Abstract
A compact bulk liquid dispenser was designed and constructed to enable the interleaved execution of fully automated assays on space-constricted workstations. Due to its design, multiple instances of dedicated dispensers can be integrated on the same system, providing independence, redundancy, and preservation of reagents with no carryover risk. The performance and impact of the integrated dispensers on the throughput of an automated enzyme-linked immunosorbent assay system are demonstrated in this article. (JALA 2007;12:166–71)
Introduction
Full automation of biochemical and cell-based assays is enabled through the integration of components with specific functionality, such as liquid handlers, plate handlers, plate washers, and readers. In the past, large automated systems with external robotic plate handlers have been used for the execution of fully automated assays. However, in recent years, workstations with internal plate handlers have become available with the advantage of a smaller footprint, higher throughput, and lesser complexity than larger integrated systems.
A small reagent dispenser is introduced to alleviate the size of commercial bulk dispensing devices from dominating the deck space of such workstations. Multiple dispensers can be arranged in a space-saving, stacked configuration to present plates efficiently within the work envelope of the internal robotic arm. Each bulk dispenser is fully independent, freeing workstation resources, thereby optimizing run times. This concept was realized with the integration of four dispensers (QuadDispenser) into the working envelope of a robotic workstation.
Methods and materials
Bulk Dispenser Design
Each bulk dispenser consists of two major components, a syringe pump connected to a dispensing manifold and a robotic stage that moves the microtiter plate columnwise under the fixed position dispense manifold. Each bulk dispenser is independently controlled by a small programmable logic controller (PLC) that sequences the robotic stage and syringe pump through high-level system commands.
