Abstract
Cell-based assays are widely used in the various stages of drug discovery. Advances in microfluidic systems over the past two decades have enabled them to become a powerful tool for cell-based assays to achieve both reliability and high throughput. The interface between the micro-world and macro-world is important in industrial assay processes. Therefore, microfluidic cell-based assays using pressure-driven liquid handling are an ideal platform for integrated assays. The aim of this article is to review recent advancements in microfluidic cell-based assays focusing on a pressure-driven perfusion culture device. Here, we review the development of microfluidic cell-based assay devices and discuss the techniques involved in designing a microfluidic network, device fabrication, liquid and cell manipulation, and detection schemes for pressure-driven perfusion culture devices. Finally, we describe recent progress in semiautomatic and reliable pressure-driven microfluidic cell-based assays.
Introduction
The cost of drug development has increased exponentially in accordance with Eroom’s law, 1 and the attrition rate in clinical trials is increasing year by year. 2 In addition, bridging the gap between experiments in animal models and clinical trials is difficult because of the inherent differences between animals and humans, making extrapolation of results difficult. Therefore, in vitro assays using cultured cells are becoming an increasingly important tool. High-throughput screening systems using a microplate and dispensing robot are widely used for cellular assays in drug development research. 3 Recent advances have been made in the application of inkjet and acoustic systems for liquid handling, and assay throughput is increasing. In addition, technological advances now allow more information to be obtained from a single assay. 4 Further advances in microfluidic technology are expected to realize even more efficient throughput.
The drawbacks for using traditional cell-based assays for screening in drug development are the differences between the environment surrounding the cells and the animal body and the fact that the cultured cells have lost many of the functions found in the body. 5 These limitations have the potential to be overcome by the application of microfluidic technology to drug discovery applications by miniaturized assays and to increase experimental throughput and reliability in drug discovery applications.6–8
In the past decade, microfluidic devices have been progressively used as versatile tools for many types of cell-based assays.9–13 One of the key advantages of using microfluidics for cell-based assays is the miniaturization of each assay. Miniaturized high-throughput and high-content cell-based assays have been reported14–17 and applied to analyses of liver drug metabolism and toxicity.18,19 Assay miniaturization provides the opportunity for creating a variety of microenvironments, such as soluble factors and extracellular matrixes, within the microfluidic devices.20,21 The fabrication of microfluidic devices with biomimetic tissues is another approach for cell-based assays.22–24 This approach has been reported for lung,25,26 renal, 27 cardiac, 28 artery, 29 gut, 30 and corneal tissue 31 and is expected to increase the reliability of in vitro assays.
Microfluidic devices have the advantage of reducing the amount of valuable sample and reagents required for cell-based assays and can be fabricated with engineered tissues to increase the reliability of results. The application of microfluidic devices to high-throughput industrial drug discovery has led to the development of the pressure-driven perfusion culture microchamber array chip. 32 Prior to the development of pressure-driven perfusion culture devices, microfluidic chips required the connection of tubes and external pumps (e.g., syringe pump and peristaltic pump) so that culture media could flow into the microfluidic network. This type of system proved to be too complicated and impractical for microfluidic chips to be used for cell-based assays designed to simultaneously screen multiple drug candidates. These issues were addressed by the pressure-driven perfusion culture microchamber array chip. 32
Table 1 summarizes the comparison of technical features in the reported liquid delivery system for perfusion culture microfluidic devices. Most of the liquid delivery systems, including syringe pumping, pneumatic pressure, gravity force, and so on, require external tube connection. One exception that does not need tube connection is the surface tension–driven liquid delivery system, in which loaded liquid is spontaneously delivered by the intrinsic energy. Compared with these liquid delivery systems, the pressure-driven liquid delivery system requires only one to a few tube connections to pressure sources. In addition, fluid flow rate is easily calculated from the microchannel geometry and applied pressure. Considering these advantages of pressure-driven perfusion culture, we recently developed a user-friendly pressure-driven perfusion culture microchamber array chip for high-throughput drug dose-response assays. The design of this system allowed multiple liquids to be introduced under pressure into the microchip. We further improved this system by integrating a serial dilution microfluidic network to generate logarithmic concentration profiles. 51 We then demonstrated the efficacy of this microchamber array system by successfully evaluating the dose response of a model anticancer drug, paclitaxel, in HeLa cells. 40 Here, we introduce recent technology developments of our pressure-driven perfusion culture microchamber array chip and highlight its simple interface and well-designed microfluidic network. This system has potential as an efficient platform for high-throughput cell-based microchip assays in drug discovery applications.
