Abstract
Study Design
Systematic Review.
Objective
To review the effect of NSAID administration on postoperative outcomes following posterior spinal surgery, with specific attention to pain control, opioid reduction, bleeding risk, and fusion outcomes.
Methods
PubMed, Google Scholar, Cochrane Library, Embase, Scopus, and Web of Science were queried through September 2025. Studies were included if they included adult patients undergoing posterior spinal procedures, including both fusion and non-fusion surgeries. In addition, studies had to report perioperative NSAID use in posterior spinal surgery with relevant clinical outcomes: Postoperative pain, opioid consumption, blood loss, wound complications, nonunion, and hardware failure. Bias was assessed with MINORS and Newcastle–Ottawa Scales.
Results
Eleven studies (396,206 patients) were included. Across posterior fusion and non-fusion procedures, short-term NSAID use consistently reduced postoperative opioid consumption and improved early pain control, with no clinically meaningful increase in bleeding or wound complications. However, prolonged NSAID use was associated with increased risks of nonunion, hardware failure, and wound complications in fusion cohorts. Significant variability in NSAID type, dose, timing, and study design exists.
Conclusions
Short-term NSAID use appears safe and effective as an opioid-sparing strategy in non-fusion posterior spinal surgery. Prolonged or high-dose administration may compromise fusion outcomes. Future high-quality studies are needed to definitively define the optimal postoperative NSAID window, dosing, and administration route.
Introduction
Effective postoperative pain management is critical to optimizing surgical outcomes, 1 as in orthopedic surgery, inadequate analgesia has been linked to increased complications, prolonged hospitalization, and reduced patient satisfaction.2,3 While opioids remain a cornerstone of postoperative pain control, their extensive side effect profile has driven the shift toward opioid-sparing strategies. Several studies suggest that reduced opioid exposure may improve patient outcomes, as opioids do not always enhance immediate postoperative pain relief and are associated with increased adverse events.4,5
Nonsteroidal anti-inflammatory drugs (NSAIDs) are commonly used as adjuncts to opioids, providing analgesic and anti-inflammatory benefits without sedative or addictive effects. Their use across orthopedic surgery has been associated with reduced opioid consumption and mitigation of opioid-related complications.6-9 However, concerns persist that NSAIDs may impair bone healing, based on both preclinical models and clinical observations. 10 NSAIDs inhibit cyclooxygenase enzymes, lowering prostaglandin production — a mechanism that, while beneficial for pain and inflammation, may negatively affect osteogenesis. 11 Prior systematic reviews have explored NSAID use in spinal surgery, highlighting potential impacts on bone healing, fusion rates, pain control, and opioid consumption in biochemical, animal, and human models, but emphasize the need for higher-quality, targeted studies. 12
Posterior spinal fusion remains among the most frequently performed surgeries for degenerative, traumatic, and deformity-related spinal conditions. Existing literature demonstrates that anterior, posterior and transforaminal approaches to surgery may have different risks and outcomes, with anterior approaches being associated with mortality, DVT and GI events, while posterior approaches are associated with an increased need for blood transfusion.13-15 Given this complication profile, clarifying the risks and benefits of NSAID use, a class of drugs that may worsen bleeding, in this population is critical to improve outcomes. Previous systematic reviews have examined the impact on NSAIDs on spinal surgery broadly, not considering these different complication rates.
This systematic review isolates posterior procedures and provides procedure-specific risk stratification of NSAID use by (1) distinguishing fusion from non-fusion surgeries, (2) separating short term and prolonged dosing-an approach not taken in previous spine NSAID reviews.
Methods
We conducted a systematic review to evaluate the safety and efficacy of perioperative NSAID use in adult patients undergoing posterior spinal surgery. The review was conducted in accordance with the PRISMA 2020 (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) and MINORS (Methodological index for non-randomized studies).
Search Strategy
Literature review utilized the following databases: PubMed, Google Scholar, Cochrane Library, Embase, Scopus, and Web of Science. The search included studies published in English or translated, with no date restrictions, and was last updated on 9/23/2025.
Eligibility Criteria
This study included human studies on patients undergoing posterior spinal surgeries. Surgeries included those conducted in the lumbar, thoracic and cervical regions. Perioperative NSAID use of any type, dose or route were included. Study types included randomized controlled trials, prospective cohort studies, and retrospective cohort studies. Included studies were required to report at least 1 of the following relevant outcome measures: blood loss, pain control, or postoperative complications. Studies that were either explicitly focusing on anterior spinal approaches or lacked clear specification of surgical approach were excluded. In addition, case reports, editorials, narrative reviews and letters were excluded. Pediatric (as defined by age <18 years at the time of surgery), animal, and in-vitro studies were also excluded. Studies not reporting relevant clinical outcome measures were excluded. Lastly, studies without English or translated versions, as well as conference abstracts without accessible data, were excluded.
Study Selection
Two independent reviewers screened all titles and abstracts for relevance. Full-text reviews were conducted for studies that met the inclusion criteria or where eligibility remained unclear. Discrepancies were resolved through discussion.
