Abstract
We present a noncontact liquid dispenser that uses a disposable cartridge for the calibration-free dosage of diverse biochemical reagents from the nanoliter to the microliter range. The dispensing system combines the advantages of a positive displacement syringe pump (responsible for defining the aliquot’s volume with high accuracy) with a highly dynamic noncontact dispenser (providing kinetic energy to detach the liquid from the tip). The disposable, noncontact dispensing cartridge system renders elaborate washing procedures of tips obsolete. A noncontact sensor monitors the dispensing process to enable an online process control. To further increase confidence and reliability for particularly critical biomedical applications, an optional closed-loop control prevents malfunctions. The dispensing performance was characterized experimentally in the range of 0.25 to 10.0 µL using liquids of different rheological properties (viscosity 1.03–16.98 mPas, surface tension 30.49–70.83 mN/m) without adjusting or calibrating the actuation parameters. The precision ranged between a coefficient of variation of 0.5% and 5.3%, and the accuracy was below ±10%. The presented technology has the potential to contribute significantly to the improvement of biochemical liquid handling for laboratory automation in terms of usability, miniaturization, cost reduction, and safety.
Keywords
Introduction
During the past two decades, microfluidic applications that require the precise and accurate dispensing of liquid aliquots in the form of free droplets have grown rapidly and are used in many commercial applications today. Examples for the use of liquid droplets in life sciences are biochemical dispensing applications in drug discovery, in vitro diagnostics, high-throughput screening, combinatorial chemistry, microarrays, lab-on-a-chip systems, cell sorting, and bioprinting.
In vitro diagnostics is the diagnosis of disease in the form of assays in a test tube (i.e., in a controlled laboratory environment outside a living organism). By definition from the Food and Drug Administration, the goal of in vitro diagnostic devices is “the diagnosis of disease, or other conditions, including a determination of the state of health, to cure, mitigate, treat or prevent disease or its sequelae. Such products are intended for use in the collection, preparation and examination of specimens taken from the human body.” 1 For the different assays, such as immunoassays or molecular diagnostics assays, the sample liquid (i.e., pretreated body fluids) and also the respective reagents have to be handled and dispensed with high precision and accuracy down to a few percentages for aliquot volumes down to the sub-microliter range.
Drug discovery and drug development is one of the most important fields of biochemical dispensing today. Pharmaceu-tical companies and research facilities handle a large number of biochemical compounds and try to identify screening hits for potential drug candidates. The quantity of possible combinations is unmanageably high for manual liquid handling by, for example, hand pipettes. That is where high-throughput screening comes into play. In high-throughput screening, assays are performed in standardized containments called microplates. A continuing demand for increasing throughput has scaled biological assay procedures into 96-, 384-, and 1536-well formats.2–6 This, of course, requires a miniaturization of the assay volume: a single assay volume typically was in the range of 250 µL in 96-well microplates by 1995; in 2003, the same assay volume ranged from 50 to 2 µL. 4 Today, 384-well or even 1536-well microplates are state of the art, with assay volumes moving down to the nanoliter range. Another important field of application of liquid handling for drug discovery is combinatorial chemistry. Combinatorial chemistry synthesizes new compounds by using already known educts, which enables the efficient generation of large screening libraries for the needs of high-throughput screening. 7
Using microarrays for drug discovery pushes the advancement in assay miniaturization one step further. A microarray is a 2D grid of a multitude of immobilized reagents with aliquots down to the nanoliter or even picoliter range, enabling the detection of thousands of addressable elements in one single experiment.8–11
Other examples of modern biochemical liquid handling are lab-on-a-chip systems. Lab-on-a-chip systems (including lab CDs and miniaturized liquid chromatographs) enable a point-of-care analysis by combining micromachined structures, actuators, and sensors on a miniaturized and therefore mobile level.4,12–15 Droplets are used here to load reagents or samples and in rare cases for dispensing the final product. 16
Finally, cell sorting and bioprinting technologies can be used to isolate (single) cells from their surrounding and analyze and dispense them or, in other words, print them.17–20 A small droplet of several pico- to nanoliters can serve as a convenient carrier for cells and for dispensing them on different targets such as microplates, test tubes, or in the form of arrays on coated glass slides, for example.
Due to the wide range of tasks, diverse dispensing technologies have been developed to address different needs of specific fields of application.2,21 Dispensing systems can be categorized into contact and noncontact dispensers.
