Abstract
This brief report describes a novel tool for microfluidic patterning of biomolecules and delivery of molecules into cells. The microdevice is based on integration of nanofountain probe (NFP) chips with packaging that creates a closed system and enables operation in liquid. The packaged NFP can be easily coupled to a micro/nano manipulator or atomic force microscope for precise position and force control. We demonstrate here the functionality of the device for continuous direct-write parallel patterning on a surface in air and in liquid. Because of the small volume of the probes (~3 pL), we can achieve flow rates as low as 1 fL/s and have dispensed liquid drops with submicron to 10 µm diameters in a liquid environment. Furthermore, we demonstrate that this microdevice can be used for delivery of molecules into single cells by transient permeabilization of the cell membrane (i.e., electroporation). The significant advantage of NFP-based electroporation compared with bulk electroporation and other transfection techniques is that it allows for precise and targeted delivery while minimizing stress to the cell. We discuss the ongoing development of the tool toward automated operation and its potential as a multifunctional device for microarray applications and time-dependent single-cell studies.
Keywords
Introduction
Micro- and nanofluidics have affected many areas of science and technology in the past three decades. The ability to manipulate and dispense small volumes of liquid is particularly useful for biological applications such as high-throughput bioassays,1–4 cell manipulation and sorting, 5 and precise delivery of molecules into cells.6–8 In particular, high-throughput microarray assays were a breakthrough in biomedical engineering because they provide accurate detection and rapid gene expression monitoring. 9 Because the natural state for most cells and biological materials is aqueous, the ability of a microfluidic tool to operate immersed in a liquid environment is particularly advantageous. 8 Common methods for generating microarrays include mechanical robotic pin printing, 2 microstamping, 10 ink-jet printing, 11 and nanoprobe-based methods.12,13 Dip-pen nanolithography is a common scanning probe-based method for patterning proteins and other materials with submicron resolution, but only a couple examples of in-liquid patterning exist14,15 because the open-tip design limits these applications. Previously, glass micropipettes were used to produce liquid droplet arrays under oil.16,17 However, the glass pipettes are individually prepared by a pulling technique, limiting utility for batch processing and efficient patterning. The most robust method for patterning of biomolecules would combine high-resolution parallel patterning with automated and/or batch operation. Toward this end, we have developed a multifunctional microfluidic device with multiple parallel cantilever probes that can be packaged to form a closed fluidic system for operation in a liquid environment. The device easily couples with an atomic force microscope or micro-/nanomanipulator for precise position and force control that can be automated.
