Abstract
To assess the impact of preanalytical variables of time and temperature on prothrombin time (PT), activated partial thromboplastin time (aPTT), dilute Russell viper venom time (DRVVT), activated protein C resistance (APCR), and
Introduction
Coagulation assays performed by clinical laboratories are integral tests which guide vital patient care decisions in multiple medical disciplines including hematology, cardiology, and hepatology to name a few. The University of Texas Medical Branch (UTMB; Galveston, Texas) completes an average of 5 000 tests per month; of these, approximately 55% are received from satellite clinics. The furthest off-site clinic is located approximately 120 miles from the central laboratory necessitating ground transportation time of approximately 3 to 4 hours. Current guidelines for coagulation testing require stringent specimen transportation and storage conditions that challenge laboratories receiving specimens from distant sites. The decentralization of phlebotomy services and the multitude of off-site clinics sending specimens to our central laboratory compel the standardization and validation of guidelines to ensure the integrity of samples and accuracy of results.
Current Clinical Laboratory Standard Institute (CLSI) guidelines require sample processing to be completed within 24 hours of collection for prothrombin time (PT) and 4 hours for activated partial thromboplastin time (aPTT) when stored at room temperature as whole blood or plasma aliquots.
1
Storage for
Lack of consensus in the literature has led to many different recommendations for processing PT and aPTT. Some studies have considered PT testing in plasma or whole blood acceptable for up to 24 hours at room temperature or 4°C, 4 whereas others recommend only 6 hours under the same conditions. 5 Similarly, processing conditions for aPTT have also been debated. Studies have shown aPTT to be stable for up to 8 hours at room temperature or 4°C 6 and ambient temperature, 7 while others extended the acceptable time interval to 12 hours at room temperature or 4°C. 4 In addition, the majority of related studies obtained blood samples from healthy volunteers, while few studies have analyzed the effect of storage conditions on samples collected from individuals with elevated PT and aPTT values. 4,8 Furthermore, the validation of dilute Russell viper venom time (DRVVT) and activated protein C resistance (APCR) ratio following storage at −20°C is limited or nonexistent.
The goal of our study was to assess the preanalytical variables of time and temperature on 5 common coagulation studies in order to determine whether the storage of plasma at −°20°C causes clinically relevant assay changes using data from both healthy volunteers and individuals with elevated PT and aPTT levels. Analytes studied included PT, aPTT, DRVVT, APCR, and
Material and Methods
Blood specimens were collected from 23 healthy individuals without known coagulation defects or receiving anticoagulants or antiplatelet medications. Blood specimens were also taken from 18 patients with a known abnormal PT and aPTT (henceforth referred to as the “coagulopathic group”). Participants with coagulopathy included individuals receiving vitamin K antagonist (VKA) therapy with warfarin who underwent international normalized ratio (INR) monitoring at an anticoagulation clinic (n = 15) and patients hospitalized for liver disease (n = 3) at the UTMB, Galveston, Texas. The study was approved by the UTMB Institutional Review Board, and informed consent was provided by all participants.
Whole blood samples were drawn from peripheral venipuncture sites in five to seven 2.7 mL siliconized evacuated tubes containing 3.2% buffered trisodium citrate (Becton Dickinson, Franklin Lakes, New Jersey). Blood samples from each individual were gently pooled, mixed, and divided into 5 equal aliquots using polypropylene tubes (Thermo Scientific, Massachusetts). The samples were then centrifuged at 1500 g for 15 minutes at room temperature to separate the plasma. For each individual, the first 2 aliquots were stored at room temperature and analyzed at 2 or 4 hours post-collection. The remaining aliquots were stored at −20°C for 48 hours, 1, or 2 weeks in a temperature-monitored, frost-free freezer (range: −18°C to −25°C). The frozen samples were thawed in a water bath for 5 minutes at 37°C before analysis. Platelet counts were measured periodically in batches after centrifugation to ensure a count less than 10 × 109/L. The PT, aPTT, DRVVT, APCR, and
Mean and standard deviations of all assays were calculated for each time point. The 2-hour sample was considered to be the baseline. A paired t test was used to compare the 4-, 48-hour, 1-, and 2-week results to the baseline 2-hour sample. A P value of less than .05 was considered to be significant. Percentage change compared to the baseline 2-hour sample was calculated and considered clinically insignificant if less than 10%.