For each of the biochemical dispensing applications described above, a liquid handler faces a typical set of requirements. Among these are preventing cross-contamination, maximizing throughput, implementing an online process control, addressing the continuing drive for assay miniaturization, and being capable of handling liquids with diverse rheological properties.2,4,21 In a nutshell, these are the requirements the dispensing system presented in this work had to fulfill as further described in the following.
To acquire reliable data from an assay, it is crucial to exclude the possibility of cross-contamination during the highly automated liquid handling process. A very efficient way to prevent cross-contamination is to use noncontact dispensing systems with disposable tips. 4 Noncontact dispensing systems are carryover free per design since they do not need to contact the target to release the liquid aliquot. Using low-cost disposables, such as pipetting tips, eliminates the need to wash contaminated tips, which again reduces the risk of cross-contamination. Another advantage of using disposable tips or noncontact dispensing systems is that the omission of time consuming washing procedures can significantly increase the system’s throughput. This is especially beneficial for automated high-throughput applications.
In addition, applicants often call for an inline and/or online process control of the liquid handling process to improve the level of safety. Greatly desired is the quantitative volumetric measurement of each liquid aliquot while it is dispensed.4,22–25 Since nondestructive, quantitative measurements of small liquid volumes are difficult to realize and time consuming anyway, qualitative dispensing confirmation and failure detection26,27 are often accepted as means to increase the reliability of liquid handling processes. Online process control, however, requires sensors that again should make no contact with the liquid aliquot to prevent cross-contamination as well as loss of the liquid within the measurement process. Also, the dispensing process should feature a high level of automation to minimize sources of error and personnel costs.
Finally, a continuing drive for miniaturization2,4,5,28,29 calls for target volumes down to the sub-microliter range to be dispensed without compromising on volumetric precision and accuracy.
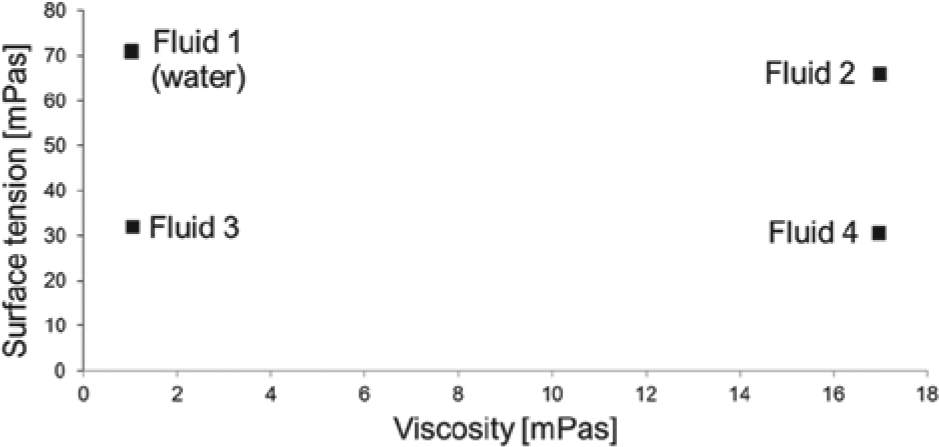
Considering these requirements, it is obvious that disposable, noncontact dispensing systems have very favorable properties for biochemical liquid handling applications. However, operating noncontact dispensing systems with high volumetric precision and accuracy—especially when using unknown liquids or liquids with very different rheological properties—is a challenging task. In contrast to inkjet printing, where special inks with known and optimized rheological properties are used, dispensing of biochemical liquids means handling fluids with very diverse properties.30–32 This prototype system was designed to handle approximately 600 different diagnostic reagents from a leading in vitro diagnostic company. Viscosity and surface tension of the liquid formulations range from 1.03 to 16.98 mPas and 30.49 to 70.83 mN/m (at 20 °C). To characterize the liquid handler’s dispensing performance for such a large portfolio of reagents, four representative test fluids were developed by Losleben et al. 31 These test liquids encompass the range of viscosity and surface tension as depicted in Figure 1 .
Prototype System, Materials, and Methods
To address the requirements described in the introduction, we have developed a noncontact dispensing system consisting of two main modules: a disposable dispensing cartridge with three reagent reservoirs and a base station consisting of actuators, a sensor, and control electronics. All low-cost polymer components that are contaminated by the reagent are assigned to the disposable cartridge (see Fig. 2 ). The base station (see Figs. 3 and 4 ) is considered a permanent, reusable (i.e., inline) part of the liquid handling prototype system.