In addition to patterning, the nanoprobe device presented here is also capable of delivering molecules into single cells by electroporation. The nanoscale physical dimensions of the probe tip, volume of the microfluidic channel that is compatible with individual cells, and three-dimensional (3D) positioning with nanometer resolution render this device particularly useful for single-cell applications. Recent technological advances in cell biology techniques have reached single-cell resolution, making possible single-cell genomics and proteomics studies.18,19 Considering that heterogeneity is an intrinsic property in even seemingly similar populations of cells, studying cellular response to a particular gene or drug with single-cell resolution is advantageous to ensure that biological noise is not concealing the true cellular behavior being studied.20,21 Consequently, in addition to single-cell analysis, single-cell molecular delivery is needed to further the accuracy and precision of single-cell biological studies. 22 Thus, we demonstrate here application of our closed system device for precise and gentle delivery of molecules into targeted single cells.
Nanofountain Probe
Nanofountain probe chips are batch fabricated on silicon wafers23–26 and have cantilever probes that can be optimized for stiffness, tip shape, and size by modifications to the fabrication process. As described elsewhere,23–26 the fabrication process starts with silicon on insulator wafers and involves several oxidation, lithography, etching, and deposition steps. The final nanofountain probe (NFP) chip is composed of silicon, silicon oxide, and silicon nitride. The NFP chips described here and shown in Figure 1a have 12 probes with stiffness of ~1.5 N/m and two microreservoirs on each side of the chip such that 6 probes are connected to each independent microreservoir through built-in microchannels. These probe arrays have fully sealed microchannels, which are essential for biological applications using the NFP in liquid. The liquid solution to be patterned on a substrate or delivered to a cell is stored in the on-chip microreservoirs and is fed through the sealed microchannels to apertured dispensing tips ( Fig. 1b and 1c ) by externally applied pressure. Precise control of liquid flow rate through the microchannels can be achieved with variation of the applied external pressure, and the pressure directly affects the resolution of in-liquid surface patterning. Position control of the NFP probes can be achieved with either an atomic force microscope (AFM)6,27 or micromanipulator. 28 Details of the design, microfabrication, theoretical analysis, and applications of the NFP are reported elsewhere.6,27–29
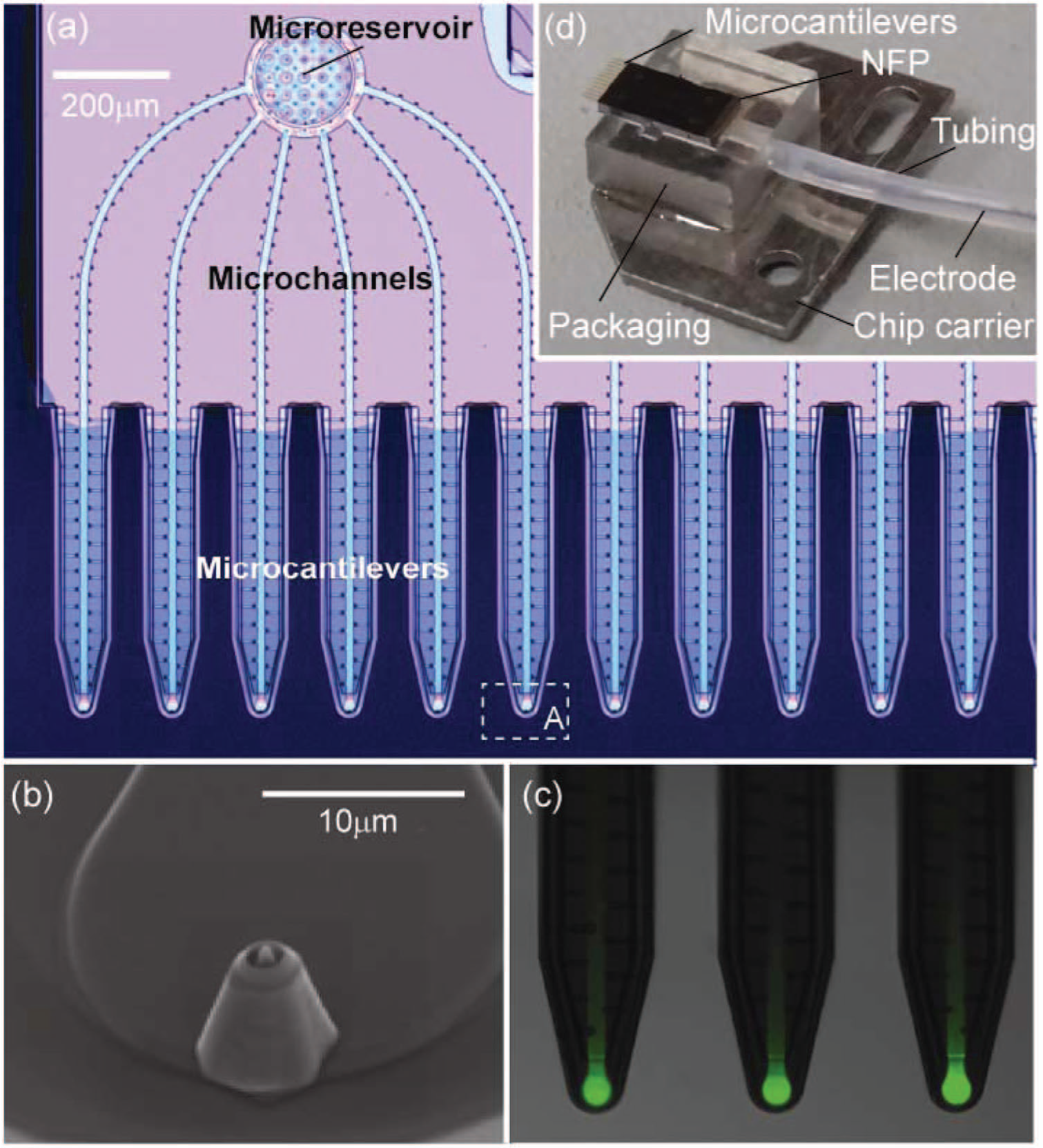
Images of the nanofountain probe (NFP) chip and packaging that enable operation in a liquid environment. (
