Abstract
Bovine viral diarrhea virus (BVDV) is a pathogen in cattle and alpacas (Vicugna pacos), causing acute and persistent BVDV infections. We characterized the effect of acute BVDV infection on the immune system of alpacas by determining lymphocyte subpopulations in peripheral blood and gut-associated lymphoid tissues (GALT) as well as serum interferon levels. Alpacas were experimentally infected with BVDV-1b (strain CO-06). Peripheral blood leukocytes were isolated at 0, 3, 6, and 9 d postinfection (dpi), and leukocytes of GALT at 9 dpi, and evaluated using flow cytometry. Serum interferon levels were determined daily. Flow cytometric analyses of peripheral blood leukocytes showed a significant decrease in CD4+, CD8+, and αβ T-lymphocytes at 3 dpi. CD8+ lymphocytes were significantly increased, and activated lymphocytes were significantly decreased in the C3-stomach region in BVDV-infected alpacas. Serum interferon concentrations significantly increased in BVDV-infected alpacas at 3–6 dpi, peaking at 3 dpi. Our study confirms that BVDV can be a primary acute pathogen in alpacas and that it induces an interferon response and alters leukocyte subset populations. The changes in the proportion of T-lymphocytes during the early stages of BVDV infection may result in transient immunosuppression that may contribute to secondary bacterial and viral infections, similar to cattle.
Introduction
Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus 29 ) is an economically significant viral pathogen of cattle in the United States. 3 BVDV also causes significant economic loss and persistent infections in alpacas (Vicugna pacos).2,6,33 Evaluation of 63 alpaca herds in the United States in 2006–2007 showed that 6.3% of the herds contained persistently infected (PI) crias, confirming the significance of BVDV infections in alpacas in the United States. 33
Acute, chronic, and persistent BVDV infections in cattle have been well described.8,18,22,23,28,34,36 In cattle, BVDV is lymphotropic, contributing to immunosuppression and enhanced pathogenicity of secondary viral and bacterial pathogens.4,5,10,12,17,18,27 The predominant biotype and subspecies reported in feedlot cattle with respiratory disease is noncytopathic BVDV-1b. 13 Experimental and naturally occurring BVDV infections in alpacas have been described and have shown that BVDV is a primary viral pathogen in alpacas and may contribute to secondary bacterial and viral infections as a result of immunosuppression, similar to what has been shown in cattle.2,6,32 The only BVDV genotype (and subgenotype) reported to infect North American alpacas is BVDV-1b.21,33
Lymphocytopenia occurs during the first 3–7 d following acute BVDV infection in cattle18–20 and alpacas. 32 Microscopically, viral antigen deposition and lymphoid depletion occur in the gut-associated lymphoid tissues (GALT).8,10,18,22,23,28,32,34 Using immunohistochemistry and primary T- and B-cell–specific monoclonal antibodies (mAbs), B-lymphocyte depletion was documented in the GALT of alpacas following acute BVDV infection. 32 In calves, following experimental BVDV-1b infection and using flow cytometric (FC) analyses, CD4+ and CD8+ lymphocyte subpopulations were depressed in thymic tissue and the B-lymphocyte subpopulation was depressed in Peyer patches.5,24 To our knowledge, the effect of acute BVDV-1b infection on the immune system of alpacas based on FC analyses of peripheral leukocytes and GALT following acute BVDV infection has not been reported. The objective of our study was to determine the effect of acute BVDV-1b infection on the immune system of alpacas by determining lymphocyte subpopulations in peripheral blood and lymphoid organs using FC analyses.
Materials and methods
Animals
Fifteen clinically normal alpacas, ages 6–24 mo, negative for BVDV antibody by serum neutralization, were housed indoors in a biosecurity level 2 animal research facility under Institutional Animal Care and Use Committee–approved conditions. The animals were housed in age-matched groups of 2 and acclimated for 7 d prior to the initiation of experiments.