A total of 1101 studies were identified through database searches. Following title and abstract screening, 61 studies were selected for abstract review, of which 11 met all inclusion criteria and were analyzed to be included in the final analysis. The study selection process is illustrated in a PRISMA flow diagram, with detailed documentation of exclusion criteria at each stage (Figure 1). PRISMA 2020 flow diagram depicting study identification, screening, eligibility assessment, and inclusion for systematic review of NSAID use in posterior spinal surgery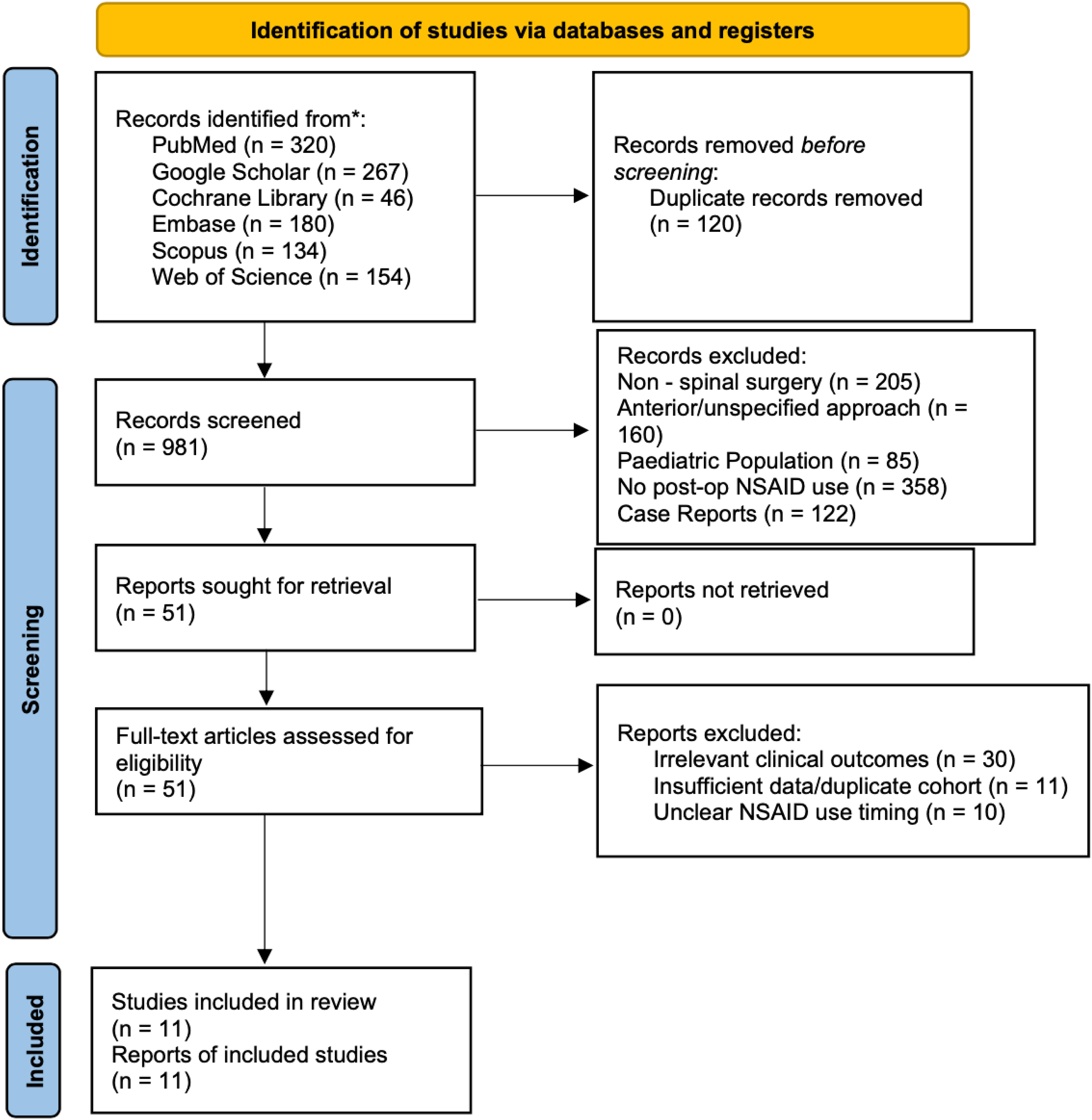
Data Extraction
Data extraction was performed independently by 2 reviewers using a standardized collection form. Information collected included. • Study title, year, authors, design • Reported clinical outcomes: Bleeding metrics, including estimated blood loss (EBL) and transfusion rates; Pain control, including Visual Analog Scale (VAS) scores