Results
Healthy Participants
In the healthy group, PTs were within normal ranges for each time interval for all 23 participants. Changes seen at 48 hours (12.9 ± 0.7 seconds; mean ± SD), 1 week (12.6 ± 0.6 seconds), and 2 weeks (12.9 ± 0.7 seconds) compared to baseline (12.5 ± 0.7 s) were statistically significant; however, the percentage changes for all data were less than 8% (therefore clinically insignificant), and all results were concordant (Table 1). No significant changes were observed for aPTT up to 1 week. Two patients had slightly elevated aPTT values (41.8 and 45.1 seconds) when assayed at 2 weeks; however, the mean percentage change was less than 10%. No significant changes were observed for DRVVT at any time point, and percentage change was less than 3% for 4-hour assays but varied from 0% to 26.5% for 48-hour through 2-week results. Dilute Russell viper venom time values converted from normal range to borderline high levels in 2 participants: 1 individual had elevations at 1 and 2 weeks (50.2 and 42.2 seconds, respectively), and the second individual had mild elevations at 48 hours and 2 weeks (43.2 and 43.8 seconds, respectively). In the healthy group, APCR was within normal ranges at each time point for 21 participants. A statistical decrease was observed at 48 hours (2.5 ± 0.2) and 2 weeks (2.4 ± 0.2) compared to baseline (2.6 ± 0.3), but these changes did not affect the interpretation of assay results. For
Mean and Mean Percentage Change in Coagulation Studies Stored at −20°C for 2 Weeks.
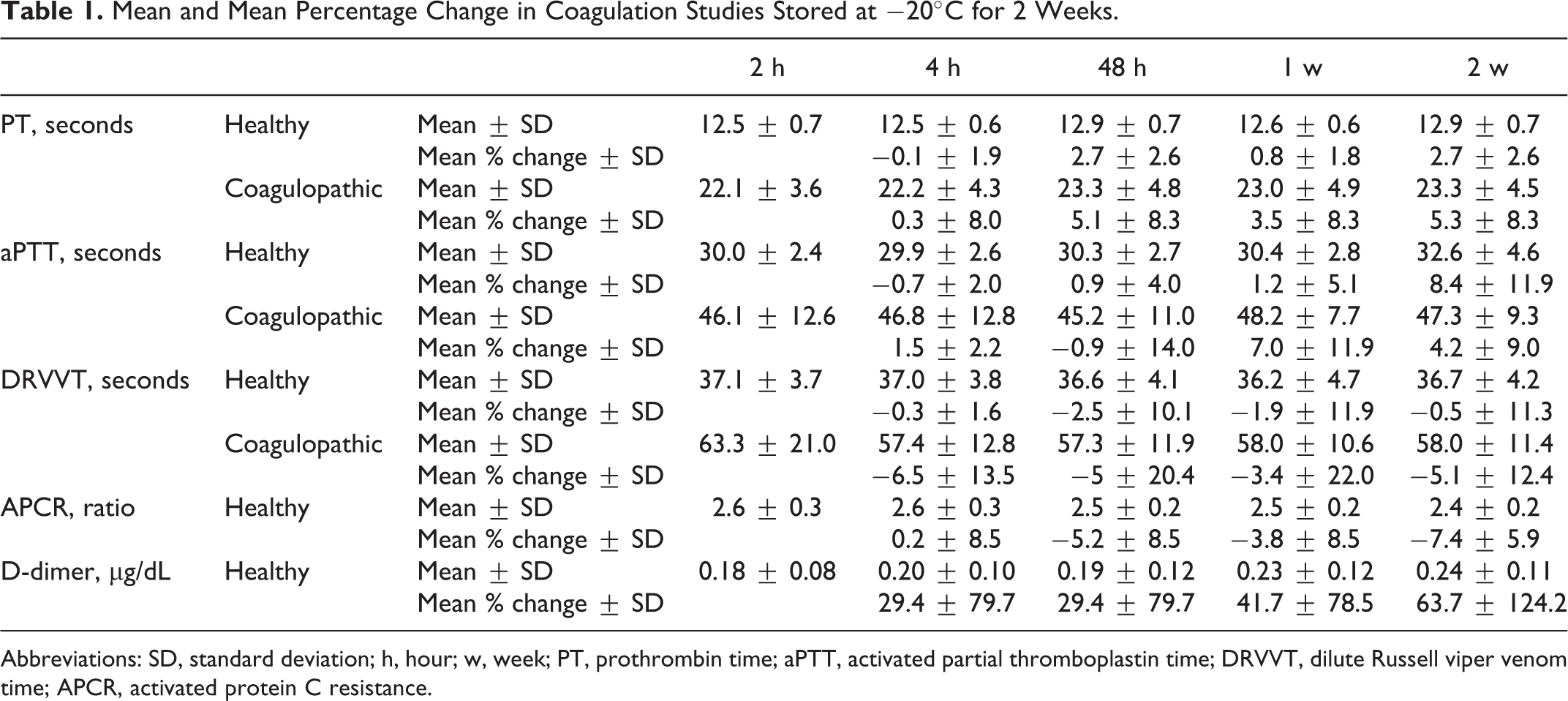
Abbreviations: SD, standard deviation; h, hour; w, week; PT, prothrombin time; aPTT, activated partial thromboplastin time; DRVVT, dilute Russell viper venom time; APCR, activated protein C resistance.
Participants With Coagulopathy
Participants with subtherapeutic VKA therapy attending the outpatient coagulation clinic were excluded from statistical analysis. The remaining participants had elevated PTs with a 2-hour baseline of 22.1 ± 3.6 seconds. There was a statistically significant prolongation occurring only at 48 hours (23.3 ± 4.8 seconds). One individual had a 2-hour baseline PT of 15.3 seconds converted to 14.7 seconds (the upper limit of normal reference range) at 4 hours and 48 hours. Overall percentage