Abstract
Compound management faces the daily challenge of providing high-quality samples to drug discovery. The advent of new screening technologies has seen demand for liquid samples move toward nanoliter ranges, dispensed by contactless acoustic droplet ejection. Within AstraZeneca, a totally integrated assay-ready plate production platform has been created to fully exploit the advantages of this technology. This enables compound management to efficiently deliver large throughputs demanded by high-throughput screening while maintaining regular delivery of smaller numbers of compounds in varying plate formats for cellular or biochemical concentration-response curves in support of hit and lead optimization (structure-activity relationship screening). The automation solution, CODA, has the capability to deliver compounds on demand for single- and multiple-concentration ranges, in batch sizes ranging from 1 sample to 2 million samples, integrating seamlessly into local compound and test management systems. The software handles compound orders intelligently, grouping test requests together dependent on output plate type and serial dilution ranges so that source compound vessels are shared among numerous tests, ensuring conservation of sample, reduced labware and costs, and efficiency of work cell logistics. We describe the development of CODA to address the customer demand, challenges experienced, learning made, and subsequent enhancements.
Introduction
Compound management (CM) has the task of supporting numerous projects in various stages of the drug discovery process, demanding a range of technology, capabilities, and delivery capacities. Reviewing the demand profile of projects across a typical drug discovery site, it is clear that there are significant differences in demand and delivery time for compounds. A project in lead optimization will require small numbers of compounds to be tested very quickly as the data are required for subsequent rounds of chemistry. 1 Within any drug discovery organization, there may be many projects in this phase, and therefore CM must deliver many small sets of compound plates, typically in a dose-response format, very quickly to meet demand. For projects earlier in the pipeline, chemistry plans may be more predictable, and synthetic strategies may not require biological data to trigger work as they may be based on structure-activity relationships. In this environment, compound testing may be relatively high, but turnaround times may be longer (1000s of compounds per month). For high-throughput screening (HTS), compounds are required in very high numbers but generally only in single-dose formats; again, these may be generated only intermittently. It is this variation that provided a challenge to design a solution to address all the customer requirements in the different science groups while maintaining flexibility to cope with future changes. 2 AstraZeneca’s CM group at Alderley Park was tasked to deliver a system that would address the aforementioned issues.
Existing systems and processes were fragmented as laboratories invested in their own liquid-handling capabilities and satellite storage facilities, often with inappropriate environmental conditions to protect sample integrity. 3 The different research areas each had their own bespoke processes to achieve the same goal of creating assay-ready plates (ARPs) for their specific target areas. Hence, there was an opportunity to centralize and standardize such processes into one platform within CM. This platform would have to provide ARPs on demand for just-in-time delivery to the equivalent or better quality of the existing systems. This centralization would negate the need for research area scientists to undertake ARP preparation, releasing resources back to perform bioscience-based activities. Future capital spending on equipment procurement and maintenance would be reduced with one system to maintain. In addition, the need for expensive satellite stores would be minimized and replaced with environmentally monitored tactical solutions. The concept of multiple research areas accessing a centralized system created an opportunity to conserve liquid sample stocks by using one sample source to deliver to several projects simultaneously. This was in contrast to revisiting the same sample source numerous times, thus creating delays in accessing the sample, excess waste, and possible contamination. The centralization of the ARP facility was defined to address all customer groups that generated ARPs. These groups included DMPK (drug metabolism and pharmacokinetics) and safety screening in addition to early hit and lead optimization. Centralization complemented local delivery for cell-based and separation-based assays. These standalone cell platforms delivered sample (compound) acoustically to the cell media without the need for aqueous dilution or media removal. Thus, the investment to centralize ARP provided AstraZeneca with the ability to provide compound to assays using the same technology and process across all phases, from HTS to lead optimization and to safety screening, while the ability to provide samples to external collaborators was a key design factor.
Acoustic droplet ejection (ADE) technology 4 provided the means to dispense very low volumes of liquid to previously unattainable levels of precision without the requirement for tip washing, eradicating any possibility of cross-contamination that exists with conventional contact liquid dispensers incorporating tips. The ability to dispense such low volumes also provided an opportunity to exploit delivery to clinical research organizations using their screening capacity without compromising intellectual property as structural information could not be gleaned from such small volumes of liquids. The use of ADE underpinned the AstraZeneca strategy for the centralization and delivery of ARPs across the drug discovery process for internal and external customer groups.
Here we outline the process of defining the demand, scoping the required hardware, and building a software package to support the hardware solution, as well as details of delivery and validation of a functioning capability.
Development
During the system development phase, a business engineering technology assessment (BETA study) was undertaken to gather clear customer requirements of the new system incorporating business drivers, benefits, risks, and understanding. Due to the aforementioned requirement to centralize the production of ARPs, it was essential to define the process flows (demand profiles) required by the different customer groups. Four main demand profiles were identified.
For primary screens (collection orders—demand profile 1), the system had to deliver ARPs to HTS at the same rate at which the plates could be screened. In the case of the Alderley Park customer base, this equated to the support of up to four assays running concurrently at a rate of 100 000 samples per day. The samples would be screened at a single concentration with separate control plates produced. Such control plates are defined as a microplate consisting entirely of predefined standards or controls; these are known as whole-plate controls. These predefined controls are listed within the ARP production system as Total [max], Null [min], and Reference. The screening collection is held in 1536-well working plates sourced from tubes stored in the primary liquid store (PLS). 5,6 This collection is currently remade periodically, factored by depletion rates and sample integrity.
In addition, HTS has the demand to screen bespoke subsets of the screening collection based on hits from stratified primary screens and near chemical neighbors. These orders are known as single-concentration orders (demand profile 2a), typically ranging in size from 5000 to 150 000 samples sourced directly from the PLS with the intention of such collections being screened only once. Controls are handled in the same way as with the collection orders.
Following all primary and single-concentration screens, HTS is tasked to perform secondary screening on active and chemically interesting samples selected by the project chemists—in this instance, these are known as HTS multiple-concentration orders (demand profile 2b). For secondary screens, the system had to deliver ARPs to HTS at the same rate at which the plates could be screened. In the case of the Alderley Park customer base, this equates to the support of up to four assays running concurrently at a rate of up to 5000 samples per week per assay, typically producing curves between 7 and 12 different concentration points. To generate the desired ARP, the requested samples are picked from the PLS and plated into the first quadrant of an ADE-compatible 384-well plate using an Agilent 96 fixed-tip VPrep (Agilent, Santa Clara, CA). To optimize efficiency, a maximum of 84 samples are picked per 384-well plate. The samples are then diluted dependent on the target concentrations for the assay using 100% DMSO in the remaining three quadrants of the plate, as shown in Figure 1 .
