Abstract
This article describes a novel microfluidic 3-dimensional encapsulation method via the self-assembling peptide hydrogel. The microfluidic immobilization strategies using a peptide hydrogel have been designed for microfluidic cell-based assays, cocultures, and biomimetic micro blood vessels. A sol-gel transition peptide hydrogel, Puramatrix, is adopted for use in the microfluidic device fabricated by photolithography and a poly(dimethylsiloxane) replica molding process. The peptide hydrogel was hydrodynamically focused by sheath flows of distilled water and cell culture media, and gelled by diffusion of media. After being transitioned from a sol to gel phase, the fabricated scaffold in the middle of the main channel was not washed away via fluid flows. The diffused chemicals in a stripe-shaped peptide scaffold of microchannel formed a linear concentration gradient within the scaffold. Based on application in an in vivo-like 3-dimensional microenvironment, this microfluidic system could be applied to cocultures, angiological research, cytotoxicity tests, cell viability monitoring, and continuous dose-response assays as well as drug-drug interaction studies.
Keywords

Introduction
Establishing an appropriate environment and condition is critical to the cultivation of cells as these factors affect and control cellular function and behavior in animal cell cultures. 1, 2 Recently, a variety of techniques have been proposed for realizing suitable cell culture conditions. One of the most promising approaches is microtechnology. Microfluidics can provide in vivo-like cellular conditions and microenvironments 3 and support the sophisticated tools for cell studies including chemotaxis, 4 patch clamps, 5 and cell differentiation. 6 Also, as 3-dimensional cultures are preferred over conventional 2-dimensional cultures, 7 tissue engineering approaches have been attempted for 3-dimensional cell cultivation. Although a combination of both technologies could lead to the realization of a desirable cell culture system, synthetic polymers widely used in tissue engineering such as polylactide-glycolide (PLGA) and polylactic acid (PLA) are not easily applied to microfluidic channels. Furthermore, it is difficult to observe cell morphology by optical microscopy because of their opaque characteristics.
In this study, we report a novel microfluidic 3-dimensional encapsulation system by the self-assembling peptide hydrogel, a material that is known to be highly biocompatible to cells. 8 Sol-gel transition of a peptide hydrogel is the basis for various 3-dimensional encapsulation processes such as 3-dimensional cell immobilization, cocultures, and sidewall immobilization mimicking a blood vessel. This process requires a sol-gel transition material, a material to induce gelation, and a material to control the sol-gel transition. In this study, a sol-gel transition peptide hydrogel, Puramatrix, is adopted for use in the microfluidic device. The microfluidic device was fabricated by photolithography and a poly(dimethylsiloxane) (PDMS) replica molding process. Here, we focus on the fabrication of various micro peptide scaffolds adequate for application as a biomedical cell culture platform, such as microfluidic cell-based assays, cocultures, and biomimetic micro blood vessels. The concentration gradient profile in the peptide scaffold is also described.
