Abstract
Although microscopy still represents the gold standard for cytometric analysis of peritoneal fluids, automated flow cytometry may improve throughput and accuracy. We evaluated the performance of total nucleated cell (TNC), white blood cell (WBC), polymorphonuclear cell (PMN), and mononuclear cell (MONO) counts of Sysmex XE-5000 on peritoneal fluids. The imprecision was excellent, being always lower than 11%, whereas linearity studies yielded correlation coefficients of 1.00 for all parameters. The carryover was always lower than 0.2%. The comparison between XE-5000 and microscopic analysis of 117 ascitic fluids yielded correlation coefficients always greater than 0.96, with mean biases <11/µL. The diagnostic accuracy versus manual microscopy was greater than that of XE-2100, especially at thresholds for septic ascites (100 versus 98% for ≥500 WBC/µL; 98 versus 93% for ≥250 PMN/µL). The correlation with manual microscopy for macrophages and mesothelial cell count was also higher for XE-5000 than for XE-2100 (0.63 versus 0.55). The results of this evaluation show optimal performance of XE-5000 for routine analysis of ascitic fluids, which are combined with the advantages of automated analysis such as high throughout, shortened turnaround time, no need of sample preparation and trained staff, reduced sample volume, and less likelihood of transcriptional errors.
Ascites, a term that conventionally defines the accumulation of fluid within the peritoneal cavity, is a frequent complication of localized and systemic disorders. The leading conditions in which ascites may significantly develop include cirrhosis (especially of alcoholic origin), heart failure and constrictive pericarditis, Budd-Chiari syndrome or other veno-occlusive disorders, malnutrition (especially Kwashiorkor), nephrotic syndrome, malignancy (both primary peritoneal carcinomatosis and metastasis), infections such as tuberculosis or bacterial peritonitis, pancreatitis, serositis, hereditary angioedema and Meigs syndrome, as well as hypothyroidism, renal dialysis, or peritoneum mesothelioma, among other less frequent causes. 1
The identification and differential diagnosis of ascites is an essential part of clinical decision making and encompasses paracentesis, with chemical and morphological analysis of the peritoneal fluid, which basically includes assessment of proteins and albumin, along with cell counting and classifcation. 2 In particular, a white blood cell (WBC) count ≥500/µL 3 and/or a polymorphonuclear cell (PMN) count ≥250/µL 2 are required for establishing a diagnosis of spontaneous bacterial peritonitis.
Although the gold standard for cell counting and classification in ascitic fluid is still represented by manual microscopic analysis, entailing total cell and cytocentrifuged differential counts, the automated flow cytometry by means of urine 4 or hematology analyzers 5 may be effective to overcome most of the drawbacks of manual microscopy, with the advantage of examining rapidly and accurately an almost limitless number of characteristics of individual cells. The diagnostic accuracy of body fluid analysis has been assessed with a variety of hematological instruments, some of which revealed unacceptable performances because of unsuitable detection limits and interference or mismatch due to the presence of macrophages and mesothelial cells, which are typically lacking in the whole blood. 6 Therefore, the aim of this study was to assess the performance of the novel and fully automated Sysmex XE-5000 for flow cytometric analysis of peritoneal fluid samples as compared with data obtained with manual microscopy and Sysmex XE-2100.
Materials and Methods
Instrument Characteristics
The Sysmex XE-5000 (Dasit SpA, Cornaredo, Italy) represents the latest development of Sysmex Corporation hybrid, fully automated hematology instrumentation. Besides basic characteristics shared with the former XE-2100, and which have been comprehensively described elsewhere, 7 the novel analyzer is equipped with additional features, including a dedicated body fluid mode with extended linearity of cell enumeration and increased precision in samples with lower cell values (i.e., imprecision of 20% at a cell value of 10/µL versus 50/µL for XE-2100). 8 This specific mode is based on an innovative software, wherein WBC, PMN, and mononuclear (MONO) cells are enumerated and classified in the “DIFF” channel according to side scatter and intensity of fluorescence staining. Cells with increased fluorescence such as macrophages and mesothelial cells are also counted in the DIFF scattergram and finally subtracted from the total WBC count. The number of total nucleated cells (TNCs) is thereby the sum of WBC and “other” cells.
