Abstract
Cell-based assays are essential tools used by research labs in a wide range of fields, including cell biology, toxicology, and natural product discovery labs. However, in some situations, the need for cell-based assays does not justify the costs of maintaining cell culture facilities and retaining skilled staff. The kit-on-a-lid assay (KOALA) technology enables accessible low-cost and prepackageable microfluidic platforms that can be operated with minimal infrastructure or training. Here, we demonstrate and characterize high-density KOALA methods for high-throughput applications, achieving an assay density comparable to that of a 384-well plate and usability by hand with no liquid-handling equipment. We show the potential for high-content screening and complex assays such as quantitative immunochemistry assays requiring multiple steps and reagents.
Introduction
Cell-based assays and in vitro models are essential tools for a wide range of biological and chemical applications such as cell biology, drug discovery, and toxicology. 1 However, traditional assays, including microfluidic assays, suffer from significant limitations that prevent more widespread use.2,3 Among these barriers to adoption are the cost of cells and reagents, the limited scalability of the assays due to the reliance on a serial (vs. parallel) operation process, and the requirement of both preparation and equipment, such as pipettes and centrifuges ( Fig. 1 ). Perhaps more importantly, traditional assays require trained staff, which limits accessibility of these assays to labs that do not have cell culture expertise and/or facilities, such as natural product discovery labs, educational labs, engineering labs, or environmental research labs. 4 The need for simplified assays has been highlighted by assays such as the SlipChip. 5 Yet there remains a dearth of technologies for cell-based assays that are low cost, are simple to operate, and integrate the ability to perform sequential and complex fluid replacements.
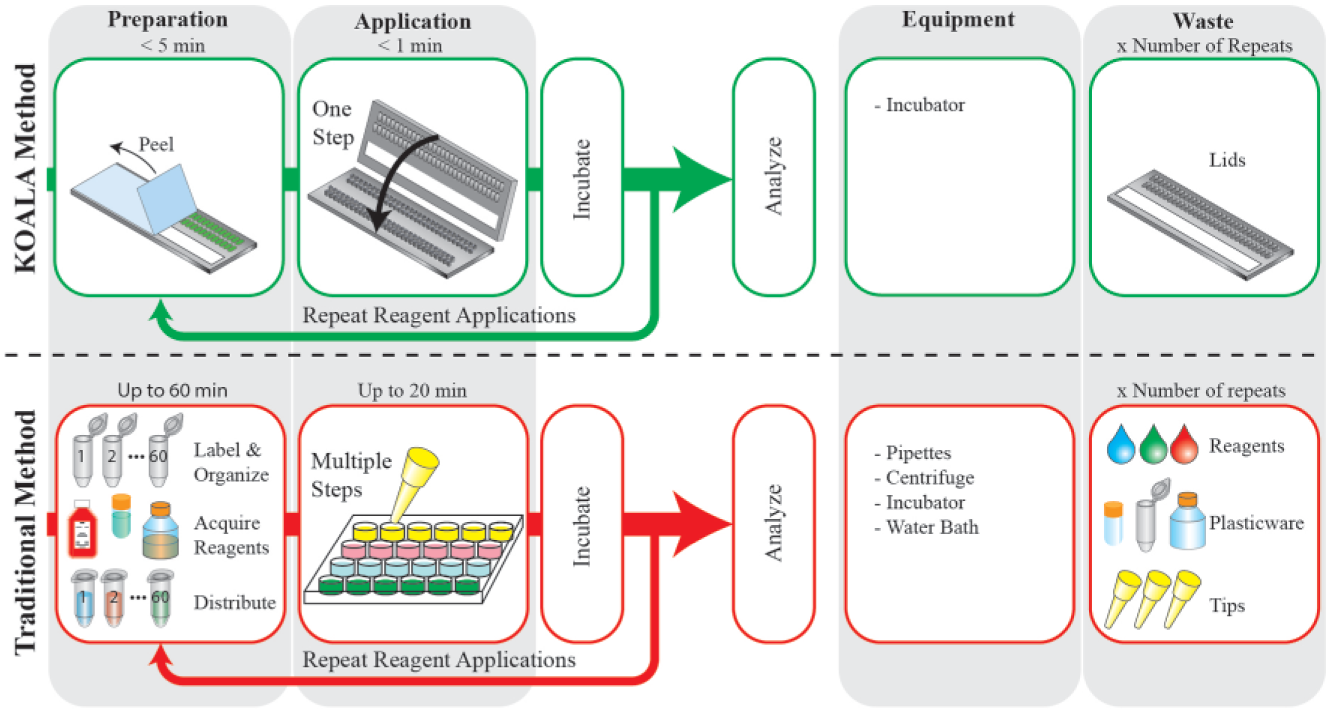
Schematic represents the workflow differences between a kit-on-a-lid assay (KOALA) microfluidic platform (top) and a traditional assay (bottom). KOALA enables prepackaging of reagents into lids, parallel fluid applications, minimal demands for equipment, and minimal waste. Traditional assays often require significant preparation, a serial process for fluid application, several pieces of equipment, and yield large amounts of waste.
