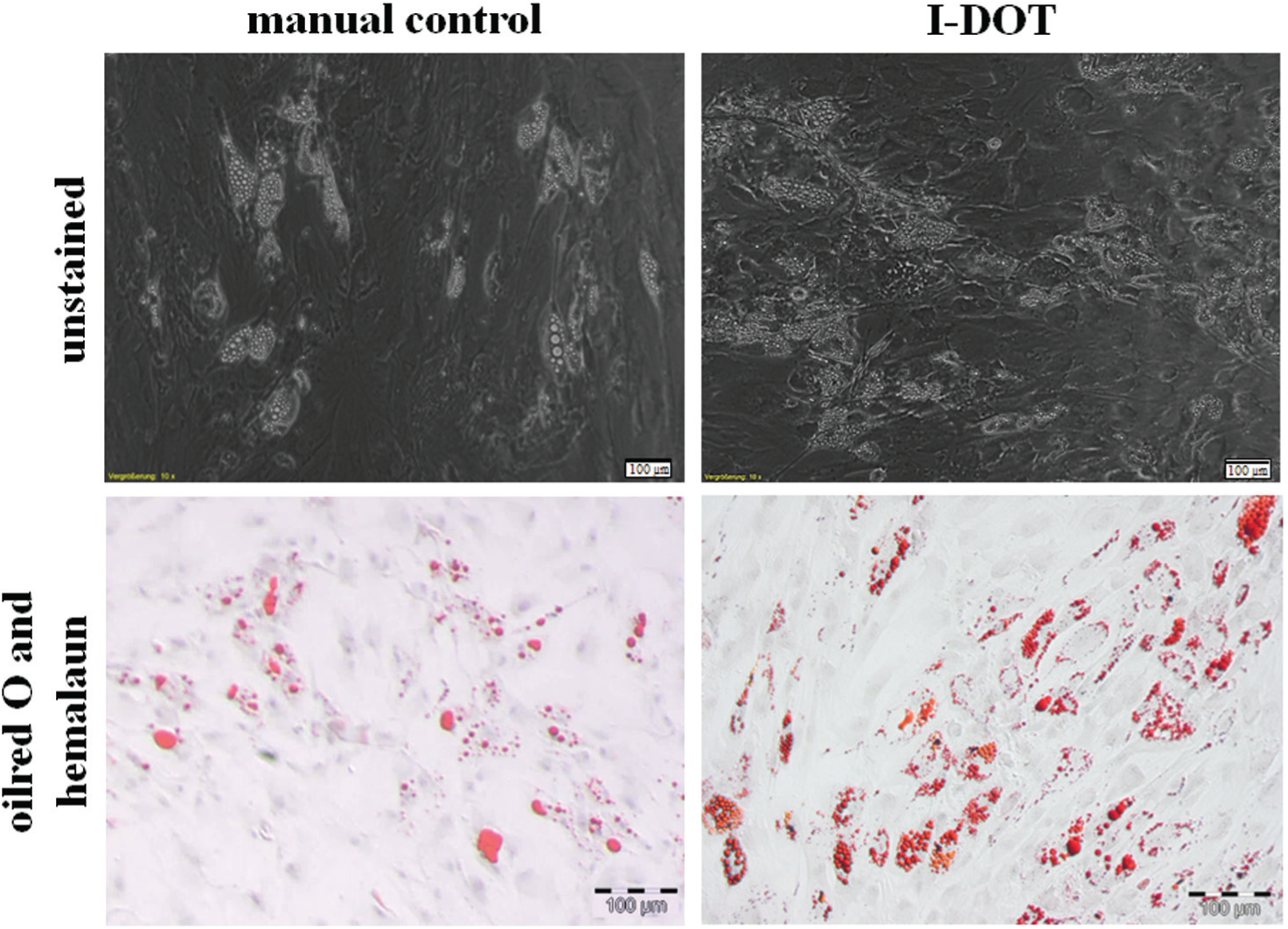
Adipogenic differentiation of manually seeded and dispensed human mesenchymal stem cells (hMSCs). (
Confirmation of osteogenic differentiation
About 9 days after starting the culture with osteogenic differentiation medium, first crystalline structures around the cells could be observed under the microscope. After 4 weeks of culture, large accumulations of crystalline tricalcium phosphate (TCP) appeared all over the hMSC monolayer. However, the nondifferentiated group cultured with standard cell culture medium DMEMr appeared unchanged and showed no signs of differentiation during the experiment. Histological staining with alizarin red S stained the TCP crystals around differentiated cells pink ( Fig. 8 ). Dispensed as well as manually pipetted hMSCs were successfully differentiated to adipogenic and osteogenic lineages.