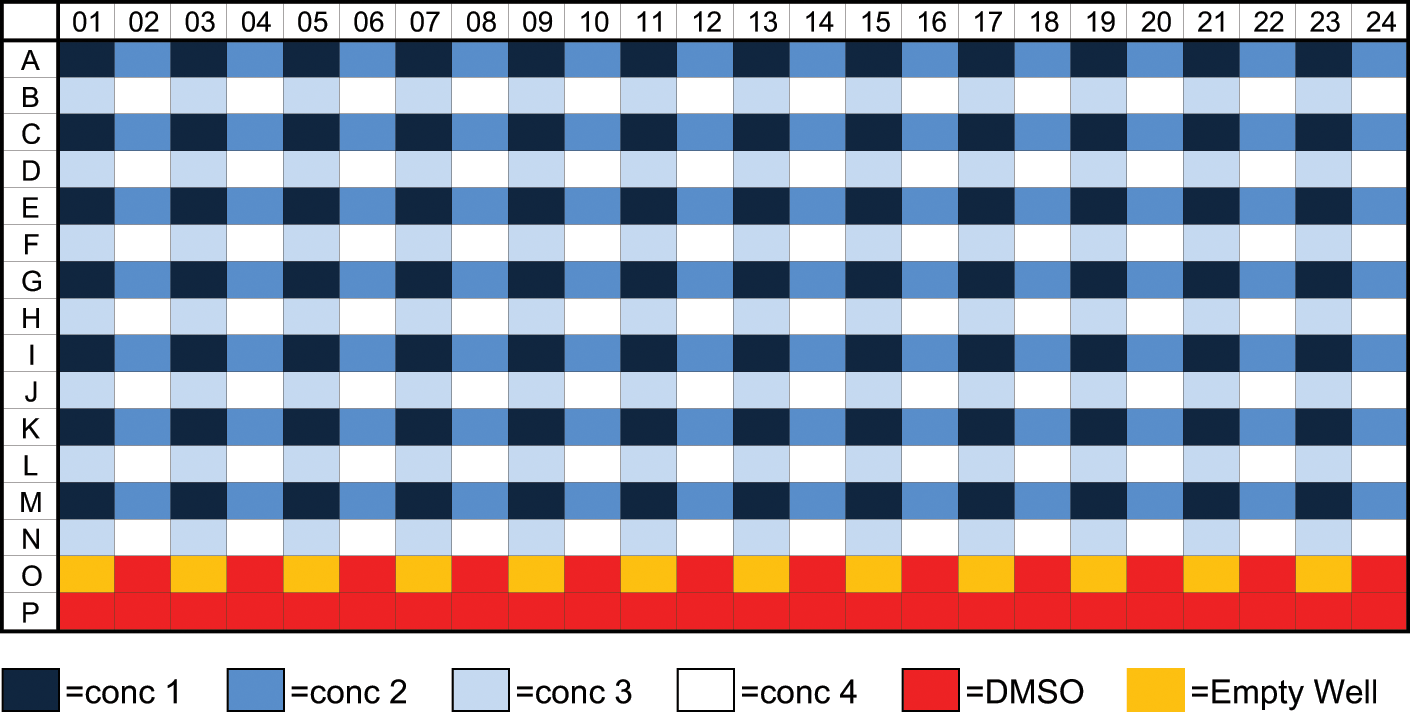
A quadrant diluted intermediate plate with the last two rows containing DMSO for later use as source for acoustic back filling.
To produce this plate, the 84 samples are dispensed to quadrant 1 (e.g., A01, A03, A05, C01, C03, C05, etc.). Then approximately half of the required final volume of DMSO is added to quadrants 2, 3, and 4. A volume of sample from Q1 is dispensed into Q2, and the remaining volume of DMSO is placed on top to create the final volume and concentration in Q2. Sample from Q2 is then transferred to Q3 as above, then Q3 to Q4, producing a plate of 84 samples at four progressively lower concentrations. This process creates 36 wells in rows O and P with 100% DMSO to serve as source wells for acoustic back filling of <50 nL DMSO to normalize the ARP. Back-fill requirements of >50 nL need to be performed by an integrated nanoliter dispensing unit. Such requests can accommodate whole-plate controls as described for HTS primary screens or collection orders and/or onboard controls. These onboard controls consist of control samples that are dispensed into the same output plates in predefined wells at predefined concentrations and volumes specified by HTS or research area that contain the serially diluted samples. Again, these predefined controls are to be listed within the system as Total (max), Null (min), and Reference.
In addition to supporting HTS requirements, the system is tasked with maintaining the regular delivery of smaller numbers of compounds in varying plate formats for either cellular or biochemical screens. This can equate to up to 140 assays per week at an average throughput of 120 samples per day per assay. Requests for such screens are known as multiple-concentration orders (demand profile 3) and follow the same process flow as HTS multiple-concentration orders but do not incorporate whole-plate controls, instead using onboard controls only.
One of the outcomes of this study was a stipulation for full flexibility in the choice of plate map layout and alignment with existing customer sample tracking and data-handling software.
The BETA study also highlighted that to meet the needs of screening projects requiring simultaneous access to the same samples for different tests, it was necessary to design a solution whereby multiple tests could be supplied from multiple sources with a large number of the sources being shared across the tests.
The project was complex with multiple customers and diverse requirements. The project included organizational change, significant software development, and integration into existing systems, new technology, infrastructure, and hardware changes. After the BETA study had collated the risks, customer requirements, processes, and technologies required and had identified and evaluated the options open to AstraZeneca, the project moved into an equipment and vendor selection process. It was believed, after all the information had been reviewed, that it would take more than one company to deliver what was and is a complex solution.
AstraZeneca produced a User Requirement Specification document to provide potential vendors with an initial specification to work to in producing a proposal for the desired system. A key purpose of the document was to define requirements for each of the demand profiles, allowing vendors to submit proposals that were designed to meet the requirements of one, two, three, or all four demand profiles (collection orders, single-concentration orders, HTS multiple-concentration orders, and multiple-concentration orders).
A number of vendors proposed solutions, and The Automation Partnership (TAP, Royston, UK) was successful in this process. The TAP solution, known as CODA (Compounds on Demand for Assay), involved integrating third-party equipment into a TAP platform and included an “off-the-shelf” operating system known as Concerto. In addition, a bespoke collation tool (The Collator) was designed, based on the TAP Concerto software, which reduced the demand for source sample picks placed on the PLS, maximizing the life span of the liquid stocks. This software collates multiple orders for a sample into a single source vessel pick on the PLS, and it then uses the cherry-picking functionality of the acoustic dispenser to dose that single sample pick back out to the multiple sample orders. This system had to be integrated into the existing processes operated by CM—notably, the in-house ordering system, COSMOS, and the automation system from RTS Life Science (RTS, Manchester, UK). The integrated third-party hardware was provided by Labcyte (Sunnyvale, CA), Agilent, and Beckman Coulter (Brea, CA). All hardware is driven using Overlord 2 software from Process Analysis Automation (PAA, Farnborough, UK) linked into the TAP Concerto operating system.