Comparison of Liquid Delivery Systems for Perfusion Culture Microfluidic Devices.

Perfusion Culture Microchamber Array Chip
Figure 1 shows our pressure-driven perfusion culture microchamber array chip with its simple interface. 32 The microchamber array chip has macroscopic liquid reservoirs, which can stock a small volume (<1 mL) of culture media containing different drugs or cell suspensions loaded by a micropipette ( Fig. 1a ). The culture media can be delivered simultaneously through the microfluidic network from the liquid reservoirs by applying pressure through the air vent filter ( Fig. 1b ). In addition, each microchamber was designed to have a unique geometry and independent perfusion microchannel to prevent cross-contamination between neighboring microchambers and for permitting easy air-bubble removal, uniform cell loading, and pressure-driven long-term perfusion culture in quantitative cell-based assays ( Fig. 1c ). We examined the cytotoxic effect of seven anticancer drugs in parallel to demonstrate the functionality of this pressure-driven perfusion culture microchamber array chip ( Fig. 1d ).

Photographs of the pressure-driven perfusion culture microchamber array chip. (
Microfluidic chips are usually fabricated from poly(dimethylsiloxane) (PDMS), which is a flexible, durable, transparent, and inexpensive polymer that allows microstructures to be easily molded by soft lithography.52–54 In our previous study, we designed a PDMS microchip with a microfluidic network composed of microchannels at different depths by means of multilayer photolithography and replica molding.