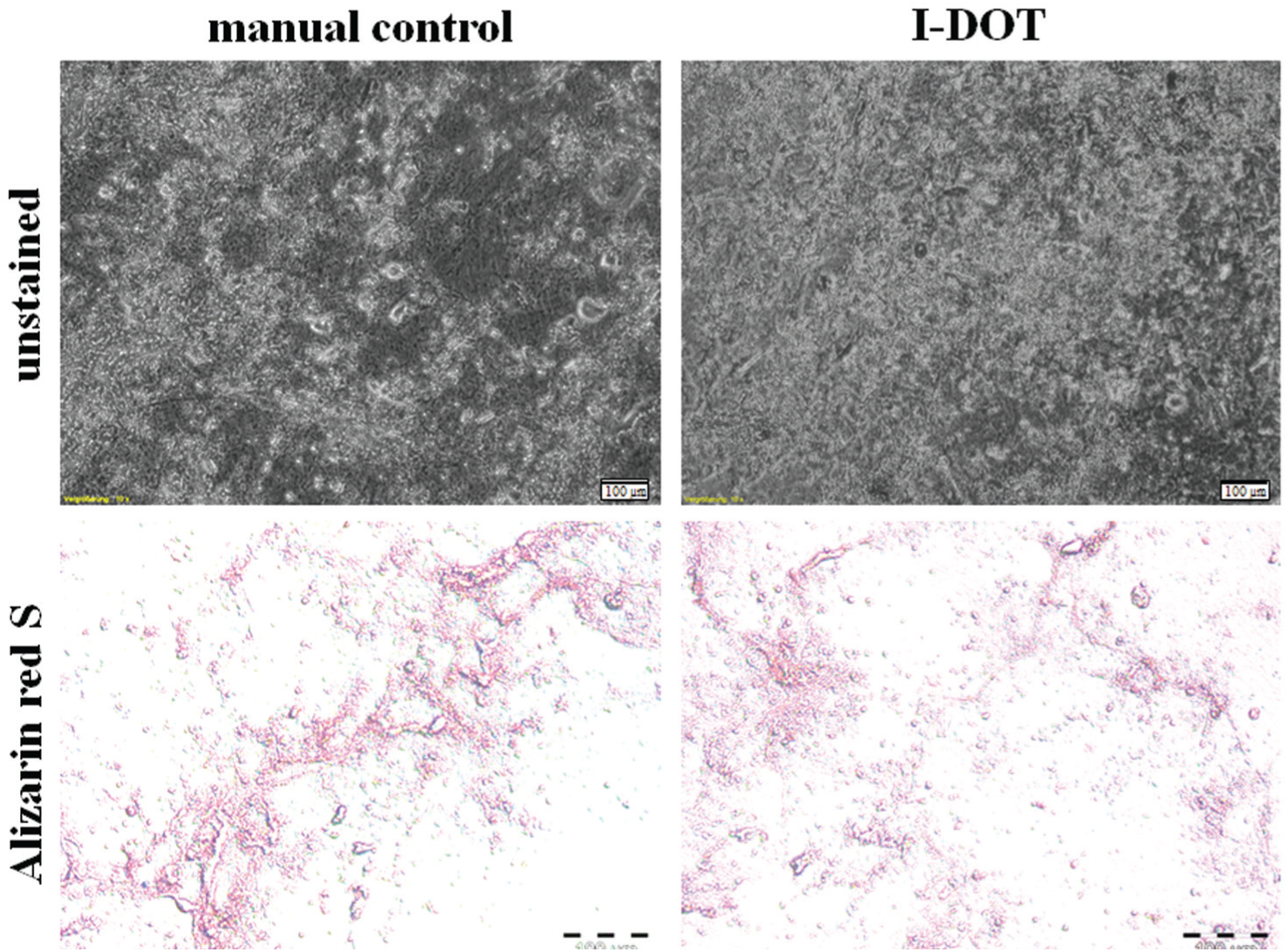
Osteogenic differentiation of manually seeded and dispensed human mesenchymal stem cells (hMSCs). (
Discussion
The applicability of the I-DOT technology for dispensing viable cells was demonstrated, proving the survival and proliferation rate of treated cells. Also assessed was the possibility of cell death due to cytotoxic or apoptotic events after an application with the I-DOT. Due to the promising findings and the complexity of cellular functions of stem cells, the differentiation potential of hMSCs after dispensing with the I-DOT was investigated.
