Abstract
Conventional methods to prepare sperm have been amenable to the investigation of outcomes such as rates of recovery and conventional semen parameters. The standard preparation of sperm for assisted reproduction is criticized for its centrifugation steps, which might either recover motile sperm in variable proportions or increase the probability of damage to sperm DNA. An microfluidic system was designed to separate motile sperm according to a design whereby nonmotile spermatozoa and debris flow along their initial streamlines and exit through one outlet-up, whereas motile spermatozoa have an opportunity to swim into a parallel stream and to exit through a separate outlet-down. This chip was fabricated by microelectromechanical systems technology with polydimethylsiloxane molding. The hydrophilic surface, coated with poly (ethanediol) methyl ether methacrylate, exhibits enduring stability maintained for the microchannel. Microscopic examination and fluorescent images showed that the motility of sperm varied with the laminar streams. To confirm the sorting, we identified and quantified the proportions of live and dead sperm before and after sorting with flow cytometric analysis. The results on the viability of a sample demonstrated the increased quality of sperm after sorting and collection in the outlet reservoir. The counted ratio of live sperm revealed the quantity and efficiency of the sorted sperm.
Introduction
Since the first in vitro fertilization (IVF) birth in 1978 in England, 1 with the increasing clinical utilization of assisted reproductive technology (ART), scientists and clinicians have acquired insight into basic gamete and embryo biology and translated that knowledge into improved IVF. Conventional methods of preparing sperm have led to investigations of outcomes such as the rate of recovery and conventional parameters of the semen.2,3 Motility is regarded as an important parameter related to sperm quality. Procedures that safely and efficiently select motile sperm are desirable; the swim-up method and density-gradient centrifugation serve this purpose. 4
The standard preparation of sperm in assisted reproduction has been criticized for its centrifugation step, which either increases the probability of damage to sperm DNA by reactive oxygen species5,6 or recovers motile sperm to a variable extent through swim-up preparation.4,7,8 Attention to sperm processing and isolation to increase the recovery of motile sperm and to diminish sperm damage has improved the rates of fertilization and the development of embryos.9–11
Intracytoplasmic sperm injection (ICSI) offers the opportunity to allow fertilization even in cases of severely compromised quality or number of sperm. 12 Sperm screening is an important step for successful fertilization. Semen are still processed with the swim-up method even for those patients with a severe male factor, resulting in a limited number of viable spermatozoa available after processing for use in ICSI. 13 Speculation about new techniques to isolate sperm is hence required for future application in ART.
Microfluidics is a promising new technology. Growing attention is thus being paid to microfluidics in the field of reproductive medicine. Microfluidics, based on the physical principles of fluid behavior in a microenvironment, serve widely for applications in chemistry and molecular biology.14–20 Many advances in nano- and microtechnology are applied to study biological systems. Microfluidic devices are being developed for biological assays with the potential to perform analysis with efficient throughput, use samples or reagents in small amounts, and analyze single cells.21–25 Several microfluidic devices have been explored for diagnosis of sperm.26–28 These devices have demonstrated feasibility and might replace conventional methods of motility assessment and semen analysis. These studies have demonstrated that sperm movement within a microchannel to reach the end of the channel correlates with forward progression. 26 A microfluidic device based on a sperm-sorting method embodies the use of a parallel laminar flow stream to distinguish motile sperms from those of poor quality. 28
Developments in biomedical engineering and the fabrication of micromachined devices have opened avenues of investigative research. Microfluidic technology is used in numerous biological applications specifically for miniaturization and simplification of laboratory techniques, because the control of fluid transport is useful for systems, including cell analysis, drug delivery, and ART. 29 The field of andrology has benefited from application of the evaluation of human sperm. The unique property of sperm, with a nature of progressive motility, makes them a suitable candidate for study and manipulation in this environment. The main reasons for human male infertility are morphology abnormality and insufficient quantity of sperm. These problems might be solved with ART. The enhancement of fertilization is accomplished with either IVF or ICSI. Traditional methods of sorting sperm, such as swim-up and Percoll gradient separation, typically require manual selection and are tedious. The probability of DNA damage is increased with these methods. 2 Using microfluidics to isolate motile sperm eliminates centrifugation and the related detrimental effects of reactive oxygen species from the centrifugal pelleting of a sperm sample.
