Abstract
Cells process various mechanical cues in the microenvironment to self-organize into high-order architectures during tissue morphogenesis. Impairment of morphogenic processes is the underlying cause of many diseases; as such, understanding the regulatory mechanisms associated with these processes will form the foundation for the development of innovative approaches in cell therapy and tissue engineering. Nevertheless, little is known about how cells collectively respond to mechanical cues in the microenvironment, such as global geometric guidance, local cell-cell interactions, and other physicochemical factors, for the emergence of the structural hierarchy across multiple length scales. To elucidate the mechanoregulation of tissue morphogenesis, numerous approaches based on biochemical, biomaterial, and biophysical techniques have been developed in the past decades. In this review, we summarize techniques and approaches for probing the mechanoregulation of tissue morphogenesis and illustrate their applications in vasculature development. The potential and limitations of these methods are also discussed with a view toward the investigation of a wide spectrum of tissue morphogenic processes.
Introduction
Tissue morphogenesis is a fundamental multicellular activity that is essential for various developmental and regenerative processes. Vasculogenesis, for instance, leads to de novo blood vessel formation from mesodermal precursors overlying the endoderm. 1 Organs of ectodermal origin, on the other hand, are vascularized by angiogenesis—the outgrowth of new capillaries from the existing vasculature. 2 The formation of new blood vessels also contributes to numerous malignant, ischemic, inflammatory, infectious, and immune disorders.3,4 Particularly, tissue engineering, a rapidly emerging field in regenerative medicine, has great potential to restore, maintain, or improve tissue functions. Skin and cartilage tissue engineering, for example, has gained significant success because of the relative simplicity of the tissue architecture. Other tissues with extensive vasculature and heterogeneous cell arrangements, however, present significant hurdles in creating functional tissues that mimic their physiological counterparts.5,6 A fundamental understanding of the mechanisms and regulatory factors involved is required for creating functional tissue constructs for regenerative medicine.
The chemical basis of biological pattern formation and tissue morphogenesis is best understood in the reaction-diffusion model, which is also referred to as the activator-inhibitor system or the Turing pattern. 7 Moreover, the theoretical model describes the roles of autocatalytic reactions and lateral inhibition in tissue morphogenesis and has been applied to explain a wide spectrum of morphogenic processes.8–11 In addition to the reaction and diffusion of morphogens, physical factors are also important throughout the developmental process. The regulatory role of cell mechanics (e.g., cell contractility, intercellular tension, and cell-matrix mechanical interactions) is increasingly recognized in tissue morphogenesis. 12 As an example, geometric confinement has been demonstrated to regulate capillary network topology via cell-matrix mechanical interactions. 13 Tissue deformation has been shown to modulate vascular endothelial growth factor gradients and endothelial cell proliferation in deformable tissue constructs, coupling biochemical and mechanical tissue regulation. 14 Cell traction force has been demonstrated to control capillary network formation in vitro and in vivo by applying a Rho inhibitor and modulating extracellular matrix (ECM) elasticity. 15 Alternation of the ECM composition also revealed the importance of endothelial cell traction force in network stabilization. 16 These studies highlight the mechanical basis of tissue morphogenesis and encourage the development of novel mechanoregulation techniques.
Cells in tissues interact with their physical environment and generate endogenous contractile forces via multiple feedback mechanisms.17–19 The cell traction forces are coordinated by the cytoskeleton dynamics as well as cell-cell and cell-ECM interactions. Advances in biochemistry, material science, microfabrication, and nanotechnology have created new opportunities in modulating these interactions and probing the mechanoregulation of tissue morphogenesis systematically.20–24 In this article, we summarize advances in techniques and approaches for studying the roles of mechanical factors in tissue morphogenesis. First, pharmacological and biochemical approaches for perturbing the cytoskeletal structures and cell adhesion molecules are discussed. Second, biomaterial and microfabrication techniques for modulating the cell-matrix mechanical interactions are presented. External physical perturbations with mechanical, optical, magnetic, and fluidic techniques are then reviewed. Examples of the development of vascular and other tissues are described to illustrate the applicability of these techniques. Finally, the potential and limitations of these techniques for probing the mechanoregulation of tissue morphogenesis are discussed.