8
The dimensions of the microchamber and connected microchannels were determined from calculations based on mass transfer and hydrodynamics.
Table 2
shows the parameters to be considered for designing dimensions of the microchambers and microchannels. These calculations were used to predetermine diffusive and convective mass transfer rates and velocity distribution at medium flow (
Parameters to Be Considered for Designing Dimensions of the Microchambers and Microchannels.

µ denotes the fluid viscosity,
ρ denotes the fluid density and
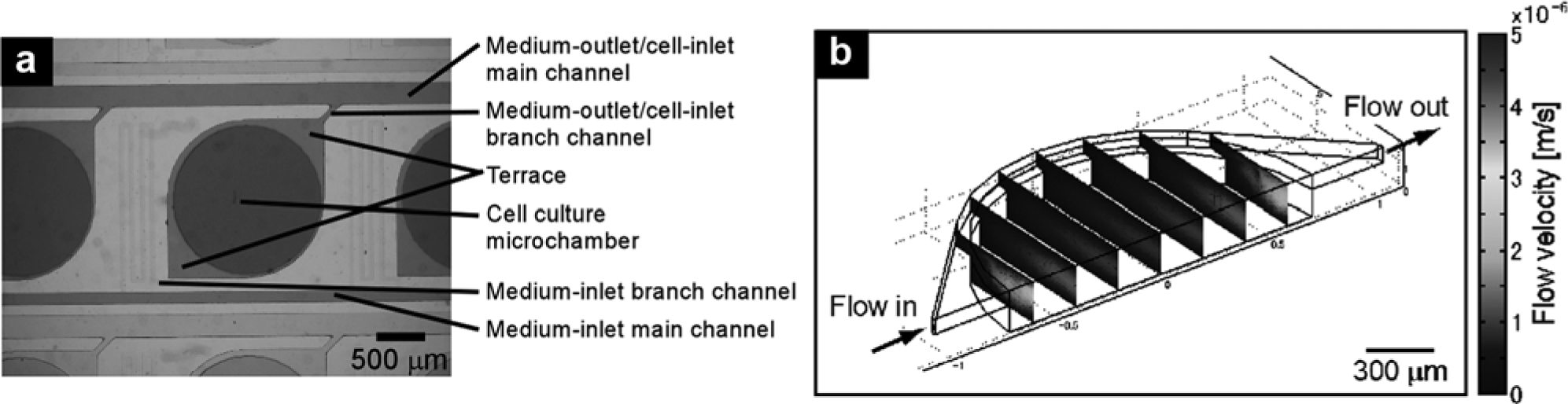
Design of the microchamber array. (
Dose-Response Assay by Perfusion Culture Microchamber Array Chip
Dose-response assays are crucial in the field of drug discovery. Scientists often prepare stepwise drug concentrations in a linear55–59 and logarithmic60–65 dilution series and evaluate the 50% growth inhibitory concentration (IC50) or 50% effective concentration (EC50). Generally, the dose-response assays are carried out in multiwell plates and performed either manually or semiautomatically with expensive robotics.66,67 The preparation of a large number of drug solutions at different concentrations is, however, tedious and time-consuming. In addition, the dilution process sometimes causes experimental errors, since serial dilution using a micropipette can induce accumulation of dilution errors. To address these issues, serial dilution microfluidic networks for generating various concentration profiles have been developed.34,68–72