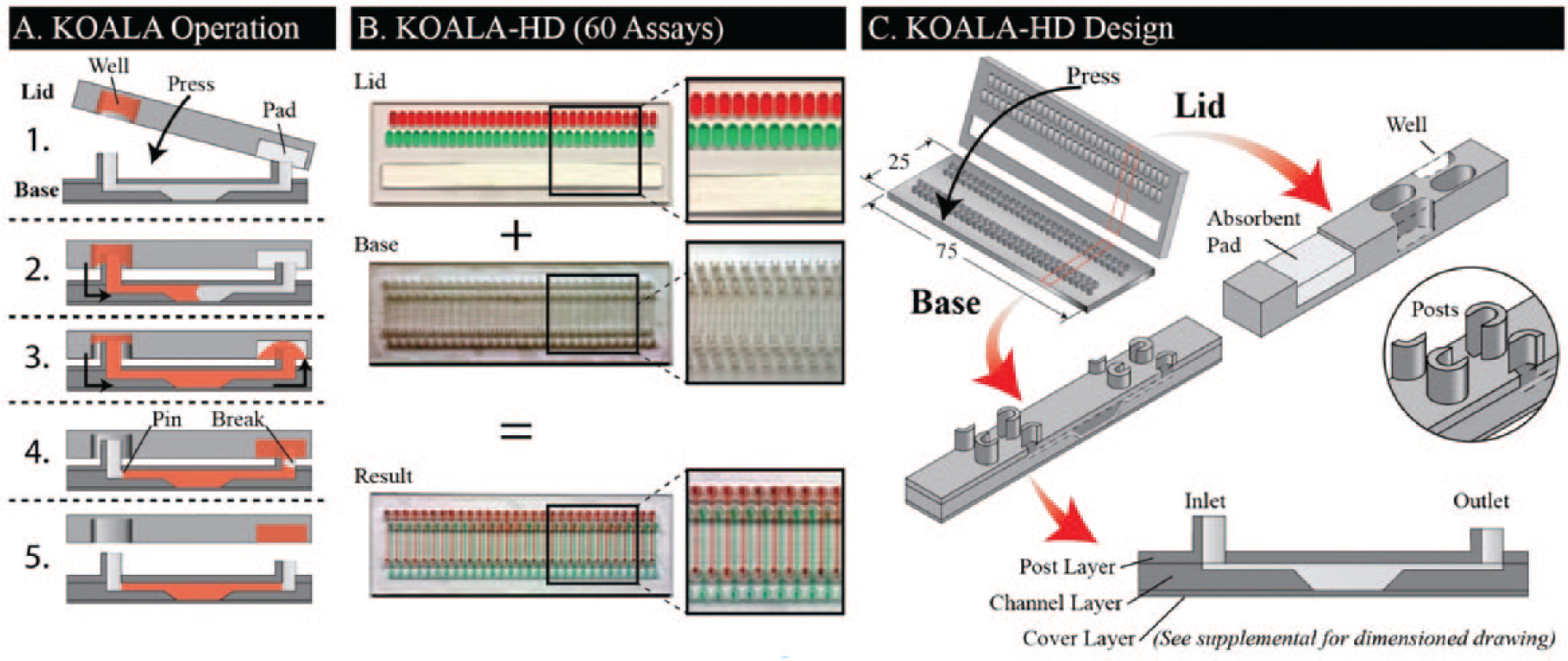
The kit-on-a-lid assay (KOALA) technology is a novel microscale platform that aims to reduce barriers to adoption of cell-based assays by enabling precise and advanced fluidic handling in a self-contained, user-friendly interface.6,7 KOALA integrates several features to simplify complex assays. First, prepackaging eliminates the need to prepare reagents, label tubes, and distribute reagents to each assay, a time-consuming and often underestimated part of traditional assays. Second, KOALA is a micro-to-micro platform, reducing waste by eliminating the need to dilute/prepare reagents for the assay via macroscale tubes (as is currently done even for microscale platforms). Finally, the integrated fluid-handling system of KOALA eliminates the need for fluid-handling equipment, such as pipettes, mixers, and centrifuges. The KOALA fluid-handling system uses a series of lids that are used in conjunction with a single base containing a set of microchannels. Different sets of lids are used for different assays. Lids are prepackaged with only the necessary volumes of prediluted reagents required for each step of the assay. Each step (i.e., fluid exchange step needed to apply a treatment or reagent on the cell culture) is then performed by removing a lid from the freezer, thawing it, peeling a protective layer covering microwells containing reagents, and “clicking” the lid onto the base ( Fig. 2A ).
(
Current KOALA methods have described the basic principles for exchanging fluid between a lid and a base. However, significant limitations remain for using these methods for high-density and robust screening applications, 6 including modes of cross-contamination between neighboring wells and channels, fluid replacement efficiency and overall fluid characterization, and validating the technology for functional cancer cell assays. We address these