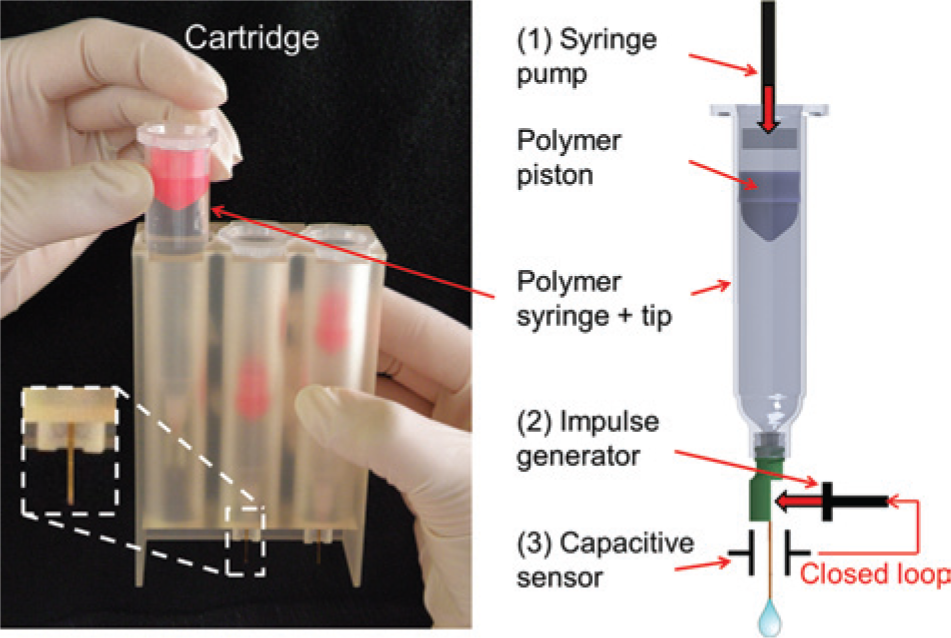
Left: The cartridge holding three syringes. Right: Sketch of a syringe with a polymer tip and piston. For dispensing, the syringe first displaces the target volume (1). Subsequently, the impulse generator detaches the droplet forming at the orifice due to low Weber numbers (2). The noncontact sensor monitors the detachment and retriggers the impulse generator if necessary (3).

Front and rear view of the prototype base station. The cartridge is not loaded (left).The touch panel for controlling and monitoring the dispensing process can be taken out of the base station and used as a handheld device.

The complete prototype system with a loaded cartridge. The aluminum cartridge fixture (left) provides space for three positions to contact one syringe at a time. In the left picture, tip 2 is contacted by the impulse generator and ready for dispense.
Cartridge
The cartridge (70 × 30 × 111 mm), shown in Figure 2 , is equipped with up to three polymer syringes that function as reagent reservoirs. Each distinct syringe reservoir (10-mL Optimum syringes from Nordson EFD, Pforzheim, Germany) is sealed by a polymer piston. A specially developed low-cost tip comprising a polymer nozzle (PipeJet dispensing pipe with a length of 18 mm and a diameter of 500 µm from BioFluidix GmbH, Freiburg, Germany) is connected to the outlet of the syringe by a standard Luer-Lock thread. All components of the cartridge feature an injection-moldable design to enable low-cost mass fabrication. The tip and the cartridge are currently fabricated by 3D rapid prototyping (material “Visijet EX 200”).
Base Station
The main components of the base station (240 × 283 × 443 mm) are a 14:1 neMESYS syringe pump from Cetoni GmbH (Korbußen, Germany), a piezoelectric noncontact PipeJet P9 dispenser 33 from BioFluidix GmbH, and a capacitive sensor described elsewhere 22 (see Figs. 3 and 4 ). The PipeJet P9 dispenser, referred to as an impulse generator hereafter, incorporates a mechanical impulse into the liquid column inside the tip by hitting the polymer nozzle with a piezoelectric plunger (stroke, 30 µm; impulses, 1). This mechanical impulse leads to the detachment of the liquid aliquot from the orifice of the tip at the end of the dispensing process.
The capacitive sensor consists of a standard printed circuit board with a sliced-through connection (via) with a diameter of 3 mm functioning as the sensing capacitor. The capacitor’s electrical stray field is affected by the liquid meniscus forming at the orifice of the tip. The resulting change in capacity is amplified by the electronics and sampled at a rate of 100 Hz for an online process control of the dispensing process. Additional components such as a miniaturized PC, pneumatic automation components, electronics, power supply, and a touch panel are integrated into the base station to make it work as an automated stand-alone prototype system.