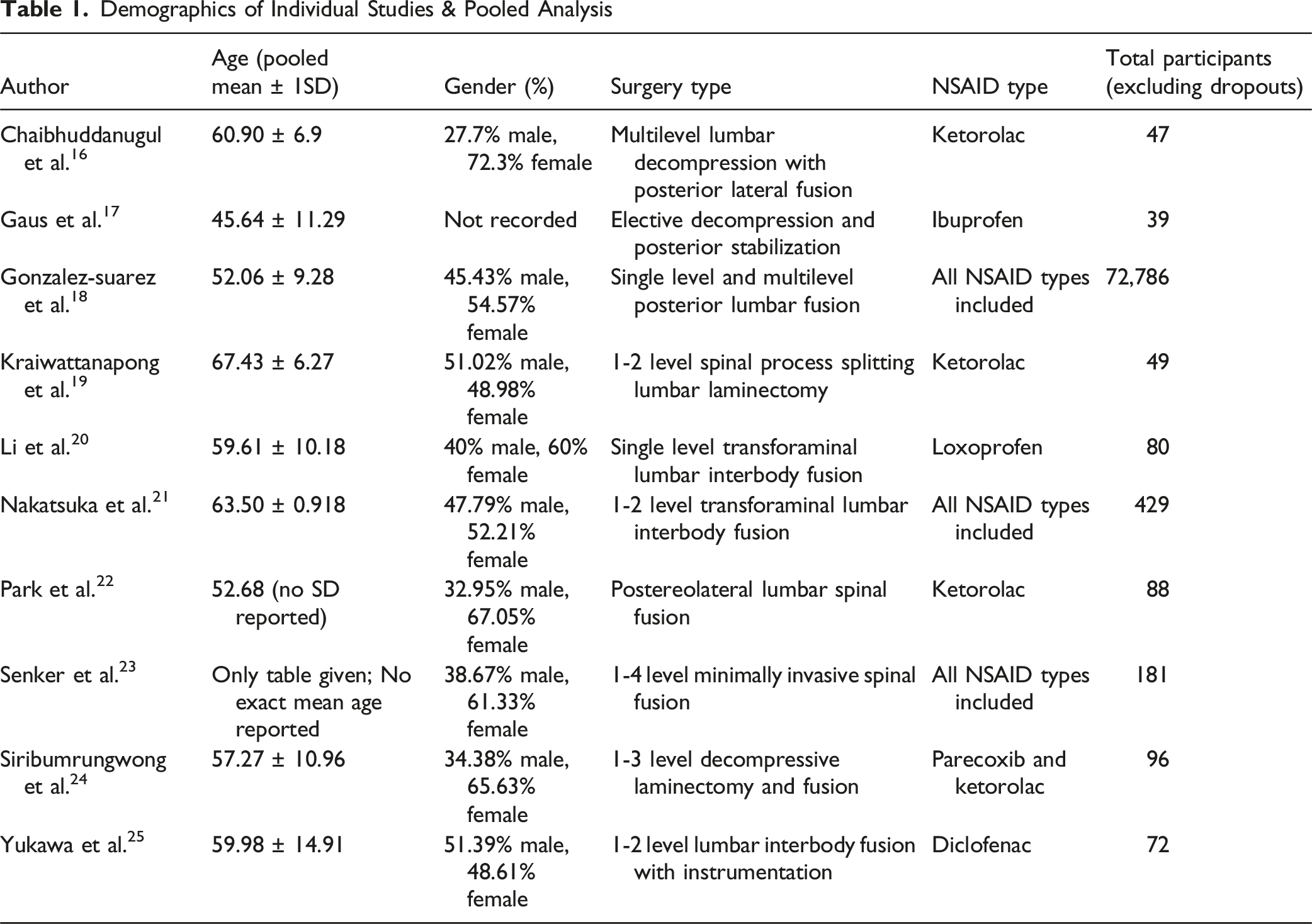
Demographics of Individual Studies & Pooled Analysis
Risk of Bias Assessment
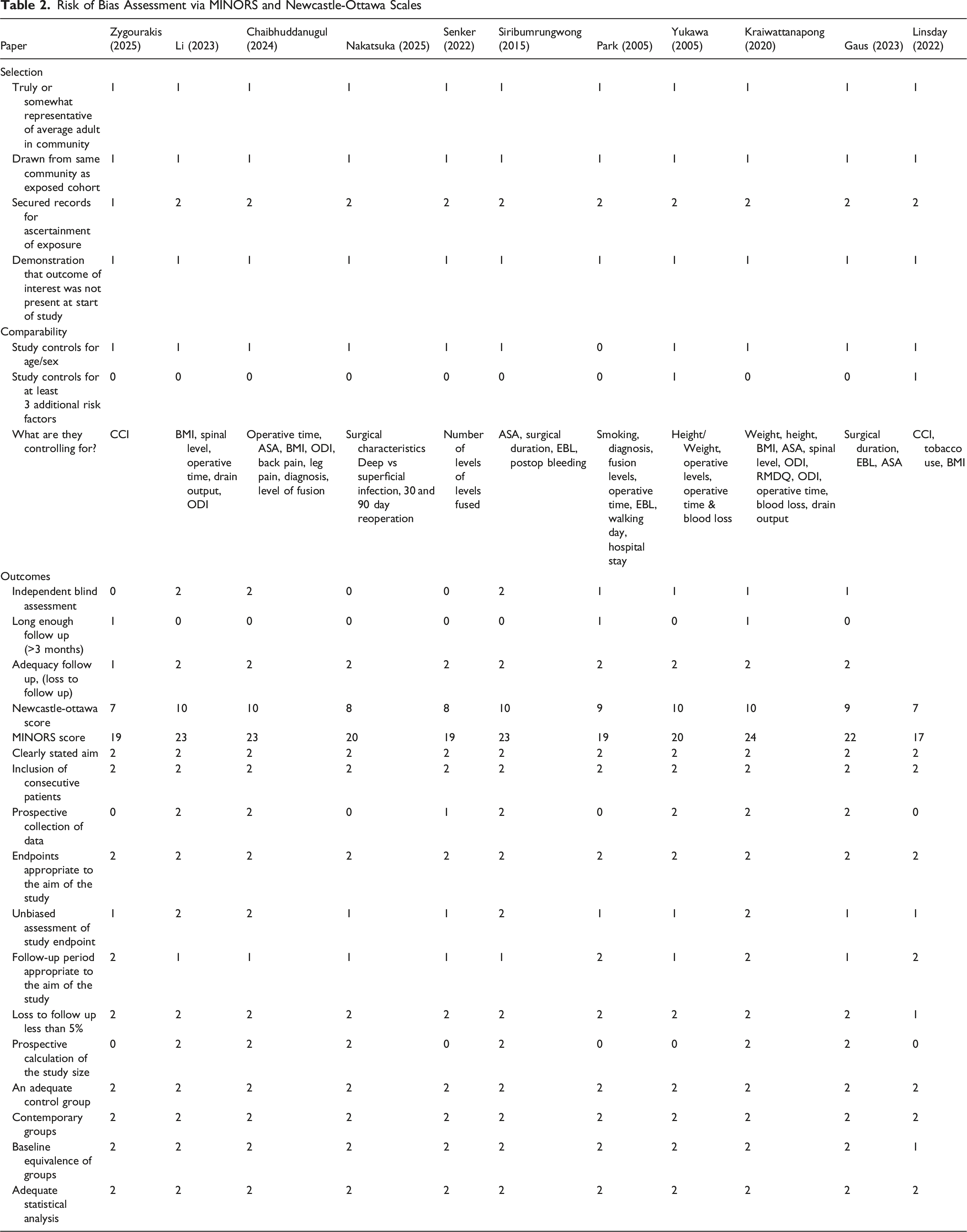
Risk of Bias Assessment via MINORS and Newcastle-Ottawa Scales
Data Synthesis
Studies were grouped based on clinical outcome findings. Quantitative pooling was deemed inappropriate due to substantial clinical and statistical heterogeneity among studies. Specifically, included trials varied in NSAID class (ketorolac, ibuprofen, diclofenac, parecoxib), administration route (topical, infiltration, systemic), duration of therapy, and outcome reporting metrics (VAS vs NRS pain scales, inconsistent nonunion definitions). This heterogeneity violated assumptions of homogeneity required for meta-analysis; therefore, results were synthesized qualitatively using a structured narrative framework emphasizing consistency of effect direction.