To optimize the use of the NFP for surface patterning or localized electroporation, two different types of NFP tips have been fabricated: (1) core-shell structure with protrusion and (2) shell-only structure without protrusion, as shown in Figure 2b and 2c , respectively. For surface patterning, type (1) is used because we found that a sharp tip with an optimal protrusion length of ~500 nm is necessary to achieve nanoscale resolution of the pattern features. 27 Conversely, for NFP electroporation (NFP-E), it is important to have reliable electrical sealing between the probe and cell without applying any excessive force on a target cell. Therefore, type (2) is used for single-cell electroporation to optimize electrical sealing and focus the applied electrical field to a small region of cell membrane ( Fig. 2c ). Furthermore, the improved probe-cell sealing that occurs with type (2) tips allows the onset of probe-cell contact to be detected by monitoring the electrical response of the NFP circuit. The electrical current through the gap between the probe and cell significantly decreases at the onset of probe-cell contact, as shown schematically in Figure 2c . 28
(
Packaging of NFPs for In-Liquid Operation
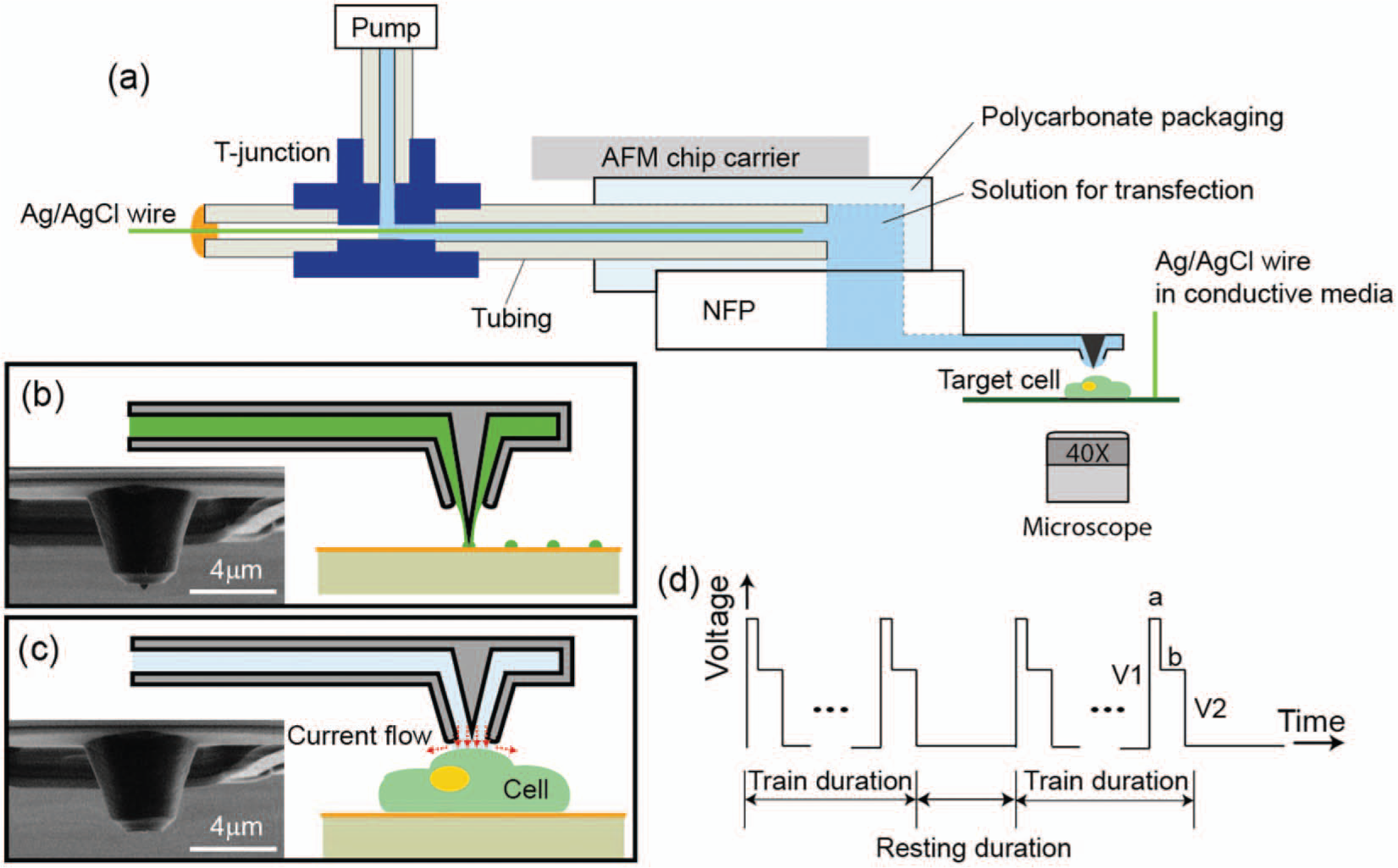
To enable in-liquid operation of the NFP for biological applications, it is necessary to isolate the solution in the NFP chip from the ambient liquid environment. To achieve this unique capability, the NFP chip is integrated with a polycarbonate packaging unit with a built-in fluid channel (see Fig. 1d and Fig. 2a ) and an inset that is the same size and shape as the NFP chip. The packaging unit is connected to tubing for loading the NFP chip using a microfluidic pump to apply external pressure. The solution can be continuously delivered to the NFP tips with precise flow rate control, enabling large-area patterning without the need to refill and reposition the NFP chip. Here, we demonstrate use of the NFP loaded with one solution, but the two independent microreservoirs ( Fig. 1a ) make possible the simultaneous use of two different solutions with a slightly modified polycarbonate packaging unit. For ease of assembly to a manipulator or AFM, the packaging unit is glued onto a metallic AFM chip carrier so it can be mounted on any magnetic holder. For use requiring application of an external electric voltage, the packaging is connected to plastic tubing in which an Ag/AgCl wire electrode and conductive solution are inserted, as shown by the schematic in Figure 2a . Using a T-junction, one end of the tubing is connected to a microfluidic pump while the other end is sealed with the inserted Ag/AgCl wire. This integrated packaging creates a built-in electrical circuit such that the NFP can be readily used for electrophoresis-enhanced surface patterning 27 or NFP-E of single cells. 28 Further details on the packaging protocol are described in the Methods section.