change did not exceed 10%. Activated partial thromboplastin time was statistically prolonged from baseline (46.1 ± 12.6 seconds) at 4 hours (46.8 ± 12.8 seconds), but all 4-hour assays had a percentage change less than 7%. A single result for aPTT dropped within the normal range at 48 hours (25.3 seconds). The discordant results seen with PT and aPTT would impact the clinical management of the participants.
For 3 individuals receiving VKA therapy and 1 patient with liver disease, DRVVT results were within the normal reference range and not included in statistical analysis. No significant differences in DRVVT occurred at any time point. In the coagulopathic group, APCR was within normal ranges at each time point for all participants with a baseline mean comparable to the healthy group (2.6 ± 0.3), and no significant changes were observed at any time point. Of the 3 patients with liver disease, 2 had elevated
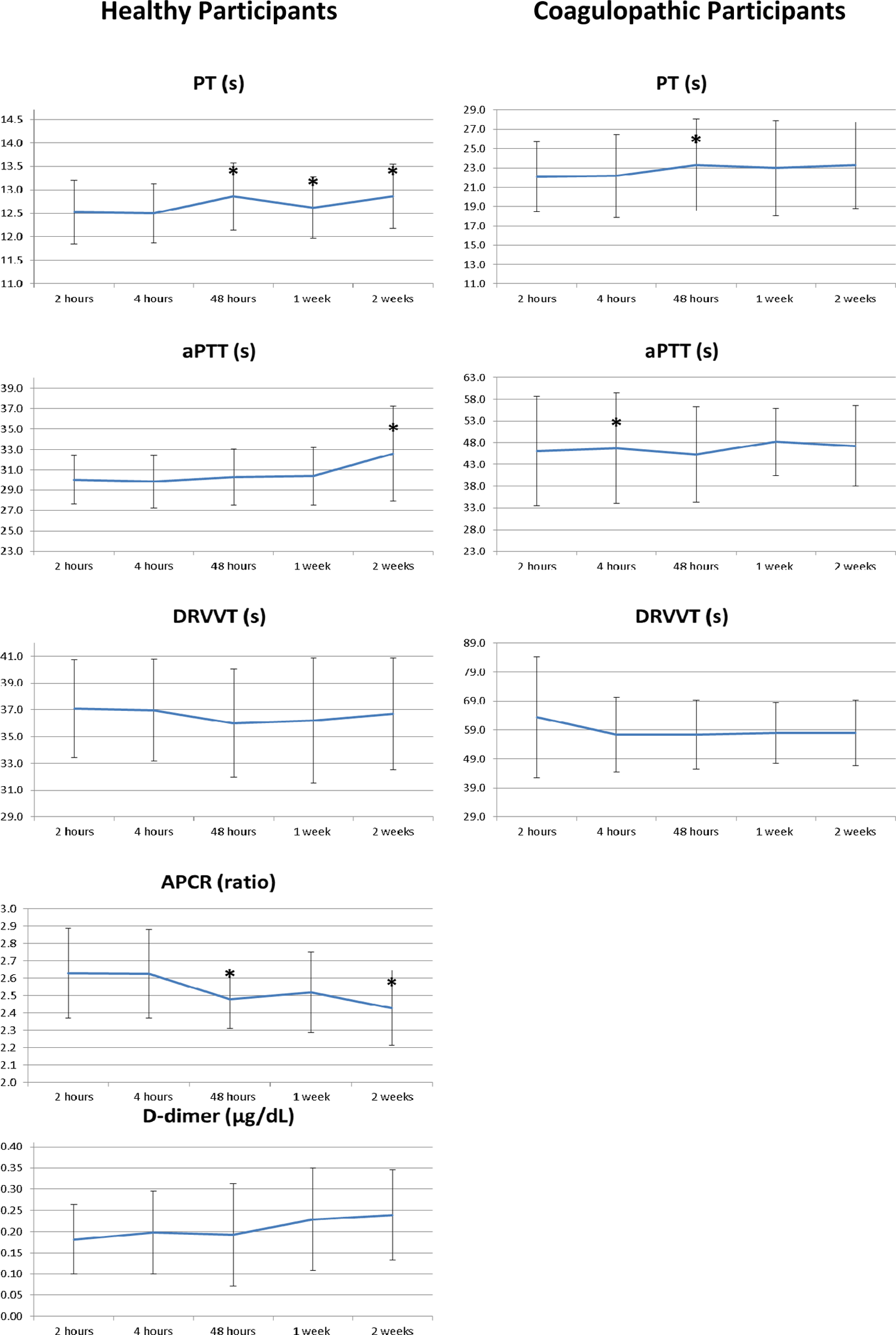
Coagulation assay stability following storage at −20°C for 2 weeks in healthy participants and participants with coagulopathy. Results reported as mean and standard deviations. *P < .05.