limitations through multiple design enhancements that enable a high-density KOALA (KOALA-HD) format with throughput on par with 384-well plates in a platform that can be operated by hand without any fluid-handling equipment ( Fig. 2B ). The following describes the geometrical parameters (i.e., feature characteristics, orientation, and dimensions) and fabrication processes that are necessary to enable KOALA-HD while preventing cross-contamination. We present and characterize the washing efficiency, operation, and performance of the KOALA-HD platform for repeated and higher-density applications in terms of fluidic function and for facilitating cell-culture applications lasting more than 24 h. Finally, we demonstrate the utility of KOALA-HD for high-throughput cell-based applications by performing a screening assay with complex immunohistochemistry (IHC) endpoints.
Materials and Methods
Device Fabrication and Preparation
The lids and bases are fabricated by CNC milling (PCNC 770, Tormach, Waunakee, WI) with standard microscope slide dimensions (25 mm × 75 mm). Bases are composed of three layers: (1) a post layer, (2) a channel layer, and (3) a cover layer (
Fig. 2C
;
Lids are milled from 2 mm PS. Each lid contains a microwell for each channel (i.e., 60 microwells) and a slot for absorbent pads. Each microwell has an elliptical shape, which is 1.5 mm wide, 4 mm long, and 2 mm deep and is spaced to match the pattern of the input posts. The slot for the absorbent pad is 10 mm wide, 72 mm long, and 2 mm deep. The lids are assembled by first applying an optical adhesive cover (#04 729 757 001, Roche, Basel, Switzerland) to the back of the lid and then placing two layers of absorbent pad (#CFSP223000, Millipore, Billerica, MA) in the designated slot. Both bases and lids are ultraviolet sterilized for 30 min, prior to cell culture, and lids are loaded with reagents prior to each application.