Working Principle
To operate the system, the prefilled cartridge is placed into the base station manually. Hereby the nozzle self-aligns concentrically inside the sensing capacitor of the noncontact sensor. The sensor surrounds the nozzle concentrically approximately 2 mm above the nozzle’s orifice to prevent its contamination by dispensed droplets or jets. After the cartridge has been inserted, the operator initiates the dispensing process by a simple click on the touch panel (see Fig. 3 ) and by entering the target volume. Subsequently, the base station engages automatically with the cartridge by bringing the impulse generator and the syringe drive into mechanical contact with the cartridge. From here onwards, the base station processes the cartridge automatically and delivers and monitors the liquid aliquots as requested by the operator.
For dispensing a liquid aliquot, the syringe pump determines the volume by positive displacement of the piston inside the syringe body. Thus, the displaced volume 
In particular, it is independent of rheological properties of the respective liquid. The nominal positioning accuracy of the applied syringe pump is
The described dispensing process is calibration-free, meaning that the operator does not need to calibrate the system’s control parameters (stroke and stroke velocity of the syringe, parameters of the impulse generator, etc.) for a specific liquid by reference measurements. The only input parameter the operator has to enter is the desired target volume. The positive displacement principle makes the prototype system particularly insensitive toward temperature fluctuations that influence the liquid’s viscosity. The total dispensing process, shown in Figures 5 and 6 , typically takes 1.5 s. The actuators (syringe pump and impulse generator) and the sensor are not contaminated by the liquid during the dispensing process.

Phases of the dispensing process. The syringe pump delivers the liquid aliquot by positive displacement (phases A–C). After an idle time of 1.0 s (D), a pending droplet has formed at the orifice, mainly due to capacitive effects caused by the elasticity of the polymer syringe. Finally, the droplet is released (E) by the impulse generator. 37
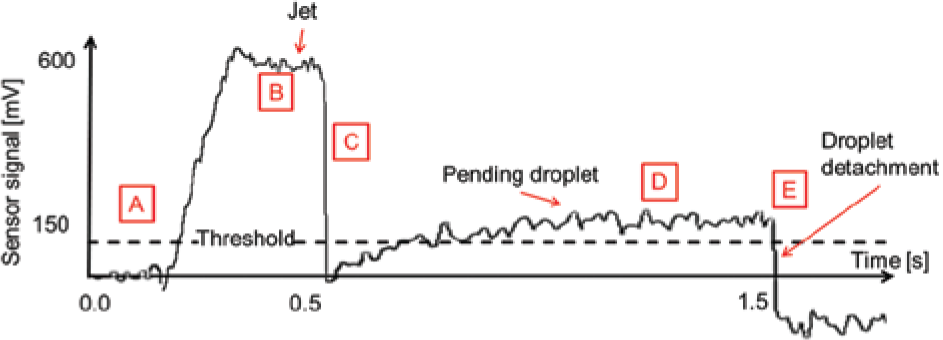
Exemplary diagram of the sensor signal corresponding to the phases of the dispensing process shown in Figure 5 . The threshold for detecting the presence and absence of a pending droplet (dashed line) can be adjusted in the graphical user interface.
The sensor at the nozzle (see Figs. 2 and 4 ) detects potential failures of the dispensing process such as clogging or failure of the droplet detachment. The threshold for detecting a successful dispense, indicated by the dotted line in Figure 6 , is set by the operator via a simple click onto a teach button on the graphical user interface displayed on the touch panel. If the closed-loop control also is activated, the impulse generator is retriggered if the droplet is not detaching properly. In this case, the impulse is reissued up to a maximum of five times within 1 s. Prior to each loop, the number of impulses per detachment attempt is increased by one (i.e., from one to a maximum of five impulses per detachment attempt). Also, the stroke of the piezoelectric plunger is increased from 30 to 34 µm. After five loops, the droplet detachment effort is stopped, and an error is reported and documented. In case of a successful droplet detachment, a successful dispensing process is confirmed, reported, and documented.