Experimental
Microfabrication of a Microfluidic Device
We have designed a microfluidic device for 3-dimensional encapsulation via hydrodynamic focusing. The configuration of a representative microfluidic device is shown in Figure 1A. Inlets 1, 2, and 3 are for the peptide hydrogel, distilled water, and Dulbecco's modified Eagle's media (DMEM; GIBCO-BRL, Gaithersburg, MD), respectively. Because the peptide hydrogel is transitioned by media, distilled water is used to control the gelation by flowing at the middle of the hydrogel and media.
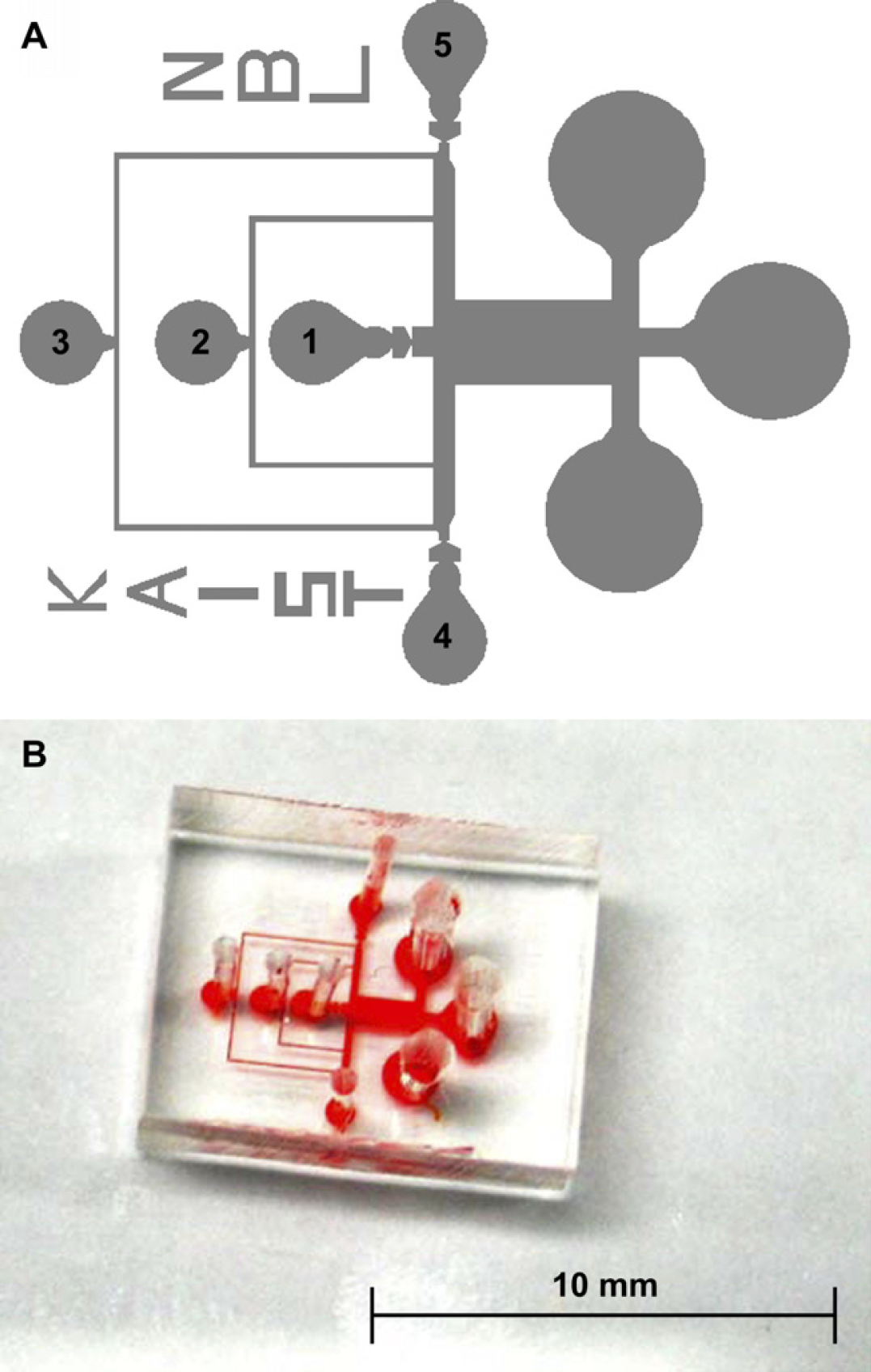
(A) Design and (B) fabricated PDMS device for a microfluidic 3-dimensional encapsulation device.
The microfluidic device was fabricated by photolithography and a PDMS replica molding process. Monitoring 3-dimensionally immobilized cells or particles in a microchannel with a height exceeding 100 μm is difficult due to limitations in optical focal length; accordingly, the microchannel was designed to 50 μm thickness. A negative photoresist (PR), SU-8 (Microlithography Chemical Co., MA), was spin-coated on a bare Si wafer at a speed of 500 rpm for 10 s and at 1600 rpm for 30 s. After flattening and soft baking, the PR was patterned by UV exposure of 350 mJ cm−2. A mixture of PDMS prepolymer and curing agent (Sylgard 184; Dow Corning, MI) was poured onto the fabricated mold and cured at 60 °C for 3 h in a convection oven to complete cross-linking. Holes for inlet and outlet ports were punched and the PDMS replica and a slide glass were sonicated in 70% ethanol for 30 min as a sterilizing and cleaning process. After drying the replica and slide glass, air plasma (200 mTorr, 200 W) using an expanded plasma cleaner (Harrick Science, Ossing, NY) was applied for irreversible bonding. Finally, the microfluidic 3-dimensional encapsulation device was assembled as shown in Figure 1B.
Procedure for Microfluidic 3-Dimensional Encapsulation
Sol-gel transition of a peptide hydrogel for 3-dimensional encapsulation requires a sol-gel transition material, a material to induce gelation, and a material to control the sol-gel transition. In this study, self-assembling Puramatrix peptide hydrogel (BD sciences, MA), media, and distilled water were used for the materials, respectively.
Figure 2 shows the 3-dimensional encapsulation process, which occurs in the middle of the main channel. After removing air in the microchannels, distilled water is flowed followed by the media, which is focused to the sidewalls of the channels. When the streams are stabilized, a mixture of Puramatrix and polystyrene microbeads (Fluka, Germany) is flowed along the middle of the main channel, as shown in Figure 2A. After the mixture reaches the middle outlet, the distilled water flow is immediately turned off and both sides of the media contact the mixture within a few seconds (Fig. 2B). When the media approach the mixture, the mixture flow is also turned off. At this time, the media are diffused into the mixture and the peptide hydrogel is transitioned from sol to gel phase. The microbeads in the mixture are thus 3-dimensionally encapsulated within the peptide hydrogel (Fig. 2C). Finally, distilled water is flowed again, and a 3-dimensionally immobilized peptide scaffold can be seen in the middle of the main channel and is not washed away (Fig. 2D).