The successful proposal by TAP was based on its bespoke hardware and off-the-shelf sample order and process management software package (Concerto), which was already in use within the pharmaceutical industry. Using this software product allowed development costs to be kept to a minimum. Likewise, integrating the Overlord 2 software with the reliability of TAP hardware (previous AstraZeneca collaborations) and the postinstallation support model presented by TAP resulted in a preferred partner for AstraZeneca on this project.
There were a number of reasons why other vendors were ruled out: They were unable to provide the sample conservation demonstrated by the TAP collation software, previous experiences of kit supply were not robust on delivery, software required integration by third parties, software support was deemed inadequate, and delivery schedules were not in line with AstraZeneca requirements.
Hardware and Process Control
The proposed solution incorporated two work cells, one for HTS and one for hit and lead optimization, allowing for peaks in demand and cell redundancy for maintenance.
The work cells were designed and built by TAP, which partnered with PAA and Labcyte to deliver a fully automated acoustic dispensing platform. Existing software used to communicate with an AstraZeneca compound batching process and assay data analysis software was incorporated into the project, minimizing changes required to downstream assay data analysis tools.
Each of the two cells (see Figures 2 and 3 ) is made up from three modules: a plate handling module, an Echo feeding module, and a carousel unit.
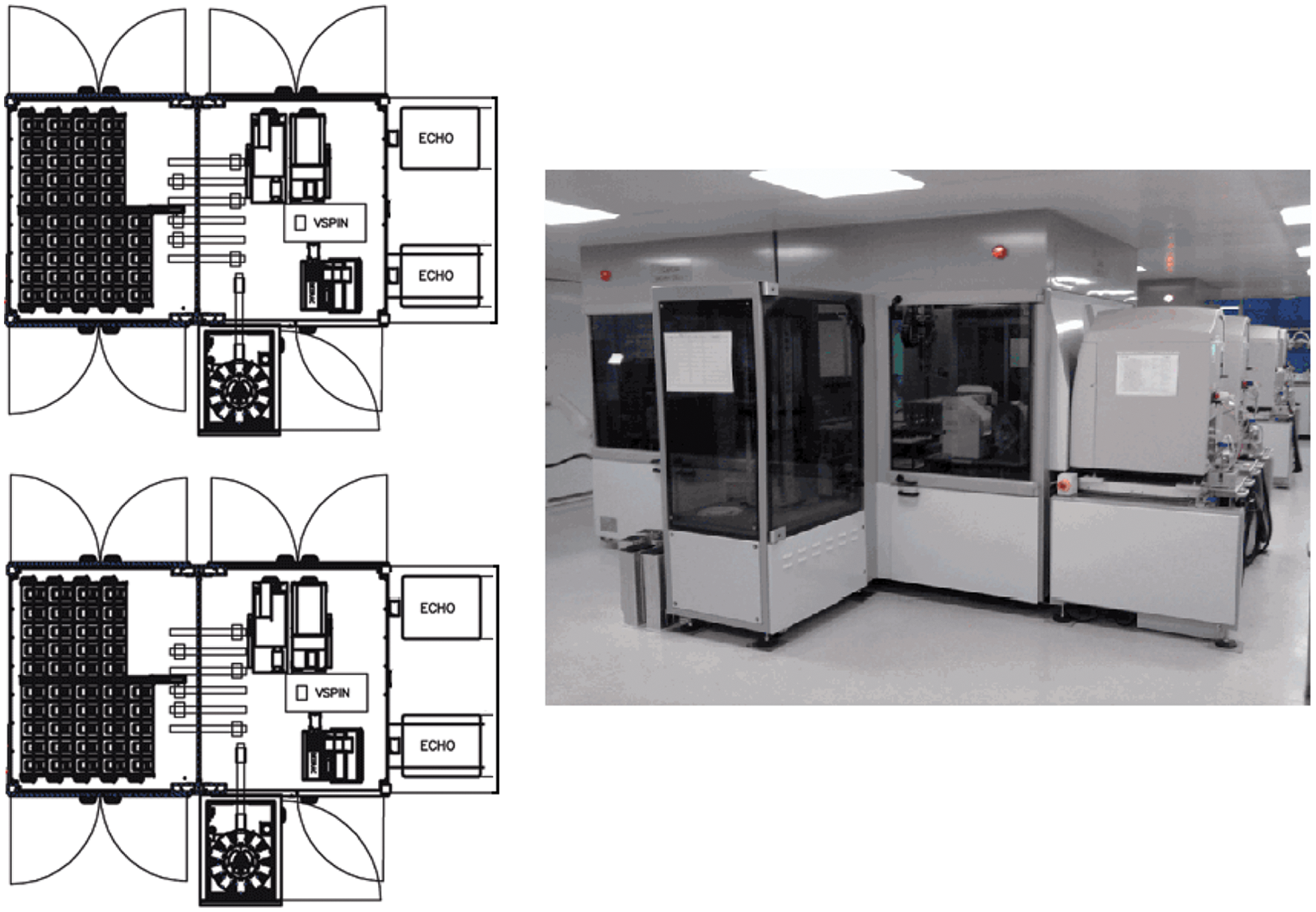
A plan view and a photograph of the two CODA cells at Alderley Park.

Internal view of CODA cell showing Echo feeding module and plate handling module.
The plate handling module consists of 18 shelves designed to take the TAP Platesafe magazines and 2 shelves for temporary and permanent Platesafe lids. A Platesafe is a plate storage magazine sealed with a polypropylene base and lid and designed to contain up to 30 standard footprint microplates. The Platesafes are accessed by the plate handling module robot, which is an anorad linear arm robot designed to pick and place microplates, read barcodes, and lid and de-lid Platesafes.
The Echo feeding module, which is linked to the plate handling module by six shuttle conveyors, houses all third-party equipment, comprising an Agilent PlateLoc sealer, an Agilent VSpin centrifuge with an Access 2 robot feeding the VSpin, a Labcyte LX Deerac nanoliter dispensing unit with antistatic capability, and a Beckman Coulter labeler, and it can access up to three Labcyte Echo 555 ADE liquid-handling units (two initially supplied). The Echo feeding module robot is a second anorad linear arm robot that transfers both source and destination microplates to all third-party equipment, shuttles, and a carousel unit.