Study Design
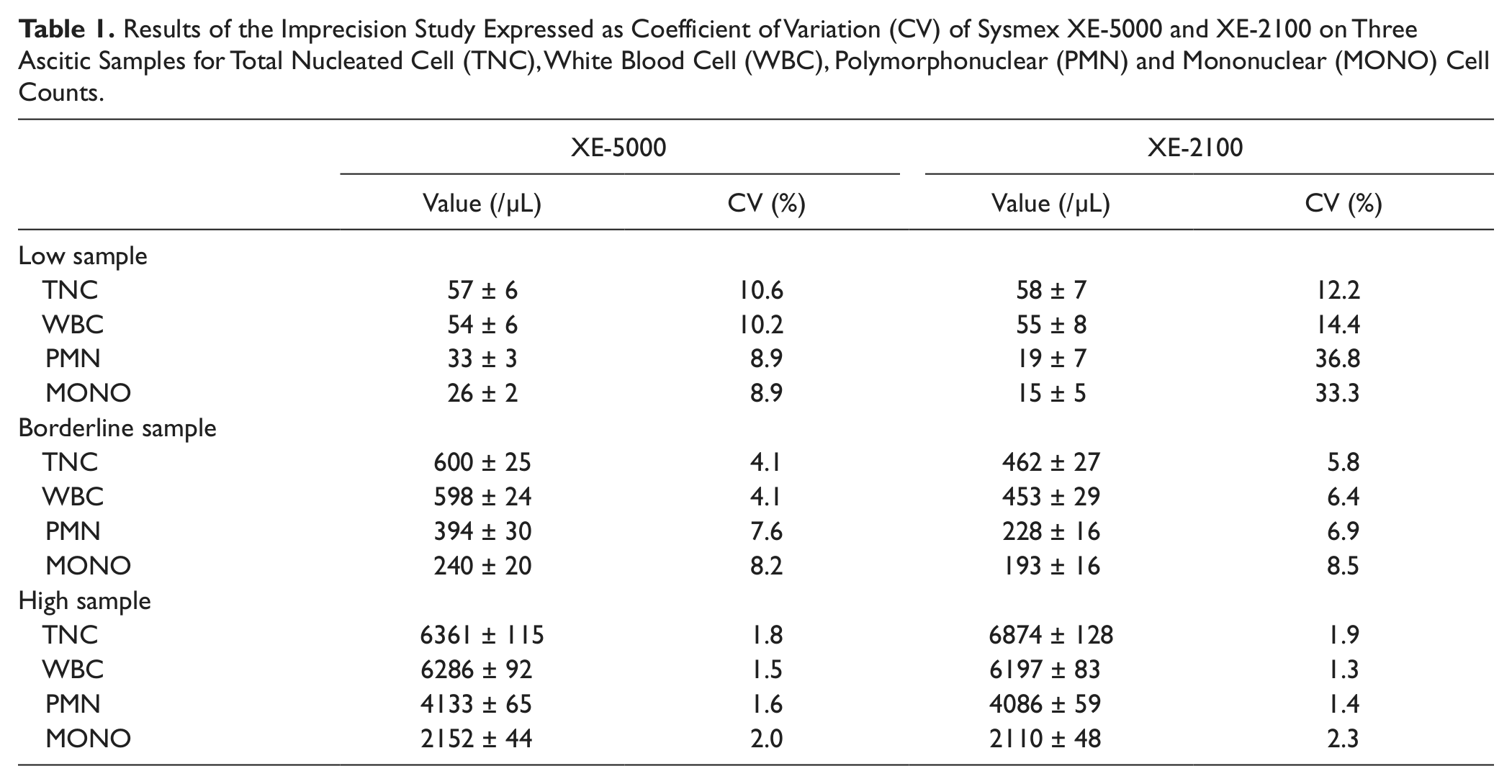
All experiments were carried out using peritoneal samples collected in 2.0 mL, K2EDTA anticoagulated tubes (Becton Dickinson Italia S.p.A., Milan, Italy). The imprecision of the XE-5000, as expressed by the coefficient of variation (CV), was evaluated according to the Clinical and Laboratory Standards Institute (CLSI) document EP5-A29 by analyzing in 20 consecutive runs three peritoneal fluid specimens selected among those referred for routine analysis and displaying low, borderline, and high values of TNC, WBC, PMN, and MONO counts ( Table 1 ). The linearity was evaluated according to the CLSI document EP6-A, 10 by analysis of serial dilutions in triplicate (from 135 to 1162 TNC/µL, from 134 to 1158 WBC/µL, from 100 to 873 PMN/µL, and from 33 to 288 MONO/µL). The carryover was evaluated by assessing the sequence A1-A2-B1-B2-B3 in duplicate, where A was a peritoneal fluid sample with a high number of cellular elements (i.e., 2431 TNC/µL, 2412 WBC/µL, 643 PMN/µL, and 1769 MONO/µL) and B a peritoneal sample with a low number of cells (i.e., 6 TNC/µL, 5 WBC/µL, 4 PMN/µL, and 1 MONO/µL). The final results were calculated according to the validated formula proposed by Broughton, 11 as follows: carryover, % = 100 × (B1 − B3)/(A2 − B3).