The heart of the bulk dispenser is composed of the Tecan XLP6000 microstepping digital syringe pump (Research Triangle Park, NC) fitted with a 10-mL syringe. This pump is fully programmable to aspirate and dispense measured quantities of liquid with high accuracy and precision. Connected to the syringe pump output is an 8- or 16-channel manifold from Titertek (Huntsville, AL), which features interchangeable 0.016-in. ID glass lined tips and a see-through Kel-F body. Teflon tubing connects the syringe pump directly to the manifold and reagent reservoir. The fluid path for each dispenser is considered inert consisting of glass, Kel-F, Teflon, and polypropylene.
The fluidics layout (Fig. 1) for each bulk dispenser consists of a syringe pump, the manifold, prime station, waste, and reagent reservoirs. A prime station is located on the backside of the plate stage, such that when the stage is in the plate loading position the manifold can be primed. The prime station allows for a “clean in place” procedure to be implemented on the manifolds. Additionally, the manifolds are readily removable for maintenance operations. Waste is removed from the prime station into a common vacuum waste vessel.
Fluidics layout for the single bulk dispenser.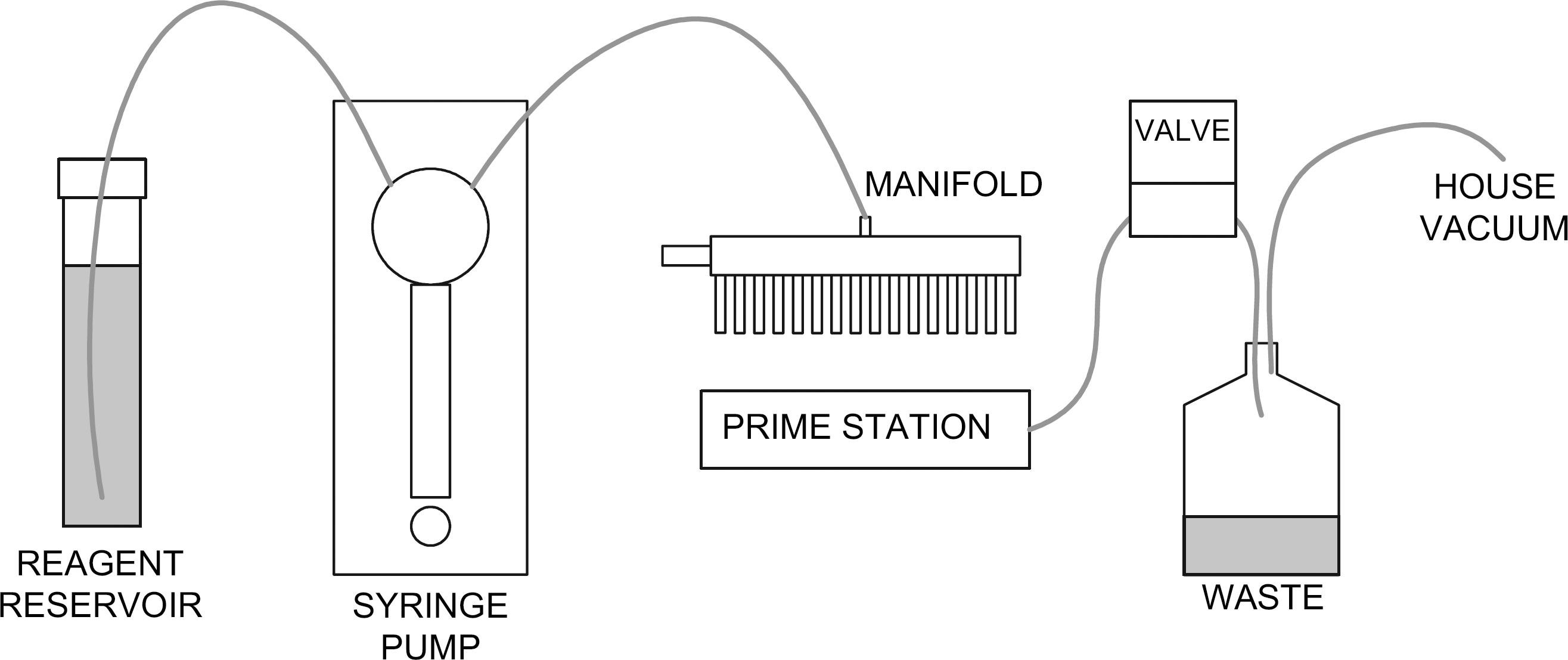
The unit design of a single dispenser is shown in Figure 2. Here the dispense manifold is confined to a fixed location whereas the robotic stage moves the microtiter plate and prime station underneath it. The robotic stage from Yamaha Motor Co. (Hamatsu, Japan) features a controller with an I/O interface programmable through 32 pre-defined positions.
Single bulk dispenser design. The robotic stage (1) moves the microtiter plate or the priming station (2) underneath the manifold (3).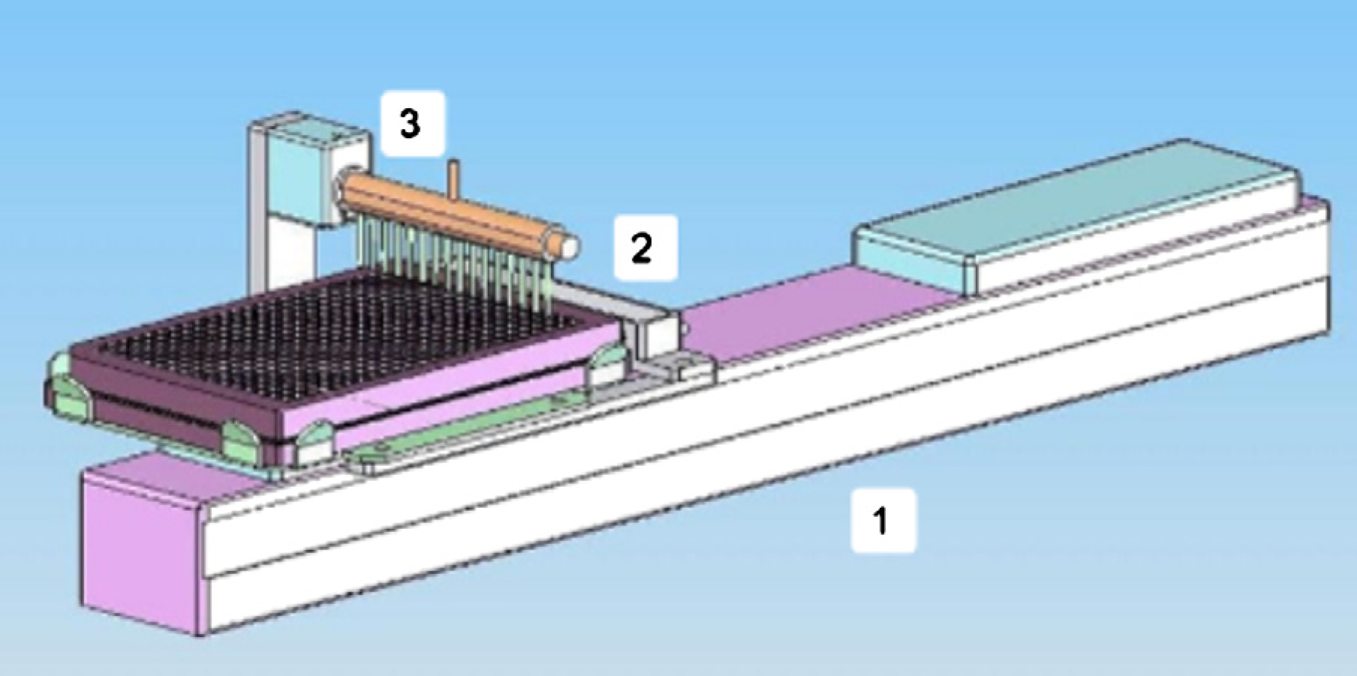
The QuadDispenser combines four independent single bulk dispensers into a system footprint 66 cm deep, 24 cm wide, and 20 cm high. The syringe pumps, associated reservoirs, waste vessels, and the control cabinet are stored beneath the workstation.