The carousel unit is connected to the Echo feeding module via a shuttle conveyor and has a capacity for 360-compound and/or control source microplates.
The cells were delivered with appropriate seals and ducting to enable a temperature-controlled and dehumidified atmosphere to be provided by air-handling units. This minimizes water ingress and evaporation for samples handled in the cells, which has been highlighted as an issue for repeated use of compound collections. 7,8
Correlation Data
To establish the accuracy and robustness of the Echo compared with conventional contact dispensing, the project team ran numerous correlation trials between CM and the research areas.
The first of these trials involved a centralized biochemical screening team (BST). BST had been tactically operating an Echo prior to the CODA centralization of acoustic dispensing in AstraZeneca. To ascertain the accuracy and reliability of this new equipment, BST ran trials against the predominant conventional contact liquid handler used in BST at the time, RSP (robotic sample processor).
BST found that overall, the data quality significantly improved switching to an Echo from RSP compound handling.
9
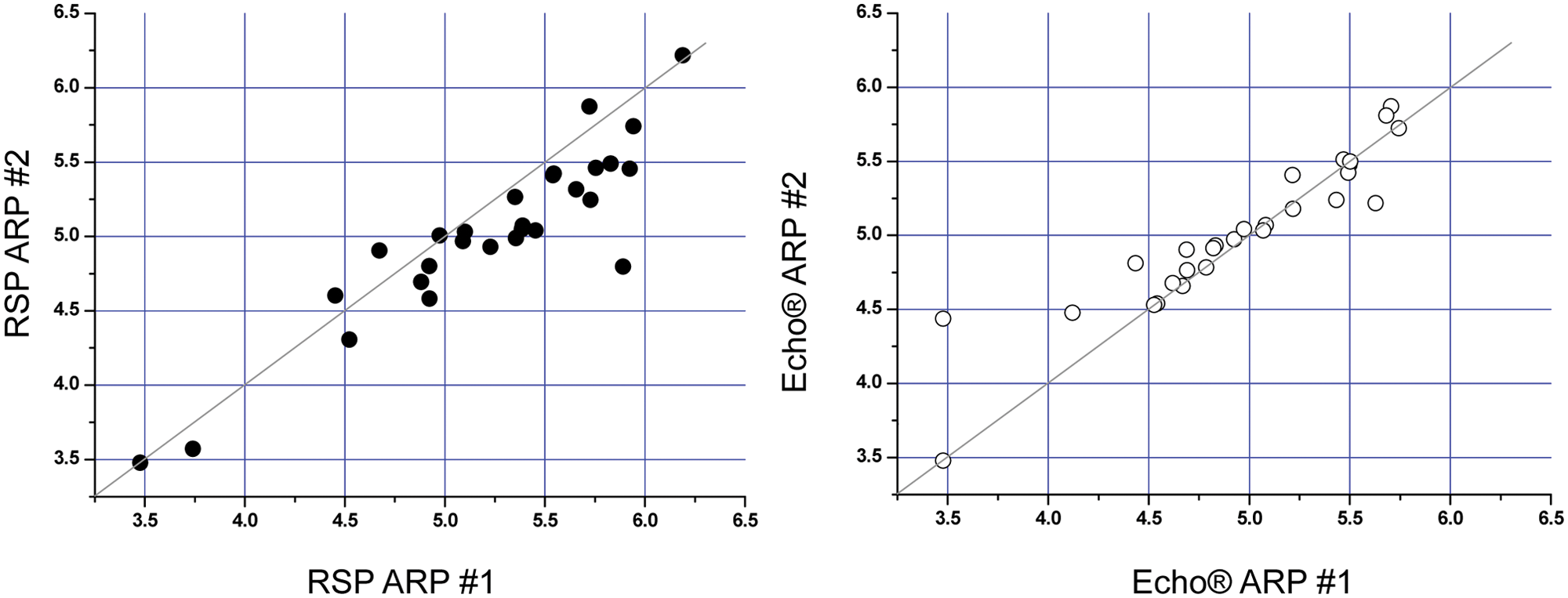
Furthermore, the correlation between enzyme and cell assays clearly demonstrates the data cluster more closely to the 1:1 line (see
Fig. 4
). The graph shows that the Echo presents a tighter correlation than a conventional RSP. One would generally expect to see a higher potency in the enzymatic assay compared with the cell assay (i.e., higher pIC50 value on the
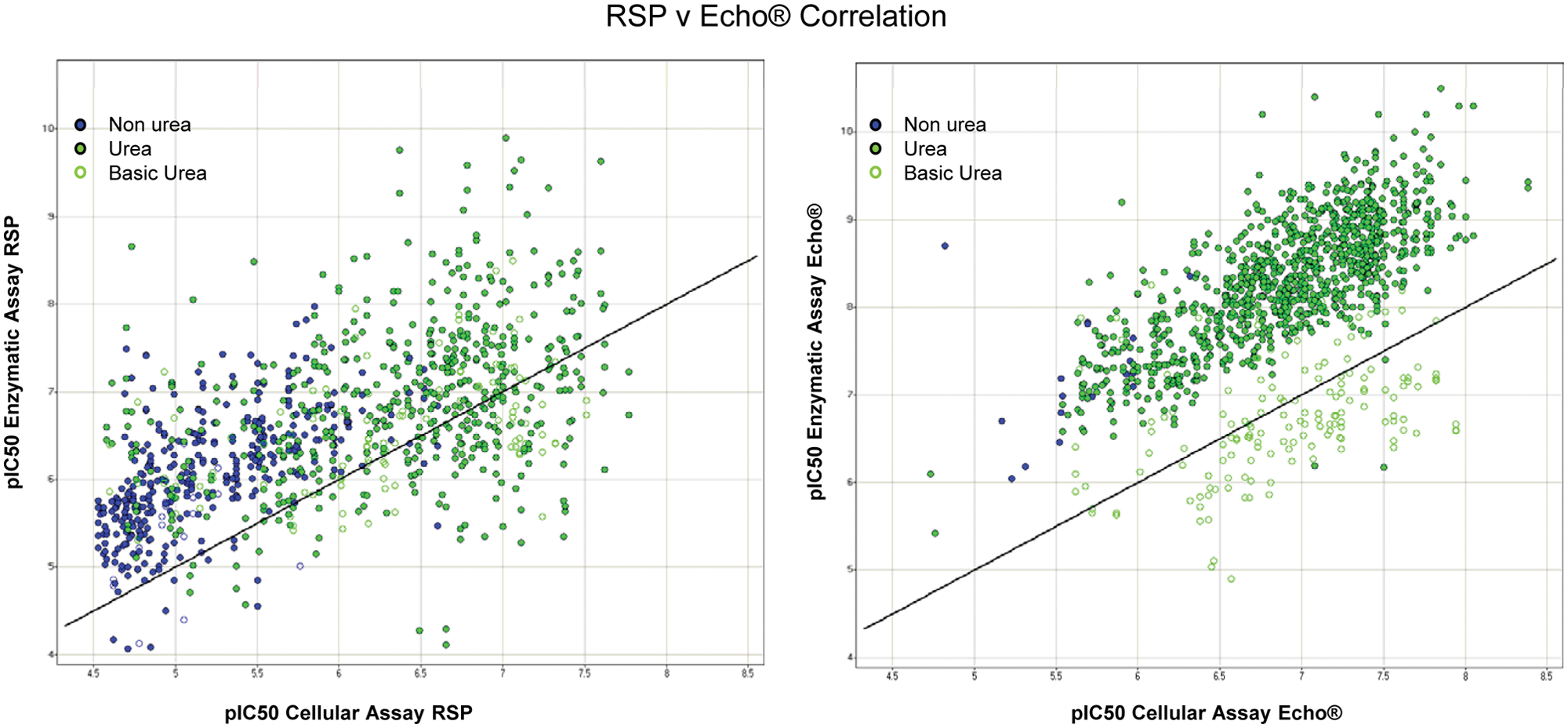
Correlation between RSP (robotic sample processor) and Echo dispensing in enzymatic and cellular assays.
