Abstract
Plasmacytomas are localized neoplastic proliferations of monoclonal plasma cells. When multifocal, the process is referred to as multiple myeloma. These lesions exhibit a pattern of antigen expression and cytomorphology that usually leads to a ready diagnosis. However, potentially troublesome variations in immunophenotype occur. We describe a case of a plasmacytoma from a patient who presented with sudden onset of pain and a lytic lesion of the left proximal humerus. Hematoxylin and eosin-stained sections showed a lymphoproliferative lesion composed of large lymphoid cells, some with plasmacytoid and immunoblastic features. The lesion also showed significant mitotic activity. Immunohistochemical staining was positive for CD45 (LCA), CD56 (N-CAM), CD43 (MT1), and cytokeratin CAM5.2. There was also clonal staining for λ light chains. In addition, flow cytometric analysis showed positivity for myeloid markers such as CD13, CD33, CD38, and CD138. Significant negative markers include CD20 (L26), CD45RO (UCHL-1), and CD79α. The unusual phenotypic features of this plasmacytoma illustrate potential diagnostic pitfalls. It is important to fully study such lesions to correctly classify them, because this has significant impact on prognosis and management.
We present a case of a plasmacytoma with an atypical immunophenotype and histologic features. The biopsy material for this report came from a patient with sudden onset of pain from a lytic lesion in the left proximal humerus. Flow cytometric and paraffin immunoperoxidase studies were performed on the tissue.
Hematoxylin and eosin-stained sections showed a lymphoproliferative lesion composed of large lymphoid cells, some exhibiting plasmacytoid and immunoblastic features (Figure 1a). There were many mitoses. Immunohistochemical staining was positive for CD45 (LCA; Figure 1b), CD43 (MT1; Figure 1c), CD56 (N-CAM; Figure 1g), and cytokeratin CAM5.2 (Figure 1h). There was also clonal staining for λ light chains (Figure 1f). In addition, flow cytometry showed positivity for CD13, CD33, CD38, and CD138. Significant negative markers include CD20 (L26), CD45RO (UCHL-1; Figure 1d), and CD79α (Figure 1e).
Neoplastic plasma cells, like normal plasma cells, usually lack the surface B-cell antigen. The absence of staining for leukocyte common antigen (LCA) and B-cell surface antigens CD19 and CD20 is highly characteristic (Strickler et al. 1988; Wotherspoon et al. 1989). Conversely, the B-cell antigen CD79α, normally expressed in plasma cells, is demonstrable in a significant proportion of cases. Staining with CD38 is variable. CD56 (N-CAM) is a natural killer cell-associated antigen commonly expressed in multiple myeloma and typically absent in monoclonal gammopathy of undetermined significance, thus being a reliable discriminator between myeloma and MGUS (Van Camp et al. 1990). Moreover, low CD56 expression predicts high levels of marrow and blood tumor involvement, both poor prognostic factors (Davies et al. 1997).
Staining with T-cell markers is unusual but reports of plasmacytomas staining with CD43 and CD45RO do exist (Petruch et al. 1992). In a study using a standard antibody screening panel that includes L26 (CD20), Leu22 (CD43), and UCHL-1 (CD45RO), 17 cases with a “CD43 only” phenotype were identified, with two of these cases being plasmacytomas (Segal et al. 1992). Cytokeratin immunoreactivity in plasmacytomas is generally considered to be rare (Sewell et al. 1986). However, in a study looking at 14 plasma cell tumors, including solitary plasmacytomas and multiple myelomas, five of the cases showed reactivity for cytokeratin, thus confirming that anti-cytokeratin reactivity by plasma cell tumors is more common than was originally believed and may represent an important diagnostic pitfall (Wotherspoon et al. 1989).