Cells and virus
Bovine turbinate a (BT) cells were grown in Dulbecco modified Eagle medium b (DMEM) supplemented with 10% equine serum. c BVDV CO-06 is an alpaca-origin noncytopathic BVDV-1b isolate recovered from a PI alpaca at the University of Nebraska. 31 BT cell monolayers at 90% confluency were infected with BVDV CO-06 at a multiplicity of infection of 1 in tissue culture flasks and incubated at 37°C in a humidified incubator with 5% CO2 for 4 d. Following a single freeze–thaw cycle, aliquots of viral-infected BT cell lysates were stored at −80°C until used. All cells used in our study were tested for extraneous BVDV, bacteria (including mycoplasma), and fungi. Alpacas received 106.9 TCID50 of BVDV CO-06 in 10 mL of DMEM by intranasal aerosolization using a nebulizer.d,25
Experimental design
Nine alpacas were experimentally infected on day 0 with BVDV CO-06. Six alpacas served as sham-inoculated controls. All animals were examined twice daily for evidence of clinical disease and to ensure patient welfare. Clinical signs (respiratory rate, dyspnea, nasal secretion, cough, feces, and body condition) were scored as described previously. 20 Starting on day 0 and continuing on 3, 6, and 9 d postinfection (dpi); blood was collected via jugular venipuncture in sterile tubes containing ethylenediamine tetra-acetic acid (EDTA). e Peripheral blood leukocytes (PBLs) were isolated from blood for FC analyses. Blood samples for serum were collected daily. Alpacas were euthanized at 9 dpi. Postmortem examination of gross and histologic changes has been described previously. 32 GALT from the stomach region C3 and Peyer patches from the jejunum were collected and stored on wet ice until processed for FC analyses. Euthanasia was performed by injection with a sedativef,g followed by an overdose of a barbiturate. h All animal housing, care, and research was performed in compliance with protocols established by the University of Nebraska Institutional Animal Care and Use Committee.
Preparation of cells from peripheral blood and lymphoid tissues
A density separation gradient was prepared by carefully overlaying 5 mL of sodium diatrizoate i (density 1.119 g/L) with 5 mL of a solution containing polysucrose and sodium diatrizoate j (density 1.077 g/L) in a 50-mL conical tube. Ten mL of unclotted blood was carefully layered over the separation gradient and centrifuged for 45 min at 20°C at 2,060 × g. Isolated PBLs were removed, washed once in calcium- and magnesium-free, phosphate-buffered saline with 0.05 M EDTA (CMF-PBS-EDTA) and centrifuged for 15 min at 4°C at 913 × g. PBLs were washed twice more in CMF-PBS-EDTA for 10 min at 4°C followed by centrifugation at 514 × g. Following 2 additional washes in CMF-PBS to remove residual EDTA, the cells were resuspended in 10 mL of 0.01 M PBS with 1% bovine serum albumin and 0.1% sodium azide (PBS-BSA-NaN3) and adjusted to a concentration of 5 × 106 cells/mL.
Tissues were processed for isolation of leukocytes immediately after autopsy. The isolation of lymphocytes from lymphoid tissues has been described previously. 24 Briefly, cell suspensions were prepared by teasing the tissues apart with a scalpel blade in sterile Petri dishes in cold isotonic saline. The cell suspension was filtered through nylon mesh k with a 70-μm pore size to remove large tissue debris. Filtered cells from lymphoid tissues were then processed as described for PBLs.