The QuadDispenser is integrated on a Tecan EVO 200 (Research Triangle Park, NC) with an eight-channel liquid handling arm and a robotic manipulator (RoMa) arm, together with two plate washers, an absorbance plate reader, a Guava PC96 cytometer (Hayward, CA), and a plate carousel (Fig. 3). The RoMa arm serves as the internal plate handler within the workstation providing plate transport to all devices. Once a plate is placed by the RoMa on one of the robotic stages of the QuadDispenser, the plate is moved column by column underneath the manifold, as described below.
Integration of the QuadDispenser into a Tecan EVO workstation. The workstation is composed of the QuadDispenser (1), absorbance plate reader (2), Guava PC96 cytometer (3), Tecan RoMa arm (4), dual plate washers (5), and plate carousel (6).
Each bulk dispenser is controlled by a Unitronics 120 series PLC (Quincy, MA), which can accept high-level serial commands and orchestrates the syringe pump and robotic stage through low-level serial commands that control each dispenser. With 2 serial ports, 6 outputs, and 10 inputs, the PLC is considered small (Fig. 4); however, it provides a seamless built-in man-machine interface (MMI). We found the MMI useful to display the current dispenser mode and the writable dispense parameters.
Single dispenser control diagram.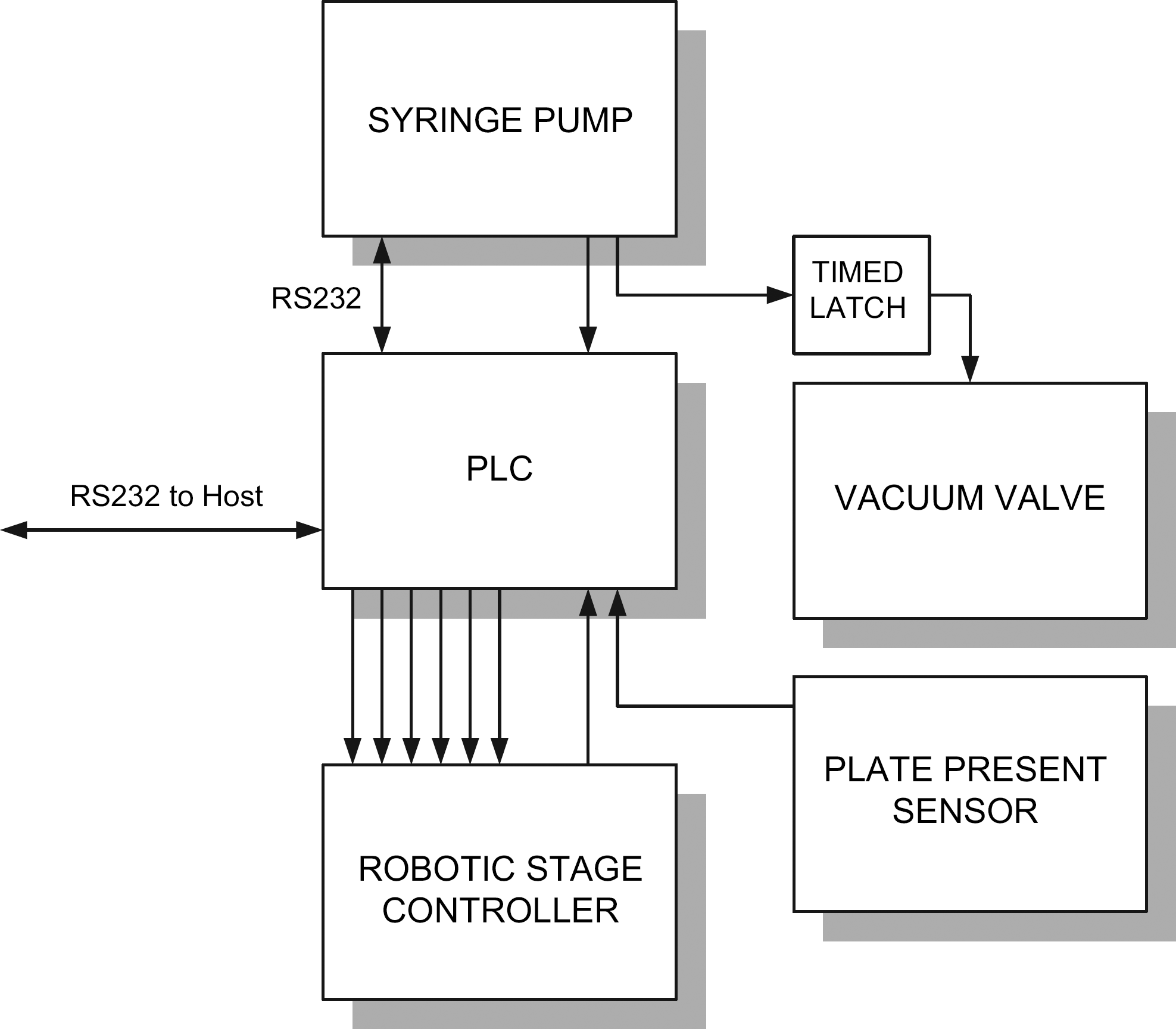
The high-level command set is composed of three types of commands.