CM ran trials using Molecular Devices IonWorks data on CHO-Kv1.3 ion channel currents to investigate whether the Echo- versus RSP-distributed ARP comparison was favorable. Twenty-eight compounds were chosen ranging from no/weak activity (pIC50 < 4) to strong inhibition (pIC50 > 6) of the Kv1.3 ion channel assay. IonWorks was used to measure CHO-Kv1.3 ion channel cell line current inhibition and then used to compare the distribution accuracy for RSP- or Echo-distributed ARPs for 10-point ½-log dose responses.
Figure 5 shows good alignment between mean pIC50 values from compound dose response distributed via Echo and RSP. For the RSP, there is a slight increase in apparent potency; this is anticipated due to built-in overdispense.
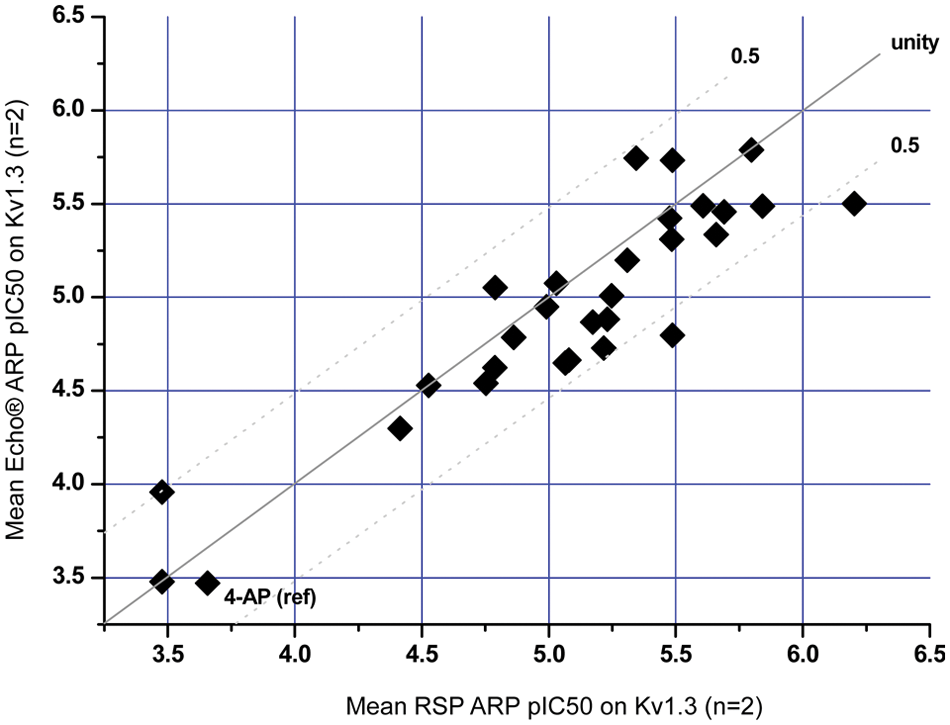
IonWorks correlation data: RSP (robotic sample processor) vs Echo, 28 compounds distributed in 10-point ½-log dose response. ARP, assay-ready plate.
Run-to-run variability is shown in Figure 6 . There was minimal variability between two separate ARPs assayed on separate IonWorks experiments/runs. The Echo showed slightly less variability than the RSP.
Minimal variability is shown between two separate assay-ready plates (ARPs) assayed on separate IonWorks experiments. Echo shows less variability than the RSP (robotic sample processor).
To ensure key partners that the CODA system was not only dispensing accurate amounts of compound but also placing the correct compound in the correct wells of the target microplate, liquid chromatography/mass spectrometry (LC/MS) analysis was conducted on trial CODA-dispensed target plates. Known compounds were dispensed into specific target wells. LC/MS analysis confirmed that the correct compounds were dispensed into the intended target wells.
Changes Prior to Implementation
Delivery of the project was more than the implementation of acoustic technology. For the throughputs deemed necessary to make CODA a success, there had to be an increase in throughput from the PLS to ensure it could cope with existing demand and the new demand expected from CODA. 11 In addition, new functionality was required to ensure downstream processes could progress efficiently. To facilitate this, a number of changes to existing processes were required.
Primary Liquid Store Caching
Previously, the PLS would progress one order at a time. Certain storage trays could potentially be picked and placed for each consecutive order, which was logistically uneconomical. To alleviate this problem, caching was introduced to enhance the picking efficiency in collaboration with RTS.
Caching was an add-on to the single-order approach. As previously, the PLS will only pick for one order, but when a tray of source tubes arrives into a picking station, it will also pick tubes required for subsequent high-priority orders (priority 1–3) within the system into a cache of empty racks in unused slots inside each picking station. When the next order starts, the picking station will pick from the cache racks into the rack at slot 1 while it waits for the first tray to arrive.
In total, there is room for 1536 tubes to be picked into cache locations across our four picking station system. When tubes have been used in an order and are being returned back to the store area, if any are required for a later order, these will be moved directly into the cache rather than a high-density source tray. To further improve the speed and performance, the nonassembly picking stations’ idle time will be used to populate the cache while assembly/compression is performed downstream.
Note that tubes are not cached for specific orders; they are allocated to the cache. The tubes are moved from the storage tray to a rack in the picking station, but they are not set to “in use.” This means that the mechanism of reserving tubes for orders treats these tubes just the same as tubes in storage trays. If a tube is cached because it appears in a priority 3 order that has not yet started, but then a priority 2 order is issued, this order can reserve and pick the tube.