Results of the Imprecision Study Expressed as Coefficient of Variation (CV) of Sysmex XE-5000 and XE-2100 on Three Ascitic Samples for Total Nucleated Cell (TNC), White Blood Cell (WBC), Polymorphonuclear (PMN) and Mononuclear (MONO) Cell Counts.
Comparison studies were performed by analysis of all consecutive peritoneal samples referred from the hospital wards to the laboratory for routine chemical and morphological analysis over 1 month (i.e., June 2012). The results of WBC, TNC, PMN, MONO, and other cell (including macrophages and mesothelial cells) counts were compared with those obtained with either manual microscopy on Nageotte chamber and Fuchs-Rosenthal hemocytometer after staining with Turk’s and May-Grunwald-Giemsa reagents and 1:10 or 1:20 dilution (depending on the number of cells in specimen) or on Sysmex XE-2100 (Dasit SpA). Two independent experts performed microscopic analyses, and results were finally averaged. The correlation between methods was assessed by Pearson correlation. The bias (and 95% confidence interval [CI]) was calculated by means of Bland-Altman analysis. The statistical analysis was performed using Analyse-it for Microsoft Excel (Analyse-it Software Ltd, Leeds, UK). The study was carried out in accordance with the Declaration of Helsinki, under the terms of all relevant local legislation.
Results
Optimal performance in terms of imprecision was obtained on XE-500 for all parameters tested, as shown in Table 1 . In particular, the CVs were between 1.8% and 10.6% for TNC, 1.5% and 10.2% for WBC, 1.6% and 8.9% for PMN, and between 2.0% and 8.9% for MONO. In all cases, the performance was better or at least not inferior to that of XE-2100, with remarkable improvement at lower values ( Table 1 ). Imprecision on macrophages and mesothelial cells could not be calculated because the values were always below the analytical sensitivity of the instrument. The XE-5000 also exhibited optimal linearity in the range of values of the peritoneal specimens tested, with all correlation coefficients being 1.00 (p < 0.001). No analytically and clinically significant bias was observed according to Broughton’s protocol, 11 with carryover on XE-5000 always lower than 0.2%.
For comparison with manual microscopy and XE-2100, 118 peritoneal fluid specimens were received over the 1-month study period. One sample was excluded because of the presence of a visible clot, so 117 specimens were included in the final statistical analysis. The comparison between XE-5000 and microscopic analysis yielded the following results (with mean bias and 95% CI): TNC, r = 1.00 (mean bias, 8/µL; 95% CI, 5 to 12/µL); WBC, r = 1.00 (mean bias, 5/µL; 95% CI, 2 to 9/µL); PMN, r = 0.98 (mean bias, 8/µL; 95% CI, −2 to 19/µL); MONO, r = 0.96 (mean bias, 1/µL; 95% CI, −3 to 6/µL); and other cells, r = 0.63 (mean bias, −3/µL; 95% CI, −4 to −2/µL), with all correlations being p < 0.001 ( Fig. 1 ). The corresponding figures for comparison between manual microscopy and XE-2100 were as follows: TNC, r = 1.00 (mean bias, 6/µL; 95% CI, 2 to 10/µL); WBC, r = 1.00 (mean bias, 5/µL; 95% CI, 0 to 9/µL); PMN, r = 0.99 (mean bias, 6/µL; 95% CI, 0 to 12/µL); MONO, r = 0.97 (mean bias, −6/µL; 95% CI, −11 to −1/µL); and other cells, r = 0.54 (mean bias, 10/µL; 95% CI, 2 to 20/µL), with all correlations being p < 0.001. At the WBC diagnostic threshold for peritonitis (i.e., ≥500/µL), the agreement between XE-5000 and manual microscopy was absolute (kappa statistics, 1.00; p < 0.001), thereby displaying 100% sensitivity, specificity, and overall agreement. At the identical threshold of ≥500 WBC/µL, a slightly lower agreement was found between manual microscopy and XE-2100 (i.e., kappa statistics, 0.98; p < 0.001), yielding 100% sensitivity, 96% specificity, and 99% overall agreement. The diagnostic accuracy as compared with manual microscopy at the ≥250 PMN/µL cutoff was greater for XE-5000 (kappa statistics, 0.94; 98% sensitivity, 100% specificity, and 98% accuracy) than for XE-2100 (kappa statistics, 0.78; 93% sensitivity, 95% specificity, 93% accuracy). Using a cutoff of ~10 WBC/µL, wherein Perné et al. 12 found most mismatches in cerebrospinal fluids, we found a 98% agreement (p < 0.001) between manual microscopy and XE-5000, with false-positive and false-negative ratios lower than 1%. At the identical WBC threshold, the agreement between manual microscopy and XE-2100 was also 98% (p < 0.001), with 2% false-negative but no false-positive results.