The integrated NFP and packaging offers precise control of the loaded solution over time because of the hermetic packaging unit. This is an important feature for reliability and repeatability of surface pattering over large areas because, without the air-tight seal between the NFP chips and packaging unit, the solution loaded into the NFP would be exposed to ambient air at the opening of the microchannels into the microreservoirs, similar to dip pen nanolithography. 12 Thus, the closed packaging prevents variation of the concentration and/or viscosity of the solution over time from evaporation or condensation of water vapor from ambient air, which could negatively affect resolution and uniformity during surface patterning.
Experimental Setup
Figure 3 shows an example of the experimental setup required for surface patterning or intercellular delivery by NFP-E. This setup includes (1) an inverted fluorescence microscope, (2) a 3D nanomanipulator or, equivalently, an AFM (not shown here), (3) a microfluidic pump, and (4) (for electric-field–assisted patterning and NFP-E) electronics including signal generator, voltage amplifier, multimeter, and oscilloscope. For nanoscale displacement control, the packaged NFP is mounted on the AFM (see the Surface Patterning of Biomolecules section) or nanomanipulator (see Surface Patterning in Liquid and NFP-E for Single-Cell Intercellular Delivery sections). The nanomanipulator is installed on an inverted fluorescence microscope (Eclipse Ti-U; Nikon, Tokyo, Japan) equipped with a charge-coupled device camera (Neo sCMOS; Andor, Belfast, UK) for optical observation of samples. For NFP-E, 3D displacement of the probes is controlled with 40 nm step size resolution. Once the probes are positioned, the electric potential is applied between the two Ag/AgCl electrodes: one inserted in the packaging and the other submerged in the conductive cell culture media on the sample plate ( Fig. 2a ). The external signal is generated by a pulse generator (DS345; Standard Research Systems, Sunnyvale, CA), amplified by a voltage amplifier (OPA445; Texas Instruments, Dallas, TX) and monitored using an oscilloscope (9384L; LeCroy, Chestnut Ridge, NY). The detailed protocol for NFP-E is described in the section NFP-E for Single-Cell Intercellular Delivery.

Example of the experimental setup required for surface patterning or intercellular delivery by nanofountain probe electroporation (NFP-E). For position control, a three-dimensional nanomanipulator with a custom-made NFP mounting arm is positioned over an inverted fluorescent microscope. The inset (lower left) shows a magnified view of the packaged NFP mounted on the manipulator arm and connected to the microtubing of the external fluidic pump. The electric pulses are applied using a function generator.
Biological Applications of NFP
Surface Patterning of Biomolecules
Patterning of biological materials on a substrate is frequently used in biological studies for preparing protein and antibody microarrays used for multiplexed analysis of biological samples.30,31 AFM-based surface-patterning technologies including NFP and dip-pen nanolithography are suitable for creating such biomolecular micro- and nanoarrays with orders of magnitude higher pattering resolution than any other alternative method (e.g., robotic spotting or photolithographic-based techniques).27,32,33 In particular, NFP offers an attractive advantage for continuous surface pattering by directly delivering the biomolecular solution from a microreservoir to the probe through the built-in microchannels. 31
Figure 4 displays parallel surface patterning of ferritin protein arrays on silicon dioxide using the NFP mounted on an AFM. To achieve sub–100 nm resolution pattering, NFPs with the core-shell structure with protrusion (type 1) were used. High-throughput 10 × 20 arrays of ferritin protein dots with nanoscale resolution were achieved by implementing simultaneous deposition with the multiple probes on the NFP chip in parallel. We observed some variation in the size of patterns and found that such variation strongly depends on two key parameters: (1) variations in tip shape and (2) differences in applied force between tip and substrate. A detailed protocol for AFM-based surface patterning is discussed in the Methods section.

Surface patterning using an atomic force microscopy–controlled nanofountain probe (NFP) to deposit ferritin protein on a silicon dioxide substrate by parallel deposition from the multiple NFP probes on the chip. (
Surface Patterning in Liquid
Figure 5 shows a schematic and demonstration of in-liquid patterning using the packaged NFP system (see the Methods section for the surface preparation protocol). For visualization of the liquid dispensing, we used mineral oil as the medium and deposited droplets of fluorophore-labeled streptavidin in a water solution onto a biotin-coated glass surface. The bond formed between the streptavidin and biotin secured the fluorophore to the surface even after removal of the oil media, as shown in Figure 5b and 5c . It is worth mentioning that when we dispensed the water droplet on a bare glass surface under the mineral oil environment using a manual micromanipulator, we observed a decrease in the drop size over time. We also performed similar experiments under phosphate-buffered saline (PBS). Although we achieved protein patterning under PBS, we found that the biomolecular solution mixes with the PBS and makes it challenging to maintain a sufficiently high localized concentration of the biomolecules at the probe tip for precise patterning.