Fluid Handling
Fluid exchanges are characterized by measuring (1) duration, (2) washing efficacy, and (3) cross-contamination during fluid replacements. Fluid replacements are initiated by mating the lid with the base, thereby inserting the series of posts into the microwells. (1) To assess time required for fluid replacement, the microwells are filled with 15 µL of deionized (DI) water, and the lid is clicked on the base. Time is measured from the point of initial fluid contact between the post and the fluid to the point at which the microwell is emptied. (2) Washing efficacy is assessed by measuring the depletion of a fluorescent dye after individual wash steps. Channels are preloaded with a fluorescent dye (Alexa Fluor 488, #D-22910; Life Technologies, Carlsbad, CA) and then washed by applying lids containing 15 µL of DI water per microwell. Channels are imaged after each wash is completed (IX-70; Olympus, Shinjuku, Tokyo, Japan), and fluorescence intensities are measured using ImageJ. The background autofluorescence, inherent to the PS, is measured, averaged (
Cell Culture for IHC Assay
In this section, all fluid transfers, including cell seeding, are executed by filling each of the 60 microwells with 15 µL of the respective reagent and then mating the lid with the base. NMuMG cancerous epithelial cells are seeded in KOALA-HD channels at a surface density of 400 cells/mm2 and cultured at 37 °C for a total of 72 h. For the first 24 h, cells are cultured in DMEM media with 4.5 g/L glucose (#10-017-CV; Corning Cellgro, Manassas, VA) supplemented with 10 µg/mL insulin and 10% fetal bovine serum. For the next 48 h, the cells are cultured in the same media supplemented with a 0, 2, 20, or 200 pM concentration of transforming growth factor β1 (TGF-β1; #100-21; Pepro Tech, Rocky Hill, NJ). The media are replaced every 24 h. Cells are washed with 1× phosphate-buffered saline (PBS), fixed with 4% paraformaldehyde (#43368; Alfa Aesar, Ward Hill, MA) for 15 min, and permeabilized with 0.1% Triton X-100 (#807426; MP Biomedicals, Santa Ana, CA) for 30 min. Channels are washed with 1× PBS and then blocked with 1× PBS supplemented with 3% bovine serum albumin (PBS+BSA) for 30 min. Primary antibody solutions are prepared by separately diluting anti-β-catenin (rabbit) to 2 µg/mL, anti-E-cadherin (mouse; #610182; BD Biosciences, San Jose, CA) at 1 µg/mL, anti-FAK (rabbit; #ab40794; Abcam, Cambridge, England, UK) to 2 µg/mL, anti-N-cadherin (rabbit; #ab18203; Abcam) to 6 µg/mL, or anti-vimentin (mouse) at 1:50 into PBS+BSA supplemented with 0.1% Tween 20 (PBST+BSA; #P1379; Sigma Aldrich). Primary antibody solutions are added to appropriate channels (with
Results and Discussion
KOALA-HD represents a novel class of high-throughput microfluidic platforms that can be operated quickly, reproducibly, and reliably with minimal demands for training or equipment. The KOALA-HD compresses 60 microfluidic assays into the 25 mm × 75 mm footprint of a traditional glass slide, enabling medium- to high-throughput applications by hand without the need for an automated liquid handler. The number of assays that can be performed in this footprint (i.e., assay density) is comparable to that of a 384-well plate. However, unlike assays performed in 384-well plates, fluid replacements for KOALA-HD are performed in all 60 assays simultaneously. The high-density format of the channels in the base resulted in design challenges that needed to be resolved to prevent cross-contamination of reagents and support reliable operation of the device. These design challenges and solutions are discussed here.
Fluid Exchanges in KOALA-HD
All fluid-handling mechanisms necessary to operate KOALA are integrated into the KOALA lids and bases. Each lid is a one-time-use component composed of 60 microwells, which houses reagents and an absorbent pad to drive fluid flow (
Fig. 2C
). Similarly, each base is composed of 60 microfluidic channels; however, unlike the lids, the base is used for the duration of the assay. Each fluid replacement—performed by clicking a lid onto the base—is completed for all 60 channels within 15 s (see
To make KOALA suitable for HD applications, however, several obstacles must be overcome, including (1) determining a configuration suitable for densification, (2) eliminating cross-contamination, and (3) achieving complete and defined fluid exchanges in the new HD format.