Results
Eleven studies, including 5 randomized controlled trials and 6 retrospective or database-based cohort studies, were included. Outcomes were categorized into 3 major domains: (1) Opioid-Sparing and Pain Control, (2) Postoperative Complications and Healing (3) Bleeding and Drainage. Across studies, NSAID type, administration route, timing, and duration varied. Several studies reported significant findings across several domains.
Grouped Outcomes
Summary of Opioid Consumption and Pain Control Studies
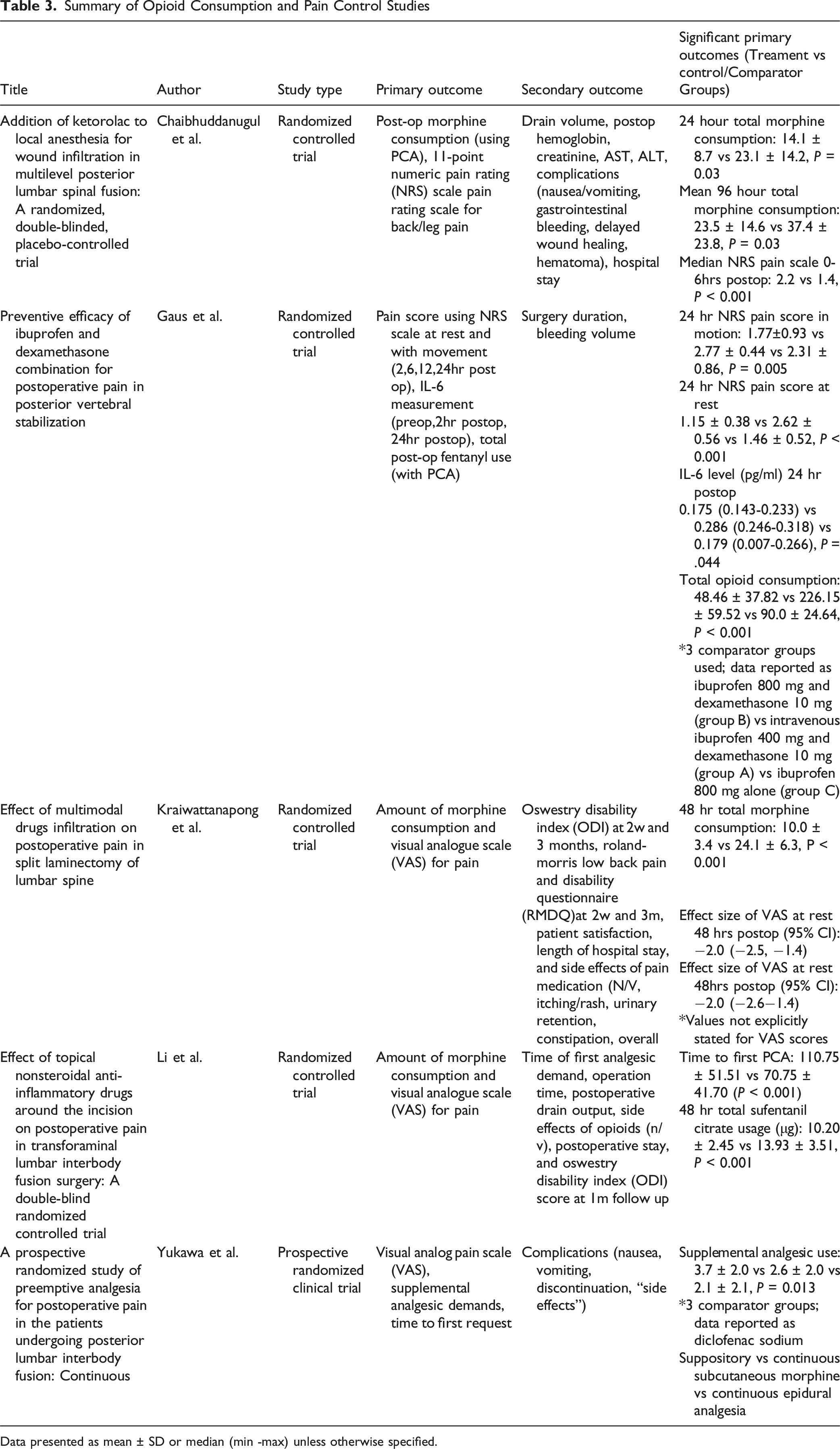
Data presented as mean ± SD or median (min -max) unless otherwise specified.