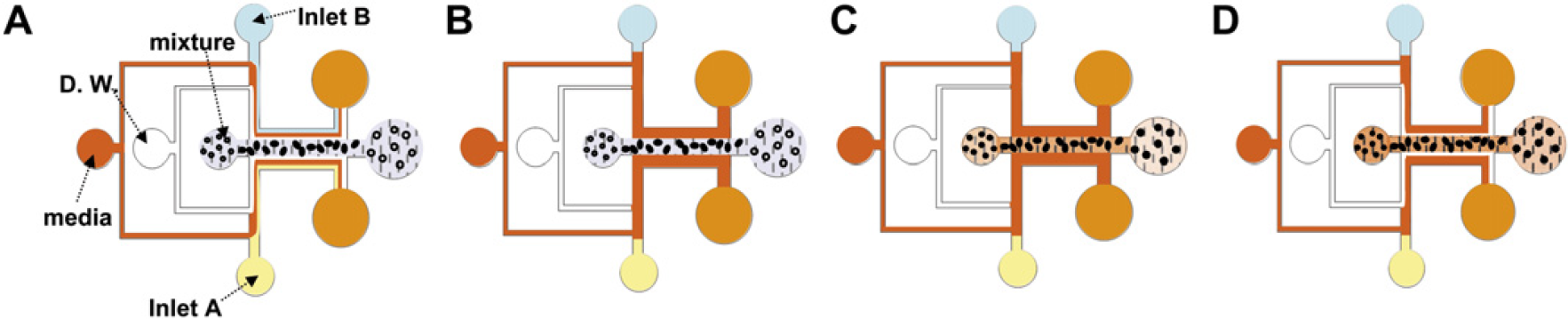
Procedure for 3-dimensional encapsulation in the middle of the main channel (A). Distilled water (D. W.) focuses media to side-walls of the main channel and mixture is flowed into the main channel. (B) D. W. is stopped and both sides of the media contact the mixture. (C) The mixture is also stopped, and the media diffuse into the mixture. The hydrogel is transitioned to a gel phased stripe-shaped peptide scaffold and microbeads (or cells) are 3-dimensionally encapsulated in the scaffold. (D) As D. W. is flowed, the presence of a 3-dimensionally immobilized peptide scaffold, which is not washed away, can be confirmed.
Procedure for 3-Dimensional Coculture in a Microchannel
By modifying the mixture inlet part, the system can also achieve 3-dimensional coimmobilization in a microfluidic channel. Fig. 3 shows the modified design and process for the coimmobilization system. The procedure for coimmobilization is similar to that described earlier for 3-dimensional encapsulation in the middle of the main channel.
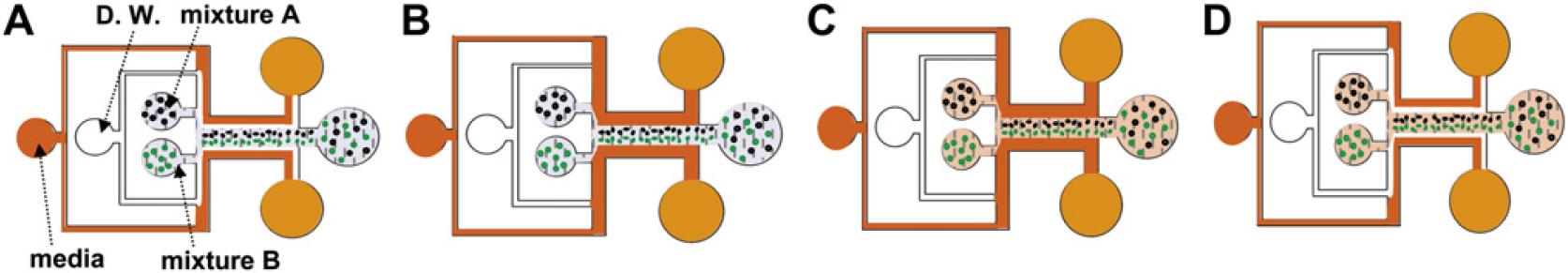
Procedure for microfluidic 3-dimensional coculture. (A) Both mixtures A and B are flowed instead of the mixture of Fig. 2A. (B) D. W. is turned off. (C) Mixtures A and B are also turned off and they are transitioned from sol to gel by the media. (D) A stripe-shaped coimmobilized scaffold was fabricated.
After distilled water directs the media to the sidewalls of the microchannel, mixtures A and B are flowed and focused to the middle of the main channel by sheath flows. The streams of the two mixtures contact each other and laminar flows are formed (Fig. 3A). When all streams are stabilized, distilled water flow is stopped and the media contact mixtures A and B from both sides (Fig. 3B). The two mixtures are then transitioned from sol to gel and mixtures A and B are 3-dimensionally encapsulated in the very vicinity of each scaffold (Fig. 3C). By flowing distilled water, the formation of 3-dimensionally encapsulated microbeads or cells can be confirmed (Fig. 3D).
Procedure of 3-Dimensional Immobilization for Mimicking a Blood Vessel
Three-dimensional immobilization to the sidewalls of the main channel can be accomplished by exchanging mixture and media flows, as shown in Figure 4. The mixture and media are flowed by inlets (3) and (1), respectively. When streamlines are formed, as shown in Fig. 4A, the mixture flow is stopped and the media are diffused to the hydrogel (Fig. 4B). As distilled water is flowed again, hydrogel patterning suitable for application as a biomimetic micro blood vessel can be seen (Fig. 4C).