To determine a setup that best supports the densification, we test two designs: a format that uses individual posts as described above and a common rail configuration (
To eliminate cross-contamination, hydrophilicity between the posts and fluid displacement in the microwells is decreased. After plasma treatment, we implement a novel approach, described by Guckenberger et al. 9 to recover surface hydrophobicity between the posts, thus mitigating hydrophilicity-based cross-contamination. Inadequate contact between the outlet posts and the absorbent pad lead to cross-contamination via rapid displacement of the fluid in the microwells. This issue is circumvented by ensuring proper connection between the outlet posts and the absorbent pad prior to inserting the inlet posts into the microwells. In the future, a small, reusable device for repeatably aligning and mating the lid and base (i.e., a jig) will be designed to facilitate this connection and prevent the possibility of user error.
To ensure complete and defined fluid exchanges for each channel, we leverage the designs of the microwells, the posts, and the absorbent pads. The volume allotted for each fluid exchange is dictated by the volume of the microwell. However, maximizing use of the fluid in the microwell, and thus minimizing dead volume, depends on the orientation of the inlet post and the size of the absorbent pad. We found that to minimize dead volume, the open side of the horseshoe should be directed toward the end of the microwell (
Evaluating KOALA-HD Function
To make a highly functional platform to be used for toxicology and cell-based studies, we assess the platform for its ability to (1) ensure proper fluid replacement in the channels; (2) ensure no cross-contamination between adjacent channels, a difficulty particularly compounded by higher throughput; and (3) perform several fluid exchanges without failure.