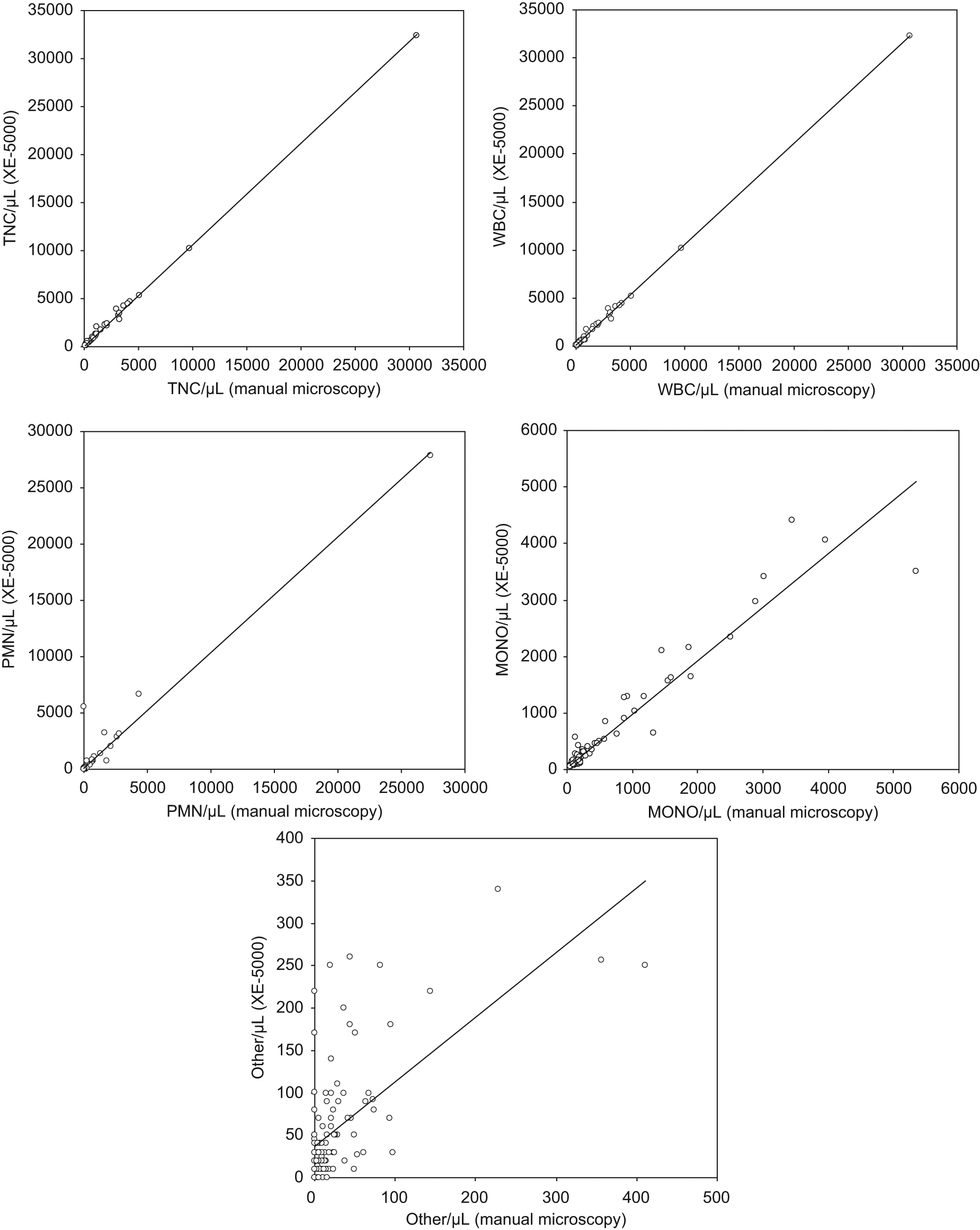
Person’s correlation between manual microscopy and Sysmex XE-5000 for total nucleated cell (TNC), white blood cell (WBC), polymorphonuclear cell (PMN), mononuclear cell (MONO), and other cell (i.e., macrophages and mesothelial cells) counts in 117 inpatients specimens of peritoneal fluid.