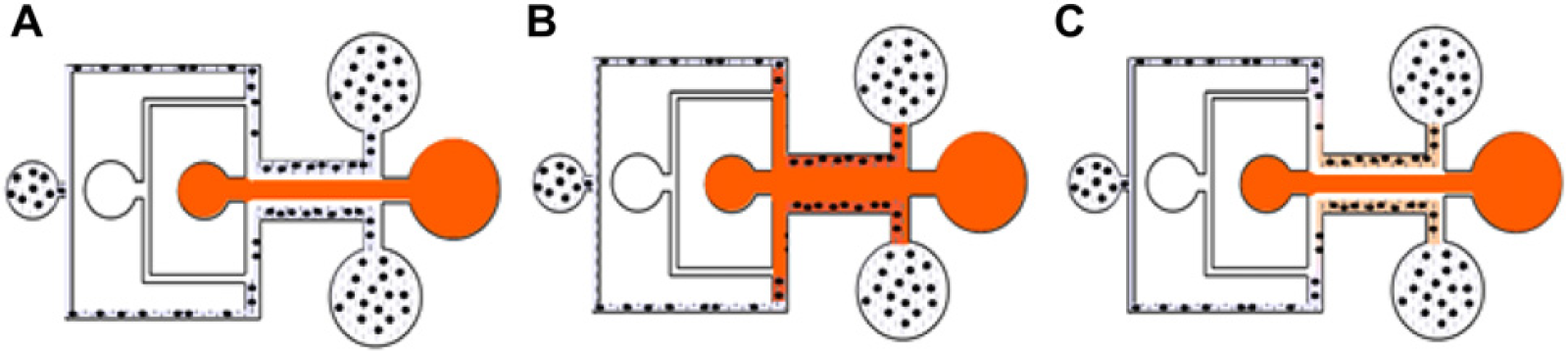
Procedure of sidewall immobilization for biomimetic micro blood vessel. (A) The inlets for media and mixture are exchanged. (B) After formation of stable streamlines, D. W. is stopped. The media are diffused into sidewall located mixtures. (C) By flowing D. W., immobilization for the micro blood vessel is verified.
Measurement of Concentration Gradient Profile in the Peptide Scaffold
Alexa Fluor 488 goat anti-mouse IgG (molecular weight: ∼ 150 kDa) was selected to examine the concentration gradient by fluorescence intensity. The fluoropore solution was diluted 10:1 with distilled water and injected to the microfluidic device via inlet (4). At the same time, pure water was also injected at the other side (5). The fluorophore was exited by a 50-W mercury lamp with an illumination wavelength of 490 nm. An image analysis was performed with the Java-based image analysis program ImageJ (W. Rasband, ImageJ 1.29x freeware, rsb.info.nih.gov/ij/). After converting the images from 32 bits to 8 bits gray scale by a type conversion mode, the fluorescence intensity profile was measured on the basis of the integrated density value. These raw data were subtracted from the background and normalized by the maximum value.
Results and Discussion
3-Dimensional Encapsulation Strategy for Cell-Based Assays
Until now, some efforts making functional 3-dimensional tissues have been made by improvement of synthetic polymers such as PLGA, PLA, and polyglycolic acid. The pore sizes of these polymers range from tens to hundreds of micrometers. However, because peptide hydrogel mostly has pore sizes from tens to hundreds of nanometers, an authentic 3-dimensional environment for cells can be made by using natural polymers. In this study, we have used a biologically inspired self-assembling peptide hydrogel having a pore size of several tens of nanometers. This material minimizes the risk of carrying biological pathogens relative to animal-derived biomaterials, 9 and it also provides an appropriate environment for the retention of cell phenotype, as well as for cell differentiation and tissue formation. 10 Self-assembly takes place when it is exposed to electrolyte solution, salt solution, or pH, where the net charge of the peptide molecule is near zero. 9 As a preliminary study, the sol-gel transition degree of Puramatrix was conducted. Because the distilled water does not undergo sol-gel transition of the solution to the best of our knowledge, it was used as a sol-gel control solution for Puramatrix whereas cell culture medium was used for gelation of the peptide hydrogel. Before flowing distilled water, 70% ethanol was flowed for 30 min. This step contributes to sterilization and sustenance of hydrophilicity in a micro-fluidic channel. In addition, bubbles entrapped in microchannels can be removed.