Figure 5 shows the different phases of the dispensing process as recorded by high-speed imaging (high-speed camera MC 3070 from Mikrotron GmbH [Unterschleissheim, Germany], with a frame rate of 5826 images per second, shutter time 20 µs). Phase A represents the initial state of the system. Between 0.0 and 0.5 s, the major part of the liquid volume is dispensed by the positive displacement of the syringe pump as a free continuous jet (phase B). The displacement velocity is adjusted by default to displace the complete target volume within 0.5 s. After the positive displacement stops (0.5 s, phase C), a pending droplet starts to form at the nozzle mainly due to capacitive effects caused by the elasticity of the polymer piston and syringe. Such mechanical effects in the hardware lead to a liquid flow for some time, even after the syringe drive has stopped (0.5–1.5 s, phase D). After approximately 1.5 s (phase E), the impulse generator detaches the pending droplet. The droplet sensor confirms the successful detachment by comparison of the sensor signal before and after the droplet detachment process within a predefined threshold. If the droplet is not detaching, the sensor detects a signal that does not drop below the threshold. In this case, appropriate measures can be taken (e.g., the system can issue a warning, stop proceeding, or retrigger the impulse generator within a closed-loop control until the still pending droplet is released as described above).
As expected from theory, the sensor signal differs for liquids with different dielectric constants. 37 To enable the online process and closed-loop control as described above for liquids with significantly different dielectric properties than water, the sensor calibration 22 can be adjusted by a simple manual potentiometer at the rear of the system (see Fig. 3 ). However, the dispensing process itself (i.e., dispensing aliquots without the online process control) requires no calibration at all.
Experimental Setup
The dispensed volume was measured gravimetrically using a “gravimetric regression method with reduced evaporation” (GRM-R) described elsewhere.
38
The GRM-R measurement method is a further development of the gravimetric regression method (GRM),
39
which compensates for evaporation of liquid during the measurement on the basis of a linear regression analysis of continuously monitored gravimetric results. In contrast to the GRM, the GRM-R operates at reduced evaporation by covering the prefilled weighing capsule with a silicon oil layer. This additional reduction of evaporation improves the GRM-R expanded measurement uncertainty at standard conditions to 3 nL for
The dispensing performance was evaluated with respect to the volumetric precision and accuracy within a run of 24 consecutive aliquots (Intrarun Coefficient of Variation [CV] and Intrarun Accuracy), among several syringe and tip assemblies (Tip-to-Tip CV and Tip-to-Tip Accuracy) and among several loading procedures of a single syringe and tip assembly (Load-to-Load CV and Load-to-Load Accuracy). Prior to each run, three predispenses were performed to increase precision by allowing the system to adapt to a set target volume. Intrarun and tip-to-tip performance assessment has been discussed by Bammesberger et al., 2 who give the precise mathematical definitions of the intra- and tip-to-tip quantities.
The load-to-load measurements are a sensible enhancement for the performance evaluation, especially for liquid handlers with automated loading mechanisms. For these experiments, the cartridge containing a prefilled syringe and tip assembly was removed and placed back into the base station 10 times for these experiments. After each insertion of the cartridge into the base station, the system contacts the syringe automatically by a loading procedure. This feature is called
Results
The volumetric intrarun precision and accuracy for a run of 24 individual aliquots is depicted in Table 1 . All test fluids 1 to 4 (see Fig. 1 ) were dispensed with identical dispensing parameters (as described above). The Intrarun CV ranged from 1.9% to 5.3% at 0.25 µL, 1.3% to 2.2% at 0.50 µL, 0.8% to 2.8% at 1.0 µL, and 0.5% to 0.6% at 10.0 µL. Thus, the relative precision improves with increasing target volumes. This can be explained since the influence of the positioning inaccuracy for displacing the syringe’s piston becomes less dominant for larger target volumes: the displacement stroke for a target volume of 0.25 µL is 1.3 µm versus 53 µm for 10.0 µL (see equation (1)). Hence, capacitive and elastic effects of the polymer components, slip-stick effects, and mechanical positioning inaccuracies become less dominant at larger volumes.
Intrarun Dispensing Performance for the Test Fluids 1 to 4 (in Percentages).