Summary of Postoperative Complications & Healing Papers
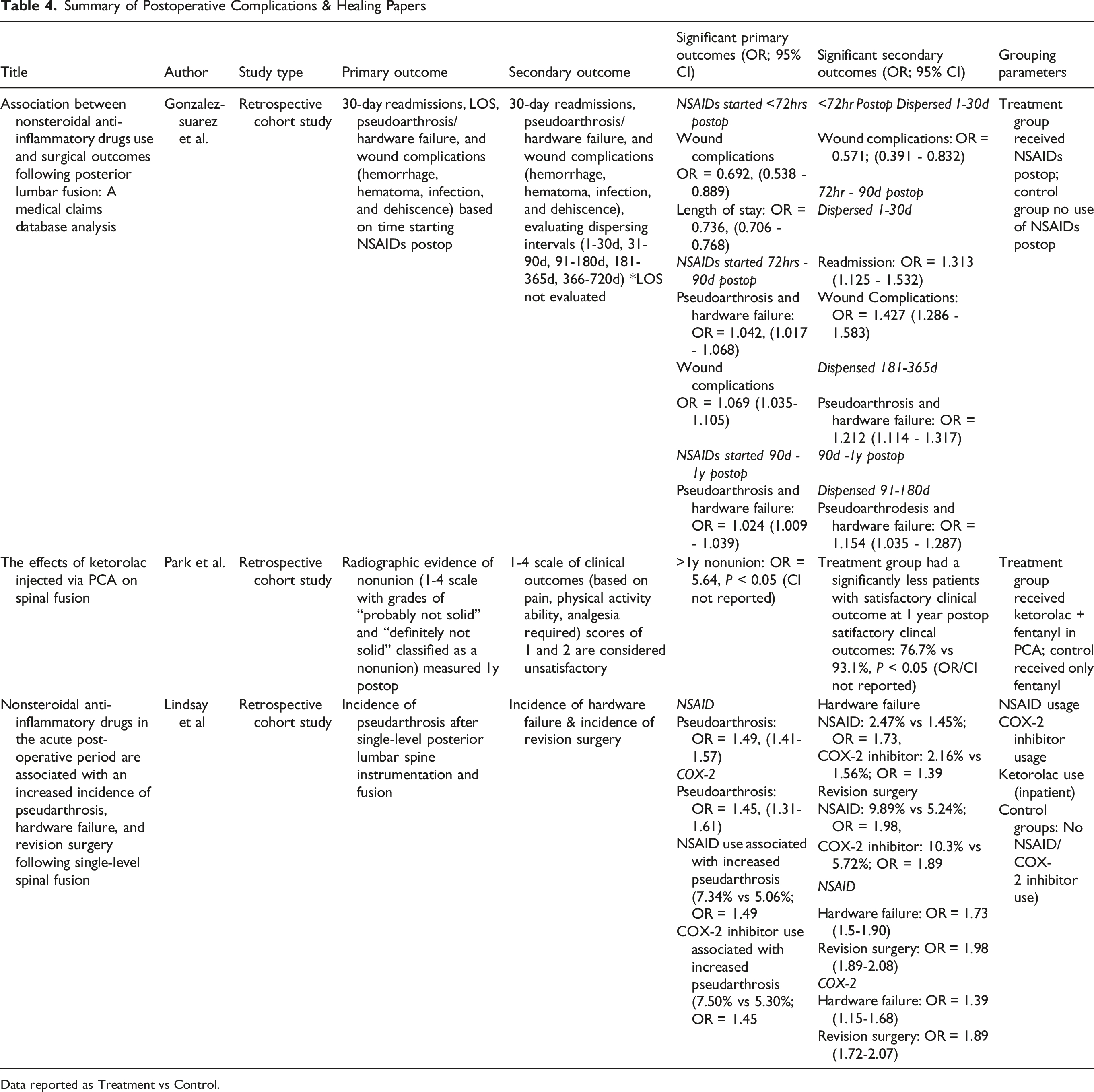
Data reported as Treatment vs Control.
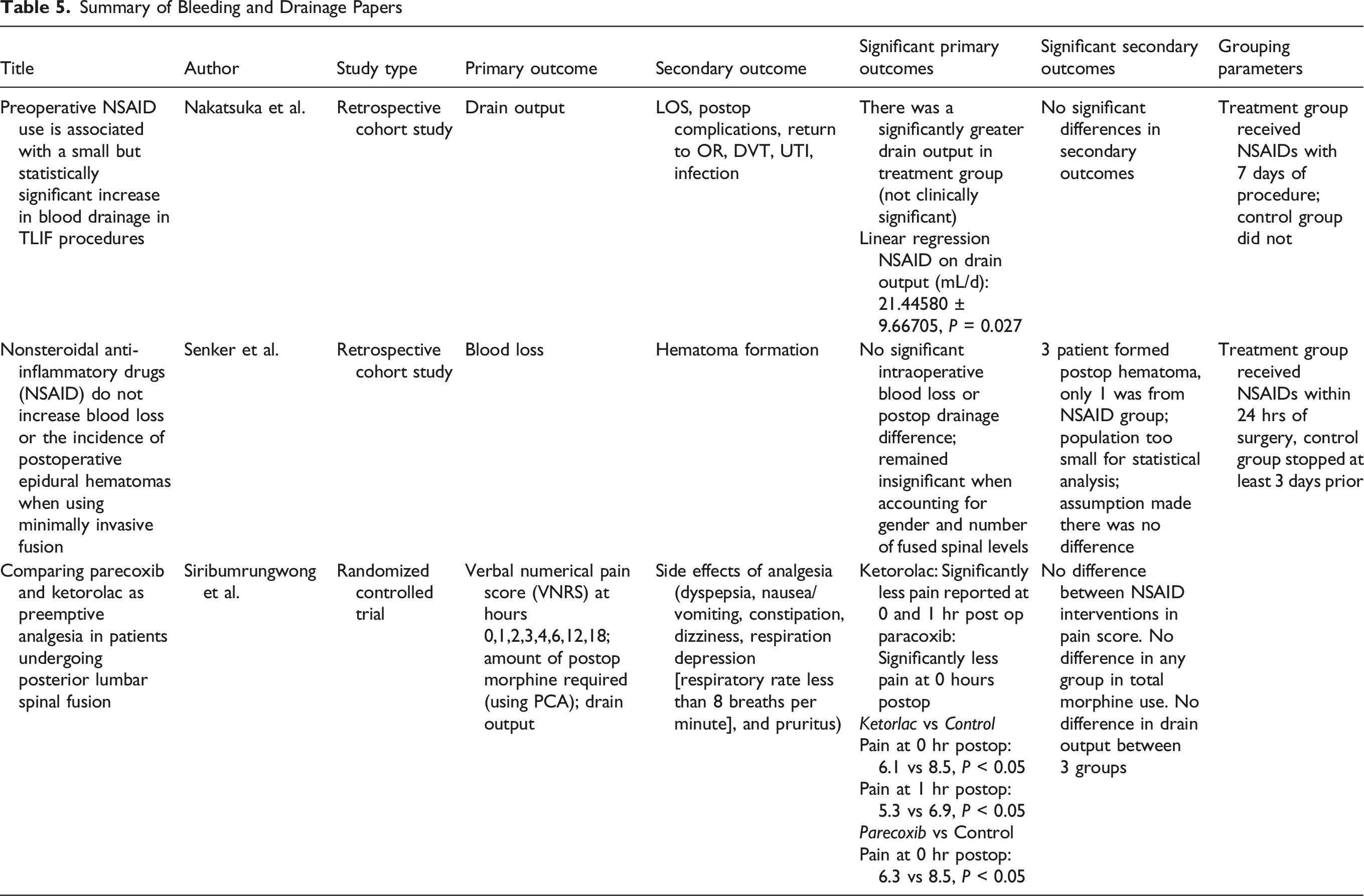
Summary of Bleeding and Drainage Papers
Opioid Consumption and Pain Control
Six studies examined the relationship between perioperative NSAID use and opioid consumption, as well as pain control, and all demonstrated statistically significant reductions in opioid use.
Chaibhuddanugul et al reported that patients receiving ketorolac with bupivacaine infiltration had a 44% reduction in morphine consumption during the first 24 h (P < 0.03) and a 35% reduction over 96 hours compared to controls (P < 0.03), with no significant differences in NRS pain scores.
Gaus et al found that patients receiving ibuprofen combined with dexamethasone had significantly lower NRS pain scores at all postoperative time points, both at rest and with movement, compared to those receiving either agent alone. This group also showed reduced total opioid consumption (48.46 ± 37.82 vs 226.15 ± 59.52 vs 90.0 ± 24.64, P < 0.001).
Kraiwattanapong et al observed that NSAID infiltration significantly reduced total morphine use and VAS pain scores at all postoperative intervals, with no significant differences in ODI scores at 2 weeks or 3 months.
Li et al demonstrated that topical NSAID administration led to significantly lower opioid consumption and VAS pain scores at all time points, a significantly longer time to first PCA demand (110.75 ± 51.51 vs 70.75 ± 41.70, P < 0.001), and fewer opioid-related side effects, though no other secondary outcomes reached significance.
Lastly, Yukawa et al found that patients receiving subcutaneous morphine with diclofenac sodium had significantly lower VAS pain scores at time zero postoperatively, though they required more supplemental analgesia within the first 72 hours. There were no significant differences in time to first analgesic request or complication rates (Table 3).