In this study, because we focus on the development of various 3-dimensional immobilization methods in a microchannel, 12 μm polystylene beads were used instead of cells. Also, media was mixed with food dye (Kemide Co., Korea) at a 1:1 ratio for visualization of clear streams. Figure 5 shows images of fabricated peptide scaffolds for a 3-dimensional cell culture and cell-based assays. Polystylene beads suspended in a 10% sucrose solution were mixed with Puramatrix at a 1:1 ratio. Distilled water was flowed to separate the media and mixture, and then the media and mixture were flowed. As the distilled water flow was turned off, the media and mixture approach each other. At this point, the flow rate of the media was doubled to maintain predetermined thickness of the mixture stream. When the media contact the mixture, the mixture was stopped (Fig. 5A). The media diffused into the mixture for a few seconds (Fig. 5B). When distilled water was flowed again, we could observe a stripe-shaped peptide scaffold tightly fixed on the surface. The fabricated nanoporous scaffold contained the media for some time (Fig. 5C). Because Puramatrix is highly viscous compared to water, the use of 50% sucrose solution enhanced the shape of a straight peptide scaffold by compensating the difference of viscosity as compared to that for pure water. The thickness of the scaffold could be controlled by the ratio of the three fluid flow rates and was reproducibly produced in a range from 50 μm to more than 600 μm thickness (coefficient of variance; 5.8%, n = 7).

Photographs of peptide hydrogel immobilization in the middle of the main channel. (A) After formation of stable streamlines of a mixture including 12-μm polystyrene beads, D. W., and media, D. W. was stopped so that the media contacted the mixture. (B) The media were diffused into the mixture where the beads are 3-dimensionally encapsulated. (C) A picture of the fabricated peptide scaffold in the middle of the main channel whose thickness is 300 μm ± 8 μm. Scale bars are 100 μm.
Fabrication of Peptide Scaffold for Coculture
Coculture systems mimicking an in vivo environment have significant potential for studying cell-cell interactions and cellular behavior 11 and have been widely used for tumor angiogenesis study, 12 a blood-brain barrier (BBB) model, 13 embryo development, 14 etc. Here we have developed a hydrodynamic-based microfluidic 3-dimensional coimmobilization method for application in a coculture. Two different kinds of beads, a 12-μm polystylene bead and a green fluorescent 7 μm streptavidin conjugated microsphere (Polysciences Inc., PA), were used for demonstration of coimmobilization. Each bead was mixed with the peptide hydrogel at a 1:1 ratio and injected via the port for mixtures A and B, as shown