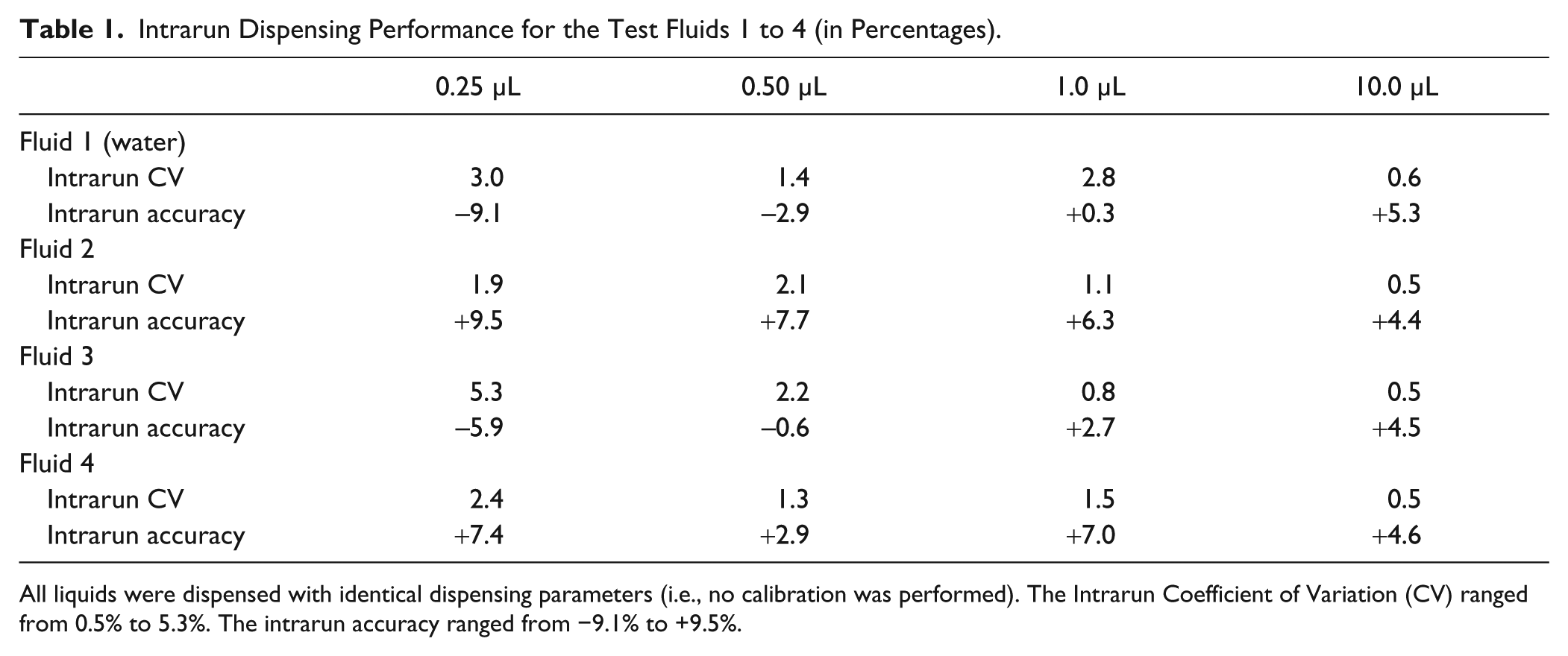
All liquids were dispensed with identical dispensing parameters (i.e., no calibration was performed). The Intrarun Coefficient of Variation (CV) ranged from 0.5% to 5.3%. The intrarun accuracy ranged from −9.1% to +9.5%.
The Intrarun Accuracy ranged from −9.1% to +9.5% at 0.25 µL, −2.9% to +7.7% at 0.50 µL, +0.3% to +7.0% at 1.0 µL, and +4.4% to +5.3% at 10.0 µL. Thus, the Intrarun Accuracy for the complete volume range under evaluation was less than ±10%. Again, it should be noted that these values were obtained without calibration. Mechanical tolerance of neither the syringe nor the syringe drive had been considered or corrected.
The tip-to-tip and load-to-load performance at a target volume of 1.0 µL with water (fluid 1) is shown in Figure 7 . For the tip-to-tip experiments, a new syringe and tip assembly was used after each run of 24 measurement points (i.e., aliquots) to evaluate variation stemming from the disposable components of the system. It turned out that the Tip-to-Tip Accuracy CV and the Tip-to-Tip Accuracy of the average volumes of 10 runs were both 2.3%. The load-to-load CV and the Load-to-Load Accuracy (relating to the variation caused by removing a cartridge from the prototype system and reinserting it again) were 0.7% and −0.3%, respectively. Thus, the dispensing performance across different tips or different loading operations is not significantly diminished if compared with the intrarun performance summarized in Table 1 . This means that neither fabrication tolerances for the cartridge nor the automated loading process introduce significant imprecison into the dispensing process and that the system operates with high reproducibility.