Based on the ability of motile sperm to move from their initial streamlines in the laminar fluid stream, a highly motile subpopulation of spermatozoa has been demonstrated to be isolated from semen using microfluidics. Approximately 40% of motile spermatozoa are demonstrated to be recovered using a microfluidic chamber device.28,30 Some authors have proposed a horizontally set-up and gravity-driven pumping system, which greatly simplified the sorting step without centrifugation. In particular, the channels and reservoirs were coated with bovine serum albumin (BSA; 1%) to improve the surface hydrophilicity.28,31 The advantages of the Cho et al 28 study are that gravity served as the driving force from the reservoirs, and operation was simple. The system could sort sperm successfully. However, fabrication of reservoirs was more difficult. The advantages of the Horsman et al study 30 are that the system could be applied to cases of sexual crime and that the motile sperm could swim themselves. The system could separate sperm with epithelial cells successfully. However, the process is time-consuming because the motile sperm may swim themselves. Our work demonstrates an integrated microfluidic system, which consists of a laminar stream–based microchannel and flow cytometric analysis, to enhance the efficiency of sorting sperm motility. This modified sorting provides a selection that advances in resolving traditional sorting problems. We have demonstrated an enhanced rate of motile spermatozoa recovered smoothly with fluency in a microfluidic device with a surface-modified microchannel. SYBR 14 staining demonstrated that more motile sperm were also recovered at the reservoir of the microfluidic chip. Staining with propidium iodide (PI) demonstrated that more dead sperm were recovered from the upper outlet of the microfluidic chip.
As our previous work demonstrated, 32 a microfluidic system effectively separates raw samples of semen into immobile and mobile groups without centrifugation; damage to the sperm is thereby avoidable, but no method has been devised to confirm the test of sperm quantity to evaluate readily the motility information. By fluorescent staining 33 and statistical methods, such as flow cytometric analysis, 34 the amount of sperm, including live and dead sperm, can be characterized; a statistical model to analyze the sorting efficiency of motile sperm was developed.
In this work, we developed an integrated microfluidic system that consists of a laminar stream–based, surface-modified microchannel that demonstrates the separation of motile sperm. In our experiments, gravity served as the driving force from the reservoirs. The control of flow rate involves having liquid surface heights of two inlet reservoirs higher than those of two outlet reservoirs; the difference in height makes liquid flow, and controlling the difference in height can make a difference in the flow rate. We used flow cytometric analysis to assess the sorting efficiency of sperm motility.
Materials and Methods
Sample Collection of Mouse Sperm
The experiments, approved by the Animal Care and Use Committee of Chang Gung Memorial Hospital (CGMH), were conducted in accordance with the principles and procedures outlined in the CGMH Guidelines for the Care and Use of Laboratory Animals. Preparing the sperm sample from ICR mice had four steps. First, cervical dislocation was used on male ICR mice. Second, the abdomen of male ICR mice was cut and the epididymides removed. Third, the epididymides were put in a culture dish full of culture media. Fourth, the culture dish was placed in an incubator for 10 min at 37 °C, and the sperm were allowed to disperse and then be collected. The special characteristic of ICR mice sperm is with the hook on its head. Thus, it usually connects motile sperm with dead sperm or impurity. The result of flow cytometric analysis may show some percentage of dead sperm in the outlet-down. Although we have successfully used ICR mice sperm to test our chip work, in the future, our study will be applied to human sperm, which should have better results.
Our experiments, which include staining, separating, collecting, and analyzing, are usually finished in 1 h. After the sperm sample is prepared from ICR mice, we make sure the environmental temperature is suitable and do the experiments quickly. We hope to decrease the percentage of sperm fading.
Microfluidic System
A schematic illustration of sperm sorting with a microfluidic system is shown in Figure 1 . In this work, the experiment developed as a process complete in three steps. Briefly, semen samples were first stained with a fluorescent dye that revealed live sperm to differ from dead sperm. After injecting the stained sperm into the microfluidic chip from the inlet reservoir of the surface-modified channel, we recorded the results with a microscope. This microscopic examination revealed sperm with varied motility. The proportions of live and dead sperm after sorting were quantified with flow cytometric analysis.