in Figure 3. Figure 6A exhibits flow streams of two mixtures in the middle of the main channel. When the streams became stable, distilled water was stopped. When the media approached the mixtures, the flows of the two mixtures were also stopped and the media diffused into the mixtures (Fig. 6B). Figure 6C shows 3-dimensionally immobilized beads in a cocultured form. The two streams formed a single straight fluid stream and fused with each other. However, because the fluids were governed by laminar flow, the two beads were not mixed, as confirmed by fluorescence microscopy (Fig. 6D).
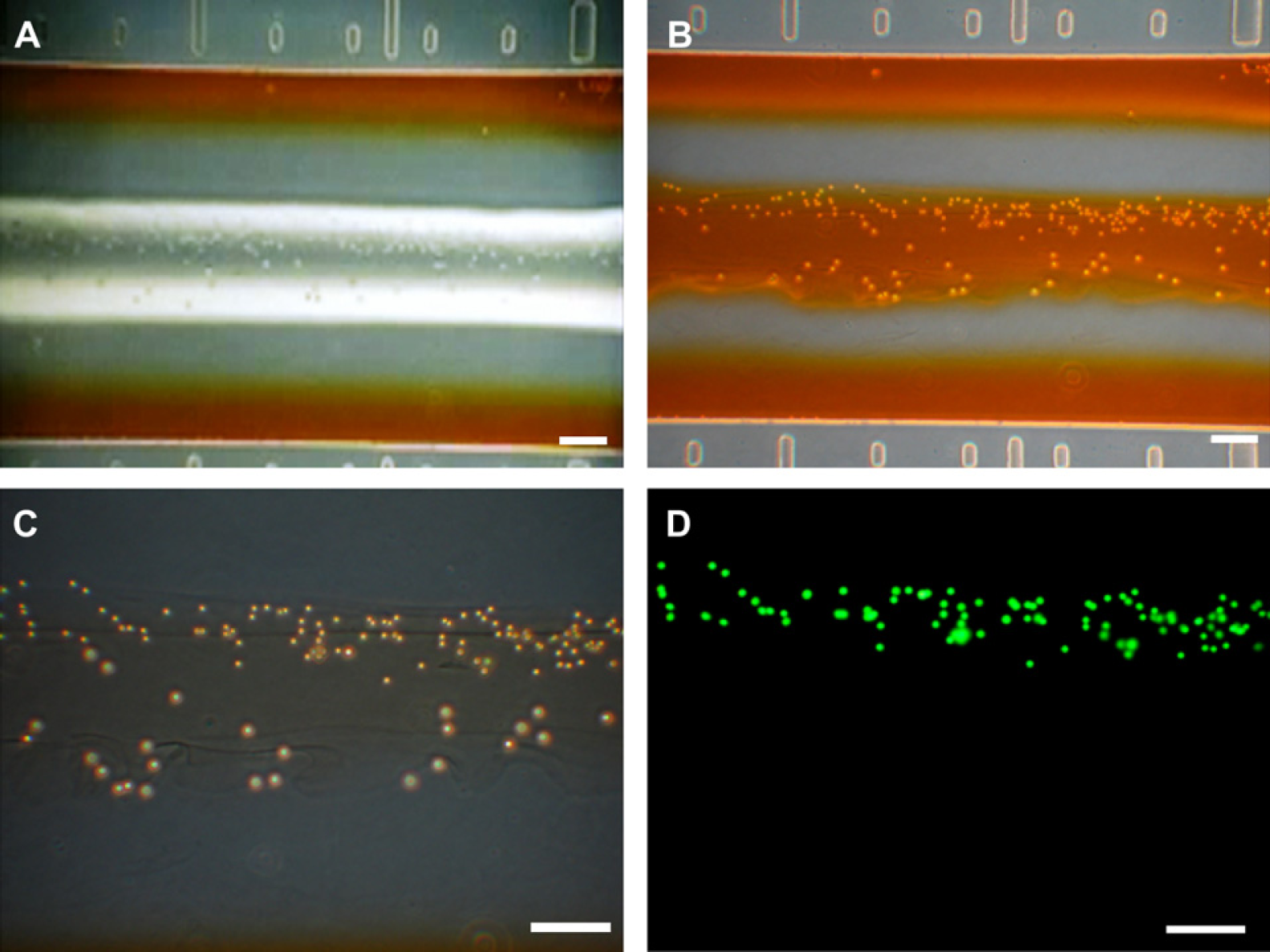
Result of microfluidic peptide hydrogel immobilization for 3-dimensional coimmobilization. (A) Flow streams of D. W., media, and two mixtures were formed in the main channel. (B) Image of a fabricated scaffold for 3-dimensional coculture. (C) Two different kinds of beads (a 12-μm polystylene bead and a 7 μm green fluorescent streptavidin conjugated microsphere) were 3-dimensionally immobilized in the vicinity. In addition, they were not mixed with each other because the flows were laminar. (D) Confirmation of bead immobilizations by fluorescence microscopy. All scale bars are 100 μm.
Direct contact between cells used in a coculture can cause a problem, especially in human embryonic stem cells and BBB model. This method has a potential advantage to cultivate two cells in the same vicinity via an indirect contact mode. Also, more credible cell-based assays could be realized in that the system expressing cell-cell communication mimics a biological microenvironment.
Sidewall Immobilization for in vitro Angiological Study at a Microscale
2-Dimensional cell patterning via a variety of methods has been extensively studied. 15 However, because the cubic blood vessel environment cannot be readily reflected in this technique, a novel 3-dimensional cell immobilization mimicking a blood vessel is described here for in vitro angiological study application. Figure 7A shows a schematic of a blood vessel. Figure 7B presents the device schematic, whereby the structure and geometry of a blood vessel are reflected using the microfluidic device. By changing the fluids between media and a mixture of Puramatrix and cells, we could simply realize 3-dimensional cell immobilization for mimicking a blood vessel structure. Figure 7C–F show images of the procedures for sidewall immobilization of the main channel. To demonstrate the validity of the concept, we used polystyrene microbeads instead of specific cells. After formation of stable streamlines (Fig. 7C), the flow of distilled water, which was dissolved with 50% sucrose to reduce the viscosity difference, was stopped. When the media was near to the side mixtures, the flow of media was doubled and diffused into the mixtures. Faster media flow contributed to clear defined hydrogel pattern. However, roughly 15% variation of hydrogel thickness occurred.
