Abstract
We planned an original study to investigate the morphological changes caused by spurious hemolysis of whole-blood samples, analyzed using an automated image analysis system. Seven whole-blood specimens anticoagulated with EDTA were pooled and divided in two aliquots. The former was left untreated, whereas the latter was subjected to mechanical hemolysis by forced aspiration with an insulin syringe. The complete blood cell count was performed on a Sysmex XE-2100, and the aliquots were then processed with CellaVision DM96. In spuriously hemolyzed samples, the main findings included a rarefaction of erythrocytes, the presence of a remarkable number of cellular debris, a greater degree of microcytosis and anisocytosis, the appearance of band neutrophils, a shift of values between lymphocytes and monocytes, and an increase in smudge cells, artifacts, and large platelets. The results of this study demonstrate for the first time that blood cell morphology may be consistently biased in spuriously hemolyzed whole blood and that the use of an automated image analysis system such as the CellaVision DM96 may be a suitable approach to identify spurious hemolysis in whole-blood specimens.
Introduction
Spuriously hemolyzed specimens represent the leading source of preanalytical problems in clinical laboratories, with a prevalence that can be as high as 3% of all samples received for routine and urgent testing. 1 The presence of cell-free hemoglobin in diagnostic blood samples is always challenging, because results of several clinical chemistry, immunochemistry, hemostasis, and even hematological tests may be biased.1,2 Nevertheless, since no information is available on morphological abnormalities that may appear in spuriously hemolyzed whole blood to our knowledge, we planned an original study to investigate these potential changes with an automated image analysis system and to establish whether an automated image analysis system may be a suitable approach to identify spurious hemolysis in whole-blood specimens.
Materials and Methods
This study was based on seven random samples collected into 13 × 75–mm, 2.0-mL blood tubes and containing 3.6 mg spray-dried K2EDTA (BD Vacutainer Plus; Becton Dickinson Italia S.p.A., Milan, Italy), which were referred to our laboratory for routine hematological testing. After routine analysis had been completed, the whole blood of the specimens was pooled and divided into two aliquots. The former aliquot was left untreated, whereas the latter was subjected to mechanical hemolysis by forced aspiration with an insulin syringe equipped with a 30-gauge, 0.3 × 8–mm needle (Picindolor; Artsana S.p.A., Grandate, Italy) to generate a mechanical injury of blood cells. 2 The complete blood cell count (CBC) was performed using a Sysmex XE-2100 (Sysmex, Mundelein, IL), and the aliquots were then processed with a CellaVision DM96 automated image analysis system (Sysmex). The CellaVision DM96 is an automated device for differential enumeration and characterization of leukocytes and erythrocytes, consisting of a slide feeder unit, a microscope with three objectives, a camera, and a computer equipped with acquisition and classification software.3,4 The cell-free hemoglobin concentration (i.e., hemolysis index) was also measured on centrifuged plasma with a Beckman Coulter DxC (Beckman Coulter, Brea, CA).
Results
In agreement with previous data, 2 the process of mechanical injury caused a substantial degree of hemolysis in pooled whole blood, as reflected by an increase of cell-free hemoglobin plasma concentration from 0.5 to 3.5 g/L. The most relevant differences in the CBC were represented by a substantial reduction of red blood cell (RBC) count (–22%), hematocrit (–23%), reticulocytes (–20%), and monocytes (–31%) ( Table 1 ). The hemoglobin value, the white blood cell (WBC), and the platelet counts were not consistently affected. The spurious lysis also caused a reduction in mean corpuscular volume (MCV), combined with a remarkable increase in mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCH), mean platelet volume (MPV), and plateletcrit. Both the RBC distribution width (RDW) and the platelet distribution width (PDW) increased after hemolysis. A modest increase was also observed in neutrophil count, which was not completely explained by the decrease of monocytes (i.e., +0.14 vs. –0.26 × 109/L). Interestingly, the number of RBC fragments on the XE-2100 remained mostly unchanged. The CBC on hemolyzed blood was also characterized by the appearance of specific flags (i.e., RBC agglutination and turbidity/hemoglobin interference), which were instead absent in nonhemolyzed blood.