We previously described a serial dilution microfluidic network for generating arbitrary monotonic concentration profiles (
Determine a microchamber size and cell density considering the target application.
Determine a desired concentration profile considering the target application.
Determine a possible applied pressure range considering available equipment.
Estimate flow rate in each outlet microchannel from the value estimated by step 1.
Calculate flow rate,
Calculate fluidic resistances,

Finally, calculate dimensions of each microchannel assuming the steady-state pressure-driven flow in a rectangular microchannel formulated as
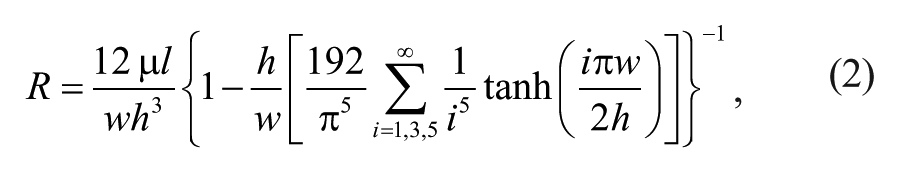
where
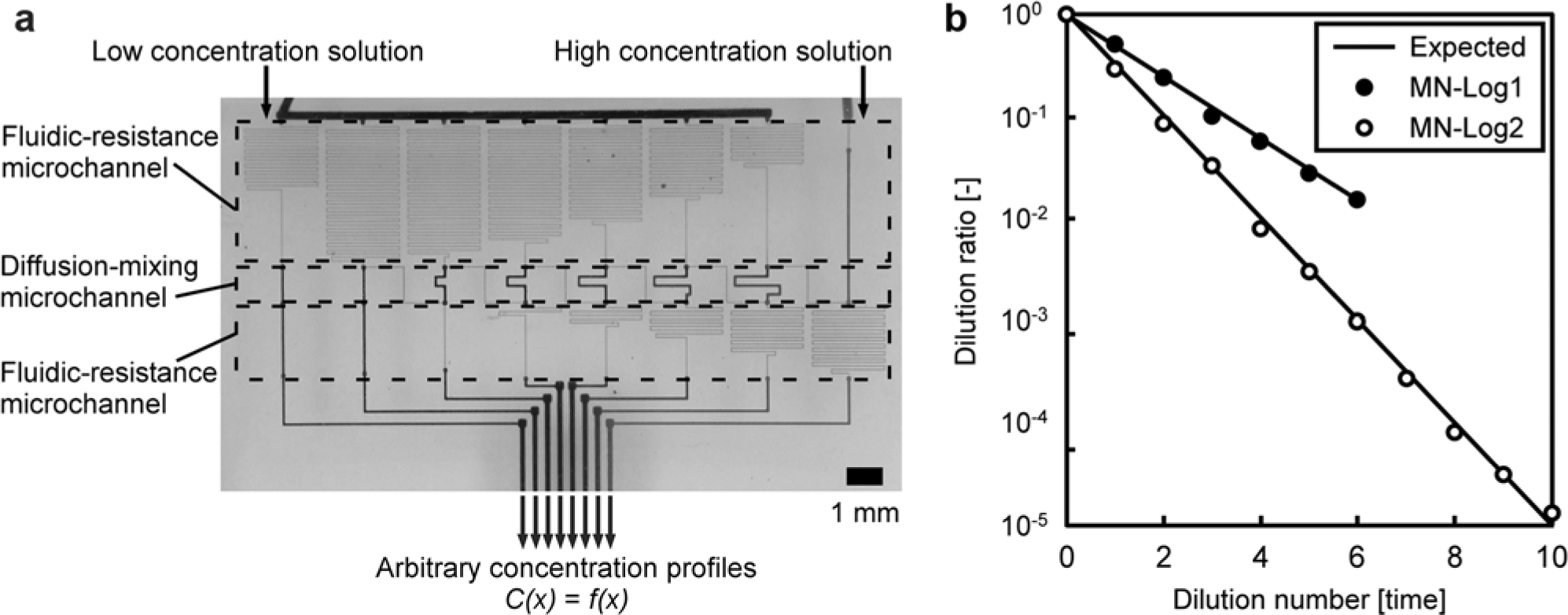
Automatic generation of arbitrary concentration profiles by the serial dilution microfluidic network. (
The microfluidic networks of this system were composed of fluidic-resistance microchannels with a cross-sectional area approximately one-tenth that of other microchannels. The fluidic resistance in these microchannels per unit length is therefore 102- to 103-fold higher than that of other microchannels. The flow rates for each diffusion-mixing microchannel are determined by the fluidic-resistance microchannels. Therefore, arbitrary concentration profiles can be generated by constructing fluidic-resistance microchannels at an appropriate length and ensuring complete mixing in the diffusion-mixing microchannels ( Fig. 3b ).