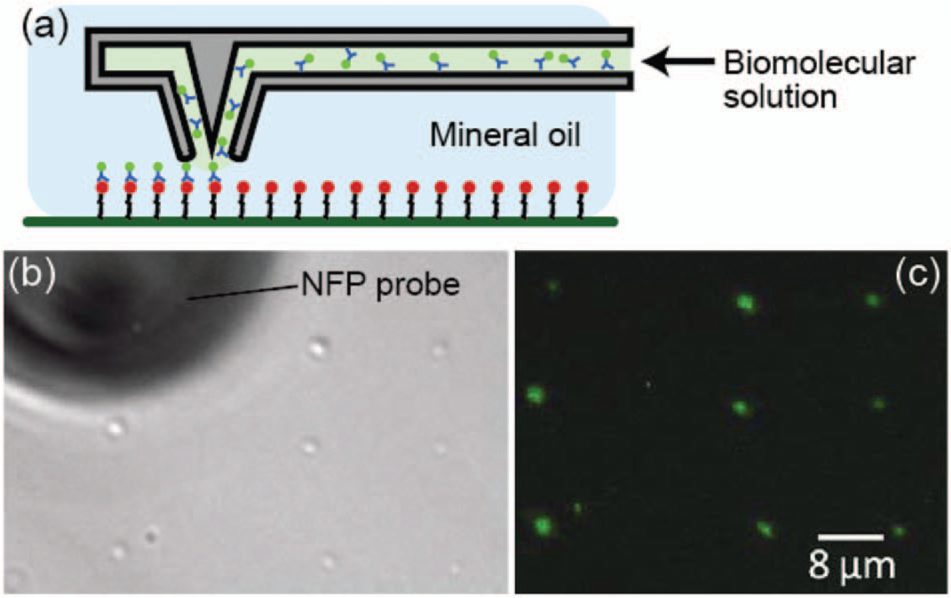
(
NFP-E for Single-Cell Intercellular Delivery
Electroporation of a cell is a multistep process that includes membrane charging, formation of nanopores, delivery of molecules, and recovery (closure) of nanopores. 34 Membrane charging occurs until the transmembrane potential reaches a certain critical threshold, 0.2 to 1 V, 35 which triggers the onset of nanopore formation on the cell membrane. The characteristic time for pore size evolution is on the order of milliseconds and is driven by energy minimization of the whole cell membrane. 34 Delivery of biomolecules into a target cell occurs through the temporary nanopores by diffusion or electrophoretic forces. 36 After the electric pulse is turned off, the cell membrane discharges through the existing pores and the transmembrane potential returns to zero. Consequently, the pores shrink to the minimum energy state and finally reseal the membrane in the order of seconds.34,35 The electroporation process is dependent on amplitude and frequency of the input voltage (e.g., theoretical models predict that the number of nanopores formed increases with the strength of the applied pulse).34,35 Although the experimental results presented next were generated using a single cantilever probe, each cantilever is electrically independent so that parallel single-cell electroporation is possible with this NFP design. Indeed, we have successfully delivered a fluorophore into two adjacent cells simultaneously.
The packaged NFP can be used to deliver molecules in liquid solution to cells with single-cell selectivity and qualitative dosage control according to the following procedure. Using an inverted microscope and nanomanipulator, a previously filled NFP probe is optically aligned over a target cell adhered to a glass-bottom petri dish placed on the microscope stage ( Fig. 6a ). The probe is then brought down to make gentle contact between the probe and cell. Gentle contact is critical to minimize mechanical stress placed on a cell and can be achieved via optical observation or electrical measurement of the NFP-E circuit ( Fig. 6b ). The detailed strategy to detect probe-cell contact based on a resistance measurement was previously reported. 28 Once the probe is positioned in contact with the cell, the electric field is applied to the target cell to trigger formation of nanopores in the cell membrane. Previous parametric studies and theoretical analysis revealed that the major potential drop occurs through the NFP microchannels, and therefore, an input voltage of 15 to 30 V provides the critical transmembrane potential drop (0.2~1 V) at the NFP tip. 28 The solution in the NFP microchannel will diffuse into the nanopores and enter the cell, the amount of which can be qualitatively controlled by the duration of the electric pulses. The small opening of the NFP tip creates a highly focused electrical field that is localized to a small region of the cell membrane, and therefore, the majority of the cell membrane remains intact during NFP-E. This gentle contact and localized electrical field result in ultrahigh cell viability (>92%) of electroporated cells using NFP-E. 28