Complete Blood Cell Count and CellaVision DM96 Data of a Normal Blood Sample and a Paired Specimen after Spurious Hemolysis.
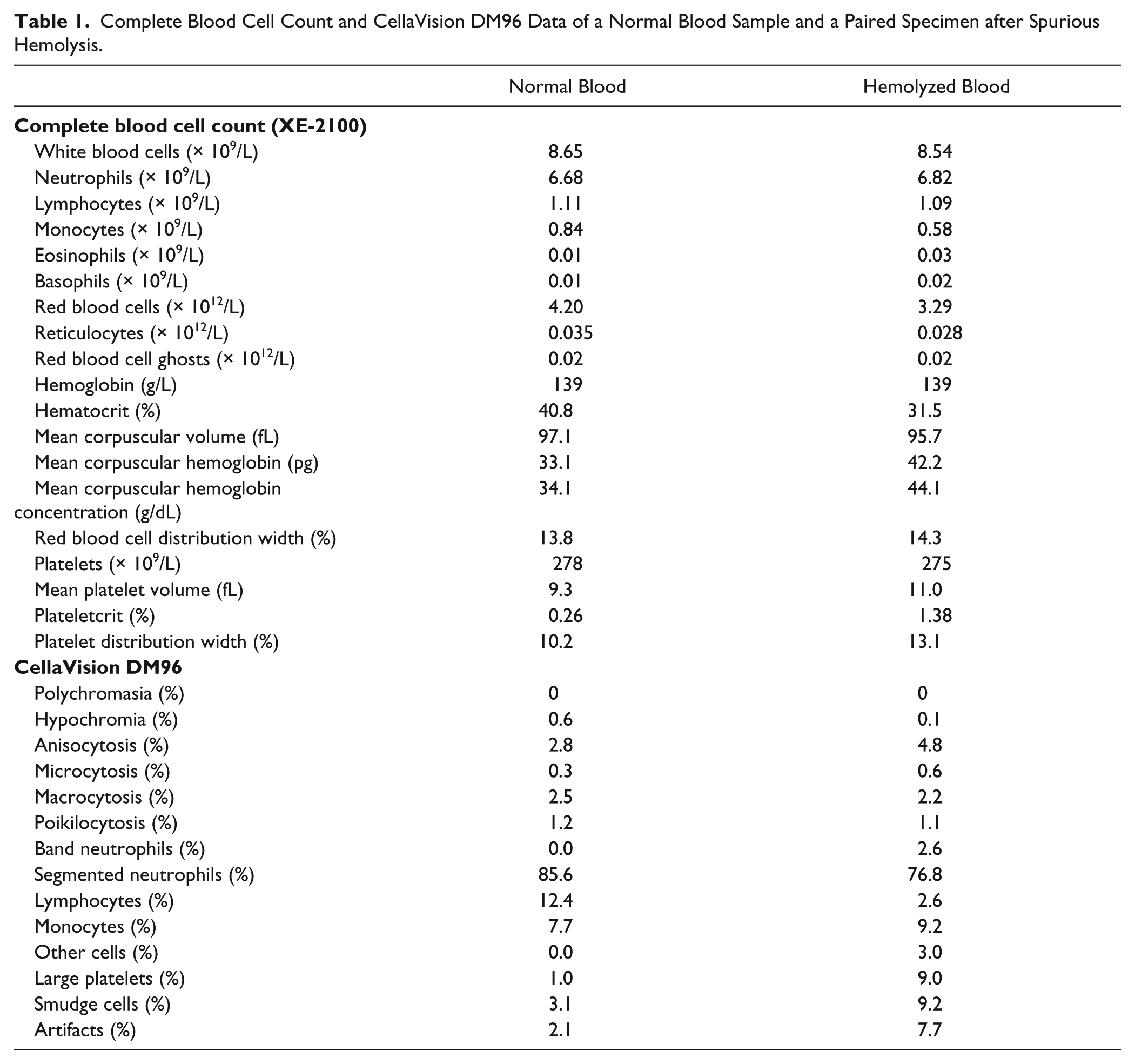
Most of these changes were mirrored by the onset of abnormalities in blood smear analysis of the spuriously hemolyzed aliquot ( Fig. 1 ). The main finding was indeed represented by a rarefaction of RBCs, attributable to the breakdown of erythrocytes and blood film preparation, which is dependent on the relative density of cells. 4 Another interesting finding was the presence of a remarkable number of cellular debris, which probably originated from injured RBCs. As reflected by an increased RDW value, a greater degree of microcytosis and anisocytosis (but not of poikilocytosis) could also be appreciated in hemolyzed blood. Changes in platelet morphology (i.e., increased size and heterogeneous shapes) were also evident. The analysis of RBC morphology in hemolyzed blood by CellaVision DM96 software was consistent with these findings, displaying a greater degree of anisocytosis and microcytosis, a lower degree of hypochromia and macrocytosis, and a substantially unchanged degree of both polychromasia and poikilocytosis ( Table 1 ). Despite the presence of a specific flag on the XE-2100, no evidence of RBC agglutination could be observed in the blood smear. With regard to the WBC subpopulations, remarkable differences emerged by CellaVision DM96 software analysis of hemolyzed blood, with the appearance of band neutrophils, considerable shift of values between lymphocytes and monocytes, and a remarkable increase in smudge cells, artifacts, other cells (i.e., metamyelocytes), and large platelets.
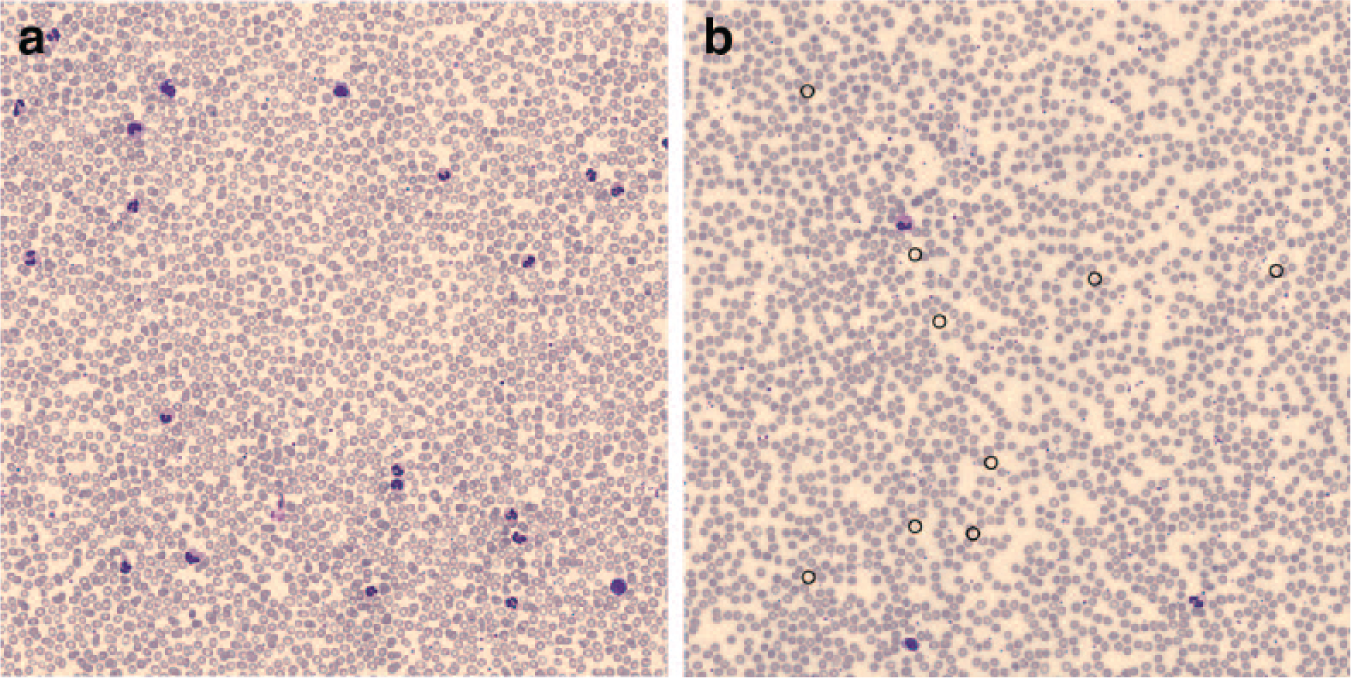
Blood smears obtained with the CellaVision DM96 in a normal blood sample (
Discussion