Online Quadrant Dilutions
As previously mentioned, the series of source concentrations required to produce the dilution curves for HTS and small-screen multiple-concentration orders are created using a quadrant dilution within the source plate. The source plates were previously created manually using an offline Agilent VPrep with manual data manipulation. Generating the plates in this way was inefficient with the potential for data errors. Any errors at this point were time-consuming with mistakes rectified via IT support. Subsequently, these issues led to the implementation of a processing route that used the online VPrep and automatically uploaded the source plate data to CTM, the CM database.
New Tip Wash Protocol
Having made the decision to incorporate the compound dilution as part of the automated process, it was critical to ensure that this augmentation did not limit the rate of plate production. A total of four wash cycles are necessary to create each quadrant diluted plate. On average, the PLS is capable of picking the tubes required for one intermediate plate every 10 min, dictating the dilution step could not exceed this time. This led to the challenge of having to reduce the duration of the tip washing protocol, which took approximately 5 min per cycle, without compromising the integrity of the product. The solution was derived by designing and analyzing several wash protocols, each one consisting of different features such as tip soaking, varying velocities of piston movements, flow rates, and quantities of DMSO. To measure the effectiveness of each approach, a volume of fluorescein dye was aspirated and dispensed before each wash protocol was executed. A volume of DMSO was then aspirated and dispensed into a microplate and analyzed for remnants of fluorescein using a Tecan SpectraFlour Plus plate reader (Tecan, Männedorf, Switzerland). The outcome of this testing indicated that a fast and aggressive movement of the pistons, coupled with a high flow rate of DMSO, was the most significant factor in effective tip washing. Focusing on these two basic principles enabled the development of a new protocol that was able to deliver a wash cycle that was 80% faster than the existing protocol, as well as being more effective. To prevent a significant rise in costs due to the increased consumption of DMSO required to maintain a high flow rate, a recycled flow system was also implemented, based on the existing model used in AstraZeneca CM Mölndal. 7
Postimplementation Enhancements
The scale of the initial project to satisfy the demands of both high-throughput and structure-activity relationship screening involved certain budget constraints, which meant that the project could not deliver every eventuality. The emphasis was therefore placed on the essential elements that were needed to meet the project goals. Certain improvements and refinements to the system could be assessed, analyzed, and implemented only after the system had been in operation and additional monies were made available. Therefore, after the implementation stage of integrating CODA into the working practices of CM and its subsequent success, there was immediate evidence of the high demands needed to be met, and in the first year, CODA produced over 13 million output wells (see Demand Profile section for more detail). The high demand for the new service during the initial year provided a large data set against which CM could evaluate how the system had performed. A number of enhancement opportunities were identified based on these findings and drivers from the business.
First, there was a drive to conserve expensive control samples used within all assays, which led to a fundamental change in control plate production calculation. Originally, the system was configured on achieving the maximum throughput possible for the CODA cells. Therefore, the formula was designed to calculate the fill volumes for the source control wells based on the maximum transfer volume. However, this caused issues with expensive reagents being used in assays as this process meant more filled wells with less volume. Since each well incurs a dead volume, each extra filled well increases the volume of sample wasted.
Changing the control well calculation to fill the control wells to maximum capacity meant a dramatic reduction in the number of wells required and so reduced the dead volume wasted per control source plate.
Second, the opportunity to process multiple orders simultaneously was identified as a major enhancement. Initially, this had been limited by having only one Process Analyser for Roles and Tasks (PART) server, and this was starting to affect the exploitation of CODA to a wider research community since CM needed to increase the volume of orders processed in the system at any one time. The PART server analyzes the tasks to be completed and schedules how the run will proceed on the CODA cell. Since PART is primarily a serial process, having two instances more efficiently uses resources that would otherwise be idle. Consequently, this improves responsiveness by allowing planning of more than one order at a time.
Third, the work cells were designed to take three Echos per system. The cost of the Echo limited this additional capacity; however, once proven, the addition of an extra Echo on each cell was seen as a priority to increase throughputs, flexibility, and redundancy. This was realized in 2011 when a third Echo per cell was incorporated. The extra Echo per cell provided CM with the opportunity to retire one Echo for offline work or scheduled maintenance without affecting throughputs.
Finally, one of the challenges when with working with multiple suppliers is that the proposed delivery is not always the most effective solution. This can be due to budget and time constraints in development, but it can also be caused by a limited understanding of the mechanics of third-party instruments by the integrator. One example of this inefficiency was found in the generation of the files that control the order of the Echo dispensing in CODA. These files were initially randomly organized, meaning that a truly efficient compound transfer was highly unlikely to occur. We found that simply reorganizing these files in a more structured manner (i.e., dispensing in rows) had a significant impact on plate transfer times. Order processing times were typically reduced by 35%, delivering a time saving of approximately 45 min for every 1000 compounds dispensed in IC50 orders.
Exploitation
The introduction of a centralized ARP platform to CM within AstraZeneca has allowed new and exciting areas of pharmaceutical collaboration to be established. Due to the nature of acoustic dispensing, using nanoliter volumes, collaborative work with leading public and private bodies has become possible. Intellectual property can be maintained as the screening samples provide only single use and structural investigation becomes impossible.
AstraZeneca Alderley Park has run productive collaborations with the Medicines for Malaria Venture and the Institute of Cancer Research and currently is collaboratively partnered with organizations such as Cancer Research Technology and Medical Research Council Technology. The CODA system is also being exploited to support compound delivery into AstraZeneca’s Neglected Tropical Disease Strategy, helping people with diseases such as human African trypanosomiasis, tuberculosis, and leishmaniasis.
Using the ability of the Echo to reformat samples in any way, CM has been able to aid in the progression of customers requesting compound mixtures. This can range from the simplest form, multiple compounds at a single concentration within a single well, to the more complex form, which is a combination of two or more compounds at multiple concentrations within a single well. 12
Previously, these types of requests would consume 200 µL of sample from stock tubes. This same service can now be delivered using just 5 µL of compound, using low dead volume source plates delivering a liquid stock saving of 195 µL, equating to multiple years of supply in the nanoliter dispensing environment. The benefits this has provided include end-user reduced labor intensity; increased quality due to improved robust dispense; reduced compound, plate, and reagent costs; ability to screen at a medium throughput; and randomized placement of combinations across plates.
Demand Profile
The figures below demonstrate the impact of CODA and the savings made on compound usage and automation availability.
Figure 7 shows an increase in the number of output wells generated by CM since the introduction of CODA, saving the research scientists valuable time.