The optimization of the mixing process is essential for the accurate performance of the serial dilution microfluidic network. In these chips, the high-concentration and low-concentration solutions are mixed with each other by diffusion in the diffusion-mixing microchannels. The mixing process can be examined by simulating the flow pattern and concentration distribution in the diffusion-mixing microchannels by commercial hydrodynamic analysis software (
Fig. 4a
; we used version 3.3a; COMSOL Multiphysics, Stockholm, Sweden). The required residence time for complete mixing in the diffusion-mixing microchannels depends on the molecular weight of the solute and the dimensions of the diffusion-mixing microchannels. We can evaluate the required residence time by calculating the dimensionless diffusion time, Fick number (

where
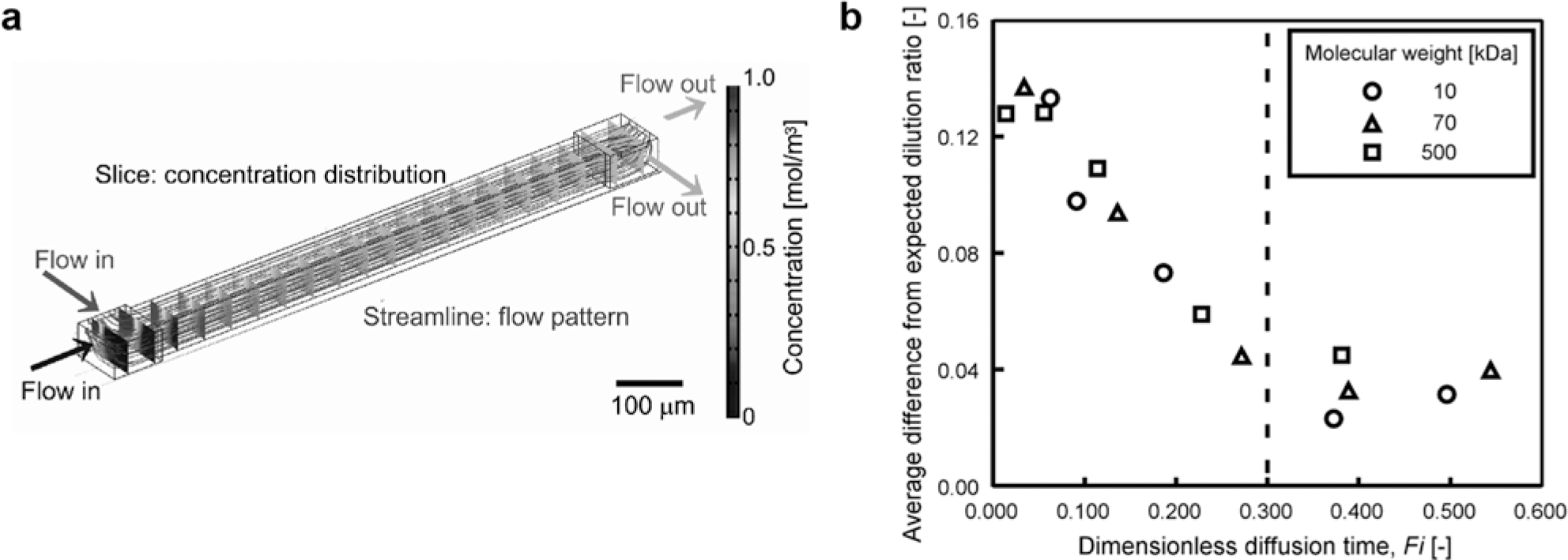
Predetermination of flow and mixing in the microchannel. (
Recently, we developed microfluidic cell-based assays for IC50 determination by applying the pressure-driven perfusion culture microchamber technology and the serial dilution microfluidic network ( Fig. 5a ). 40 In these experiments, the perfusion culture microchamber array chip was equipped with 12 perfusion culture microchambers and a serial dilution microfluidic network capable of generating 12 different logarithmic concentrations spanning six orders of magnitude ( Fig. 5b ). We successfully used this pressure-driven microfluidic perfusion culture device to perform a dose-response assay for paclitaxel in HeLa cells by fluorescent image analysis. We successfully calculated the IC50 of paclitaxel using data from three separate microchamber arrays ( Fig. 5c ).