The results of this study have two meaningful implications. First, we have demonstrated for the first time that blood cell morphology may be consistently biased in spuriously hemolyzed whole-blood specimens, and we also have confirmed that the use of specific parameters such as RBC ghost may be unsuitable to screen for spurious hemolysis, 5 even in samples with more than 20% hemolysis. Most of the changes observed in the CBC were reflected in CellaVision DM96 software analysis, but none could be considered really exclusive. Interestingly, significant morphological abnormalities could be observed in the RBC population, with a higher degree of anisocytosis and microcytosis, as well as the presence of a remarkable number of cellular debris. Platelet alterations (increased size and shape changes) were also evident in hemolyzed blood and are consistent with platelet hyperactivation consequent to mechanical injury of blood, a phenomenon that has also been previously appreciated using a platelet function analyzer. 6 The appearance of abnormal and smudge cells also impaired the accurate characterization of WBCs. We can hence conclude that spurious hemolysis may consistently impair the morphological analysis of blood cells. The second important finding of this study is that we have proven that an automated image analysis system such as the CellaVision DM96 may be effective in identifying the presence of hemolysis in whole-blood specimens referred for the CBC. This is noteworthy, since the only available means so far to check for these important preanalytical variables has been represented by centrifugation of the specimens after analysis, a practice that is essentially unsuitable or unpractical, especially for those clinical laboratories that process a large volume of samples. 7 Interestingly, the minor yet significant decrease of the MCV value recorded with the Sysmex XE-2100 has a plausible explanation and is prevalently attributable to a loss of membrane integrity accompanied by exocytic vesiculation, which is typical of a mechanical erythrocyte injury.8,9 It is also noteworthy that we failed to find significant differences in the number of RBC ghosts, and this is reasonably attributable to the previously reported low sensitivity of this parameter for identifying erythrocyte injury. 