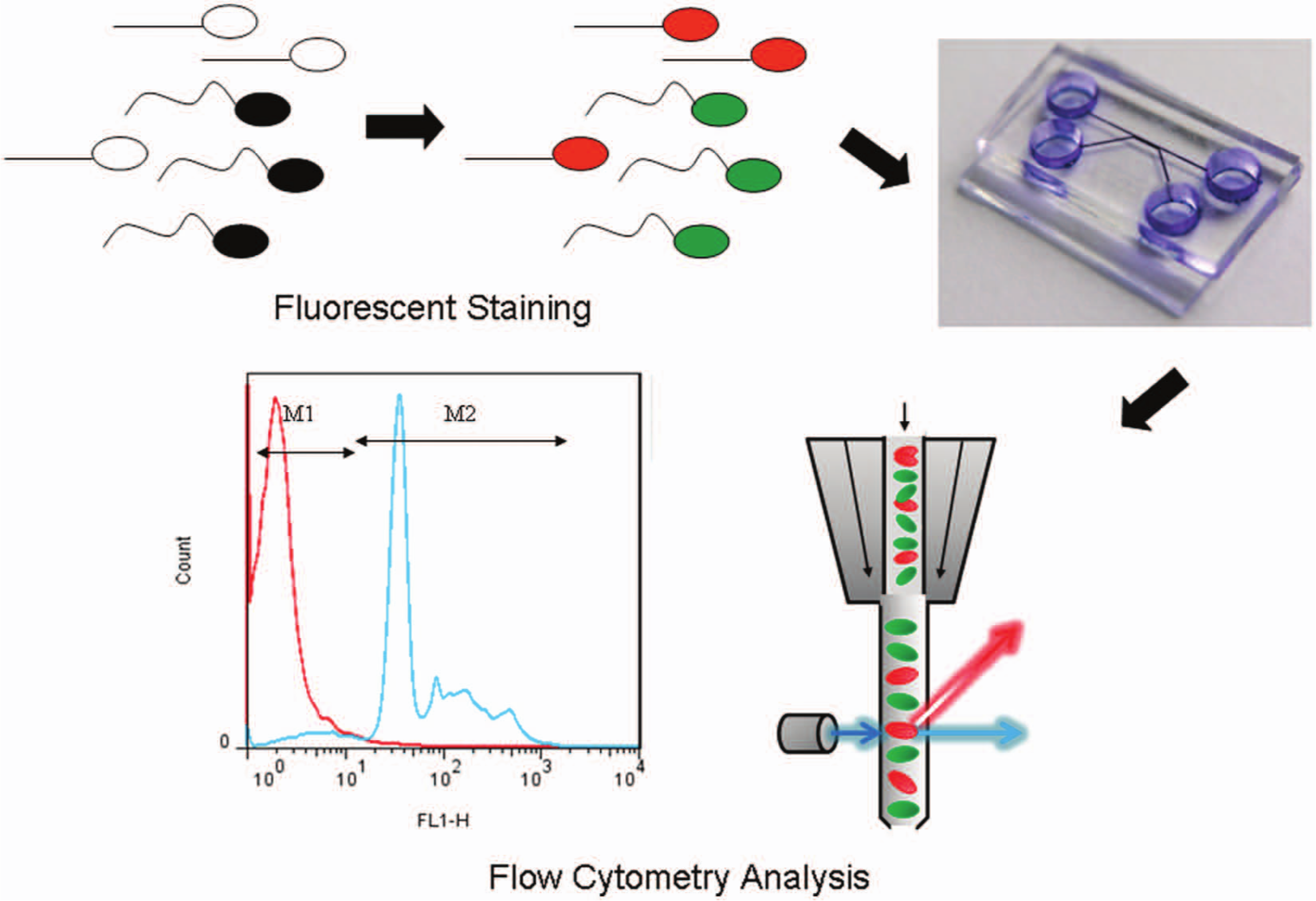
Schematic illustration of the sorting process for the microfluidic system.
A Microfluidic Chip with Multiple Microchannels
The purpose of this research was to sort motile sperm based on the effect of laminar flow in a microchannel and the motion activity of sperm samples. The idea of two parallel laminar stream–based, surface-modified microchannel microfluidic chips is presented in Figure 2 ; the dimensions of our chip have been designed carefully. The widths of the inlet-up and inlet-down are 100 µm and 300 µm, respectively. The main microchannel has a length of 5 mm and a width of 400 µm. The dimensions of the two outlets are the same as the inlets, and the whole height of the microchannel is 50 µm. We inject semen into microchannel from the inlet-up. Because the width of the inlet-down is larger than that of the inlet-up, the larger space can precollect motile sperm that vary with the laminar streams. To encourage the movement of the sperm in one direction, the height of the microchannel is less than the length of a sperm. This can reduce the opportunity of motile sperm to move in a z-direction and even can increase the sorting efficiency. Thus, we claim that the specific microchannel dimensions provide high efficiency of sorting sperm motility in the laminar flow.
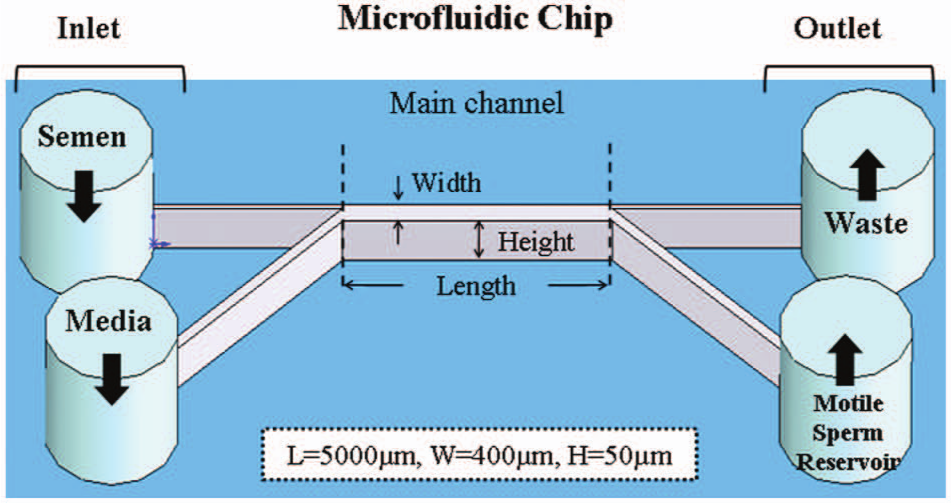
Schematics of two parallel laminar stream–based, surface-modified microchannels in a microfluidic chip. The motile spermatozoa had an opportunity to swim into a parallel stream and exit a separate outlet reservoir.