Biochemical Approaches for Mechanical Perturbation
Actin-Targeting Reagents
Cell traction forces and intercellular mechanical interactions can be modulated by reagents that target various cellular components such as actin filaments, microtubules, actomyosin, and cell junctions. The chemical inhibitors, RNA interference, forced expression, as well as genetic mutations of cellular components have been used to alter the mechanical properties of cells ( Fig. 1 ). It is well known that the actin cytoskeleton plays a major role in mechanical support, generation of cell traction force, and transmission of exogenous and endogenous cellular stress; therefore, actin-targeting drugs possess a significant capability to perturb cell mechanics. For example, cytochalasins, which are F-actin (filamentous) depolymerization reagents, bind to the barbed end of F-actin and prevent actin monomer polymerization. With cytochalasin D treatment, the contractile force and dynamic stiffness of endothelial cells are reduced significantly, whereas the intracellular structural damping remains unaffected. 25 In angiogenesis studies, endothelial cell tube formation is also inhibited by cytochalasin D. 26 Interestingly, at low concentrations of cytochalasin D, changes in the actin cytoskeleton are undetectable by fluorescence microscopy, despite significant changes in the mechanical properties of the cells. 27 Cytochalasin E, similar to cytochalasin D, inhibits endothelial proliferation and angiogenesis without disrupting actin stress fibers. 28
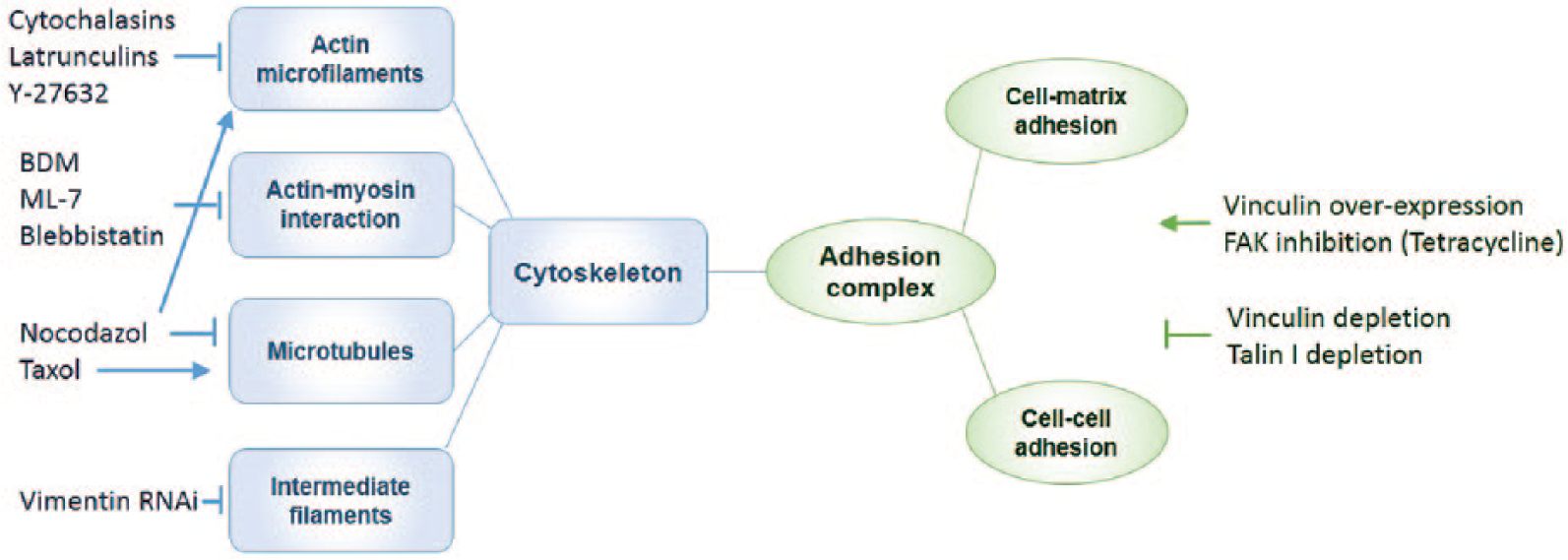
Biochemical approaches for perturbing cell mechanics.