5 In the Sysmex-2100, RBC ghosts typically appear in the “4DIFF,” “WBC/BASO,” and “NRBC” scattergrams as elements with scarce side fluorescence scatter and modest scattered light, after lysis of RBCs with a specific reagent. It is hence predictable that mechanical hemolysis, such as that used in this study, would generate RBC fragments of such a limited size to be ultimately misclassified as platelets by impedance reading rather than as RBC ghosts. This hypothesis is confirmed by the virtually identical number of platelets obtained with optical reading counterbalanced by the nearly 5× increase in the impedance-based plateletcrit measurement in hemolyzed blood ( Table 1 ). The relative decrease of the monocyte count obtained with the Sysmex XE-2100, but not with the CellaVision DM96, is another important finding. It is hence likely that mechanical injury of blood would generate morphological changes in these cell elements to impair their typical fluorescence scatter but not the morphological recognition by automated image analysis systems. Since there are no previous data about the use of this system in patients with various forms of hemolytic anemia or other similar conditions, we cannot definitely conclude that the morphological aberrancies observed are specific for hemolysis, and further studies will be needed to clarify this issue. It is also noteworthy that our experimental protocol was based on whole blood pooled from seven random patients’ samples. As such, although the morphological aberrancies that we have observed should be considered reliable and reproducible, they may not be representative of any potential hemolyzed sample received in a clinical laboratory.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