To increase the sorting efficiency of the microfluidic chip, we performed computational fluid dynamics (CFD) simulation to analyze the 3D flow field to obtain an optimal design. The parameters of CFD simulation include geometry, fluid properties, and flow rate. The geometry is the same as our chip. The widths of the inlet-up and inlet-down are 100 µm and 300 µm, respectively. The main microchannel has a length of 5 mm and a width of 400 µm. The dimensions of the two outlets are the same as the inlets, and the whole height of the microchannel is 50 µm. The viscosities of semen and media are set up as 0.0105 Pa*s and 0.0001 Pa*s, respectively. The flow velocities of the two inlets are the same at 300 μm/s. The result is distribution of concentration by simulation. Figure 3 shows that semen was kept at the upper part of the main microchannel, forming an optimal laminar flow with the media. This means that motile sperm swam across the two fluid boundaries to be sorted, and dead sperm were taken out from the outlet-up.

Computational fluid dynamics simulation was applied for proof of concept and for the design of the microchannels.
Chip Fabrication
Microfluidic devices fabricated from polydimethylsiloxane (PDMS) offer the ability to improve biological and medical capabilities. It is imperative that PDMS be used in the construction of these devices and that the materials in contact with the gametes during coincubation are not deleterious to the survival of human sperm, fertilization, and early development of the embryo.35–37 A PDMS-based sperm-sorting device can have the surface modified with a coating of poly (ethanediol) methyl ether methacrylate (PEG-MA) to create a moderately hydrophilic and nonfouling surface. 38 With fluorescent staining 33 and a statistical method based on flow cytometric analysis, 34 the amount of sperm, both live and dead, can be characterized; we developed a statistical model to analyze the sorting efficiency of motile sperm.
Standard soft lithography was modified to fabricate microfluidic channels using PDMS ( Fig. 4 ). Briefly, our microfluidic chip was made of SU-8 photoresist on a silicon wafer 50 µm in depth as a master mold to form microchannels. Subsequently, Slygard 184 and a curing agent were mixed (10:1), poured on top of the master mold and degassed in a vacuum, and cured in an oven at 90 °C for 1 h to obtain the PDMS microchannels. The resulting channels were cut from the reservoirs to connect the channels, and the device was treated with oxygen plasma for 30 s, then bonded with clear glass to form the separation microchannel. PEG-MA is hydrophilic and biocompatible. PDMS microchannels coated with PEG-MA were fabricated for the microfluidic system and to demonstrate the hydrophilicity of this biochip. Our chip is disposable. We also do sterilization before experiments. The parameter setting for sterilization includes putting the chip in 75% alcohol for 10 min.

Fabrication of the sperm sorter. PDMS, polydimethylsiloxane.
Surface Modification
A hydrophilic surface on PDMS is desirable for microfluidic channels. A novel method to create a stable and hydrophilic surface was developed in which PEG-MA bonded with PDMS ( Fig. 5 ). The surface of PDMS coated with PEG-MA was characterized by measurements of the contact angle to determine the hydrophilicity and the temporal stability. Untreated PDMS is hydrophobic; treatment with an O2 plasma yields only a temporary hydrophilicity: after 3 days, the contact angle of this plasma treatment is 100.13°. A hydrophilic surface of PDMS coated with PEG-MA has a contact angle of 35.97° and maintains its stability for at least 3 days. This modified surface allows the development of a microfluidic system that is portable and operates reliably.