Latrunculin, a class of actin depolymerizing agents,29,30 is more potent than cytochalasins. Latrunculin B binds to G-actin (globular) and inhibits nucleotide exchange on actin, while reducing cell stiffness by approximately 50%. 31 Low concentrations of Latrunculin B preferentially inhibit F-actin polymerization in filopodia. 32 Y-27632, a synthetic pyridine derivative that inhibits the Rho kinases (ROCK I and ROCK II), affects various downstream events including F-actin depolymerization and inhibition of myosin activity. As a contraction inhibitor, it relaxes contraction in smooth muscle cells and trabecular meshworks. 33 Treatment with Y-27632 abrogated vascular endothelial growth factor (VEGF)–induced softening of human umbilical vein endothelial cells in three-dimensional (3D) matrixes 34 and inhibited angiogenesis in vivo without observable effects on preexisting vessels. 35 In addition to chemically regulating the actin cytoskeleton, RNA interference of actin can also be applied to modulate mechanical forces. For example, the interference of smooth muscle actin (α-SMA) attenuated transforming growth factor β (TGF-β)–induced cell traction force in fibroblasts, and a linear relationship was also observed between the α-SMA protein expression level and the magnitude of cell traction force. 36
Microtubule-Targeting Molecules
Microtubule-targeting drugs represent another group of potent agents for modulating cell mechanics; however, the mechanical effects of microtubule-targeting drugs are often ultimately related to the actin cytoskeleton. 37 Nocodazole, a synthetic benzimidazole that destabilizes microtubules, disrupts the cellular balance between actin and microtubule networks and indirectly activates the actin cytoskeleton to improve force generation. 38 In addition, nocodazole exhibits anti-angiogenic effects in vitro along with potent inhibitory effects on endothelial growth. 39 Taxol (paclitaxel), an agent that stabilizes microtubules, facilitates microtubule polymerization and increases cell stiffness and viscosity. 40 Taxol has been actively investigated for its anti-angiogenic effects both in vitro and in vivo. 41 Interestingly, taxol, in low concentrations, has been shown to inhibit angiogenesis in vitro without affecting microtubule assembly. 42
Drugs Targeting Actin-Myosin Interactions
The modulation of cell mechanics in physiological and pathological processes is also achieved by controlling actin-myosin interactions. Myosins are a family of ATP-dependent motor molecules responsible for cell contraction and motility in both muscle and nonmuscle cells. For instance, 2,3-butanedione monoxime, an inhibitor of the ATPase activity of myosin, depressed the contractile characteristics of muscle cells 43 and decreased the stiffness of cardiomyocytes. 44 ML-7 and ML-9, agents that bind to myosin light chain kinase competitively with ATP, can also be used to reduce cell stiffness.45,46 Blebbistatin, a pharmacological inhibitor that restricts actin-myosin interaction by lowering myosin’s affinity for actin, reduces contractile force and cadherin adhesion. 47
RNA interference can be applied to inhibit actomyosin interactions and angiogenesis. 48 However, myosin inhibition through RNA interference also influences the mechanical properties of the cells. Myosin IIA small interfering RNA has been demonstrated to markedly reduce the contraction force of fibroblast cells within fibrins. 38 The inhibitor of an unconventional myosin, Myo1G, was also shown to decrease the elasticity of Jurkat cells significantly. 49
Targeting Intermediate Filaments
Another major component of the cytoskeletal system is the intermediate filament network. Intermediate filaments play essential roles in providing mechanical and structural integrity for cells, including regulating cellular tension development. 50 Multiple types of intermediate filament proteins have been identified. Among them, vimentin is one of the most widely distributed intermediate filament proteins. Vimentin intermediate filaments support cellular membranes, fix the position of some organelles, and transmit membrane receptor signals to the nucleus. Stimulation of tracheal smooth muscle strips with acetylcholine induced the increase in the ratio of soluble to insoluble vimentin in association with force development. 51 Treatment of muscle tissues with vimentin RNA interference attenuated force development in response to acetylcholine or KCl depolarization, as well as lower passive tension; the latter may be associated with the impairment of desmosomes. 52
Targeting Focal Adhesions and Cell Junctions
Cell mechanics may also be modulated by targeting cell-matrix and cell-cell interactions with genetic manipulation. Vinculin, which couples integrins or cadherins to the actin cytoskeleton at focal adhesions and adherens junctions, has been frequently used for assessing cell mechanical measurements. Cell contractile force generation is reduced when vinculin is absent or enhanced when vinculin is up-regulated. 53 Depleting the paxillin-vinculin interaction by substituting endogenous paxillin with a mutant reduced the total traction force of mouse embryonic fibroblasts. 54 Overexpression of the vinculin binding domain of αE-catenin decreased E-cadherin–mediated adhesion strength. 55 Down-regulation of talin I, an adaptor protein that links integrins to actin at the adhesion complex, decreased cellular force generation. 56 Depletion of α-actinin, which links integrins with actin, enhanced initial force generation and prevented adhesion mutation in subsequent steps. 57 Overexpression of actin-binding protein caldesmon blocked cell contractility and interfered with focal adhesion formation. 58 Chemical inhibitions of focal adhesion kinase with specific inhibitor or broad spectrum inhibitor have been shown to increase traction forces. 59 Activation of integrins with integrin-activating antibody inhibited the contractile force of muscle cells. Similar effects can be achieved by treatment with a synthetic integrin-binding peptide, whereas integrin function-blocking antibodies reversed the effect of the peptide on contractile force. 60
Targeting Signaling Pathways That Regulate Cell Mechanics
The manipulation of several signaling pathways can directly or indirectly lead to changes in cell mechanics. RNA interference of transcription factor JunB led to the inhibition of cell contractility under both basal and TGFβ1-stimulated conditions. 61 Treatment of DNA binding protein HMGB1 caused a TLR4-dependent increase in traction force, accounting for the downstream inhibition of enterocyte migration. 62 Silencing of the function-unknown gene VPS13A attenuated the F-actin network and reduced the stiffness of endothelial cells. 63 Silencing integrin-linked kinase enhanced vascular smooth muscle cell contraction under a puling force. 64 Inhibition of soluble adenylyl cyclase results in significant endothelial cell softening. 65 However, for applying biochemical approaches in studying cell mechanoregulation, achieving a pure mechanical perturbation is still a challenge because of the complex signaling network. Analyzing the results due to the force feedback–sensing, as well as force feedback–generating, mechanisms in cells is also demanding.