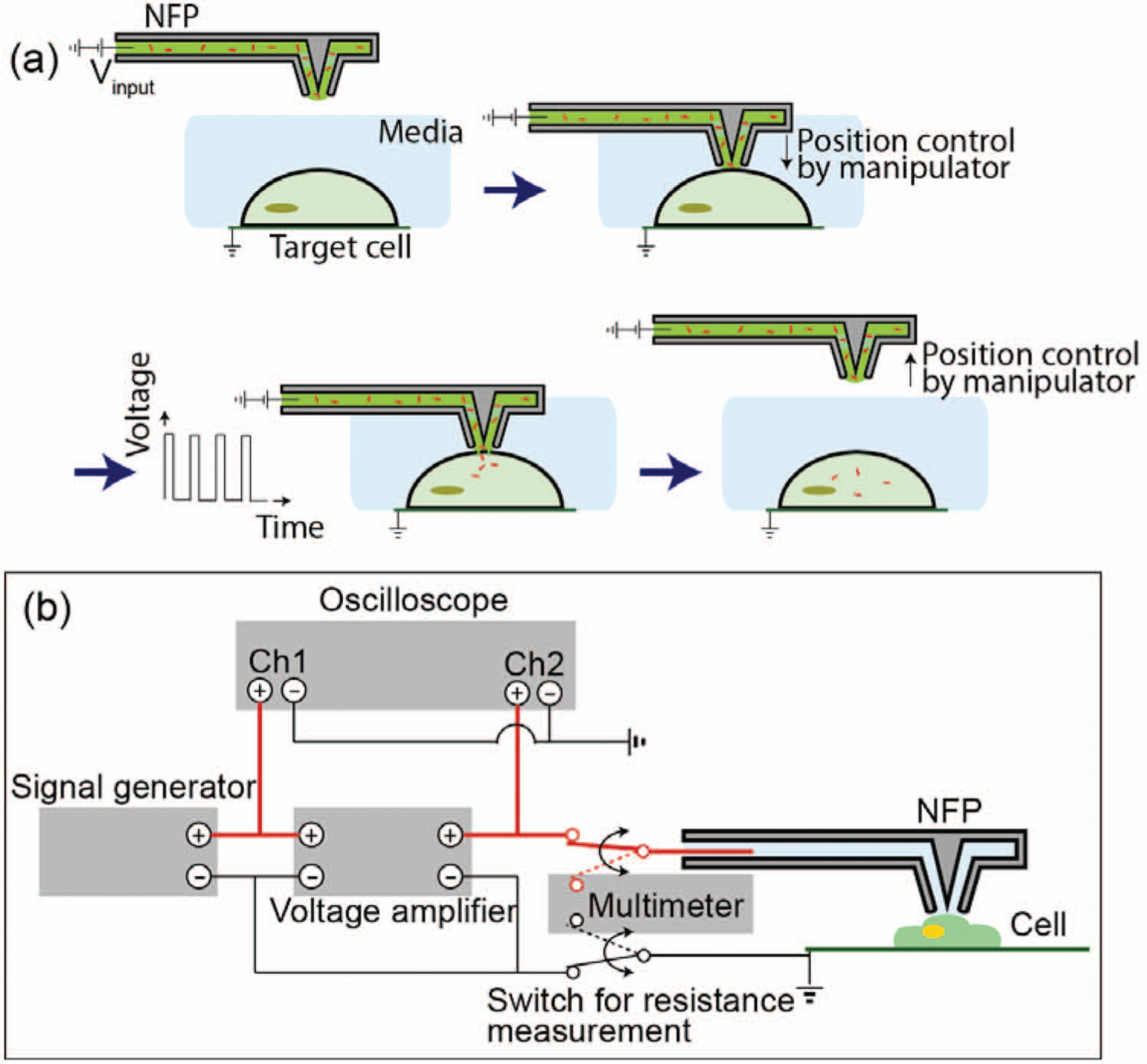
(
Figure 7 shows delivery of a 1 mg/mL solution of 3000 MW fluorophore-labeled dextran (Alexa Fluor 488) in water into several target HeLa cells using NFP-E. (The detailed procedure for cell culture is described in the Methods section.) These experiments were performed with applied voltages of 15 to 30 V and bilevel square wave signals of 200 Hz ( Fig. 2d ). Each pulse train duration was 1 s and was separated by a 1 s interval. The ratio of V2/V1 was 0.375 with a = 1 ms and b = 3 ms. No transfection of the dextran molecules was observed at lower voltages (≤7 V), whereas damage to the target cells was observed at higher voltages (40–60 V). 28 Note that the fluorophore was delivered only to the targeted cells (cell A–D), and the neighboring cells were not affected. Moreover, no extraneous fluorescence is observed in the region of the electroporated cells, indicating that the amount of solution released from the microchannel is localized to the electroporated region of the cell in contact with the probe tip. These results confirm that the electric field is focused to the tip of the probe and that the probe provides localized exposure to the solution in the NFP, both features that are essential for single-cell selectivity of NFP-E. The electroporated cells in Figure 7 exhibited consistent fluorescence intensity 4 h after NFP-E, and viability tests using propidium iodide confirmed that the cells were viable. 28
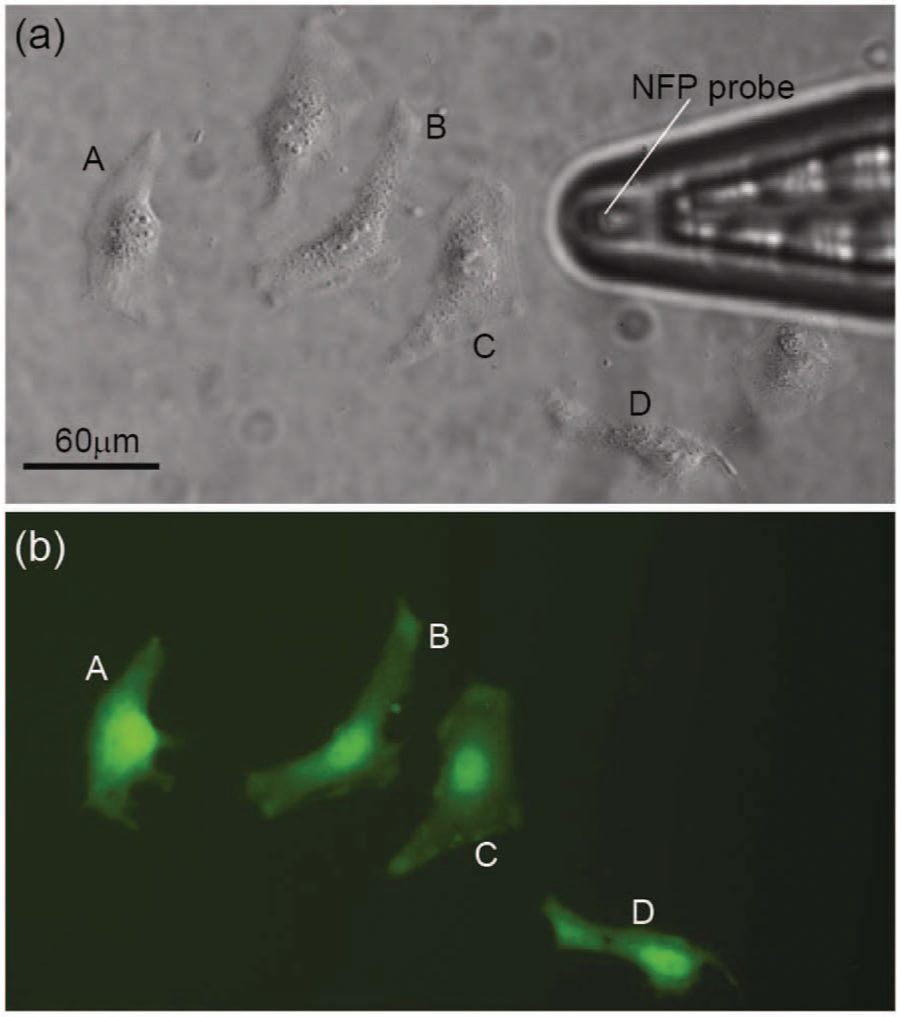
Demonstration of single-cell nanofountain probe electroporation of HeLa cells transfected with 3 kD dextran–Alexa Fluor 488. (
Discussion
The NFP is a useful tool for biological applications because the coupled packaging design described here creates a closed fluidic system that is compatible with either a manipulator or an AFM, allowing a user the flexibility to use the advantages of their platform depending on the desired application. Examples of the utility of the packaged NFP include the following: (1) parallel patterning of biomolecules such as proteins, (2) in-liquid surface patterning, and (3) delivery of biomolecules in solution into targeted single cells via localized electroporation.
The continuous flow, direct write, parallel probes, and closed system are ideal for patterning of biomolecules in liquid solution. Virtually any type of pattern and geometry can be created simply by programming the pattern of the scanner used for position control. Feature size can be controlled by dwell time on the surface or, for in-liquid surface pattering, by external pressure of the solution through the NFP. In-liquid patterning is particularly useful for preventing protein denaturing due to drying1,37 and is an alternative to other probe-based patterning methods such as dip-pen nanolithography, 12 which is predominantly performed in ambient condition.