Discussion
The accurate enumeration and classification of peritoneal fluid cells are the cornerstones in the diagnostic approach of patients with ascites, wherein identification of septic peritonitis and discrimination between transudate and exudates strongly influence the clinical and therapeutic decision making. 1 Nevertheless, routine and especially stat analysis of a large number of biological fluids represents a critical aspect, wherein clinical laboratories are currently facing the increasing pressure to automate several analytical operations due to continuous increase of workload and shortage of skilled personnel. 13 Although manual microscopy remains the gold standard for analysis of biological fluids, recent advancements in automated flow cytometry have overcome several shortcomings of manual analysis such as the need of trained staff, arbitrary cell classification, suboptimal imprecision, long turnaround time, and challenges of stat testing in several facilities. An appropriate analysis of the diagnostic accuracy of the single instrument is, however, necessary before hemocytometric analysis can be recommended for routine analysis of body fluids, inasmuch as both composition and concentration of proteins and ions in cerebrospinal, periodontal, pleural, or other cavitary fluids are rather different from those in the blood, whereas cellular elements such as macrophages and mesothelial cells are normally absent from the circulation. Finally, the typical detection limits and analytical thresholds of most hemocytometers may be unsuitable for enumeration of cells in body fluids, where these elements may be present to a much lower value than in blood.
The novel Sysmex XE-5000 is an evolution of the former XE-2100, with better performance claimed for analysis of body fluids. However, no definitive data are available for its diagnostic accuracy, especially in comparison with that of the XE-2100. In a previous investigation, Paris et al. 14 found an optimal agreement for PMN (r = 0.99) and MONO (r = 0.98) counts between the manual method and XE-5000 automated count on 81 ascitic fluid samples. Nevertheless, the accuracy of TNC, WBC, and other cells counts was not specifically addressed in peritoneal fluid specimens. The diagnostic accuracy of XE-5000 was also evaluated by de Jonge et al. 8 on 72 body fluids (i.e., pleural, ascites, and synovial samples), reporting good agreement but very large variations for WBC (mean bias, −12/µL; 95% CI, −1231 to 1207/µL), PMN (mean bias, −1/µL; 95% CI, −1187 to 1186/µL), and MONO (mean bias, −5; 95% CI, −348 to 337/µL).
The results of our investigation attest that the novel XE-5000 hemocytometer exhibits optimal performance for routine analysis of ascitic fluids, with excellent linearity and no significant carryover. The overall diagnostic accuracy was found to be even greater than that of the XE-2100, especially at values close to the diagnostic thresholds for septic ascites. Although at the lower threshold of 10 WBC/µL the accuracy was globally comparable between the two analyzers (i.e., 98% for both), at diagnostic cutoffs of ≥500 WBC/µL and ≥250 PMN/µL, the accuracy of XE-5000 was greater, being 100% versus 99% in the former case and 98% versus 93% in the latter. Another important improvement is represented by the more accurate classification of macrophages and mesothelial cells (i.e., “others”), wherein the correlation with manual microscopy was 0.63 for XE-5000 as compared with 0.53 for the former hemocytometer. The correlation and the bias observed for the other parameters (i.e., TNC, WBC, PMN, and MONO) was instead virtually identical between the two analyzers. Besides excellent diagnostic accuracy and improved precision at lower values, the Sysmex XE-5000 exhibits additional advantages over manual microscopy, which are typical of automated flow cytometric analysis and thereby include higher throughout, reduced turnaround time (i.e., the analysis is performed in less than 1 min), availability of stat testing, no need of trained or skilled staff for performing microscopic analysis of the vast majority of the specimens, as well as reduction of sample volume, no need of sample preparation, and potential for reducing transcriptional errors due to the availability of automatic transmission of data to the laboratory information system. It is also noteworthy that the good precision at very low threshold, yielding a 98% agreement with manual microscopy, confirms that XE-5000 may be reliably used for routine analysis of a vast array of biological fluids.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