Postoperative Complications & Healing
Three studies assessed postoperative complications and healing in relation to perioperative NSAID use. Gonzalez-Suarez et al evaluated 30-day readmissions, length of stay (LOS), wound complications, nonunion, and hardware failure in posterior fusion surgery, stratifying results by the timing and duration of NSAID administration. They found that NSAIDs initiated within 72 hours postoperatively were associated with decreased wound complications (OR = 0.692, 95% CI 0.538 - 0.889) and shorter LOS (OR = 0.736, 95% CI 0.706 - 0.768). However, NSAIDs started between 72 hours and 90 days postoperatively were linked to increased wound complications (OR = 1.069, 95% CI 1.035-1.105) and nonunion (OR = 1.042, 95% CI 1.017 - 1.068), while NSAIDs initiated between 91 days and 1 year postoperatively were associated with higher rates of nonunion and hardware failure (OR = 1.024, 95%CI 1.009 - 1.039).
Park et al investigated radiographic nonunion as the primary outcome, graded by CT, with secondary outcomes including pain and function. Patients receiving ketorolac and fentanyl via PCA exhibited significantly higher nonunion rates (OR = 5.64, P < 0.05) and fewer satisfactory clinical outcomes (76.7% vs 93.1%, P < 0.05) at 1 year compared to those receiving fentanyl alone.
Lastly, Lindsay et al 26 examined nonunion rates following posterior lumbar spinal fusion, with secondary outcomes of hardware failure and revision surgery. Patients who used NSAIDs or COX-2 inhibitors within 6 weeks of surgery had significantly higher rates of hardware failure (OR = 1.73, 95% CI 1.5-1.90) and revision procedures (OR = 1.98, 95% CI 1.89-2.08). However, a separate inpatient ketorolac subgroup within the same study did not show an increased risk of postoperative complications (Table 4).