Biomaterial Approaches for Modulating the Microenvironment
ECM and Substrate Properties
Cell-matrix interactions can be modulated by adjusting the matrix properties (e.g., stiffness) as well as by altering ECM geometry and/or topography ( Table 1 ). The stiffness of the matrix can be controlled by the ECM density. Increasing the density of fibrin from 2.5 to 10 mg/mL resulted in an approximately sevenfold increase in matrix stiffness. 66 Fibrin gel has been used to study 3D capillary morphogenesis, with stiff gels found to reduce the formation of capillary networks.16,67 Similarly, the extent of capillary-like network formation could be tuned by adjusting the density of a self-assembling peptide gel. 68 Despite the simplicity of this approach, adjusting the matrix stiffness via the gel density will also affect the ligand density, which could introduce ambiguity into the results.
Common Gel-Based Materials for Studying Cell-Matrix Mechanical Interactions.
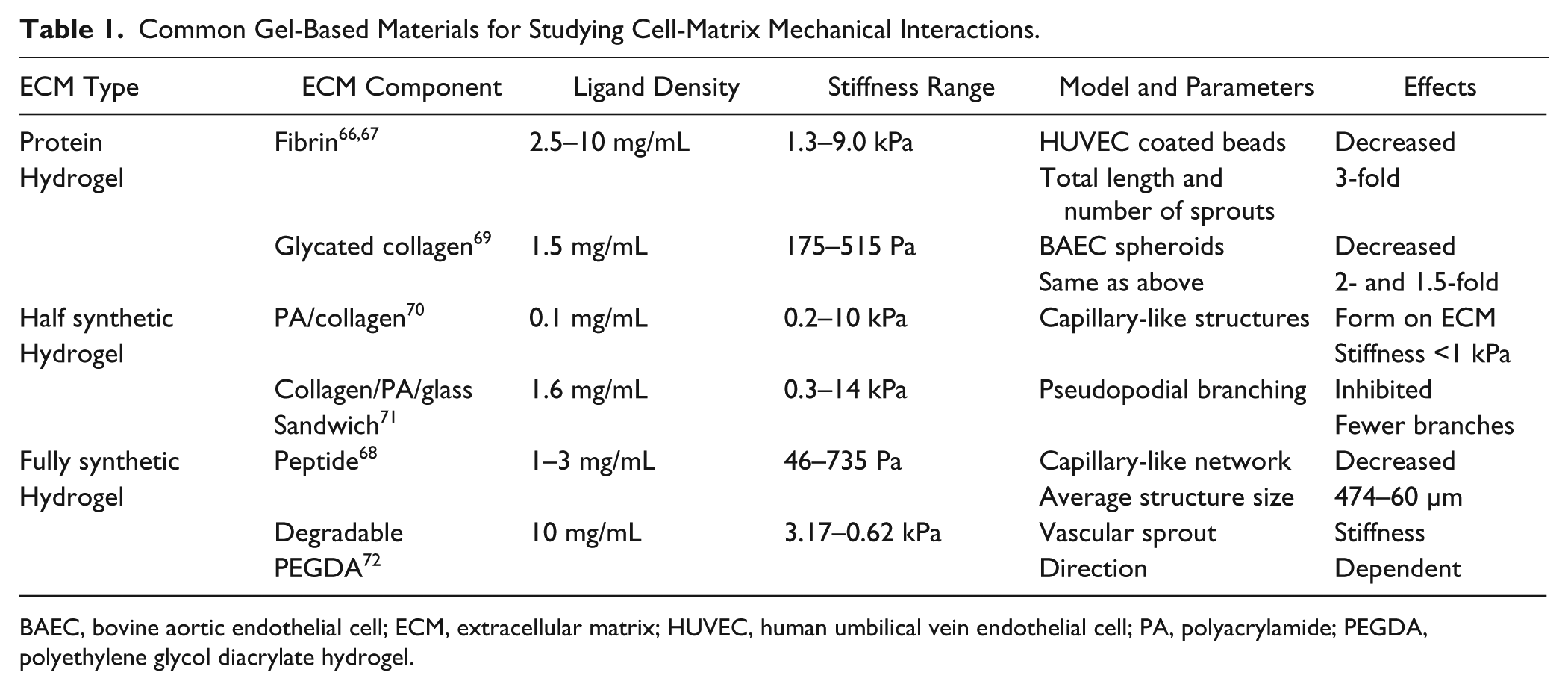
BAEC, bovine aortic endothelial cell; ECM, extracellular matrix; HUVEC, human umbilical vein endothelial cell; PA, polyacrylamide; PEGDA, polyethylene glycol diacrylate hydrogel.