Execute commands: sequence the dispenser through one of its functions.
INIT, initializes robotic stage, syringe pump, returns READY
RUN, run dispense cycle, returns DONE or ERROR NO PLATE
PRIME, prime syringe pump over prime station with current prime volume, returns READY.
Query commands; query parameters from the dispenser.
PLATE, plate present sensor query, returns TRUE or FALSE
PVOL, current prime volume query, returns PVOL #####
DVOL, current dispense volume query, returns DVOL ###
Parameter commands; write parameters to the dispenser.
SET DISP, set current user specified dispense volume
SETPRIME, set current user specified prime volume There is no reply for the parameter commands.
Enzyme-Linked Immunosorbent Assay
Nunc microtiter plates (Rochester, NY) coated with primary antibody from Sigma (St. Louis, MO), stored in blocking buffer, were used for enzyme-linked immunosorbent assays (ELISAs). ELISA plates were washed on a Tecan PW384 plate washer and standards were transferred to the ELISA plate from a Matrix Technologies 12-channel trough (Hudson, NH). ELISA plates were then incubated for 75 min at room temperature, washed, and 80 μL of secondary antibody from Pierce (Rockford, IL) was added using the QuadDispenser. ELISA plates were incubated for another 75 min, washed, and 80 μL of SureBlue TMB 1-component peroxidase substrate from KPL (Gaithersburg, MD) was added using the QuadDispenser. After 10 min, the reaction was stopped by adding 80 μL of 1 N sulfuric acid using the QuadDispenser. Finally, absorbance at 450 nm was measured on a Tecan Safire2 plate reader.
Results and Discussion
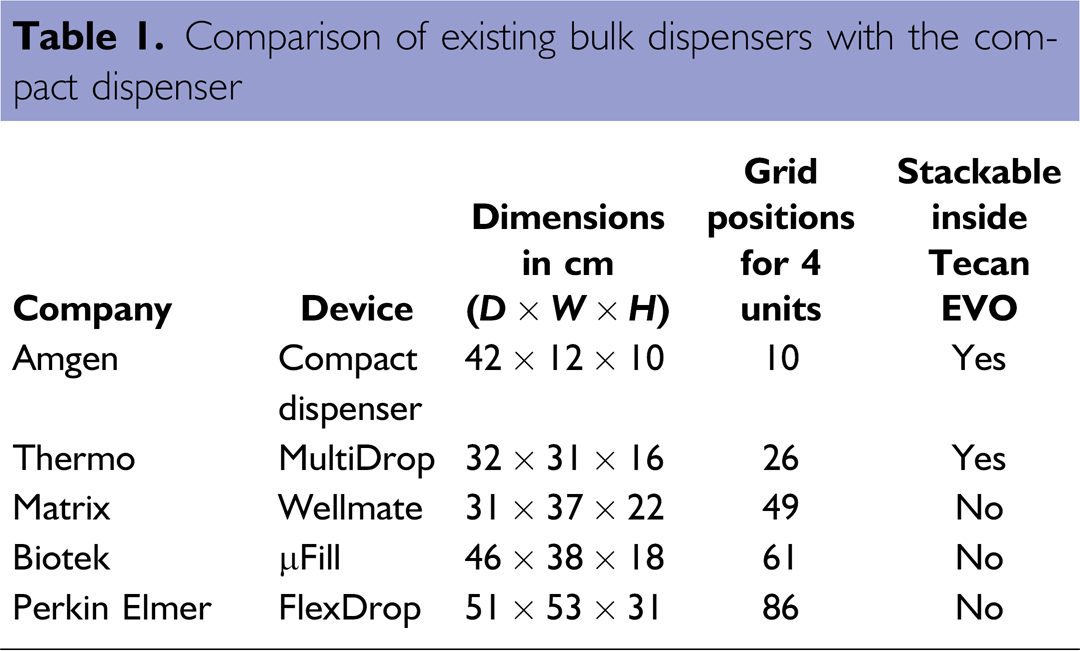
During the design phase of an automated ELISA platform, we determined that three bulk dispensers were required to achieve the expected throughput of 20 plates per 8-h shift. We also determined that commercial bulk dispensers would take up a significant portion of the available deck space on the 2-m deck of a Tecan EVO workstation. After integration of all other necessary components, such as washers, readers, and plate storage, we were left with 35 open grid positions, corresponding to 87.5 cm of open deck space at the front of the workstation. Only one commercially available bulk dispenser, the Multidrop (Thermo Electron Corp., MA), fit our requirements (see Table 1). Four stacked 2 × 2 would take up to 26 of the 35 available grid positions on the Tecan, leaving little space for future expansion. Hence, we decided to design a custom bulk dispenser that would expose only essential components to the workstation and allow stacking inside. An overview of the resulting QuadDispenser, using only 10 grid positions, is shown in Figure 3.