Monoclonal antibodies used for flow cytometry
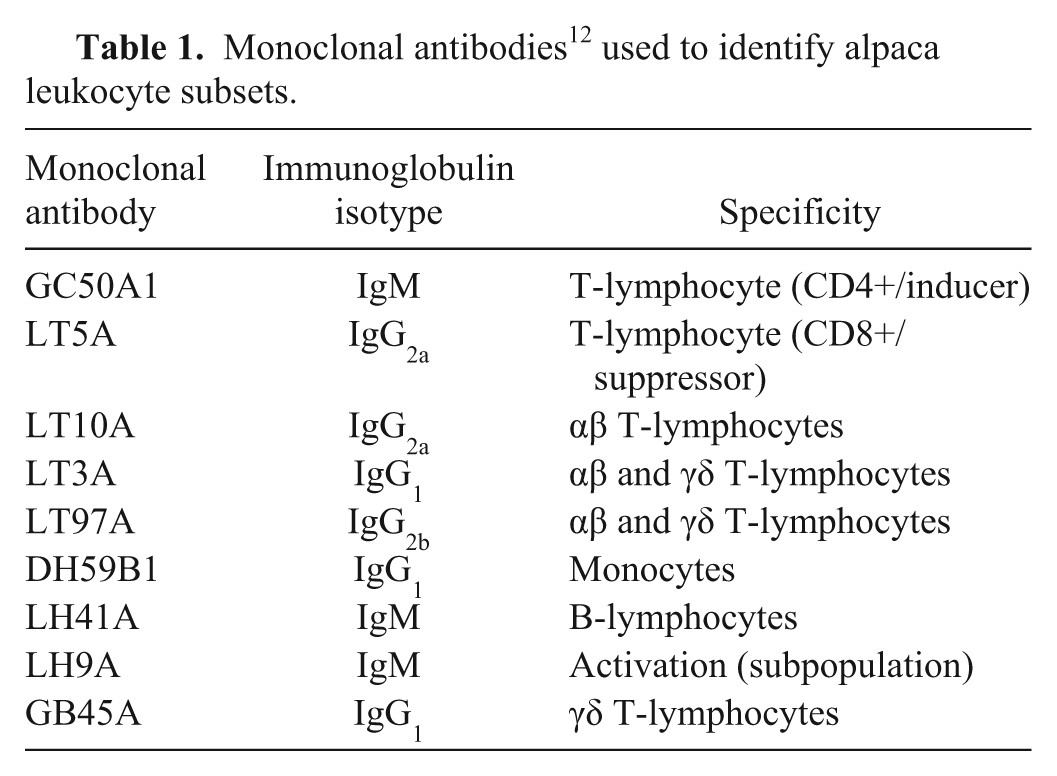
The mAbs used, their specificity, and a reference to their initial characterization are shown in Table 1 and have been described previously. 7 The mAbs included those that recognize conserved determinants on orthologous molecules in multiple species, as well as those developed by immunizing mice with llama PBLs, thymocytes, or cultured lymphocytes. 15 CD4+ and CD8+ cells are mainly αβ T-lymphocytes detected by mAbs GC50A1 and LT5A, respectively. LT97A appears to recognize a molecule on all αβ and γδ T-lymphocytes. LT3A also appears to recognize a molecule on all αβ and γδ T-lymphocytes. A comparison of LT3A with LT10A suggests that LT3A recognizes CD5. CD2 is expressed on a subset of WC1+ γδ T-cells. LT10A recognizes CD2 on αβ T-lymphocytes; it doesn’t recognize all LT3A-positive cells. GB45A recognizes an epitope expressed on most of the WC1+ subset population of γδ T-lymphocytes. This molecule is unique to Artiodactyla (ruminants, pigs, and camelids). LH41A identifies a population of cells that are Ig positive, therefore it recognizes most of the B-cells. Monoclonal antibody LH9A recognizes a molecule or molecules expressed on a subset of activated lymphocytes, and DH59B1 recognizes granulocytes and monocytes. 7
Monoclonal antibodies 12 used to identify alpaca leukocyte subsets.
Staining cells for flow cytometry
After processing, 200 μL of each cell suspension were placed in wells of a 96-well plate. Cells were pelleted by centrifugation at 185 × g for 5 min at 4°C. Incubation parameters and washes have been reported previously. 24 Briefly, cells were incubated with 1 or 2 primary mAb(s) (50 μL each) in a final volume of 200 μL of PBS-BSA-NaN3 for 60 min at 4°C. Following incubation, cells were centrifuged and washed 3 times in 200 μL of PBS-BSA-NaN3. Following the final wash, cells were incubated for 60 min at 4°C with 100 μL of diluted fluorescein-conjugated polyclonal goat anti-mouse IgG/IgM antibody or goat isotype–specific antibodies conjugated with fluorescein or phycoerythrin in a final volume of 200 μL directed against the specific isotypes of the primary antibodies. After incubation, the cells were washed and fixed in 200 μL of 3% formaldehyde in PBS, then transferred to tubes containing 300 μL of PBS–formalin, and stored at 4°C until examined.