Glycation of ECM proteins (e.g., type I collagen) has been employed to increase ECM stiffness with constant ligand density. More specifically, collagen gels were incubated in glucose-6-phopshate for 5 and 8 d to achieve different levels of glycation and ECM stiffness. Using collagen glycation, sprouting angiogenesis was shown to be delayed in stiff gels in a co-culture (endothelial cells and smooth muscle cells) sprouting model. 73 Alternatively, collagen solutions could be mixed with ribose to form glycated collagen solutions with increased concentrations of ribose and stiffness (~175 to ~730 Pa), leading to an increased number and length of sprouts from endothelial cell spheroids. 69 However, the time for complete glycation is long and the range of comprehensive moduli is small in comparison to the relevant physiological range (hundreds of kPa). Biological inert synthetic polymers have also been developed for achieving tunable gel stiffness with constant ligand density.74,75
Polyacrylamide (PA) hydrogels may achieve a large range of ECM stiffness by simply altering the relative concentrations of monomer (acrylamide) and cross-linker (bis-acrylamide). 76 However, it is not compatible with cell culture directly because of potential toxicities as well as the lack of cell adhesion capability. Proper PA hydrogel modification is required for investigating the effects of mechanical interactions at the cell-substrate interface. Endothelial cells have been shown to self-organize into capillary-like structures on compliant PA gels derivatized with type I collagen or functionalized with Arg-Gly-Asp (RGD) peptides.70,77 A collagen/PA/glass sandwich gel that enabled 3D cell culture was also shown to manipulate substrate stiffness, revealing that high ECM stiffness inhibits endothelial pseudopodial branch initiation. 71
Micropost arrays have also been used for modulating substrate rigidity, by taking advantage of microfabrication technology. The micropost arrays have been primarily used for studying cell mechanical interactions on 2D substrates and can be modified for studying 3D microtissues. 78 The deformability of the micropost can be modified by adjusting the post dimensions (e.g., height). Micropost arrays allow independent control of surface properties, decoupling substrate rigidity from cell adhesion.79,80 Furthermore, for compliant microposts (e.g., polydimethylsiloxane [PDMS]), cell traction forces can be determined by measuring the micropost displacement. It has been reported that endothelial cells showed enhanced elongation and alignment on PDMS microposts compared with stiff SiO2 microposts with similar topographical features. 81 However, the effects of ECM roughness and topology cannot be ignored and may limit micropost applications on stiffness-associated cell research.
Dynamic tuning of local ECM stiffness was demonstrated with polyethylene glycol (PEG)–based photodegradable hydrogels, in which cells exhibited a rounded morphology initially and started spreading during irradiation-induced matrix stiffening. 82 In addition, light-mediated sequential cross-linking was applied for dynamic matrix stiffening (e.g., 3–30 kPa), which mimicked the dynamic nature of tissue development, wound healing, and pathogenesis. 83 Increased cell areas and traction forces were observed in adhered human mesenchymal stem cells over a time scale of hours as the substrates were stiffened.
PA hydrogels with a photo initiator have been used for manipulating spatial control of the matrix stiffness. Stiffness gradients were generated by progressively uncovering the gel solution with an opaque mask from a noncollimated ultraviolet lamp. 84 Alternatively, a matrix metalloproteinase (MMP)–sensitivity PEG diacrylate hydrogel (PEGDA) was used to generate stiffness gradients via matrix degradation. 72 The stiffness gradients were capable of directing 3D vascular sprout formation using a co-culture angiogenesis model. In addition, magnetic beads could be embedded in collagen gels via bio-conjugation to alter the local stiffness of the ECM at the presence of an external magnetic field. 85 The magnetic force could increase the apparent stiffness of the ECM, and magnetic force gradients could induce ECM stiffness gradients, which can affect endothelial cell behaviors during angiogenesis.