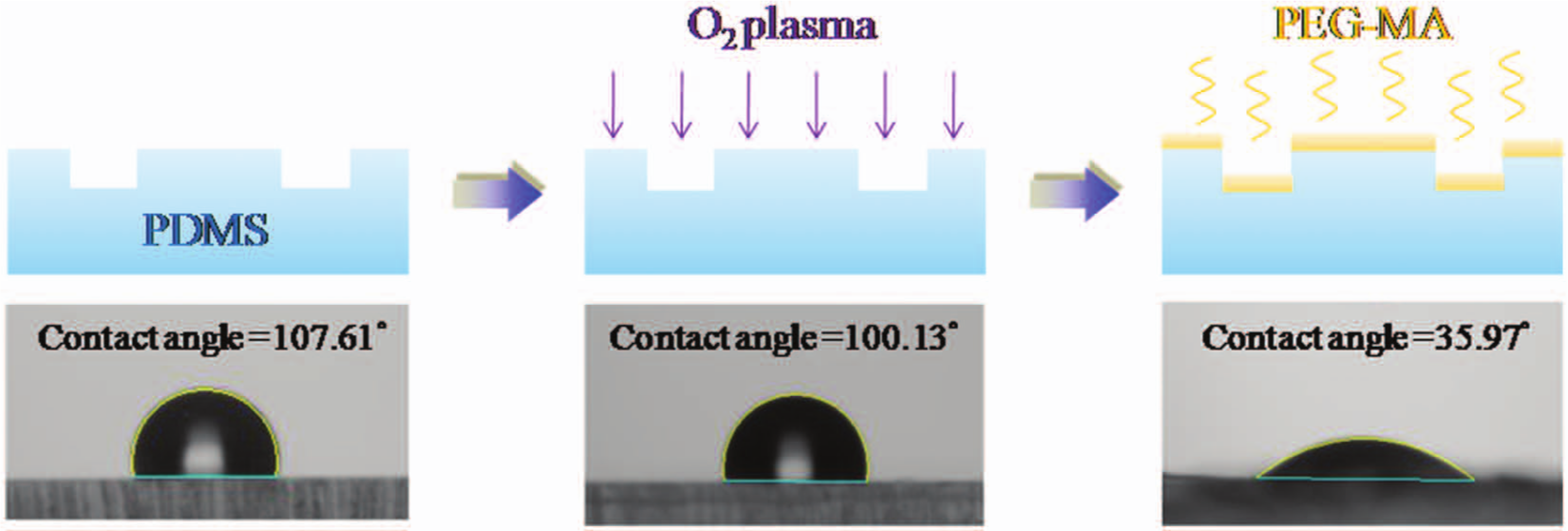
Schematic diagram of a modified process to create a hydrophilic surface on polydimethylsiloxane (PDMS) microchannels. PEG-MA, poly (ethanediol) methyl ether methacrylate.
With regard to the flow rate, we found that a flow velocity at 300 µm/s works best in many experiments. The lower flow velocity can provide more opportunity to motile sperm that are sorted. Before the experiment, the microchannels and reservoirs were coated with BSA (1%) to enhance their hydrophilicity and to decrease the nonspecific absorption of cells. The final device was tested with blue dye to prove the feasibility of the fabrication.
Staining of Motile Sperm and Flow Cytometric Analysis
In the experiment, we used fluorescent dye SYBR-14 to stain the nuclei of live sperm with a complete cell membrane. PI is a dye that stains the nucleic acid, to detect the DNA of damaged and dead sperm. These probes specific to nucleic acid stain live sperm green and dead sperm, or sperm with a damaged membrane, red. We fixed the concentration of our sperm sample in 107 sperm/mL. In this concentration, we added 5 µL PI (red fluorescence) and 0.5 µL SYBR-14 (green fluorescence) to the sperm sample and then put it in an incubator for 20 min at 37 °C. After that, we finished the staining process. The fluorescence was measured simultaneously to identify live sperm from dead sperm by flow cytometry at a wavelength of 488 nm.39,40 To confirm the real sorting situation, we identified and quantified the proportions of live and dead sperm in a sorted sample with flow cytometric analysis (FACS Caliber with CELL Quest software; BD Biosciences, Mountain View, CA).
Results and Discussion
Separation of Motile Sperm with a Microfluidic Chip and Fluorescent Tests
The swimming direction of mouse sperm indicates that sperm of all kinds, either random or with combined motility, can be selected into a desired reservoir. The images recorded from the inlet intersection of two streams revealed that motile mouse sperm passed through the straight flow boundary into the curved laminar flow with the medium. Because of the varied directions in laminar flow, the sperm injected from a surface-modified inlet are subject to a y-component force for motile and dead sperm near the flow boundary. When the semen sample (1 × 107 sperm/mL) was loaded into a straight laminar flow, the results showed that, although most motile sperm with lateral activity could swim across the two fluid boundaries to be sorted, the dead sperm easily crossed into the outlet-up reservoir.
The live sperm in semen samples exhibited bright green fluorescence for nuclei stained with SYBR-14 and excited at 518 nm. With the addition of PI, the dead sperm DNA stained bright red. The sperm stained with either SYBR-14 or PI were directly observed with a fluorescence microscope. Figure 6 is an image recorded of a stable flow field at the intersection of two streams that included a semen sample and a medium. The motile mouse sperm swam across the streamlines into the sorting channel and were collected in reservoirs, whereas the dead sperm flowed with the streamlines into the outlet-up. This phenomenon indicates an increased quality and quantity of sorted sperm.