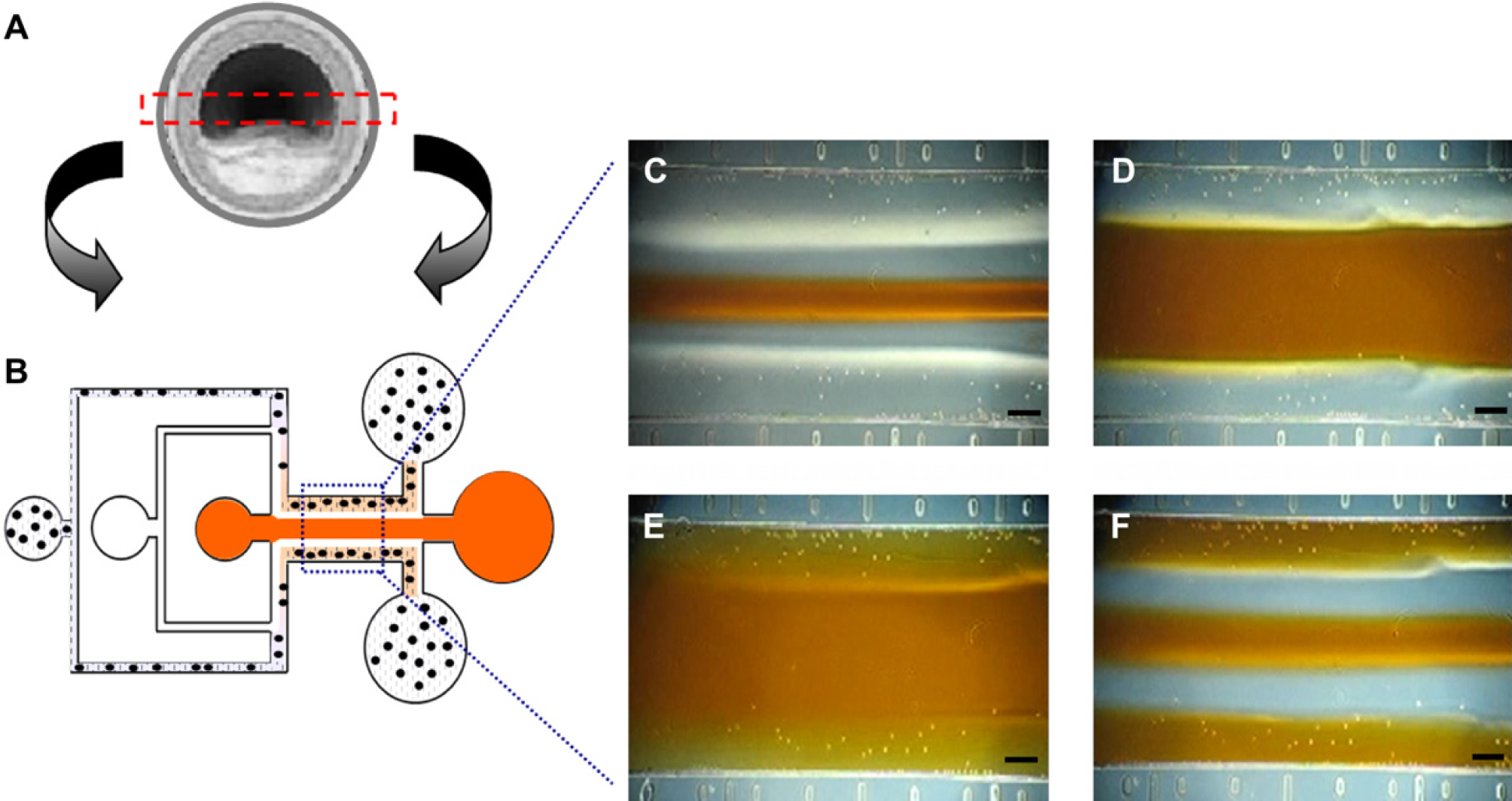
Sidewall immobilization of peptide hydrogel to mimic micro blood vessel. (A) Schematic of a blood vessel. (B) Device configuration to create biomimetic blood vessel structure. (C)–(F) Process to fabricate peptide scaffold by immobilizing the hydrogel to the side-walls. All scale bars are 100 μm.
Blood vessels vary in dimension from a few micrometers over 6 mm in the human body, and the thickness and geometry of blood vessel walls affect fluid dynamics and response of endothelial cells. Consequently, many diseases are related to the geometry of blood vessel walls. Because the thickness of blood vessel walls and width of blood stream can be controlled by the device dimensions, the mixture, and the media flow rate; the system can mimic specific blood vessels as well as models of vascular disease such as coronary sclerosis through proper geometry design. Whereas many trials to mimic blood vessel models have been conducted on a macroscale using synthetic polymers and biodegradable scaffolds, 16 few studies have been reported in microfluidic devices in relation to angiological study at a microscale. The system is expected to be helpful to study the relationships between properties of fluids and a variety of cells including endothelial cells, marrow stromal osteoblasts, and nerve cells.
Concentration Gradient Profile within Peptide Scaffold
The realization of 3-dimensional culture as well as in situ cell-based assays for drug screening systems is highly desirable. We have characterized a concentration profile within a fabricated peptide scaffold. After fabricating a 300-μm stripe-shaped peptide scaffold, the mixture of cells and peptide hydrogel, distilled water, and media were stopped and only fluorophore 488 solution and pure water were flowed at 30 μL h−1, as shown in Figure 8A. Figure 8B presents a fluorescence image of steady state in the main channel where the state was sufficiently maintained after 30 s of flowing fluids. In the area around the peptide scaffold, linear concentration gradients were formed at the entire area and did not vary according to the x-axis positions of the fabricated scaffold (Fig. 8C). Scaffolds with different thicknesses were fabricated by controlling the fluid flow rates and the profiles showed that the linear region depends on the scaffold thickness (data not shown). As a preliminary result, we found that the concentration profile using fluorescein (Sigma, molecular weight: 332.31) also showed a linear concentration gradient. Therefore, we found that the profile is not dependent on the molecular weight of the solutes.