Geometric Control
ECM geometries and physical confinements created by lithography and other microfabrication techniques can also modulate tissue morphogenesis.86,87 Photolithographic techniques were applied for micropatterned PEGDA with adhesive ligands to regulate and guide endothelial morphogenesis. 88 Endothelial cells did not assemble into cordlike structures on the stripe that was larger than 50 µm in width, highlighting the importance of geometric control of endothelial morphogenesis. Geometric control of endothelial cord formation was also demonstrated by culturing cells in microchannels that were filled with collagen gels. 89 The microchannel confinement is also used for directing the development of branches in tube formation, which may enable the production of complex capillary architectures. More recently, endothelial cells cultured on narrow (10 and 50 µm) micropatterned angiogenic Ser−Val−Val− Tyr−Gly−Leu−Arg (SVVYGLR) peptides demonstrated restricted spreading, with orientation and migration directionally guided and regulated. 90 Microwells with arbitrary shapes (e.g., star, square, and triangle) were shown to module capillary topology via cell-matrix mechanical interactions. 13 Endothelial cells can form denser networks on acute angle corners than those on reflex angle corners in a star-shaped ECM structure. The geometric control of tissue morphogenesis has significant implications in microfabrication-based tissue models and scaffold design for tissue engineering.
ECM topography and fiber alignment can also affect cell behaviors, such as the orientation of actin filaments and focal adhesions, proliferation, and migration.91–93 In particular, endothelial progenitor cells on nanotopographic substrates (600-nm-wide ridge and groove) exhibited enhanced alignment, organization, and capillary tube formation. 94 With the improvement and development of micro- and nanofabrication technologies in the future, mimicking in vivo ECM with specific topology for investigating tissue morphogenesis will be possible.
Biophysical Approaches for Probing Tissue Morphogenesis
Mechanical Perturbation
Mechanical perturbation influences various cell functions, including proliferation, migration, differentiation, and ECM remodeling ( Table 2 ). Tissue deformation that occurs naturally in muscular and cardiovascular systems regulates various tissue morphogenic processes.95,96 Mechanical stretching of rat microvascular endothelial cells enhanced the production of MMP-2, MT1-MMP, HIF-1α, HIF-2α, and VEGF at both the transcriptional and translational levels. 97 Stretching also induced an increase in intracellular Ca2+ concentration and traction forces of NIH-3T3 fibroblasts via stretch-activated ion channels. 98 In addition, physical stimulation with a mechanical probe may also be applied to create local mechanical stress and injury to individual cells in the tissue.99–101 A mechanical probe with comb-drive (capacitive) force measurement, for instance, has been used to study mechanical stimulation–induced intercellular calcium communication in microengineered endothelial networks.102,103 Endothelial cells collectively regulated their calcium levels against a large range of probing forces and repeated stimulations. Moreover, endothelial cells on elastic gels were shown to have active calcium responses when stimulated with a vibrating probe. 104 The mechanical stimulation caused deformations over a distance from the probe, and the cells sensed the local stimulation.
Biomechanical Effects on Tissue Morphogenesis.
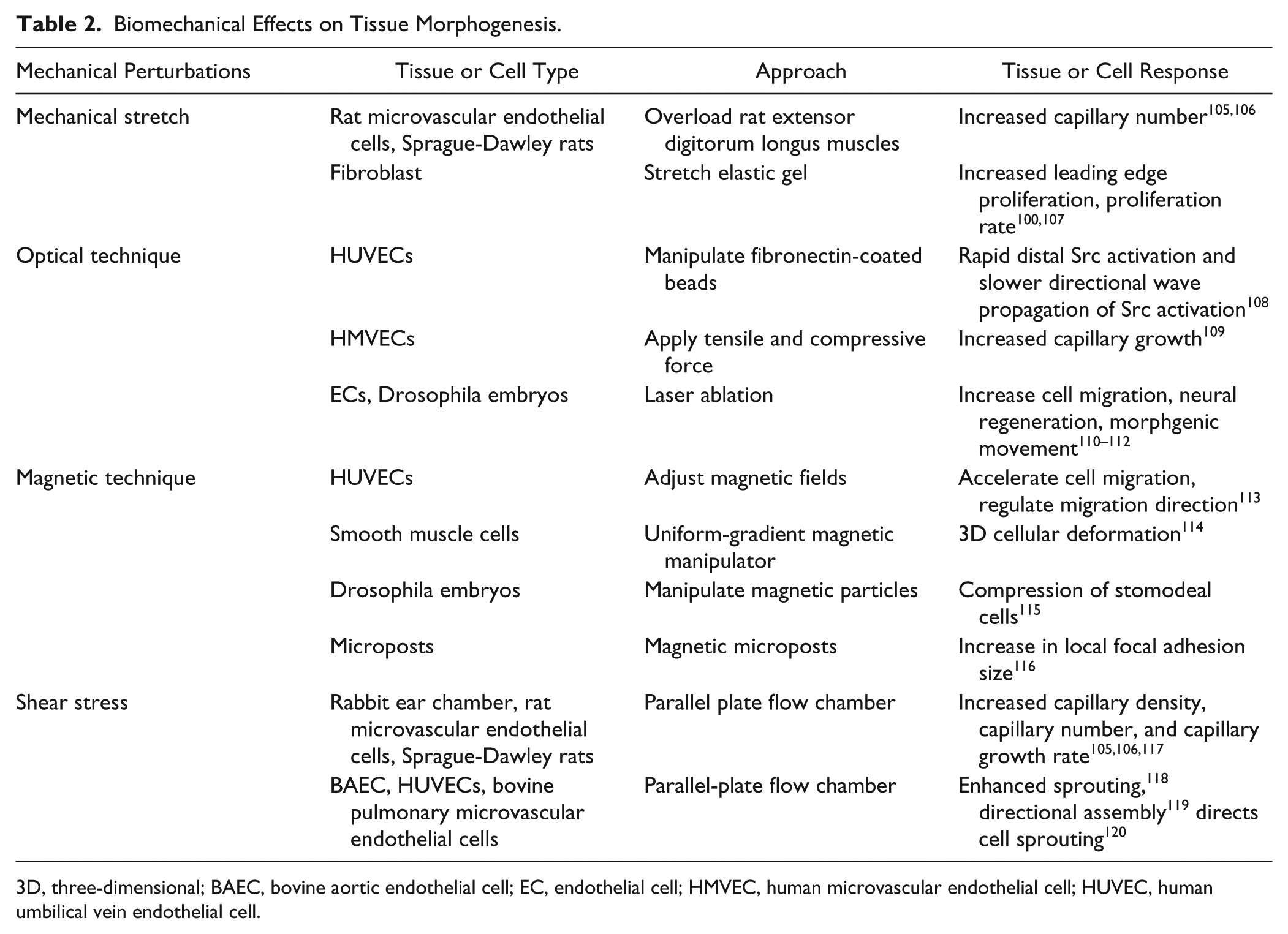
3D, three-dimensional; BAEC, bovine aortic endothelial cell; EC, endothelial cell; HMVEC, human microvascular endothelial cell; HUVEC, human umbilical vein endothelial cell.
Optical Techniques