Flow cytometry
Two-color FC analyses of cell preparations were conducted on a flow cytometer. l Controls for specificity of fluorescence in each preparation consisted of: 1) cells in PBS-BSA-NaN3 and 2) cells plus secondary antibodies only. The percentage of each positive lymphocyte subpopulation was calculated by quadrant analysis. m
Type I interferon analysis
An Mx protein–chloramphenicol acetyltransferase (Mx-CAT) reporter gene assay was used to measure interferon (IFN) in serum samples. 11 Madin–Darby bovine kidney cells stably transfected with a plasmid containing a human MxA promoter driving a CAT complementary DNA (MDBK-t2 cells) were used for the assay. 16 MDBK-t2 cells at a concentration of 1 × 106 cells per well (3.5 cm) were seeded into 6-well plates. Cell culture medium was adjusted to 2 mL per well of DMEM with 10% fetal bovine serum (FBS) and 10 µg/mL of blasticidin. n Plates were incubated for 24 h at 37°C in 5% CO2. The following day, duplicate serum samples from alpacas were heat inactivated at 56°C for 30 min. Samples were diluted 1:5 in DMEM with 2% FBS. Medium was discarded from wells, and heat-inactivated serum samples diluted with DMEM were added to the wells and incubated for 24 h at 37°C in 5% CO2 atmosphere. The next day, CAT expression was determined using a commercial ELISA kit. o The assay was performed following the manufacturer’s instructions. Cells were lysed for 20 min with Triton-X100 lysis buffer, and aliquots of 200 µL were added in duplicate to wells of the CAT-ELISA 96-well plate. A standard curve was developed using the same procedure described above with known amounts of recombinant human IFN-αA/D p using a 1:2 serial dilution (0.132–270 IU/aliquot). Interferon in test samples was calculated from the standard curve.
Statistical analyses
The mean ± standard error of the mean was calculated for each lymphocyte marker for lymphocytes harvested from PBLs, C3-stomach, and Peyer patches. A generalized linear model analysis of variance was used to determine the effect on the percent of lymphocytes from organs and on the percent of PBLs that stained positive for each marker in each treatment group. Comparisons between levels of significance (p ≤ 0.05) of treatment effects were examined using least square means.
Results
Flow cytometric analyses of peripheral blood leukocytes
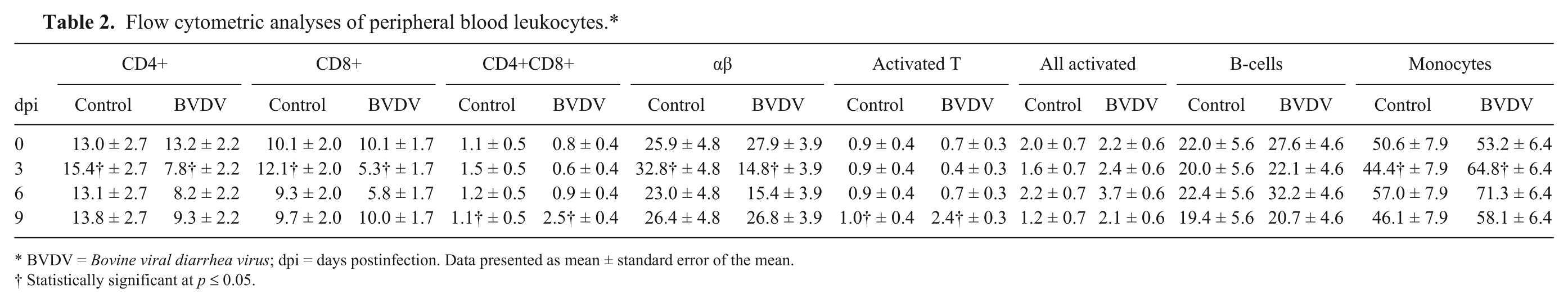
The PBLs from control and BVDV-1b experimentally infected alpacas were collected and analyzed by FC at 0, 3, 6, and 9 dpi (Table 2). The percentage of cells labeled with leukocyte subset–specific mAbs was determined. FC analyses of the PBLs showed a significant (p < 0.05) decrease in CD4+, CD8+, and αβ T-lymphocytes, and a significant increase in monocytes at 3 dpi in BVDV-infected alpacas compared with control animals. A significant increase in CD4+CD8+, and activated T-lymphocytes was observed in BVDV-infected alpacas at 9 dpi.