Bleeding and Drainage (Table 3)
Three studies evaluated perioperative NSAID use in relation to blood loss, drain output, and early postoperative recovery metrics. Nakatsuka et al identified drain output as the primary outcome and found that a 7-day postoperative NSAID taper was associated with significantly increased drain output. However, no significant differences were observed in length of stay, postoperative complications, reoperation rates, or DVT formation.
Senker et al focused on perioperative blood loss and postoperative drainage as primary outcomes, with hematoma formation as a secondary endpoint. They found no significant differences in blood loss or drainage, even when adjusting for gender and number of fused levels. Three cases of postoperative hematoma were reported, 1 of which occurred in the NSAID group, but statistical analysis of hematoma formation was not possible due to the small sample size.
Siribumrungwong et al assessed VNRS pain scores at multiple early postoperative intervals, with secondary outcomes including side effects, total morphine use, and drain output. Patients receiving ketorolac had significantly lower pain scores at 0 and 1 hour postoperatively (6.1 vs 8.5, P < 0.05 and 5.3 vs 6.9, P < 0.05, respectively), and those receiving parecoxib showed significantly lower pain scores at 0 hours (6.3 vs 8.5, P < 0.05). No significant differences were seen between NSAID groups at later time points, and there were no significant differences in total morphine use, drain output, or side effect profiles (Table 5).
Discussion
Summary
This review summarizes the current evidence around NSAID use in posterior spinal surgery, providing some evidence that short-term use reduces opioid requirements and improves pain without increasing bleeding or wound complications. In addition, in fusion cohorts, current evidence suggests that prolonged or high-dose exposure is associated with higher risk of nonunion, hardware failure, and wound complications. It is essential to note that current studies vary widely in NSAID type, route of administration, and duration, limiting the ability of researchers to make definitive claims regarding dosage or timing without further study. This posterior specific, timing aware framing is distinct from prior reviews that pooled anterior and posterior approaches.
NSAID effects on fusion are plausibly time-dependent because the biologic tasks of the hours after surgery (hemostasis, nociception, and acute inflammation) differ from those governing osteogenesis in the following weeks27,28 In the early phase, COX inhibition reduces peripheral and central sensitization and limits opioid requirements without materially impacting osteoprogenitor activity. Beyond this window, persistent suppression of PGE2 and downstream RUNX2/osterix signaling may impair endochondral ossification, angiogenesis (VEGF), and RANKL/OPG balance—mechanisms tightly coupled to graft incorporation. This phase-specific model reconciles our finding of short-term analgesic benefit with longer-term associations between extended NSAID exposure and nonunion in fusion cohorts.
Our synthesis supports an exposure–response hypothesis across 3 axes: duration (hours vs weeks), dose (standard vs high), and route (systemic vs local/topical). Trials using topical or limited inpatient dosing produce consistent analgesia with no signal for bleeding or nonunion, whereas outpatient continuation beyond discharge—particularly oral, systemic regimens—tracked with increased odds of pseudarthrosis and hardware failure in fusion. This suggests that cumulative systemic exposure in cohorts undergoing more invasive procedures, rather than a single perioperative dose in less invasive surgery, is the primary biologic driver of risk.
Fusion vs Non-fusion Procedure Distinction
Patients undergoing decompression-only procedures (eg, discectomy, limited laminectomy) typically require only brief analgesic courses, rarely have drains, and have no risk of nonunion or instrumentation failure.27,28 Decompression procedures lack a biologic fusion mandate; analgesic goals dominate and brief NSAID courses align with those goals. In this context, short NSAID courses appear low risk and beneficial. In contrast, posterior fusion procedures introduce biologic and mechanical healing demands that render the construct vulnerable to dose- and duration-dependent NSAID effects. These procedures require angiogenic ingrowth and osteoblast differentiation at the graft–host interface. Any therapy that repeatedly suppresses COX-2/PGE2 may attenuate these processes. 12 Thus, pooling decompression and fusion obscures procedure-specific biology and explains why “harm” signals appear only when fusion is required and exposure is prolonged. Stratifying by procedure type prevents dilution of fusion-specific risk signals and avoids the contradictory conclusions that arise when all posterior procedures are pooled, allowing providers to make clear clinical decisions based on a patient’s surgery specific needs.
Pain and Opioid Consumption
Six studies reported on postoperative pain and opioid consumption outcomes. Five of 6 studies found significant improvement in pain outcomes when NSAIDs were used in the perioperative period. Chaibhuddanugul et al, the only study that observed no change in postoperative pain score, still observed a decrease in total opioid usage, underscoring consistent evidence across both spine and non-spine literature that brief NSAID courses are effective opioid-sparing agents.
These findings are in line with literature outside of spinal surgery: Doleman et al reviewed 71 randomized control trials involving NSAID use in primarily abdominal, orthopaedic, and dental surgery. They found evidence that NSAIDs used in the perioperative period decreased postoperative pain up to 48 hours and decreased total morphine requirements. 29 In addition, Kein et al conducted a randomized control study in patients undergoing lumbar surgery with a treatment group receiving pregabalin and celecoxib postoperatively, leading to a significant decrease in VAS score and morphine consumption. 30
Postoperative Complications
Three fusion-focused studies (Gonzalez-Suarez, Park, Lindsay) consistently demonstrated a timing- and dose-dependent risk of nonunion and hardware complications. While early NSAID exposure may be protective, prolonged use clearly increases these risks. Notably, short inpatient ketorolac courses did not appear harmful, underscoring the importance of both timing and duration of administration in guiding NSAID safety.