Optical techniques can generate mechanical perturbations with subcellular resolution. As an example, Src activities in single endothelial cells could be activated mechanically by laser tweezers with fibronectin-coated beads. 121 Optical tweezers in a microfluidic system generated tensile and compressive forces for studying focal adhesion recruitment in endothelial cells. 122 In particular, mechanical force was applied to the ECM-integrin-cytoskeleton linkage by trapping and manipulating functionalized beads. In addition to laser tweezers, photoablation of cells and subcellular structures with lasers represented another powerful approach in modulating cell mechanics with subcellular resolution. 108 Cell ablation may be applied to disrupt the mechanical interactions between cells and perturb specialized cells in the morphogenic process. For disrupting the cytoskeleton, fluorescent proteins, such as actin-GFP, were used to visualize the cytoskeletal structure, and lasers were applied to ablate or disrupt the cytoskeleton. Femtosecond lasers with multiphoton absorption enabled fine resolution (<300 nm) and minimized damage to surrounding structures. The photoablation technique has been used for investigating various tissue morphogenic processes, such as collective cell migration, 109 neural regeneration, 108 and morphogenic movements in Drosophila embryos. 110
Magnetic Technique
Magnetic tweezers are employed to apply mechanical forces to living cells and tissues via external magnetic field gradients. 111 Intracellular forces on endothelial cells are generated under different magnetic fields after cellular uptake of superparamagnetic iron oxide nanoparticles. The intracellular magnetic force accelerates cell migration and regulates cell migratory direction via adjusting magnetic fields and increasing the cell-free culture space. 112 Moreover, 3D cellular deformation was studied by applying uniform forces to a large cell population via a uniform-gradient magnetic manipulator. 113 Magnetic particles were injected into Drosophila embryos for in vivo studies of tissue deformation. The mechanical compression of stomodeal cells in embryo mimicked the physiological deformation experienced by stomodeal cells due to germ band extension at the onset of gastrulation, which could up-regulate Twist expression in the stomodeal primordium. 114 Magnetic microposts were also employed to exert a step force that led to an increase in local focal adhesion size at the site of application but not at nearby nonmagnetic posts. 115
Shear Stress
Endothelial cells are subjected to shear stress generated by blood flow. Shear stress can modulate cell migration, proliferation, gene expression, and morphology of endothelial cells.116,123 Thus, wall shear stress can be generated to investigate the effects of dynamic shear on the development and remodeling of the vasculature. A parallel plate flow chamber may be used to study the effects of shear stress on capillary-like tube formation. Endothelial cells on matrigel experienced high shear stress, resulting in aligned tubular structures along with the direction of fluid flow. Shear stress–dependent regulation was, at least partially, induced by VEGF expression.105,117,120,124 Fluid forces generated by shear stress may also attenuate endothelial cell sprouting in a nitric oxide–dependent manner. Interstitial flow directs cell sprouting and cell morphology and therefore regulates capillary tube formation. Moreover, fluidic shear stress (3 dyn/cm2) enhanced the sprouting of bovine pulmonary microvascular endothelial cells in a 3D culture model. 120 In vivo, the effects of fluid shear stress in capillary vessel growth were first investigated in rat skeletal muscle and rabbit ear chamber.106,118 Externally applied wall shear stress was shown to stimulate angiogenesis via induction of VEGF expression by HIF-1α via the PI3K-dependent Akt phosphorylation pathway.