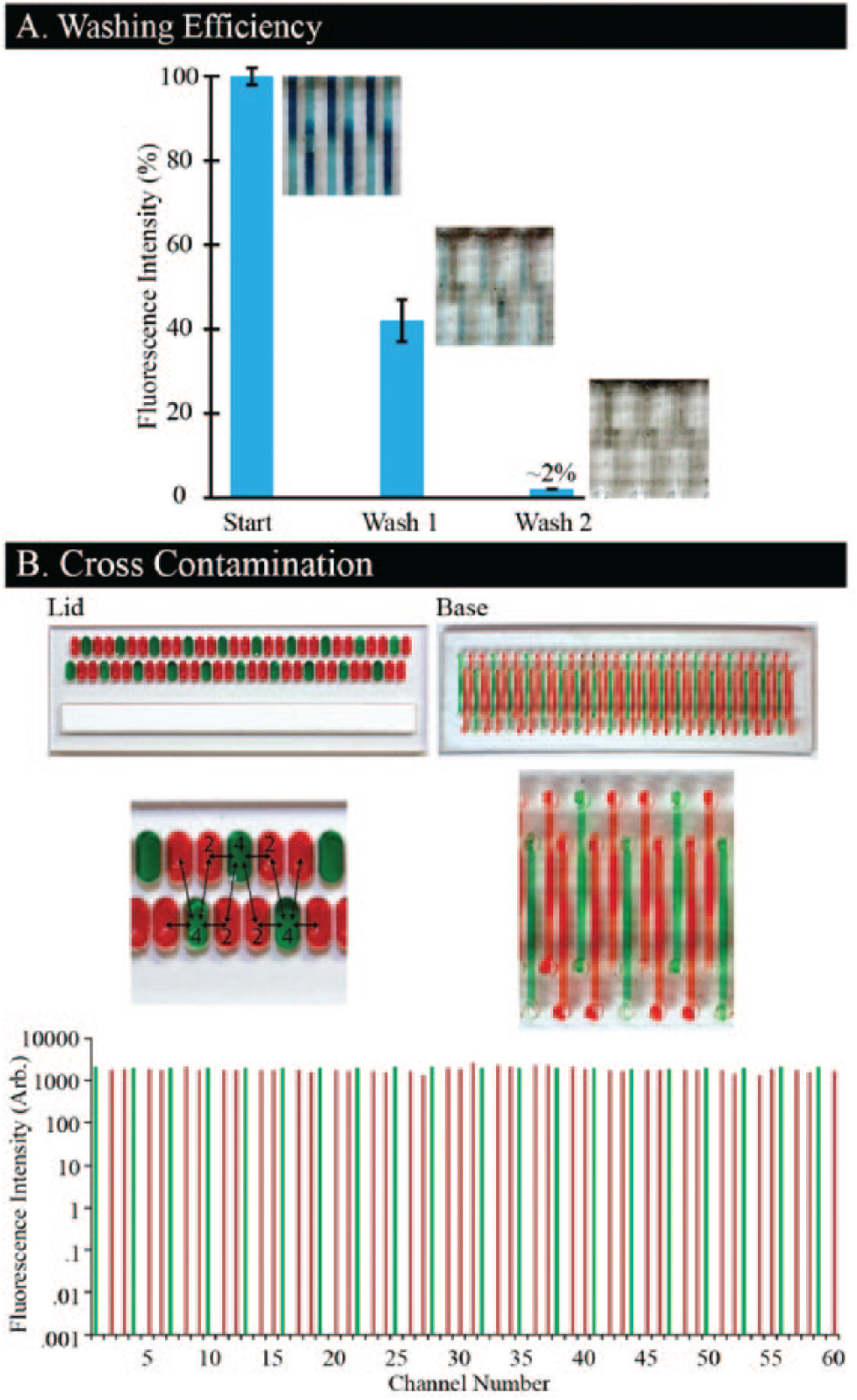
We quantify the depletion of a fluorescent dye after washing steps to assess the efficiency of fluid replacements (
Fig. 3A
). To fulfill this, a lid filled with fluorescent dye is applied to the channels, followed by two washing lids. We quantify the fluorescent depletion after each wash and normalize it to the starting intensity. We found that one washing lid is capable of removing ~60% of the initial fluid, and a second washing lid reduced the dye concentration by more than 98% (
(
Cross-contamination is assessed by quantifying fluorescence in each microchannel after applying a lid prefilled with two fluorescent dyes (
Fig. 3B
). The lid is strategically filled such that each microwell containing green dye is directly adjacent to four microwells with red dye (illustrated with food coloring), enabling detection of cross-contamination from both horizontally and diagonally adjacent microwells. This lid is applied to a base, and each channel in the base is quantified for both red and green fluorescence (
Fig. 3B
, bottom). The results reveal no cross-contamination between channels based on no increase in fluorescence intensity between channels (
Finally, we assess the ability to operate a fluidic assay without failure by applying a series of lids and verifying complete flow in each channel. A series of 20 lids are prefilled with dyed water and applied to a single base. For comparison, about 15 fluid replacements are typically required for a traditional immunochemistry staining. Each lid is filled with alternating colors to make channel failure obvious. All 20 fluid exchanges are completed without a single channel failure.