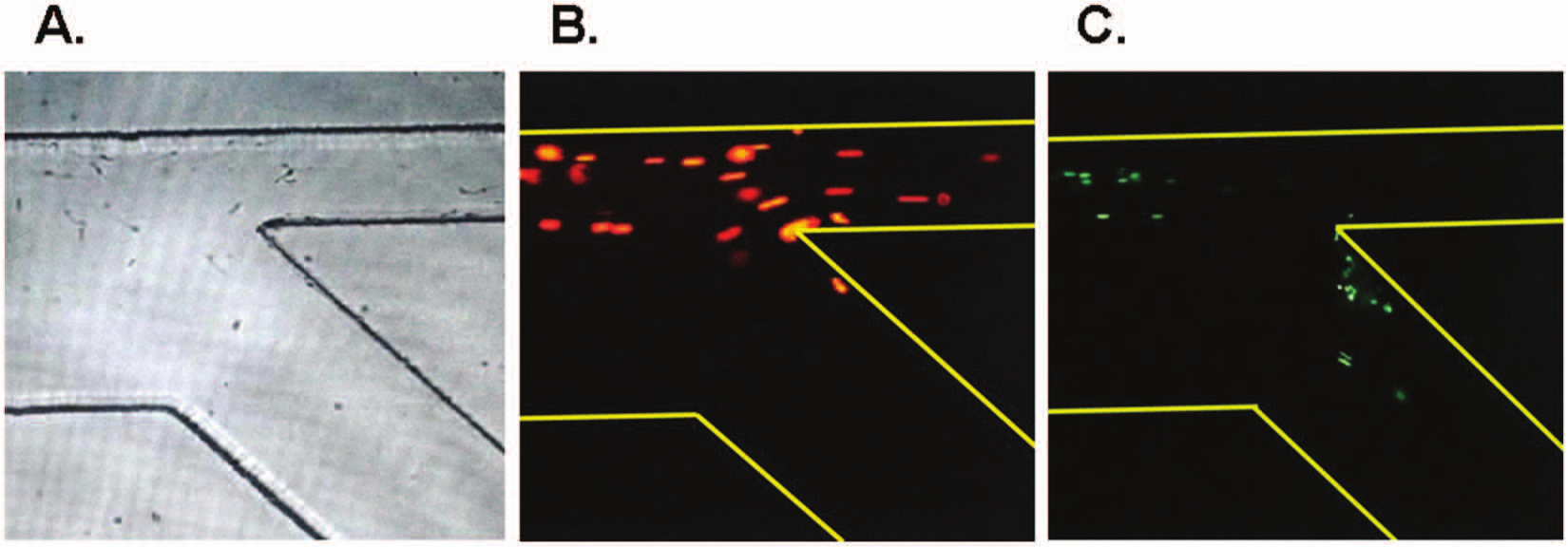
Separation of mouse motile sperm with a microfluidic chip. The recorded images from the inlet intersection of two streams revealed that motile mouse sperm passed through the straight flow boundary into the curved laminar flow with medium (
Flow Cytometry
The detection analysis technology of flow cytometry uses fluid to drive the cells or particles. The sample is stained before experiments. Fluorescence is excited by flow cytometry and receives the reflected light. The cell’s characteristics and size can be known. In our study, we use this information to understand the living and dead situation and the number of sperm. The mouse sperm were stained with SYBR-14 (green fluorescence) and PI (red fluorescence) to assess the varied sperm viability and motility. The results showed that the proportions of live and dead mouse sperm were identified via staining with a fluorescent dye and were quantified with flow cytometric analysis. Scattergram dot plots showed typical flow cytometric analysis for sperm that were stained with either SYBR-14 or PI. Quantitative data of the fluorescently stained sperm were collected with a flow cytometer (FACS Analyzer; Becton-Dickinson). For samples stained with SYBR-14, the green fluorescence (FL1-H) was collected through a filter (band pass at 525 nm band); the red fluorescence (FL2-H) was gathered through other filters (band passes at 575 and 635 nm). Histograms produced after collecting 10,000 events in a gate on sperms were generated for each replicate (n = 3). Control samples were unstained or stained with SYBR-14 alone or PI alone (data not shown). A one-parameter histogram showed a graph of cell count on the ordinate axis and the measurement parameter on the abscissa axis. With adjusted experimental parameters, the unstained sperm populations before sorting for the SYBR-14 and PI staining were located in the FL1-H− and FL2-H− regions, respectively; the stained sperm after sorting into the outlet reservoir from the microfluidic chip were located in the FL1-H+ and FL2-H+ regions ( Fig. 7 ). After sorting the sperm with the microfluidic chip, the live sperm increased and the dead sperm decreased in the outlet-down reservoir of the microfluidic chip, relative to the sperm before sorting. Figure 8 shows the sperm counts and the ratio of sorting using two-outlet channels of the microfluidic system. The viability results of the sample after sorting showed a greater ratio of live sperm in the outlet-down reservoir than in the outlet-up (62.8% ± 4.1% vs. 51.2% ± 4.3%, respectively) and correspondingly for dead sperm (37.2% ± 4.1% vs. 48.3% ± 3.6%, respectively).