For single-cell electroporation, the NFP-E system has unprecedented capabilities for targeted transfection such as single-cell selectivity, high transfection efficiency, qualitative dosage control, and ultrahigh cell viability. 28 Recent research applications of the NFP-E system have proven that it is capable of transfecting cells with large biomolecules including 70 kDa bovine serum albumin (BSA), 20 kDa GAPDH-target DNA hairpin, and 2 MDa plasmid DNA. 28 These results reveal that the NFP-E technique is an efficient method for transfecting cells with molecules of various sizes and charges while maintaining high cell viability. In addition, we observed that BSA could be delivered to the cytoplasm or directly into the nucleus by positioning the probe away from or on top of the nucleus, respectively. This localized transfection could be used to study trafficking or other spatially dependent cell responses after introduction of a molecule into cells. Currently, we are working to increase the throughput of NFP-E by implementing protein patterning for cell adhesion and/or microwells for cell alignment coupled with automated cell-probe contact detection based on a resistance measurement. In addition, the stiffness of the NFP cantilevers will be reduced to minimize damage to cells during array operation and enable parallel single-cell electroporation. Parallel operation and automation will greatly increase the throughput and robustness of the NFP-E system and make it an extremely useful new biotool for applications such as single-cell research studies, drug-screening assays, cell line development, and stem cell reprogramming/differentiation.
Methods
Protocol for NFP Packaging
For packaging of an NFP chip ( Fig. 1d ), a fabricated polycarbonate packaging unit designed specifically for use with an NFP chip (5.0 × 4.4 × 1.6 mm) is cleaned with isopropyl alcohol and deionized water to remove any residue and particles on its surface. The cleaned packaging unit is glued onto a metallic AFM chip carrier using epoxy glue ( Fig. 2a ). For simple and repeatable assembly, the polycarbonate unit has a groove that matches the dimensions of an NFP chip. The NFP chip is mounted into the groove under an optical microscope and bonded to the polycarbonate unit on the back side of the NFP chip with glue (Cemedine Super-X 8008). The groove is also designed to align the on-chip microreservoir with the fluid channel through the polycarbonate unit. There is no need for precise alignment of the NFP chip and polycarbonate unit because the fluid channel is 800 µm in diameter, which is five times larger than the diameter of the microreservoir on the NFP chip (160 µm), so a slight misalignment is compensated. Plastic tubing (ID = 350 µm and OD = 800 µm) with an inserted Ag/AgCl wire (diameter = 200 µm) is then pushed into a built-in channel on the packaging unit that connects to the microreservoirs of the NFP chip and is fixed by epoxy. As shown in Figure 2a , the plastic tubing is connected to a fluid pump and electrical input via a T-junction to apply external flow pressure and electrical input signal, respectively.