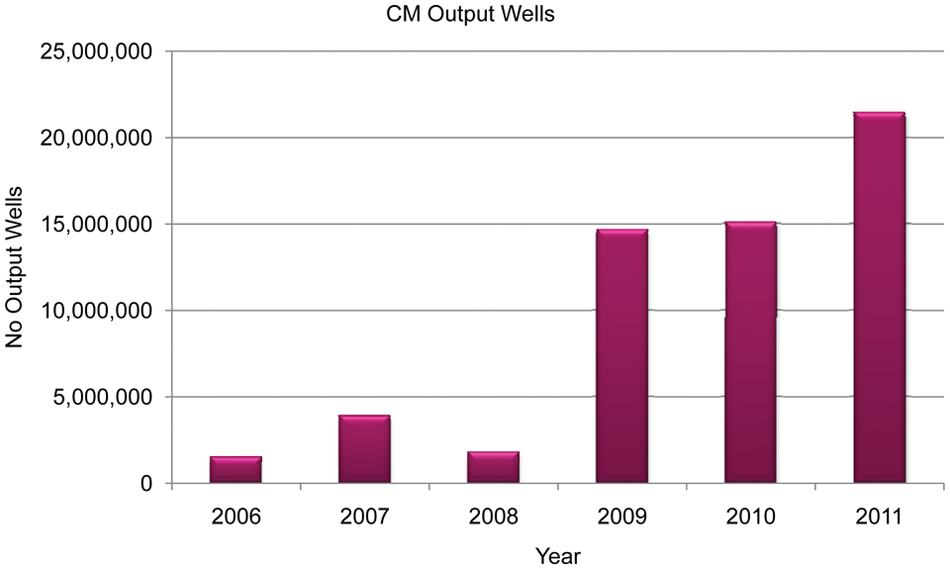
Output wells in plates generated by compound management (CM) since 2006, clearly showing the impact of the introduction of the CODA system in 2009.
Figure 8 demonstrates the actual number of samples demanded per quarter and the total number of samples post collation. These data represent a saving of processing time, logistics within the automation, and physical sample volumes.
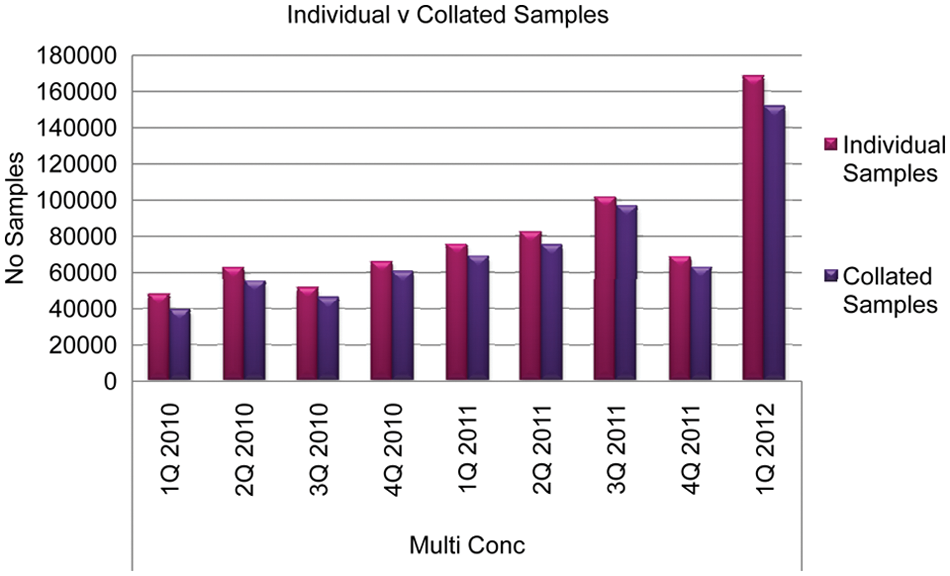
Impact of collating samples, reducing demand on the automation.
Figure 9 represents the total and collated numbers of actual orders for each order type between 2010 and 2012. For collection and multiple-concentration orders, the numbers were reduced from 1067 to 664 (38%) and 3660 to 1739 (53%), respectively. As with the number of samples, these data correspond to a significant saving in terms of physical sample, logistics within the automation, and processing time. Single-concentration orders and HTS multiconcentration orders are unsuitable for collation.
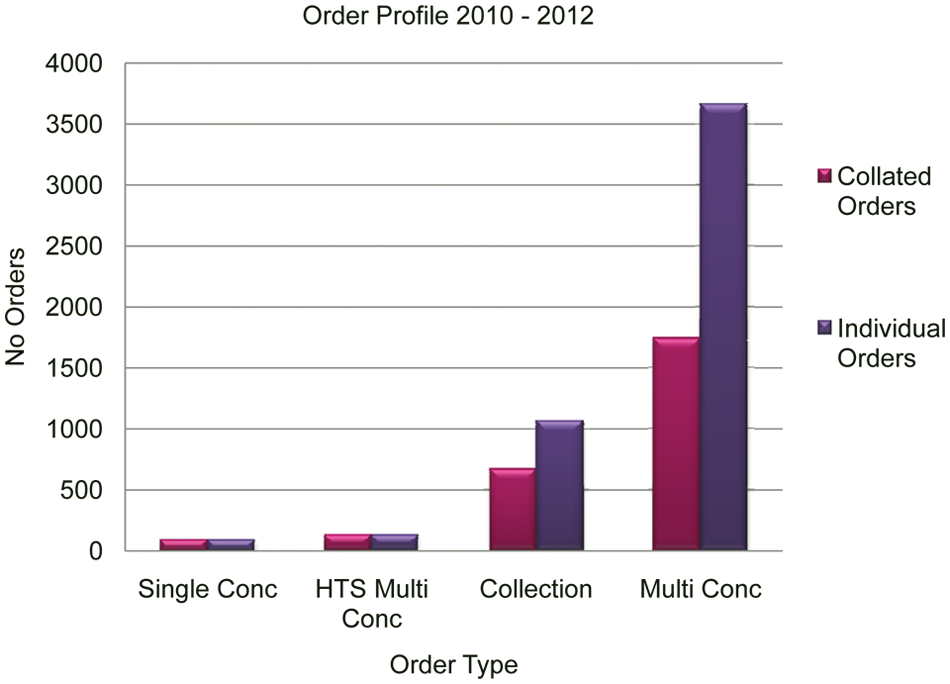
The effect of collating orders, reducing the total number of orders for the automation to process. HTS, high-throughput screening.
Figure 10 demonstrates the distribution of the number of orders across all order types per quarter. It is clear from the data that the multiple-concentration order is the most used by far; collection orders are the second most used order type, with single-concentration and HTS multiple-concentration orders being demanded infrequently. It is worth reiterating at this point that the collection orders generally dwarf the others in terms of output wells, as shown in Figure 11 .