Comparison of existing bulk dispensers with the compact Dispenser
Our targeted volume range for the 8-channel bulk dispenser is between 10 and 100 μL. A precision and accuracy test was performed to quantify performance for the following volumes: 100, 75, 50, 25, 10, and 5 μL. For this test, a buffer (Ham F12, 30 mM HEPES) containing fluorescein (1.1 μM) was dispensed into empty 96-well plates, then brought to a final volume of 100 μL with H2O using the 96-channel Beckman-Coulter Multimek (Fullerton, CA).
Fluorescence of each well was then measured on a Tecan Safire II plate reader using bottom reading (excitation at 485 nm, emission at 520 nm). A hand-pipetted calibration curve was created and used to determine average volume, percent error, minimum, and maximum dispensed volumes per plate (see Table 2). A total of 72 plates were dispensed and tabulated for this test; amounting to 12 plates per test volume or 3 plates for each dispenser at any of the tested volumes. Within the target range (10–100 μL), a coefficient of variation (CV) of less than 5% was observed for each volume (Table 2, Fig. 5).
Calibration data
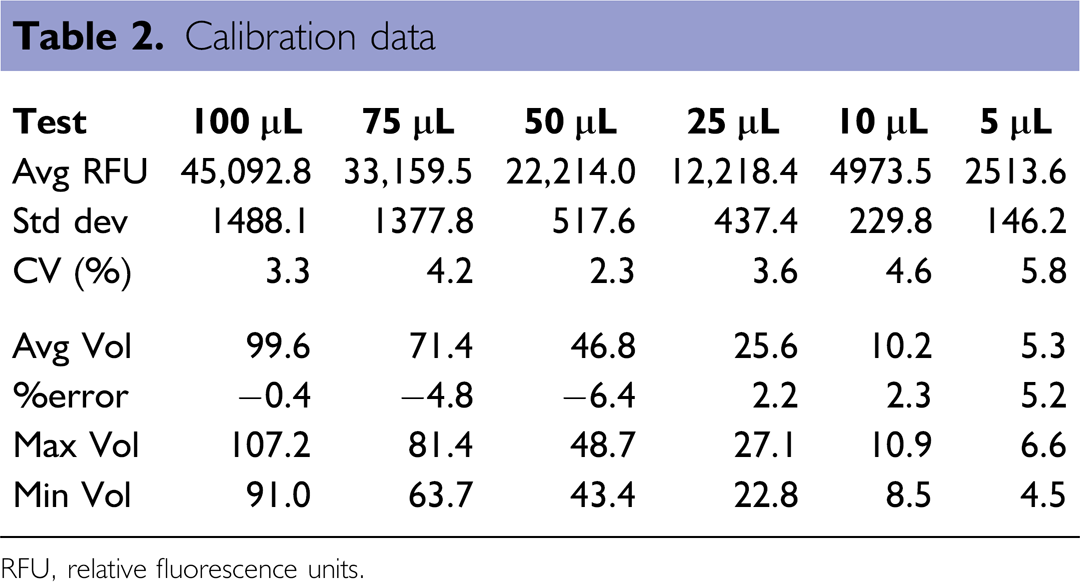
RFU, relative fluorescence units.