Flow cytometric analyses of peripheral blood leukocytes.*
BVDV = Bovine viral diarrhea virus; dpi = days postinfection. Data presented as mean ± standard error of the mean.
Statistically significant at p ≤ 0.05.
BVDV-infected alpacas had a significant decrease (p < 0.05) in peripheral CD4+ and CD8+ T-lymphocytes at 3 dpi compared with the control animals. The mean percentage of CD4+ cells were reduced to 7.8% in the BVDV-treated alpacas compared with 15.4% in the control alpacas at 3 dpi. The mean percentage of CD8+ cells were reduced to 5.3% in the BVDV-treated alpacas compared with 12.1% in the control alpacas at 3 dpi. Although not significantly different from control animals, the CD4+ and CD8+ T-lymphocytes were decreased in the BVDV-infected alpacas at 6 dpi. CD8+ T-lymphocytes returned to baseline at 9 dpi, whereas the CD4+ T-lymphocytes in the BVDV-infected alpacas were not significantly different from the control animals. The percentage of cells positive for both CD4+ and CD8+ markers was significantly increased (p < 0.05) in the BVDV-infected alpacas at 9 dpi (2.5%) compared with the control alpacas (1.1%).
Significant differences between the BVDV-infected alpacas and control alpacas were observed in the PBL subsets αβ, activated T-lymphocytes, and monocytes. At 3 dpi, a significant decrease (p < 0.05) in αβ T-lymphocytes was observed in the BVDV-infected alpacas (14.8%) compared with the control alpacas (32.8%). The αβ T-lymphocytes returned to baseline values at 9 dpi. Monocytes were significantly increased (p < 0.05) in BVDV-infected alpacas (64.8%) at 3 dpi compared with the control alpacas (44.4%). A significant increase (p < 0.05) in T-lymphocytes expressing a molecule associated with activation in the BVDV-treated alpacas (2.4%) compared with the control alpacas (1.0%) was observed at 9 dpi. No significant differences in B-lymphocytes were observed between the 2 groups of animals.
Flow cytometric analyses of leukocytes isolated from GALT
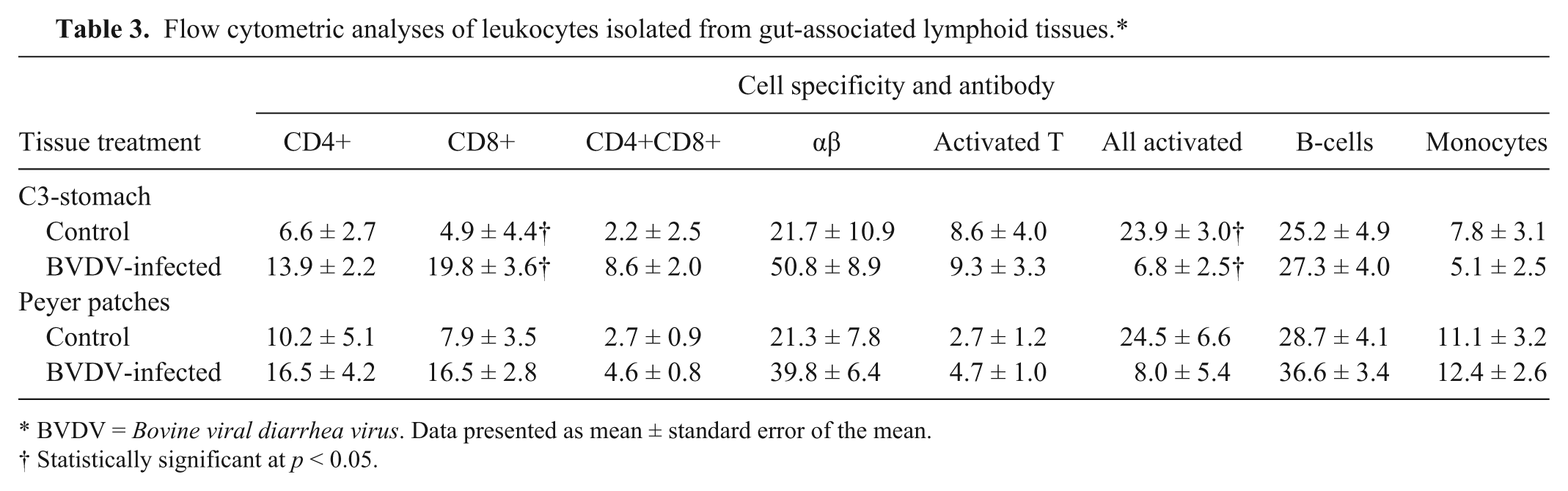
CD8+ T-lymphocytes were significantly increased in the C3-stomach region (p < 0.05) at 9 dpi in the BVDV-infected alpacas (19.8%) compared with control alpacas (4.9%; Table 3). In the C3-stomach, leukocytes expressing an activation-type molecule were significantly decreased (p < 0.05) in the BVDV-infected alpacas (6.8%) compared with the control alpacas (23.9%). There was no significant difference between treatment groups in the remaining PBL subsets from the C3-stomach and Peyer patches.