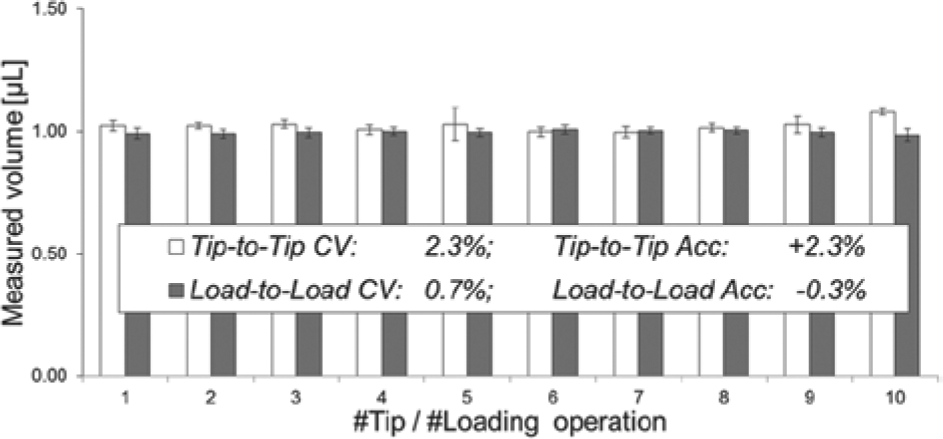
The tip-to-tip and load-to-load performance of the prototype system for a target volume of 1 µL and water (fluid 1). Each bar represents the measured mean volume of one run of 24 individual aliquots. The error bars represent the respective Intrarun Coefficients of Variation (CVs) ranging from 1.2% to 6.4% for the tip-to-tip runs and 1.3% to 2.5% for the load-to-load runs.
No failure of the droplet detachment was detected by the prototype system for the experiments reported above. This indicates a very reliable liquid handling process. Neverthe-less, detachment failures can still occur in rare cases, as shown by the example in Figure 8 . Usually, these errors result from a misalignment of the clamping of the tip by the impulse generator, which again results from a misalignment of the pneumatic linear stage moving the impulse generator or improper fabrication of the polymer tip. Alternatively, clogging due to crystallization of reagent at the orifice can be the cause of malfunctions. In the lower graph of Figure 8 , the system detects that the sensor signal does not drop below the threshold (dotted line) during the dispensing of 0.25 µL of the highly viscous fluid 2 (viscosity of 16.98 mPas). Consequen-tly, the closed-loop control increases the stroke parameters stepwise from a stroke of 30 to 34 µm and the number of impulses per detachment attempt from 1 to 5. Since the signal does not drop at the end of loop 5, an error is reported and documented.
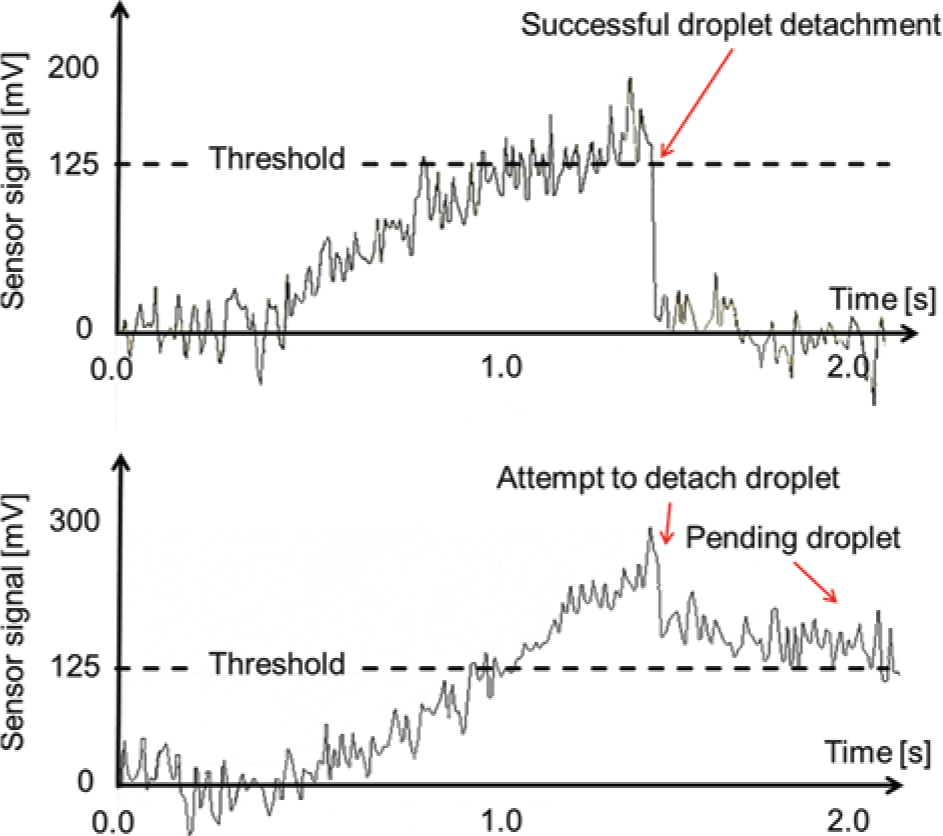
Two sensor signals (sample rate 100 Hz) for 0.25-µL aliquots of fluid 2. Top: A successful droplet detachment is confirmed by the detection algorithm due to the drop of the sensor signal below the threshold at 1.5 s. Bottom: A failure of the droplet detachment process is detected since the signal level (≈150 mV) does not drop below the threshold. Consequently, an error is reported.