Photographs of the perfusion culture microchamber array chip equipped with a serial dilution microfluidic network. (
Integrated Perfusion Culture Microchamber Array Chip
More recently, we developed a microplate-sized integrated perfusion culture microchamber array chip (
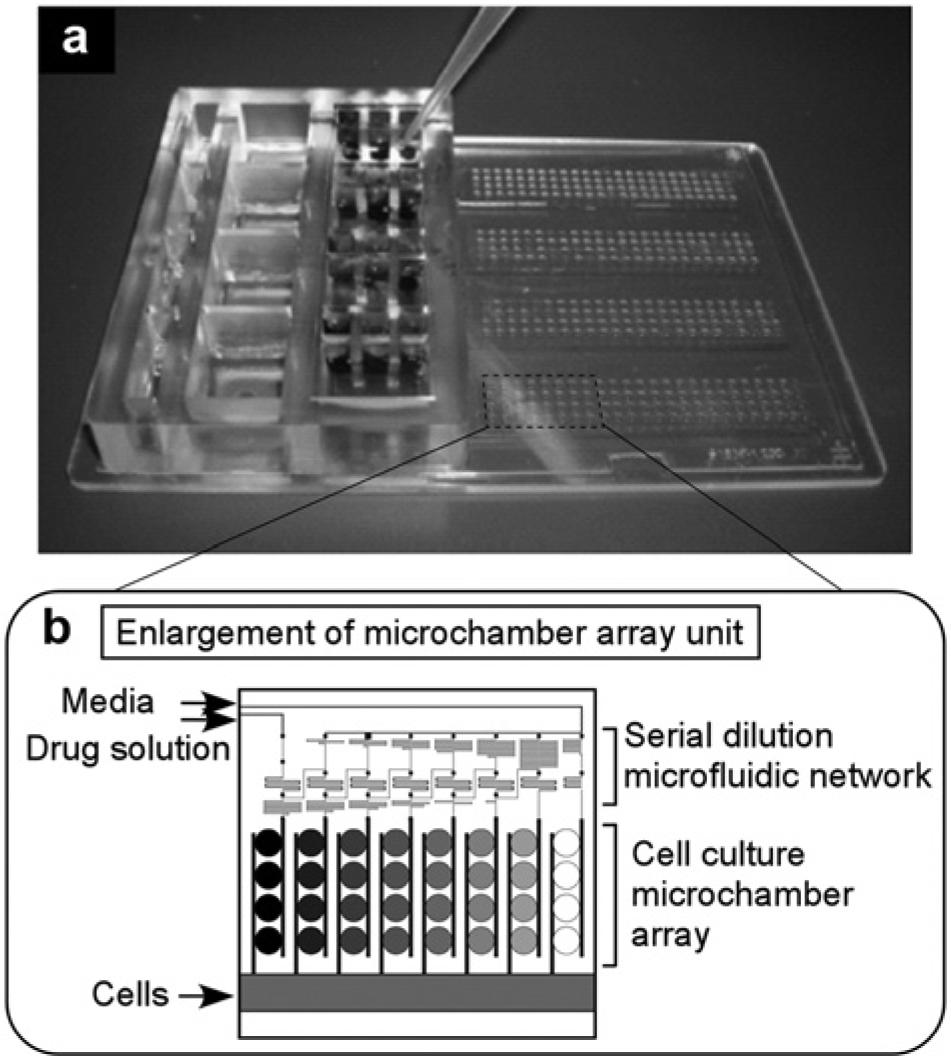
Microplate-sized integrated perfusion culture microchamber array chip. (
The serial dilution microfluidic network described here has a similar structure to that described in our previous study in that the drug solution was diluted at a ratio of 100.5 for each dilution step to create a concentration profile spanning three orders of magnitude over six dilution steps. 75 By using the integrated perfusion culture microchamber array chip, parallel quadruplicate assays for 12 drugs at eight different concentrations, which is a total of 384 assays, can be performed simultaneously.
We also developed two devices, a dispenser unit and a perfusion culture unit, for automation of the integrated perfusion culture microchamber array chip (

Peripheral devices for the automation of the cell-based assay with the integrated perfusion culture microchamber array chip. (
In conclusion, we describe an efficient pressure-driven perfusion culture microchamber array chip with a user-friendly interface and well-designed microfluidic network. Cell suspension and culture media containing different drugs can be loaded with a micropipette and can be delivered simultaneously into the microfluidic network by applying pressure through the air vent filter. We have also presented the means to design the microfluidic chips. The pressure-driven integrated perfusion culture microchamber array chip is a versatile system that can be used with standard microplate readers and dispensing robotics and has the potential to be an efficient platform for a microfluidic cell-based assay in drug discovery.
Footnotes
Acknowledgements
A part of this work was conducted in the AIST Nano-Processing Facility.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by MEXT KAKENHI (24106512).