Flow cytometric analyses of leukocytes isolated from gut-associated lymphoid tissues.*
BVDV = Bovine viral diarrhea virus. Data presented as mean ± standard error of the mean.
Statistically significant at p < 0.05.
Serum interferon response
Serum IFN concentrations were significantly increased (p < 0.05) from basal concentrations in the BVDV-infected alpacas at 3–6 dpi, with the peak IFN concentration occurring at 3 dpi (Fig. 1) in the infected alpacas. Serum IFN concentrations returned to basal level for the BVDV-infected alpacas at 8 dpi. There was no increase in serum IFN concentration in the control alpacas.
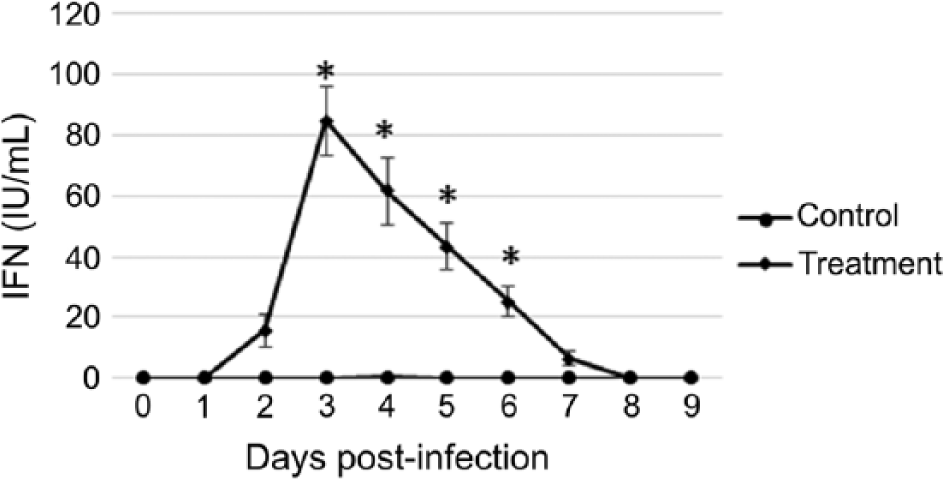
Mean ± standard error of the mean serum interferon (IFN) concentrations of alpacas (Vicugna pacos) inoculated on day 0 with Bovine viral diarrhea virus 1b (diamonds) or mock-inoculated (circles). Asterisk (*) indicates significant (p < 0.05) difference between groups.