Discussion
The presented prototype system enables calibration-free dispensing of liquid aliquots from the nanoliter to the microliter range. The novel technology combines the benefits of a highly accurate and precise positive displacement syringe pump with a highly dynamic noncontact dispenser. The intrarun dispensing performance was very precise (Intrarun CV, 0.5%–5.3%) and accurate (Intrarun Accuracy, less than ±10%) for test fluids with widely varying rheological properties (viscosity, 1.03–16.98 mPas; surface tension, 30.49–70.83 mN/m). The prototype system’s minimal target volume is 0.25 µL, which was dispensed with an Intrarun CV (Intrarun Accuracy) of 1.9% to 5.3% (less than ±10%). This is a surprisingly precise dispensing performance, taking into account that a specified positioning accuracy of the syringe pump of 70 nm alone would correspond to a volumetric variation of the aliquot’s volume of 5.2% at 250 nL (a syringe displacement accuracy of 70 nm corresponds to 13 nL in terms of displacement volume; see equation (1)). The Tip-to-Tip CV at 1 µL was 2.3%, which indicates little variation of volumetric dispensing precision across different tips. Thus, fabrication tolerances of the inner diameter of the syringe or the nozzle do not introduce significant imprecision into the liquid handling process. The Load-to-Load CV at 1 µL was 0.7%, showing that the automated contacting and priming of the cartridge is highly reproducible.
The dispensing performance of the prototype system was characterized on the basis of the measuring plan published by Bammesberger et al. 2 This measuring plan, evaluating dispensing performance on an intra-run and tip-to-tip level, is specifically designed to enable a comparable and objective assessment of dispensing performance of different dispensing technologies. A study that characterizes and compares five commercial dispensing systems for laboratory automation applications, also published in Bammesberger et al. 2 , yielded an Intrarun CV from 0.4% to 7.7% and a Tip-to-Tip CV between 1.4% and 9.9% for target volumes <1 µL. An absolute accuracy of better than ±5.0% was evaluated to be generally difficult to achieve in the sub-microliter range, even with these nondisposable dispensing technologies. Furthermore, it is important to note that each commercial dispensing system was characterized with water and DMSO (viscosity, 4.37 mPas; surface tension, 51.53 mN/m) only, whereby the dispensing parameters were calibrated to the respective rheological properties. The prototype system presented in this study features a dispensing performance for a wide range of rheological properties that is—in addition to being calibration-free—comparable if not superior to commercial dispensing systems.
The cartridge is made of injection-moldable low-cost polymer components (i.e., it is fully disposable, rendering elaborate washing procedures obsolete). This feature, in combination with the noncontact dispensing process, makes the liquid handler carryover free per design. Furthermore, there are no moving parts or complex features involved, which makes the fabrication of the disposables very cost-efficient and certainly contributes to the robust performance.
A capacitive noncontact sensor enables an optional online process control that can also be used for the purpose of a closed-loop control. Both features require in principle the adjustment of the sensor signal to the dielectric properties of the used liquids. The online process control confirms and documents each successful aliquot or detects and documents failures. The closed-loop control increases the impulse generator’s parameter stepwise to detach a pending droplet in case an unsuccessful droplet detachment was detected. Both features further increase the confidence and reliability of the dispensing process, especially for critical biomedical applications.
With the ability to hold up to three individual low-cost syringe reservoirs per cartridge, each disposable cartridge is capable of performing most assays used in, for example, in vitro diagnostic applications. Thus, the prototype system features highly accurate and precise liquid dispensing for reliable, low-cost, and calibration-free reagent handling in the in vitro diagnostic automation systems or for similar biochemical or industrial automation applications.
Footnotes
Acknowledgements
This work was developed within the “Smart Reagent Dosing” project embedded in the cluster MicroTEC Südwest, Germany.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge financial support from the German Federal Ministry for Science and Education (BMBF) (SFK 16SV5119).