Discussion
Detailed guidelines for correct labeling, patient positioning, phlebotomy technique, volume collection, tube mixing, inspection for hemolysis or clotting, and order of blood draws have been developed to ensure preanalytical factors do not compromise samples prior to processing. 9 Following collection, blood must be stored and transported often requiring samples to travel great distances from satellite clinics to central laboratories. Our study evaluated the preanalytical variables of temperature and duration of storage on the stability and validity of assay results for 5 common coagulation tests after freezing of samples at −20°C for up to 2 weeks. The CLSI guidelines indicate storage at −20°C is acceptable for samples processed within 2 weeks given other collection and temperature monitoring standards are followed. 1 When logistics delay processing beyond 4 hours from collection, samples must first be centrifuged and aliquoted into secondary polypropylene tubes, stored in a −70°C freezer or a temperature monitored −20°C freezer and then transported on dry ice or in an acceptable freezer to the final destination. 10
Prothrombin time and aPTT are 2 routinely ordered assays assessing the timing of fibrin clot formation, and generally, elevated PT and/or aPTT reflects a potential for bleeding. Prothrombin time evaluates the extrinsic coagulation cascade and is delayed in patients with vitamin K deficiency, VKA therapy, and factor VII deficiency. The INR was developed to standardize coumadin monitoring among different manufactures by raising the ratio of the patient’s PT to a reference sample by the power of the international sensitivity index, which is uniquely assigned to each thromboplastin reagent. Similarly, aPTT evaluates the intrinsic coagulation cascade and is prolonged by heparin and deficiencies or inhibitors of factors VIII, IX, and XI. Delays in transportation adversely affect PT and aPTT assays because of the short half-life of factor VII and the labile nature of factors V and VIII. The importance of using temperature-monitored freezers is because many frost-free freezers undergo freeze–thaw cycles leading to cold activation and degradation of coagulation factors. Factor VII is particularly sensitive to cold activation and can artificially decrease the PT. 10 Furthermore, prolonged sample storage leads to platelet release of platelet factor 4 from α-granules which neutralizes heparin-like molecules and interferes with heparin monitoring.
The majority of studies evaluating the stability of PT and aPTT in frozen samples have aimed at determining long-term storage capabilities over months to years. Long-term stability of frozen samples for PT has been demonstrated up to 12 months when stored at −24°C or for 2 years at −74°C, 3 and up to 3 years at −70°C. 11 Activated partial thromboplastin time could similarly be reliably stored for up to 8 months at −24°C or 2 years at −74°C. 3 However, conflicting findings have been reported under similar conditions. The PT levels were found to be prolonged when frozen at −20°C or −70°C with percentage changes exceeding 10% at 1, 2, 3, or 4 months, whereas aPTT was prolonged with less than a 10% change when tested at 1, 2, and 3 but not 4months with storage at −20°C or −70°C. 12 Freezing samples from individuals with elevated PT levels at shorter intervals found samples could be stored at −20°C for up to 24 hours and only for 12 hours in healthy individuals, whereas aPTT showed significant prolongation when stored at −20°C in both healthy and elevated aPTT participants. 4
Zürcher and colleagues demonstrated the significant change in snap-frozen PTs following transportation at ambient temperature at 48 to 52 hours was due to a decrease in factor V:C starting at 24 hours and factor VII:C at 48 hours. Similarly, a significant change in aPTT at 48 to 52 hours was attributed to a fall in factor VIII:C and factor V:C. 13 At ambient temperatures, PT is found to have statistically significant but clinically insignificant changes (percentage change < 15%) for up to 24 hours, whereas aPTT was only clinically insignificant up to 8 hours. 7 A multisite laboratory comparison found that under similar conditions, room temperature storage of PT samples varied between 6 and 24 hours depending on the testing centers. 5 This suggests other unidentified variables contribute to accuracy, and sample-processing protocols should be individualized per a laboratory basis.
The majority of preanalytical studies assessing coagulation storage have only incorporated healthy volunteers, while few studies have included participants with prolonged PT and furthermore only evaluated patients receiving VKA therapy. 7–8,11,13 One study focusing on 26 patients on VKA therapy found PT was significantly reduced, but all values were within desirable bias up to 6 hours at room temperature or up to 24 hours at 4°C following delayed centrifugation. 8 Interestingly, stability of PT varies between VKA-treated individuals compared to healthy volunteers. Percentage activity of PT started to decline at 24 hours in samples with low-normal values but was stable up to 48 to 52 hours in patients on VKA. 13 Rao and colleagues obtained samples from 16 patients not receiving VKA with elevated coagulation tests and found significant differences in PT from healthy participants when stored at −20°C for 12 and 24 hours but no significant difference in the elevated PT group for up to 24 hours. Similarly, the elevated aPTT group showed no statistical differences when frozen at −20°C for up to 24 hours, but aPTT from healthy volunteers was significantly different when frozen at the same temperature. 4 A tendency for longer storage capability of samples with elevated PT and aPTT was also observed in our findings that demonstrated fewer statistical differences at longer storage times in the coagulopathic group.