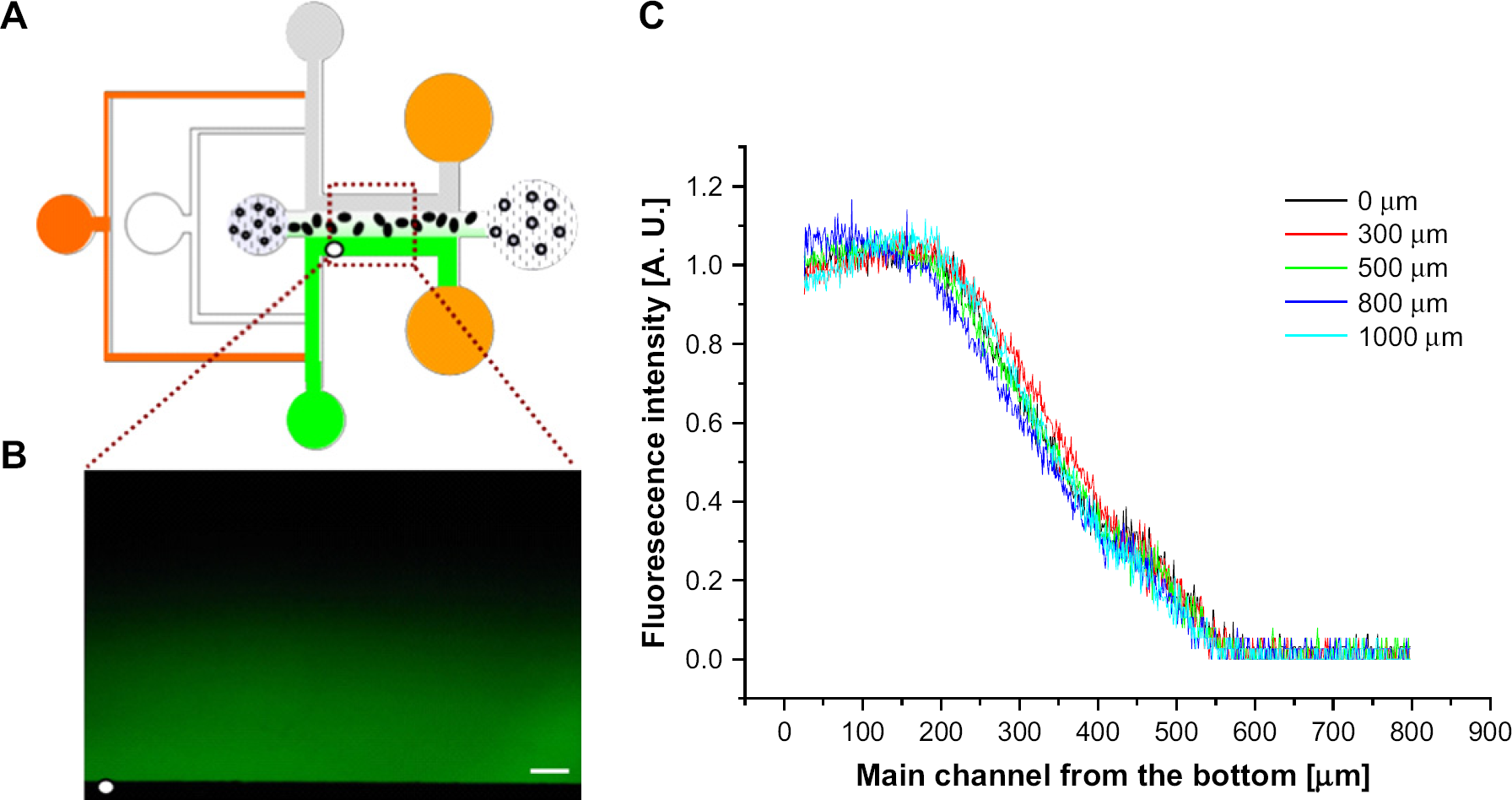
Characterization of concentration profile in the main channel. (A) Device configuration to measure the concentration profile by fluorophore 488 fluorescence solution and pure water. (B) A concentration profile formed in the main channel. Scale bar is 100 μm. (C) Fluorescence intensity at the main channel of the microfluidic device. The lines denote the intensity profiles of vertical axes away from the white dot of Figure 8B. The concentration gradient was linear at the entire area of the scaffold and did not fluctuate according to the positions of the scaffold.
Compared to other approaches requiring additional components to generate a concentration gradient such as long channels 17 and electric fields, 18 the microfluidic system described in this study enables realization of a 3-dimensional cell culture as well as in situ linear concentration gradient generation without any complicated or additional micro-channels. Detection of dose-dependent cell viability and behaviors has been widely studied for drug screening and cell study. This platform will contribute to construct a useful drug screening tool and system, using the dose-dependent responses according to the concentration gradient within a fabricated peptide scaffold. In addition, drug-drug interaction study could be accomplished by flowing different drugs instead of fluorescent solution and pure water.
Conclusions
A novel 3-dimensional encapsulation method using microfluidics was developed on the basis of various immobilization strategies. A biocompatible self-assembling peptide hydrogel, Puramatrix, was adopted in the microfluidic channel and used to fabricate micro peptide scaffolds for 3-dimensional cell cultures, cocultures, and biomimetic micro blood vessels. A highly linear concentration gradient was shown in scaffolds fabricated in the middle of the main channel. The system has potential for dose-dependent drug screening in 3-dimensional cell culture, and can be applied by a high-throughput screening method. However, high content imaging and automatic fluid injection technologies are needed to enhance the analysis ability and throughputs.
Acknowledgments
This work was supported by the Ministry of Commerce, Industry and Energy (MOCIE), Korea. The authors also thank CHUNG Moon Soul Center for BioInformation and BioElectronics Center, KAIST. The microfabrication work was performed at the Digital Nanolocomotion Center.