Discussion
Our study confirms that BVDV is a primary pathogen in alpacas. BVDV infection in immunocompetent alpacas stimulates a significant IFN response and alters leukocyte subpopulations in the peripheral blood and GALT. Mild or inapparent clinical signs observed in alpacas following BVDV infection may be attributed to the significant IFN response at 3–6 dpi in alpacas experimentally infected with BVDV compared with the noninfected controls. The increased IFN concentrations in alpacas correlate with the increased IFN concentrations observed in cattle following acute BVDV infection.16,26,30 Serum IFN concentrations have been reported to increase in calves at 3–6 dpi following acute BVDV infection, similar to what we observed in the BVDV-infected alpacas.16,30
Previously, we have shown that alpacas have mild clinical signs and lesions following acute BVDV infection. 32 We have also previously shown that in vitro alpaca cells were less permissive to BVDV infection compared with bovine cells. Additionally, when cells were stimulated with double-stranded (ds)RNA alone or infected with noncytopathic BVDV, the alpaca cells were more responsive to the stimulation than were the bovine cells, resulting in elevated IFN-1 responses. 31 Previous studies have shown that clinical signs in alpacas following acute BVDV infections are mild or inapparent.32,35 This is similar to what has been reported in cattle.4,23,25,26 The significant IFN response following acute experimental BVDV infection in alpacas reported herein and in conjunction with in vitro elevated IFN responses of alpaca cells and reduced inhibition of IFN synthesis following postinfection stimulation with dsRNA reported previously, 31 may contribute to the mild or inapparent clinical signs observed in alpacas following BVDV infection.
Using PBLs collected from BVDV-infected and control noninfected alpacas and analyzed using FC, we observed a significant decrease in CD4+, CD8+, and αβ T-lymphocytes at 3 dpi. This correlates with the decrease in total white blood cells and lymphocytes in peripheral blood at 3–7 and 3–6 dpi, respectively, previously reported following experimental infection of alpacas with BVDV-1b. 32 No significant change in the peripheral blood B-lymphocytes was observed in our study. Decreases,9,14,26 increases, 5 and no change 1 in the peripheral blood B-lymphocyte levels in calves following BVDV infection have been reported. These differing results in cattle may be a result of differences in the BVDV strain and/or the immune status of the host.
The elevated PBL subpopulations, CD4+CD8+ double-labeled lymphocytes and activated lymphocytes, and elevated CD8+ T-lymphocytes in C3-stomach GALT in BVDV-infected alpacas indicate an immunosuppressive response early during BVDV infection followed by an elevated host immune response to clear the viral infection and repopulate depleted lymphoid tissues. Therefore, during active infection and during recovery until the lymphatic tissues are repopulated, these animals putatively have increased susceptibility to infection with other viral and bacterial pathogens.
Footnotes
Authors’ contributions
CL Topliff contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. AA Alkheraif contributed to acquisition, analysis, and interpretation of data. CA Kuszynski, WC Davis, and KM Eskridge contributed to analysis and interpretation of data and critically revised the manuscript. DJ Steffen and JA Schmitz contributed to acquisition, analysis, and interpretation of data, and critically revised the manuscript. B Charleston and JN Henningson contributed to analysis of data and critically revised the manuscript. CL Kelling contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Bovine turbinate cells, National Veterinary Services Laboratory, U.S. Department of Agriculture, Ames, IA.
b.
Dulbecco modified eagle medium, Invitrogen, Carlsbad, CA.
c.
Equine serum, Hyclone, Logan, UT.
d.
Nebulizer, DeVilbiss Health Care, Somerset, PA.
e.
Kendall Monoject EDTA blood collection tubes, Tyco Healthcare Group, Mansfield, MA.
f.
Rompun (xylazine), Bayer HealthCare, Shawnee Mission, KS.
g.
Ketaset (ketamine), Zoetis, Kalamazoo, MI.
h.
Beuthanasia-D, Merck Animal Health, Madison, NJ.
i.
Histopaque-1119 (sodium diatrizoate), Sigma-Aldrich, St. Louis, MO.
j.
Histopaque-1077 (sodium diatrizoate), Sigma-Aldrich, St. Louis, MO.
k.
Tetko, Briarcliff Manor, NY.
l.
FACS caliber flow cytometer, BD Biosciences, San Jose, CA.
m.
CellQuest software, BD Biosciences, San Jose, CA.
n.
Blasticidin S HCl, Invitrogen, Carlsbad, CA.
o.
CAT ELISA assay kit, Roche Applied Science, Indianapolis, IN.
p.
Recombinant human interferon-αA/D, Biosource PHC 4045; Invitrogen, Carlsbad, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Nebraska Agricultural Research Division (grant NEB 39-152) from funds provided through Animal Health, U.S. Department of Agriculture and in part by the Flow Cytometry core facility funded by NIGMS grant P30 GM103509. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