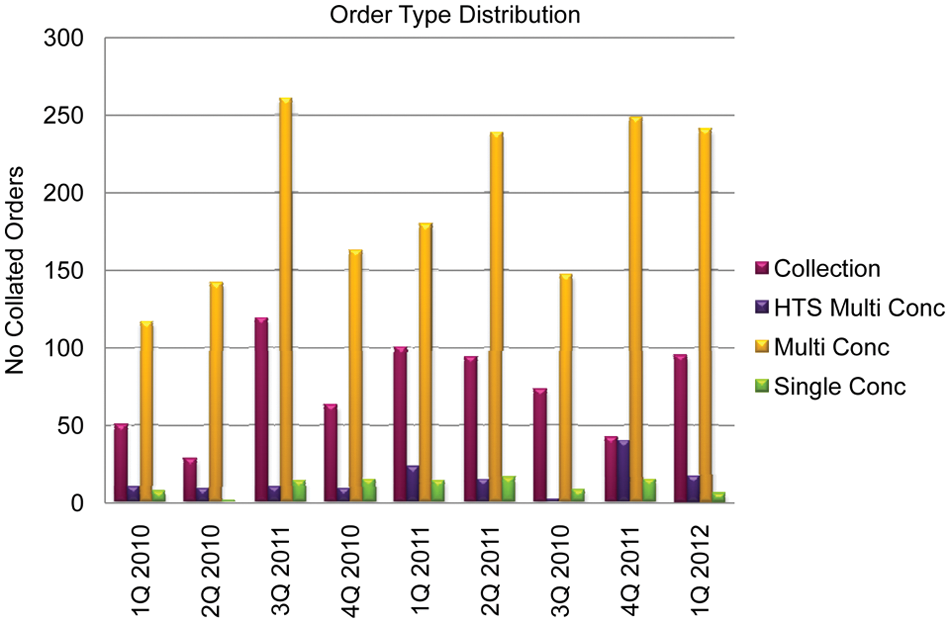
Distribution of order types processed since CODA implementation. HTS, high-throughput screening.
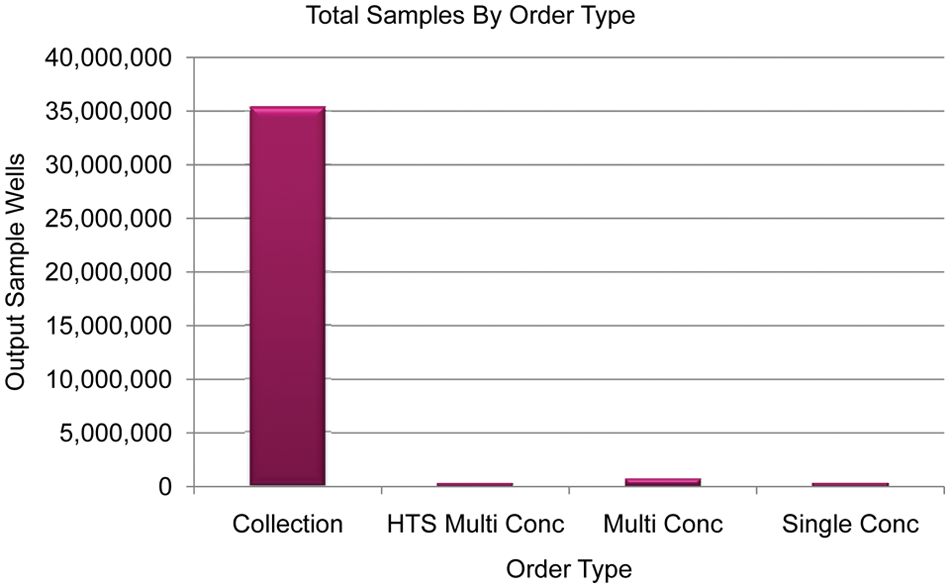
Disparity in output wells per order type. HTS, high-throughput screening.
Customer Experience
The vision to establish an innovative technology within AstraZeneca was initiated in 2002. Over a period of years and local capital investments, the technology demonstrated its value to the drug discovery process, improving data quality. A strategic decision to centralixe and provide improved sample quality to all customers was made in 2007. As previously discussed, the BETA study identified numerous risks and interdependencies for successful delivery. Importantly, this essential review highlighted what success would “look like” and established the need for the project to be owned by the CM organization concurrent with bioscience involvement. The initial proposal highlighted resource saving and improved data quality to all customers, whereas a consequence would be reduced costs associated with consumables, but this was not considered a driver at the time.
The User Requirement Specification was collated from both CM and bioscience input. The latter ensured that the tactical delivery successfully operating within the research areas was included within the document. Importantly, this engagement provided opportunities, experience, and buy-in from key customer groups. The project proactively established key user groups. These groups were a source of valuable information, were consulted throughout the project, and signed off functionality. Evidently, the local ARP satellite process generates bespoke software solutions to link compounds to screening data. As a consequence, project CODA was not only accountable for hardware implementation but also compound visibility in data analysis tools. The project addressed numerous satellite software solutions/process packages, consolidating these into a streamlined package. This ensured full end-to-end testing was undertaken from day 1, mitigating the risk of failure.
The phased delivery of ARPs to the customer base was managed to ensure customer expectation, and importantly, data quality was not compromised. Working alongside established ARP users, the CODA project team prioritized this group and successfully delivered ARPs on day 1. The rollout to HTS groups was to schedule, allowing for the first time HTS and hit follow-up, including lead optimization, to use the same compound dispensing technology.
The project plan identified the team would remain in place postimplementation for a period of time. The project withheld budget to allow for project improvements as a consequence of rollout issues and customer feedback. Importantly, the project worked with the customer user groups postimplementation and created a project improvement plan. This plan included items such as additional plate sealing and reduced “control” sample use. Likewise, for a project of this magnitude, it would be unrealistic for no issues to have surfaced during the early implementation. However, the user groups previously established helped CM when these problems arose; one example was sample carryover, but this was quickly identified and new ways of working implemented.
Finally, from the initial conceptual idea to the resultant project, CODA was a multidisciplinary success led by the engineering function within AstraZeneca, including an IS project manager, a lead vendor integrator, and the support of third-party hardware providers. The system was established within the existing CM facilities aligned to existing process workflows. The project was delivered on schedule and on budget. None of this could have been achieved without the dedication and ownership of the CM and bioscience groups and the vision they both shared.
Footnotes
Acknowledgements
The authors thank the Project Engineering, BST, and HTS teams for their vision, hard work, and expertise in the delivery of CODA.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