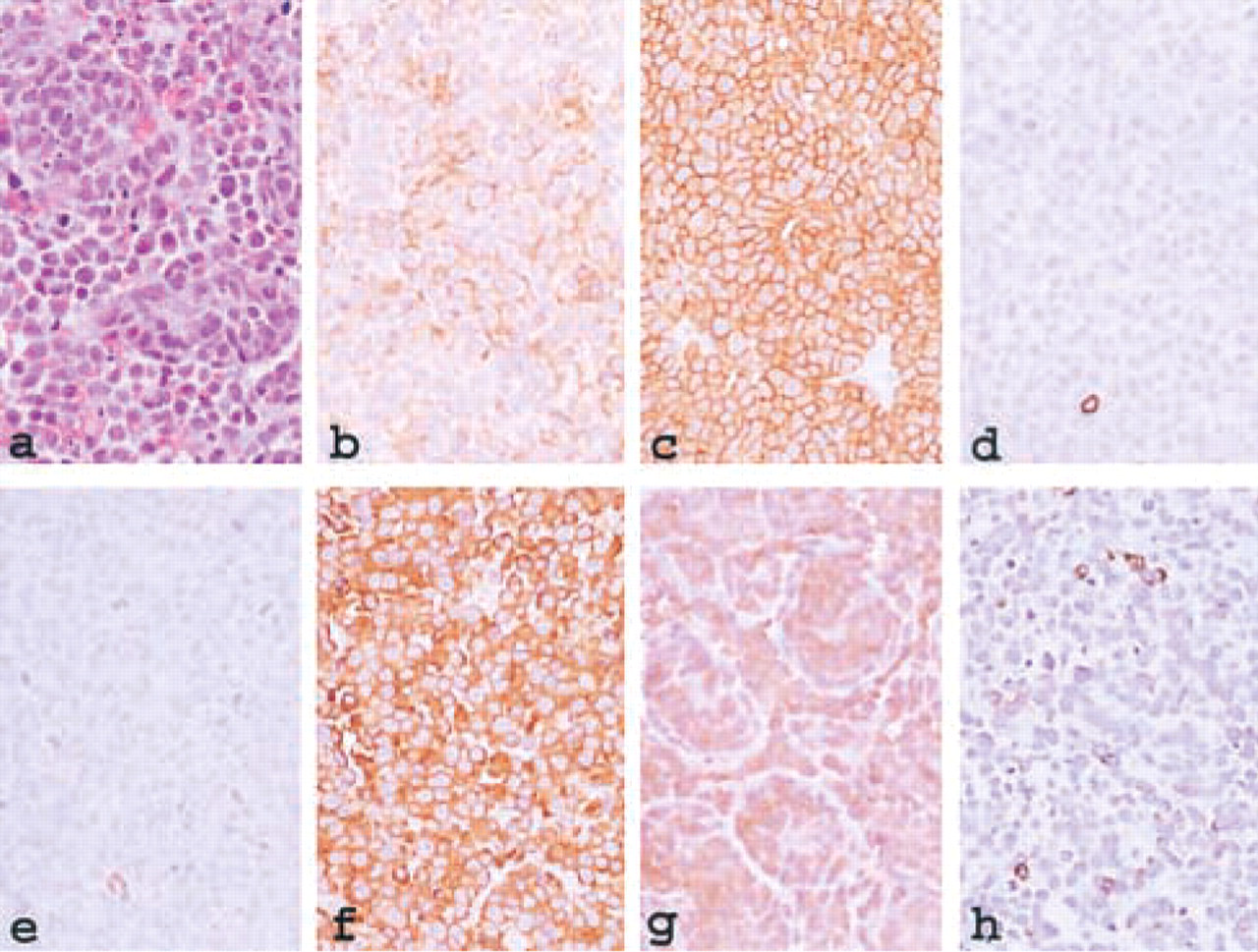
Photomicrograph of the plasmocytoma presented, stained with (
Myeloma may occasionally present with aberrant expression of myelomonocytic antigens such as CD11b, CD13, CD14, CD15, and CD33 (Grogan et al. 1989; Epstein et al. 1990). These antigens are preferentially expressed in two types of neoplasms, i.e., indifferentiated ALL and mature B-cell lymphoproliferative disorders (Nakase et al. 1996). The expression of these antigens may be related to a poor prognosis (Ruiz–Arguelles and San Miguel 1994).
The unusual features of the plasmacytoma we present illustrate potential diagnostic pitfalls. Although plasmacytomas with aberrant expression of myeloid markers, T-cell markers, and cytokeratin have been described, our case may represent a plasmacytoma with a unique immunophenotype. The significance of such an unusual lesion is not fully understood, and further classification of this lesion and other similar lesions may be useful in determination of patient prognosis and management.