Microfluidic technology provides powerful tools for investigating the effects of fluidic shear stress on tissue morphogenesis in 2D and 3D models. 125 Fluid shear can be applied to perturb diffusible gradients in the microenvironment, thereby isolating biomechanical mechanisms from other biochemical mechanisms (e.g., reaction-diffusion). For instance, microfluidics was used to identify autocatalytic alignment feedback in the long-range organization of myotube development. 126 While diffusible factors are essential in the alignment process, recirculation of the media results in normal cell fusion and long-range alignment, eliminating diffusible factors from the cell alignment mechanism. Furthermore, a microfluidic wound-healing assay was developed to wound the monolayer and test the effects of shear stress on cell migration. 127
Discussion
This review summarizes biochemical, biomaterial, and biophysical approaches for probing the regulatory roles of mechanical force in tissue morphogenesis. Several global perturbation techniques, such as modulation of ECM properties, fluid shear, and mechanical stretching, have been used extensively because of their physiological and translational relevance. These techniques are particularly useful in identifying the involvement of mechanical forces in morphogenic processes. The overall tissue architectures (e.g., orientation and network density) can be perturbed to shed light on the morphogenic process. Advances in technology have also enabled local perturbation techniques, such as single-cell probes, laser tweezers, microfluidics, and photoablation, for investigating cell-cell communication mechanisms. These techniques open new possibilities in elucidating morphogenic mechanisms by perturbing the local mechanical force distribution and observing its effects on the tissue architecture. In particular, these local perturbation approaches can be used for exploring unknown morphogenic processes, testing new hypotheses, and studying cell-cell organization. Furthermore, biochemical reagents provide invaluable insights into the molecular mechanisms involved in the mechanoregulation processes. However, biochemical reagents may disturb multiple signaling pathways and trigger nonspecific effects, which complicates the interpretation of results. Multiple reagents and approaches should be incorporated to confirm that the observation is indeed mechanically induced.
Cells and tissues represent complex networks of mechanically responsive systems with multiple hierarchical levels. Cells in tissues collectively sense and adapt to the physical microenvironment during development and regeneration. Intra- and intercellular forces are dynamically regulated through multiple cellular feedback mechanisms. While it is important to understand the molecular mechanosensing mechanism, it is challenging, if not impossible, to isolate individual factors one from the other. This represents a hurdle in understanding and interpreting study results and in elucidating mechanical regulatory mechanisms. Another challenge in understanding tissues morphogenesis is to identify the emerging behaviors that drive complex tissue architectures. Inhibiting a morphogenic process does not necessarily provide useful information in the conceptual understanding of the emerging behaviors. Careful experimental designs with multiple techniques are often required to study collective cell behavior. In addition to mechanical perturbation techniques, novel biochemical and mechanical techniques (e.g., intracellular probes100,128 and traction force microscopy129,130) should be developed and used to study morphogenic processes. Another key challenge in studying the emergent complexity of biological tissues is the difficulty in understanding the high-order structures and functions resulting from local interactions of individual cells. These emergent properties can often be counterintuitive and cannot be understood by simply extrapolating interaction between a few cells. The complex interplays between global microenvironmental cues and local cell-cell interactions further complicate understanding emergent cell behavior. A complex systems framework that incorporates biomanufacturing, microfluidics, advanced materials, biosensors, and computational modeling is ultimately required to fully understand the mechanoregulation of tissue morphogenesis. As we move forward in this field to understand this complexity, not only will basic cellular and tissue processes become clear, this insight may additionally be used in a biomanipulative way to develop novel therapeutic strategies to tackle complex disease processes that involve mechanical regulatory mechanisms.
Footnotes
Acknowledgements
The authors would like to thank Zachary Dean for constructive suggestions and English editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institutes of Health Director’s New Innovator Award (DP2OD007161).