Previous literature supports the temporal nature of NSAID’s impact on fusion in spinal surgery. A meta-analysis published by Dodwell et al isolated 7 studies and found no increased risk of nonunion after exposure to NSAID therapy. 31 Importantly, in their combined analysis, when moderate-quality long-bone studies were considered separately, NSAID exposure was associated with a markedly increased risk of nonunion (OR 4.4, 95% CI 2.5-7.8). In contrast, spine studies alone did not show a significant effect (OR 2.2, 95% CI 0.8-6.3). Further subanalyses from Dodwell et al demonstrated that high- or low-dose i.v./i.m. Ketorolac and standard oral NSAIDs were not associated with increased nonunion risk, provided exposure was short and well documented (OR 0.8, 95% CI 0.4-1.4). However, studies with prolonged or poorly defined NSAID use revealed significantly increased nonunion rates (P = 0.001). These findings reinforce that both dose and exposure duration are critical determinants of NSAID safety in the perioperative spine setting. In addition, it points to ambiguity in the current guidelines regarding NSAID administration. This supports our finding that prospective research must be conducted to clearly define the route of administration, duration, and type of NSAID and its impact on fusion rates.
Sivaganesan et al, evaluating a variety of spinal surgery fusion modalities, also found little evidence that NSAIDs increased the risk of nonunion in spinal surgery in the short term, while chronic NSAIDs resulted in a higher rate of nonunion. The authors utilized 7 publications with levels of evidence of II and III to conclude that low-dose NSAIDs at a dosing period of 48 hours postoperatively are beneficial to postoperative pain without negative effects on bone fusion. 12
This variance in findings between posterior spinal surgery and broader literature demonstrate a need for prospective studies that evaluate the risk of specific NSAIDs at specific intervals and their impact on postoperative outcomes in posterior spinal surgery.
Blood Loss & Drainage
Of the 4 studies examining these blood loss and drainage, Gaus et al and Senker et al found no increased postoperative bleeding volumes, while Li et al and Nakatsuka et al found no increased drain output.
This aligns with research outside of posterior spinal surgery. Weng et al found that dosing with parecoxib before and up to 2 days after total hip arthroplasty did not increase blood loss in the postoperative period. 32 Laoruengthana et al compared perioperative blood loss in patients receiving either ketorolac or parecoxib. They found a 117.32 mL greater amount of blood loss in the ketorolac group without a difference in blood transfusion rate, suggesting a difference that was not clinically significant. 33 A meta-analysis by Zhang et al analyzed 4 randomized control studies on spinal fusion and found that continued aspirin use in the perioperative period did not increase risk of blood loss or postoperative blood transfusion. 34 These studies in conjunction provide evidence that perioperative NSAID use carries little risk of exacerbating postoperative bleeding.
Heterotopic Ossification
While the included studies did not report heterotopic ossification (HO) as a postoperative outcome, NSAIDs are well-established in orthopedic surgery as prophylaxis against HO, particularly following arthroplasty and disc replacement.32,35-40 However, the applicability of these findings to poster, v6bior spinal fusion remains uncertain. Previous literature has found that while the incidence of HO is high in posterior fusions, (up to 81% at 2-5 years postoperatively), the clinical significance is minimal and may not warrant intervention. 41 Considering that NSAIDs negatively impact fusion rates in posterior spinal surgery, the risk of nonunion and hardware complications outweighs any potential benefit of preventing radiographically evident but clinically insignificant HO.
Strengths and Limitations
NSAID type, dose, timing, and administration route varied substantially (ketorolac, ibuprofen, diclofenac, parecoxib via systemic, topical, or wound infiltration). Timing ranged from intraoperative through 1 year. This heterogeneity, coupled with outcome reporting variability, limited comparability and precluded meta-analysis. The evidence base comprises both randomized trials and retrospective database analyses, each with distinct limitations. The randomized trials were small and underpowered for rare complications, while the retrospective and claims-based studies, though larger, relied on administrative coding that may incompletely capture NSAID exposure or nonunion events. Despite generally high MINORS and NOS scores, unmeasured confounding cannot be excluded. These factors limit causal inference and underscore the need for prospective, exposure-standardized trials. While only 11 studies met inclusion, this reflects the paucity of posterior-specific literature and underscores the need for targeted prospective studies to identify dosing and duration thresholds associated with harm. Although prior systematic reviews have faced potential duplication among national registries, the present analysis included discrete institutional and database cohorts (PearlDiver, MarketScan, and individual hospital studies) from separate countries and years. Therefore, patient-level overlap is unlikely but should be considered.
Future Directions
Future research should prioritize well-designed prospective or randomized trials to strengthen the evidence base. Studies should clearly define NSAID exposure by type, dose, route, and duration, with standardized timing windows for preoperative, intraoperative, and postoperative administration. Establishing these parameters is essential to enable valid comparisons and to identify dose- and duration-specific thresholds that may influence fusion integrity and postoperative outcomes.
Disclosures
One author has multiple financial relationships with companies involved in surgical device development and perioperative therapeutics. Please see disclosure forms for specific dollar amounts. This study was conducted independently of any of the aforementioned companies and did not receive external funding. Reporting these affiliations serves to remain transparent. ICMJE disclosure forms have been submitted by all authors. No external funding was received for this work.
Conclusion
This systematic review provides some evidence that short courses of NSAIDs may be effective for pain control and opioid use reduction in non-fusion posterior spinal surgery. Retrospective studies examining the impact of NSAIDs on fusion rates provide some evidence that prolonged use of NSAIDs may be associated with higher nonunion rates and hardware related complication risks.
Footnotes
Acknowledgements
Mitchell Ng serves as a paid consultant for Styker, Johnson & Johnson, Curafix, Pacira, Sage, Alafair, Next Science, Bonitti, Ferghana & HOF.
Ethical Considerations
This article does not contain any studies with human or animal participants.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Articles included in this review are all taken from peer reviewed, published journals.