Channel Design for Cell Culture
To ensure suitability for cell culture, KOALA-HD is designed to (1) allow precise readouts of cellular functions (proliferation, protein localization, etc.), (2) contain enough media to maintain cell viability throughout the incubation period, and (3) be negligibly affected by evaporation. The channels are designed with a lowered region in the center that serves three purposes. First, the decreased plastic thickness enables better cellular readouts (e.g., higher magnification microscopy, less autofluorescence from the plastic). Second, the increased channel size reduces the shear stress by slowing fluid velocity. Third, the lowered region increases the volume and thus increases the reservoir of nutrients that are available for the cells. With this channel design, we are able to culture cells for 24 h without media exchanges.11,12 However, for more highly active or sensitive cells, more frequent fluid replacements may be necessary. Evaporation is mitigated using previously described and characterized methods.13,14
IHC Assays
We demonstrate the ability to perform high-density IHC assays and complex staining processes reliably and without the need to prepare antibody dilutions or source-specific reagents. We chose an IHC assay based on its ubiquity in screening applications and for the multitude of steps necessary to complete the assay (i.e., cell culture, fixation, permeabilization, staining, washes, etc.). We use a relevant cancer-specific assay in which NMuMG epithelial cells are treated with various dosages of TGF-β1, inducing an epithelial-to-mesenchymal transition. 15 Markers of this transition include down-regulation and localization of β-catenin and E-cadherin to cell-cell contact areas 16 and increased expression of FAK, 17 N-cadherin, 18 and vimentin. 19 Such complex readouts require cell culture, treatment with a drug, and IHC staining. We demonstrate cell culture in KOALA-HD by culturing NMuMG cells in 60 channels for 72 h. The process requires 15 sequential fluidic steps, all performed using the KOALA platform (i.e., cell seeding, media replacements, TGF-β1 dose treatments, fixing solution, washing buffer, primary antibody staining, and secondary antibody staining). The platform is able to perform each fluidic step in all channels simultaneously without failure or cross-contamination. Each fluidic exchange requires about 15 s with a simple mating of the required lid with the base containing cells.
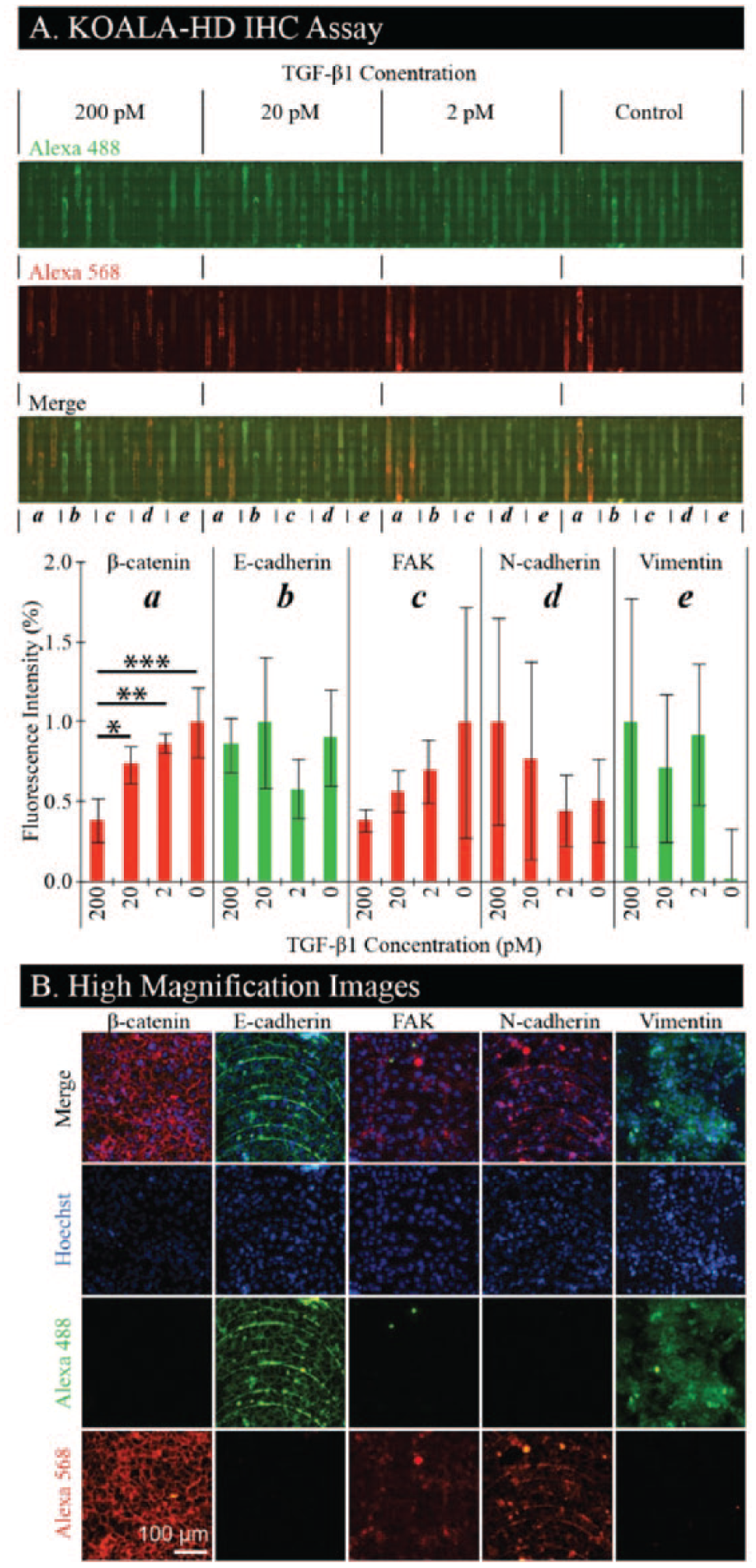
Cells are treated in four groups of 15 channels with varying concentrations of TGF-β1, after which cells are fixed, permeabilized, stained with a nuclear stain, and stained in triplicate for β-catenin, E-cadherin, FAK, N-cadherin, and vimentin ( Fig. 4A ). We found that β-catenin decreases and N-cadherin increases with increasing levels of TGF-β1, as expected. We did not observe any decrease of E-cadherin for increasing levels of TGF-β1, but we did observe localization to the cell-cell contact areas. FAK decreases with increasing levels of TGF-β1, which is inverted from typical results but has been observed in NMuMG cells expressing D119A-β3 integrin. 