Cell Count and Viability of Dispensed Cells
The cell count and viability of cells dispensed with the I-DOT were examined using different assays to get a deep understanding of cell behavior directly after dispensing (trypan blue, LIVE/DEAD Viability/Cytotoxicity Kit of cells in suspension) and after cell adherence (LIVE/DEAD Viability/Cytotoxicity Kit of adherent cells). Moreover, the possible induction of apoptosis was analyzed.
Cell count before and after dispensing with the I-DOT was comparable. Standard deviation of dispensed hMSCs was high due to a larger cell size and strong cell adhesion properties. No differences in the cell viabilities before and after dispensing with the I-DOT could be found, and similarly no difference to the manual control could be observed. These findings show the feasibility of this technique for the application of viable cells. Compared to other techniques like thermal and piezoelectric inkjet, in which post viability of 75–90% after application is described, the results with the I-DOT are in the middle of that range.19,10
Because the determination of cell viability depends on physical and biochemical properties of cells and events that do not interfere with the esterase activity or the integrity of the plasma membrane, such as the early stages of apoptosis, this will not be accurately assessed with this method. 14
Therefore, the TUNEL assay was applied to ensure no apoptosis induction after the application with the I-DOT. Again, no influence on the cell behavior could be detected.
Differentiation of hMSCs
Although viability assays showed no influence of I-DOT dispensing on these parameters, it is not clear whether sensitive cell mechanisms might be influenced during the application with the I-DOT. Because the MCF-7 cell line and the primary fibroblasts are rather robust in their ability to endure stress, MSCs might be more prone to such. The process of pressing cells through a tight orifice with high pressure and, more importantly, the sudden decompression when leaving the nozzle exposes cells to a certain shear stress. The strain might influence the cells and hamper their metabolic potential. Mesenchymal stem cells are known to be influenced by persisting shear stress, whereupon their morphology, confluence, and orientation alter.20,21 In this study, the dispensed cells displayed the same cell morphology, proliferation rate, and adipogenic and osteogenic differentiation behavior as the manually seeded hMSCs, indicating that the I-DOT represents a mild technique to also dispense large and sensible cell types. In other studies, stem cells are encapsulated to preserve differentiation potential after a printing process and for 3D tissue buildup, which could be circumvented by using the I-DOT device.22–24
Our technology can be applied either as a stand-alone device or in various robotic platforms for cell culture automation. As already described by Murphy and Atala, 25 inkjet systems struggle with similar difficulties as the I-DOT (e.g., the sedimentation of resting cells in suspension plugs the nozzle). Although cartridges or print heads of other technologies have to be cleaned or replaced in a cost-intensive and time-consuming way, the I-DOT source wells are designed as disposables and can be produced at low costs. The source wells are pressure actuated in a noncontact manner and can be easily filled by an automated liquid handling system. This facilitates a high flexibility and hitherto unknown freedom for experimental design (e.g., up to 96 independent reagents or components in a single experiment). Furthermore, in contrast to other dispensing systems, a minimal propounded sample volume of 5 µl (in a 96-well source plate) allows one to save valuable resource liquids or cell suspensions. Through the parallelization of the volume transfer, the process time for each plate can be reduced to a high degree. Acoustic systems, for example, have no parallelization option and are therefore relatively slow in transferring lots of samples, which is in demand in high throughput.
The printing frequency for piezoelectric-actuated systems is much higher than for acoustic and pneumatic-driven systems.26,27 The transfer volume for piezoelectric-, thermal-, and acoustic-actuated systems is situated in a small range. In contrast, the I-DOT can be used to transfer 1 nl to 80 µl (single drops or jet). This leads to a much higher flexibility in experimental design. Related to the use of target carriers, the I-DOT offers the highest freedom in the selection of the target. Standard plates, glass slides, as well as materials consisting of fibers or curved surfaces can be addressed.
To enable an easy and rapid integration of the I-DOT in an automated platform, SiLA (Standardization in Lab Automation) software interfaces were integrated. 29 This characteristic is a strong advantage of the I-DOT system compared to many other devices.
Conclusion
Recapitulating the findings of the viability assays and stem cell differentiation potential, the I-DOT developed at the Fraunhofer IPA is capable of dispensing viable mammalian cell lines, primary cells, and mesenchymal stem cells possessing full proliferation and, in the case of hMSCs, differentiation potential.
The I-DOT is an interesting tool for microarray formation that can be used to screen the effects of novel compounds on cell–cell interactions before application in vivo, 7 or, for example, for research on biofilm formation and antifouling applications. 28 It also allows single-cell analysis whenever the handling of low volumes is in demand. The I-DOT technology is being continuously improved to enhance performance. The parallelization of several I-DOT units and the integration into screening workstations also will be part of future developments.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V.