Expected versus actual volumes. Error bars are ±1 standard deviation for 288 determinations at each volume.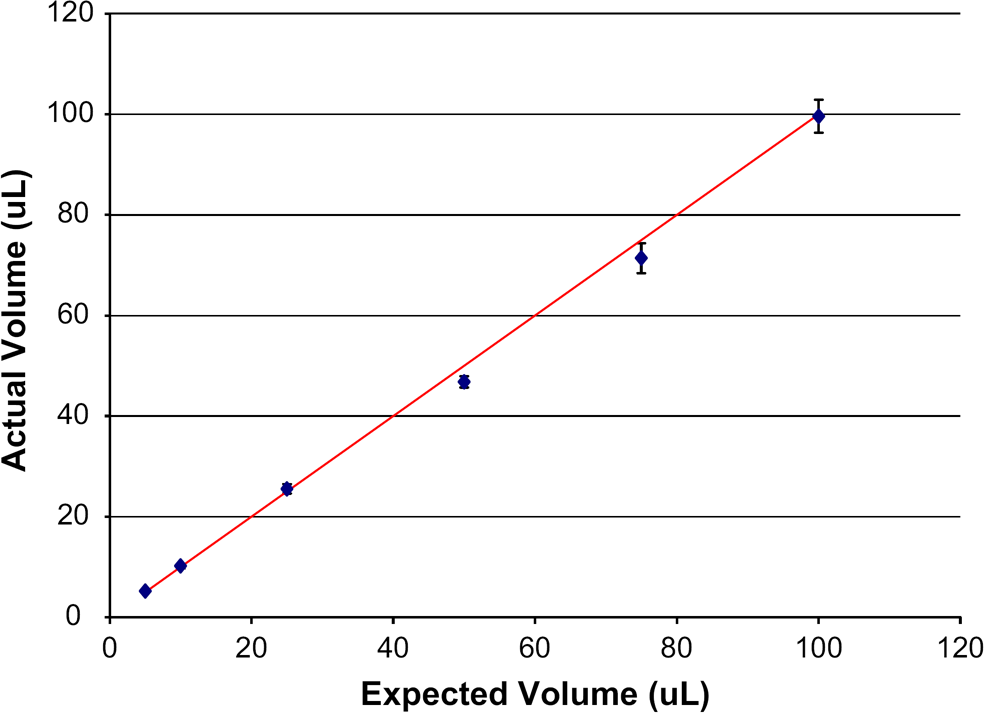
The execution time of each command for the QuadDispenser is summarized in Table 3. These results indicate that cycle times of less than 20 s for a 96-well microtiter plate can be achieved with this dispenser.
Timing performance for the QuadDispenser by command type

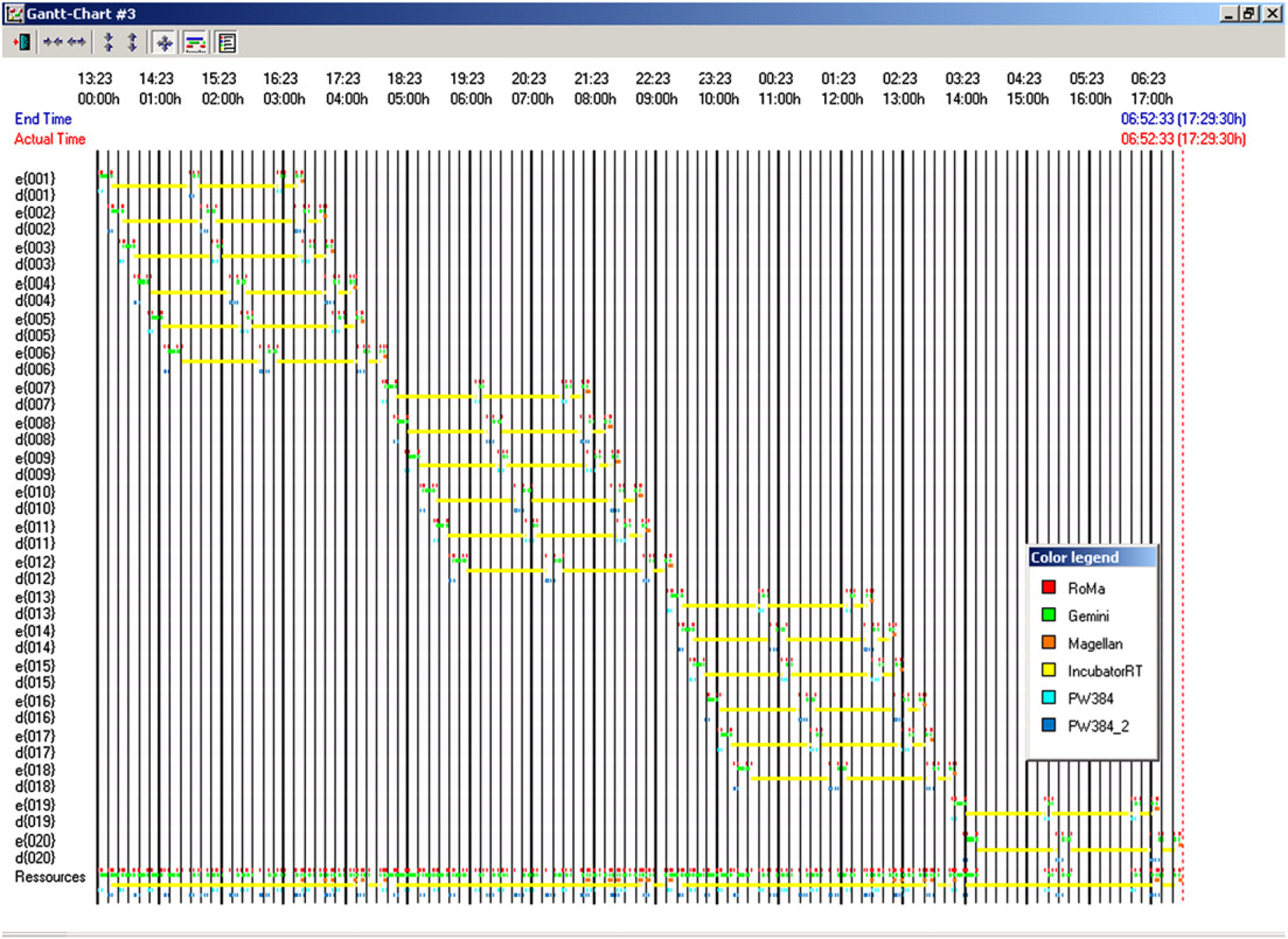
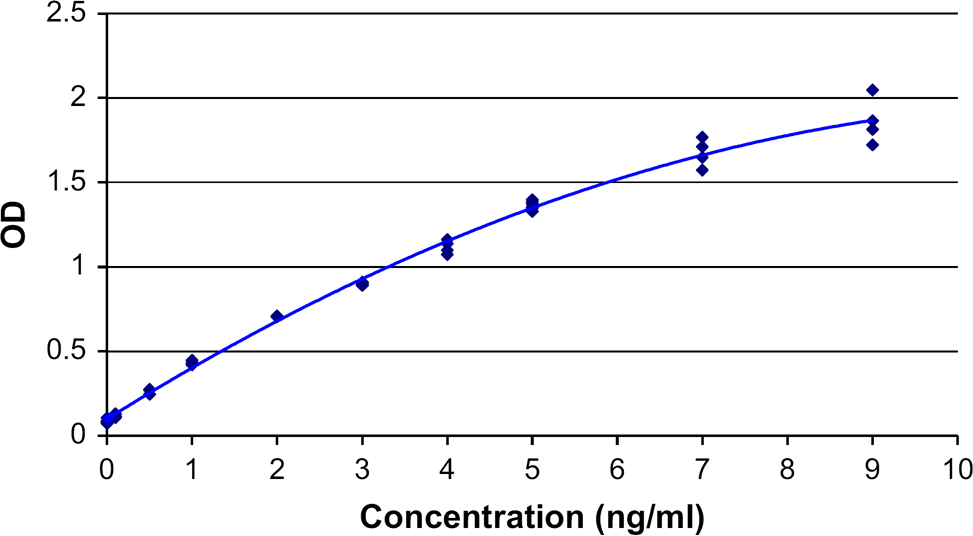
The impact of the QuadDispenser on the schedule of a 20-plate ELISA experiment is shown in Figures 6 and 7. With the QuadDispenser, a batch of 20 plates is completed within 8.5 h (Fig. 6) with the robotic arm being the limiting resource. Only three out of four dispensers are actually used in this experiment. Without the QuadDispenser, the same experiment takes more than twice the amount of time (Fig. 7). In the absence of a QuadDispenser, reagents are added using the eight-channel liquid handling arm. In that circumstance, the liquid handling arm becomes the limiting resource. The time savings for a 42-plate run was 15.5 h with the QuadDispenser versus 32 h without the QuadDispenser (not shown). Overall performance of the automated ELISA system, including the QuadDispenser, is illustrated in Figure 8, which shows the calibration curve derived from the standards of two ELISA plates. As expected, a monotonic increase in signal is observed over the measured concentration range with good agreement between replicates.