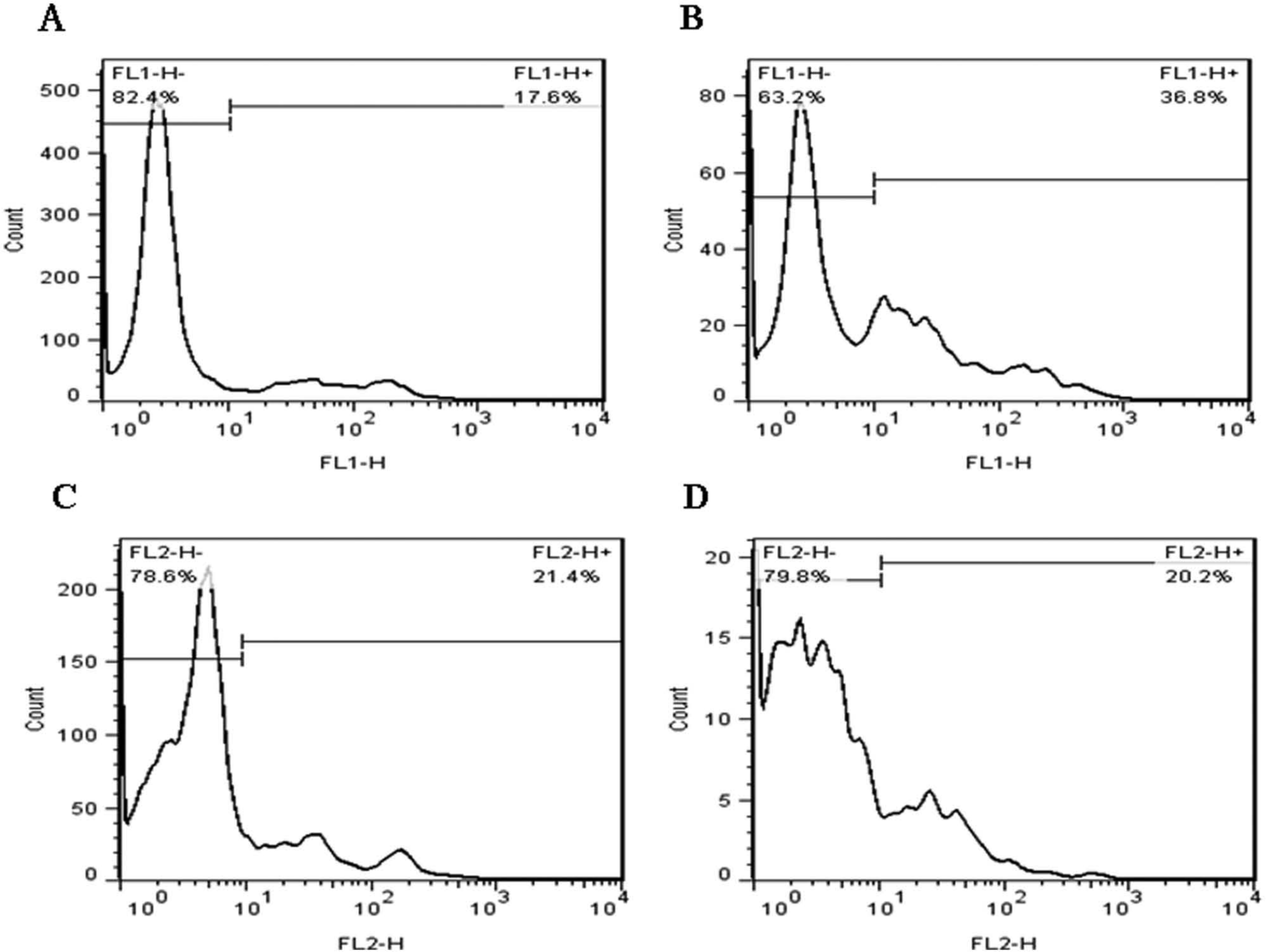
Flow cytometry analysis for the sorted sperm after the microfluidic chip. The mouse sperm were stained with SYBR-14 (green fluorescence) and PI (red fluorescence) to assess the varied sperm viability and motility. Scattergram dot plots show typical flow cytometric analysis for sperm stained with either SYBR-14 or PI. Quantitative data for fluorescently stained sperm were collected with a flow cytometer. For samples stained with SYBR-14, the green fluorescence (FL1-H) was collected through a band pass filter (525 nm); the red fluorescence (FL2-H) was gathered through other band pass filters (575 and 635 nm). Histograms produced on collecting 10,000 events in a gate on sperm were generated for each replicate (n = 3). Using adjusted experimental parameters, the unstained sperm populations before sorting with SYBR-14 and PI staining were located in the FL1-H− and FL2-H− regions, respectively. The stained sperm after sorting into the outlet reservoir in the microfluidic chip were located in the FL1-H+ and FL2-H+ regions. After sorting the sperm with a microfluidic chip, live sperm increased (
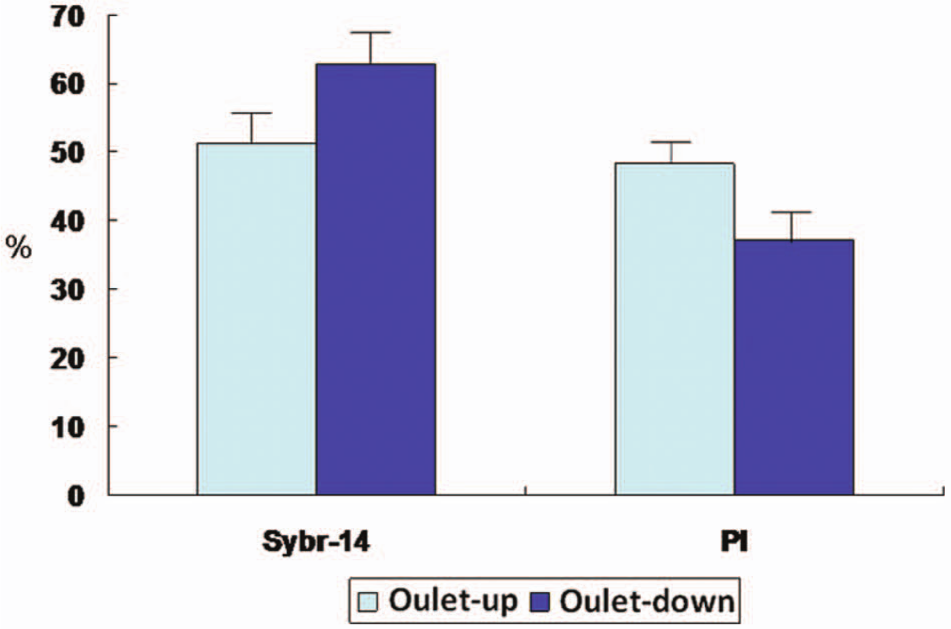
Sperm counts and ratio of sorting using two-outlet channels of the microfluidic system. The viability results of the sample after sorting showed a greater ratio of live sperm in the outlet reservoir relative to the outlet. Correspondingly, the sample after sorting showed a smaller ratio of dead sperm in the outlet reservoir relative to the outlet.
Data Analysis
The statistical analysis was undertaken with Statistical Package for Social Sciences (Stat-view Package; SPSS, Inc., an IBM Company, Chicago, IL) with p < 0.05 considered statistically significant. In the future, our study will be applied to human sperm, which is different from ICR mice sperm. The percentage of dead sperm in the outlet-down can be reduced, which will strengthen the results in the future.