17 Finally, prevalence of vimentin, a mesenchymal marker, is evident in cells treated with TGF-β1, indicating a transition to a mesenchymal phenotype. Selected channels treated with the highest concentration of TGF-β1 (i.e., 200 pM) are imaged at high magnification ( Fig. 4B ). These images demonstrate localization of β-catenin and E-cadherin to the cell-cell contact areas. A noticeable feature in some of the images is the striated pattern formed by the cells arranging themselves along fabrication artifacts. These are the result of machining marks left by the CNC machine during the channel fabrication and can be removed by using other fabrication methods such as injection molding. All channels are stained with both the Alexa 488 and Alexa 568 dyes to detect nonspecific binding. Importantly, we demonstrate that KOALA-HD can—in the footprint of a glass slide—cover a broad range of treatments and readouts, providing high-content microscopy images at a high-throughput scale, without a need for automation or trained personnel. In future applications, this platform could be imaged with a scanner for higher-throughput analysis.12,20
(
Conclusion
The KOALA-HD platform is a microscale fluidic handling technology that simplifies cell-based assays such that anyone can perform them. The only operation is to place a lid onto a base. KOALA-HD enables reagent prepackaging and cryopreservation—acting to reduce preparation time and waste, lower demands on infrastructure or trained personnel—expanding access of cell-based assays to more labs, and high-throughput microfluidic applications through the scalability offered by parallelized fluidic handling procedures. We demonstrate how feature dimensions and geometry can be altered to accommodate close packing of microfluidic channels. In addition, we characterize the robustness of simultaneously performing 60 assays within the footprint of one microscope slide. We demonstrate that the KOALA-HD would be suitable for toxicology, environmental pollutant, and natural product research by showing functional and advanced cellular readouts in the form of protein expression and morphology. However, because of the simplistic nature and device modularity, it is possible that KOALA-HD could be expanded into other areas, including patient screening in clinics,7,21 water quality tests, or even food safety testing.
Footnotes
Acknowledgements
The authors thank Dr. Joshua Lang for use of his microscope. The authors thank Brian Johnson and Ashleigh Theberge for their assistance with statistical analysis.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors hold equity in Salus Discovery LLC, which has licensed technology described in this article. All authors hold equity in Tasso, Inc. D.J.B holds equity in Bellbrook Labs, LLC.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH R01 EB010039.