Gantt chart for a 20-plate ELISA assay with the QuadDispenser. Gantt chart for a 20-plate ELISA assay with no bulk dispenser. ELISA calibration curve using QuadDispenser. Two ELISA plates were incubated with increasing concentrations of a therapeutic protein and processed as described in Methods and Materials. The solid line represents a second degree polynomial fit of the data.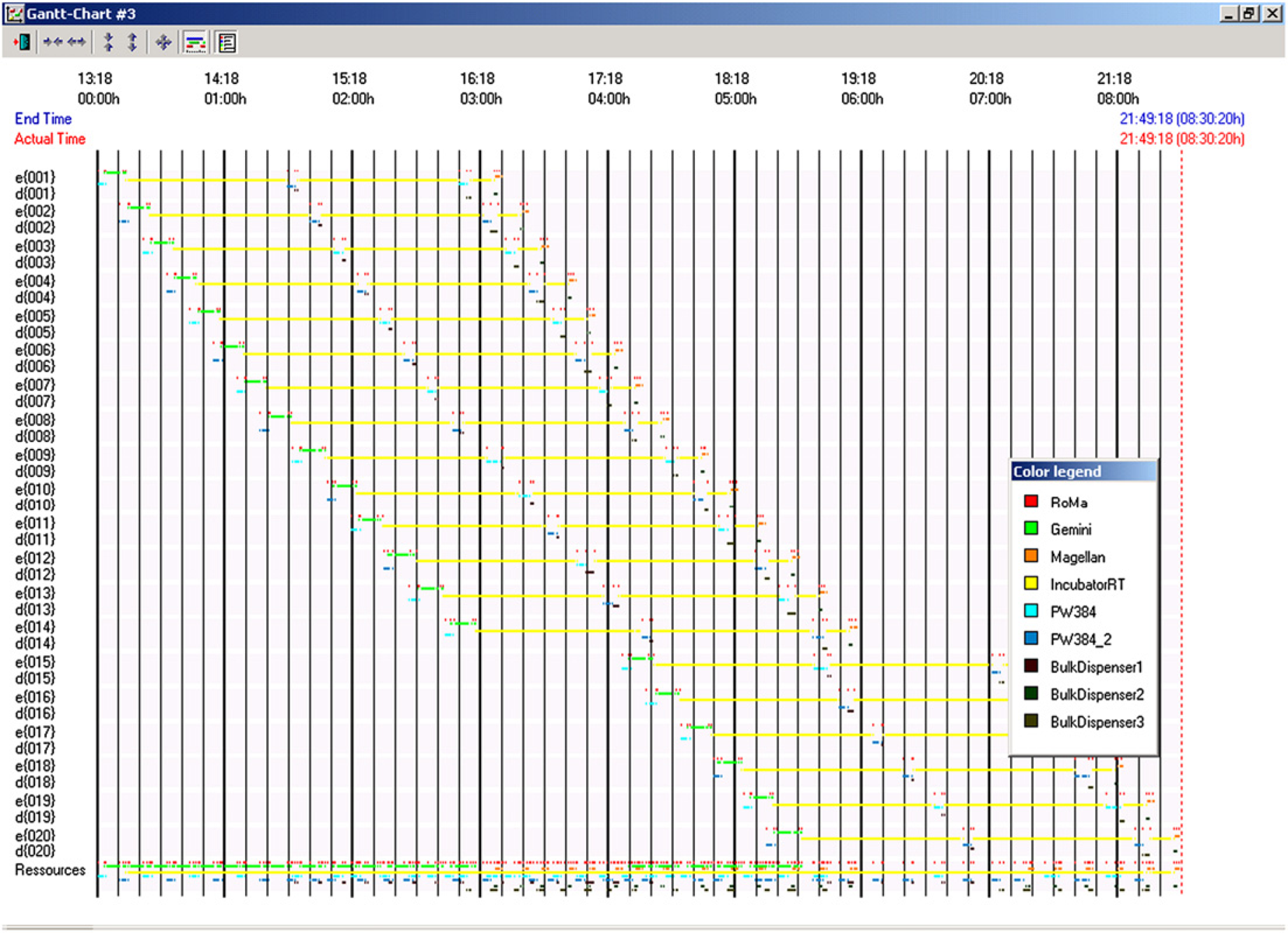
Conclusion
The compact bulk dispenser described provides significant benefits over existing commercial bulk dispensers with regard to footprint. This allows the integration of multiple, independent units in a compact space. This approach eliminates carryover and maximizes reagent preservation compared to bulk dispensers that use valves to switch between reagents. If applicable, the same reagent could be dispensed by multiple dispensers for increased throughput or redundancy. We have implemented the QuadDispenser on a workstation to perform unattended ELISA and cell viability experiments. The QuadDispenser allows us to achieve the required throughput without increasing the working envelope and complexity of the used workstation. We, therefore, conclude that the QuadDispenser is a versatile, space-saving bulk dispenser suitable for most plate-based assays requiring the addition of bulk reagents. Design modifications, such as having multiple manifolds per plate mover, could further broaden the utility of this concept.