In conclusion, an integrated microfluidic system that consists of a laminar stream–based, surface-modified microchannel has been used to demonstrate the separation of motile sperm. To increase the sorting efficiency of the sorting device, we performed CFD simulation to analyze the phenomenon of a 3D flow field. Microscopic examination revealed that sperm had varied motility. The fluorescent images indicated that the laminar streams are the same as for the CFD simulation, which revealed the distribution of the flow field. To confirm the sorting situation, through flow cytometric analysis, we readily identified and quantified the proportions of live and dead sperm after sorting. The microfluidic system with a surface-modified microchannel improves the sorting efficiency for the quality and quantity of sperm. The viability results of the sample through the outlet-down reservoir were higher in comparison to the outlet-down, demonstrating the improved quality of sorting sperm. As a result, more live sperm, meaning motile sperm, were counted in the outlet channels. The counted ratio of live sperm revealed the quantity and efficiency of the sorted sperm. With this approach, the characteristics of the laminar flow and a novel design were used to demonstrate the microfluidic system with a high efficiency to sort sperm motility. An optimized microfluidic system provides a suitable opportunity for use and an inexpensive requirement to select the most appropriate sperm before IVF/ICSI.
Footnotes
Acknowledgements
We thank Professor Fan-Gang Tseng for providing the fabrication facilities from National Tsing Hua University.
This research was presented in part at the annual meeting of the Society for Gynecological Investigation; March 24–27, 2010; Orlando, Florida.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support provided by NTHU/CGMH collaboration funds, and part of the fund comes from the National Science Council of Taiwan (NSC 101-2120-M-002-017, NSC 101-2314-B-182-041, and NSC 101-2221-E-007-099-MY3). NSC (National Science Council, Taiwan, grant 98-2314-B-182A-106) and Chang Gung Hospital (research grant CMRPG 361081 and CMRPG 380121), both to H.-Y.H., supported this research in part.