Modified aPTT assays such as APCR and DRVVT are also sensitive to storage variables and have similar constraints as the aPTT assay. Activated protein C resistance is used to assess patients for the presence of factor V Leiden, a mutant form of factor V which is resistant to degradation by activated protein C. Low levels of factors VIII, IX, and XI or platelet degranulation will prolong the modified aPTT assays of APCR; however, if the assays were exposed to similar conditions, then a significant change in the ratio would not be expected. Dilute Russell viper venom time is a phospholipid-dependent version of aPTT used to assess the presence of lupus anticoagulants associated with antiphospholipid syndrome. The first run of the DRVVT is processed in the same manner as the aPTT. Presence of a lupus anticoagulant will lead to assay prolongation by interfering with the assembly of the prothrombinase complex onto phospholipid membranes. The second run of the DRVVT is performed with excess phospholipid, and correction of the prolongation confirms the presence of a lupus anticoagulant. Increases in phospholipid, as found in platelets, can artificially neutralize the lupus anticoagulant. Therefore, extended presence of platelets within the sample due to delayed centrifugation profoundly interferes with the accuracy of the DRVVT assay. Unlike aPTT, the Russell viper venom within the DRVVT directly activates factor X and consequently is unaffected by factors XII, IX, or XI levels.
Evidence evaluating the stability of APCR and DRVVT is limited. One study demonstrated whole blood or plasma can be stored at room temperature for up to 24 hours and freezing samples at −70°C showed less deviation from original values compared to room temperature. 14 Another study found a statistically significant decline by 48 hours following storage at −80°C, but the percentage change was less than 10% and no discordant results were observed. 13 In the present study, APCR from 21 healthy individuals showed a statistical decline at 48- and 2-week intervals, but there were no discordant values and the mean percentage change was less than 10%. Also, of the 18 patients in the coagulopathic group with normal APCRs no statistical differences were seen at any time point up to 2 weeks. Activated protein C resistance evaluated by an alternative method using a thrombin generation-based assay also found samples to be stable up to 24 hours when stored at −20°C or −80°C. 15 For DRVVT, Froom and Barak demonstrated samples stored at room temperature for up to 6 to 8 hours were not significantly different nor were affected by 1 versus 2 centrifugations prior to analysis. 16 In a second study, they demonstrated samples frozen at −80°C have decreased DRVVT test ratios which lead to misclassification of 6 patients regardless of freezing time length (16-75 days). 17 In contrast, our study found no significant change in DRVVT in both the healthy and coagulopathic groups which may be due to a shorter storage interval and different storage temperature.
A difficulty in evaluating the effect of storage temperature and duration is the discrepancy between statistically significant and clinically relevant changes. For example, we found statistically significant differences at all time points compared to baseline for PT, but all data points had a percentage of change <10%; in contrast, results for
When evaluating preanalytic variables of sample processing, it is critical the participating patients reflect the spectrum of disease states typically encountered in a realistic patient setting. The INR monitoring of patients on VKA therapy comprises a large proportion of elevated PT/INR assays from outpatient clinics, while abnormal inpatient coagulation testing stems from a variety of pathological conditions. Our study included patients with coagulopathy on VKA or with liver disease to better reflect a wide range of abnormal laboratory results. However, it was found that some patients on VKA had subtherapeutic PT or normal aPTT levels and therefore were excluded from statistical analysis causing a reduction in sample size. In addition, the required volume of blood for assessment by this study prohibited inclusion of some patients with extreme elevations in PT and aPTT due to tendency for bleeding, severe anemia, or other comorbidities. To avoid patient harm, these individuals were not considered for the study; thus, only a limited number of non-VKA patients with abnormal coagulation tests were included. Similarly, low prevalence of positive APCR screens and severity of illness in patients with elevated
Footnotes
Authors’ Note
Abstract presented at the International Society for Laboratory Hematology (ISLH) 26th International Symposium, Toronto, Canada, May 2013.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