After packaging, the biomolecule solution is loaded into the tubing using a 100 µL syringe (Hamilton), and the packaged NFP is mounted on the desired platform (i.e., a nanomanipulator; Fig. 3 ). External pressure from a pump is applied through the tubing to feed the solution through the tubing, into the microreserviors, and the microchannels. For high-resolution surface pattering in liquid, a pump that allows pressure as low as 1 hPa (0.1 kPa) will facilitate flow control of several fL/s. Alternatively, for NFP-E, the tubing can be connected to a simple syringe pump because flow control is needed only to accelerate the microchannel wetting process.
To prevent waste and background fluorescence in our sample media (if applicable), we cease applying pressure immediately after completion of microchannel wetting by monitoring the wetting process via optical observation and electrical measurement. For example, see Figure 1c , which shows a bright-field and fluorescence merged image of NFP microchannels being wetted with a fluorophore-labeled dextran solution (Alexa Flour 488; Molecular Probes, Carlsbad, CA). In addition to the optical observation, completion of the microchannel wetting in the NFP-E experimental setup forms a closed electrical circuit between the two Ag/AgCl wires ( Fig. 2a ); therefore, a sudden change in the electrical response from open circuit to 10 to 20 MΩ can be measured in conductive media.
AFM-Based Surface Patterning
A 0.14 mg/mL solution of ferritin protein in 80:20 water/glycerol (v/v) was loaded in the microreservoir, and wetting of the microchannels from the reservoir to the tips was confirmed by observing the displacement of the meniscus in the microchannels under an optical microscope. The filled NFP was then mounted in an AFM (Dimension 3100; DI Instruments), and the probes were engaged on the substrate after aligning the photodetector laser on a single cantilever. Arrays of ferritin dots were patterned on a silicon substrate with a 200 nm thermal oxide layer under ambient conditions (25 °C and 30–35% relative humidity). The patterned substrates were characterized by AFM tapping mode imaging with silicon AFM tips (TAP300Al-G 300 kHz; Budgetsensors, Sofia, Bulgarai).
In-Liquid Pattering
To functionalize the glass substrate for in-liquid patterning, glass slides were cleaned in piranha solution, rinsed with nanopure deionized water, and dried under a stream of argon. The surface of the glass slides was activated by silanization in a tank containing a solution of 3-aminopropyltriethoxysilane (APTS) in ethanol 190 (EtOH:1%HCl:APTS of 98.8:1:0.2 v/v) for 90 min at room temperature. Biotin functionalization was completed by forming an amide bond between the NH2 group on the modified coverslip and the NHS ester of a PEG5000 derivative. A solution of 1.25 mg/mL NHS-PEG5000-Biotin dissolved in 100 mM carbonate buffer at pH 8.25 was prepared, and 100 µL of the active ester solution was deposited on each modified glass slide and covered immediately with another slide and incubated for 3 h at room temperature. The derivatized glass slides were washed with carbonate buffer solution and nanopure deionized water and then dried under a stream of air and used immediately. For patterning, the NFPs were filled with a solution of 20 µM FITC-streptavidin (Invitrogen, Molecular Probes) in HEPES buffer.
Cell Culture and Sample Preparation
HeLa cells (American Type Culture Collection; ATCC #CCL-2) were cultured in DMEM (Sigma, St., Louis, MO) with L-glutamine and phenol red as pH indicator, supplemented with 10% fetal bovine serum (Sigma) and 1X penicillin/streptomycin (Sigma). The cultured cells were maintained in a humidified incubator at 37 °C and 5% CO2. For electroporation experiments, the cells were plated the day before the experiment on a glass-bottom petri dish and incubated in DMEM media. On the day of the experiment, the petri dish with plated cells was rinsed multiple times with DMEM, without phenol red to avoid autofluorescence during imaging, and then filled with DMEM to keep the cells submerged in media throughout the electroporation experiments. The petri dish was then placed on an inverted optical microscope stage and imaged before and after electroporation.
Footnotes
Acknowledgements
We acknowledge Professor Punit Kohli of the Department of Chemistry at Southern Illinois University for providing biotin-coated glass surfaces and discussions. Also, we thank Professor Dean Ho of Biomedical Engineering of Northwestern University for providing the HeLa cells and use of the cell culture facility.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: iNfinitesimal LLC was founded by Horacio D. Espinosa.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science Foundation under award IIP-1142562 and the National Institutes of Health under award 1R41GM101833-01.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
