Abstract
Study Design
Retrospective comparative analysis of prospective cohort.
Objective
To examine clinical outcomes of patients with preexisting SCS after adult spinal deformity surgery.
Methods
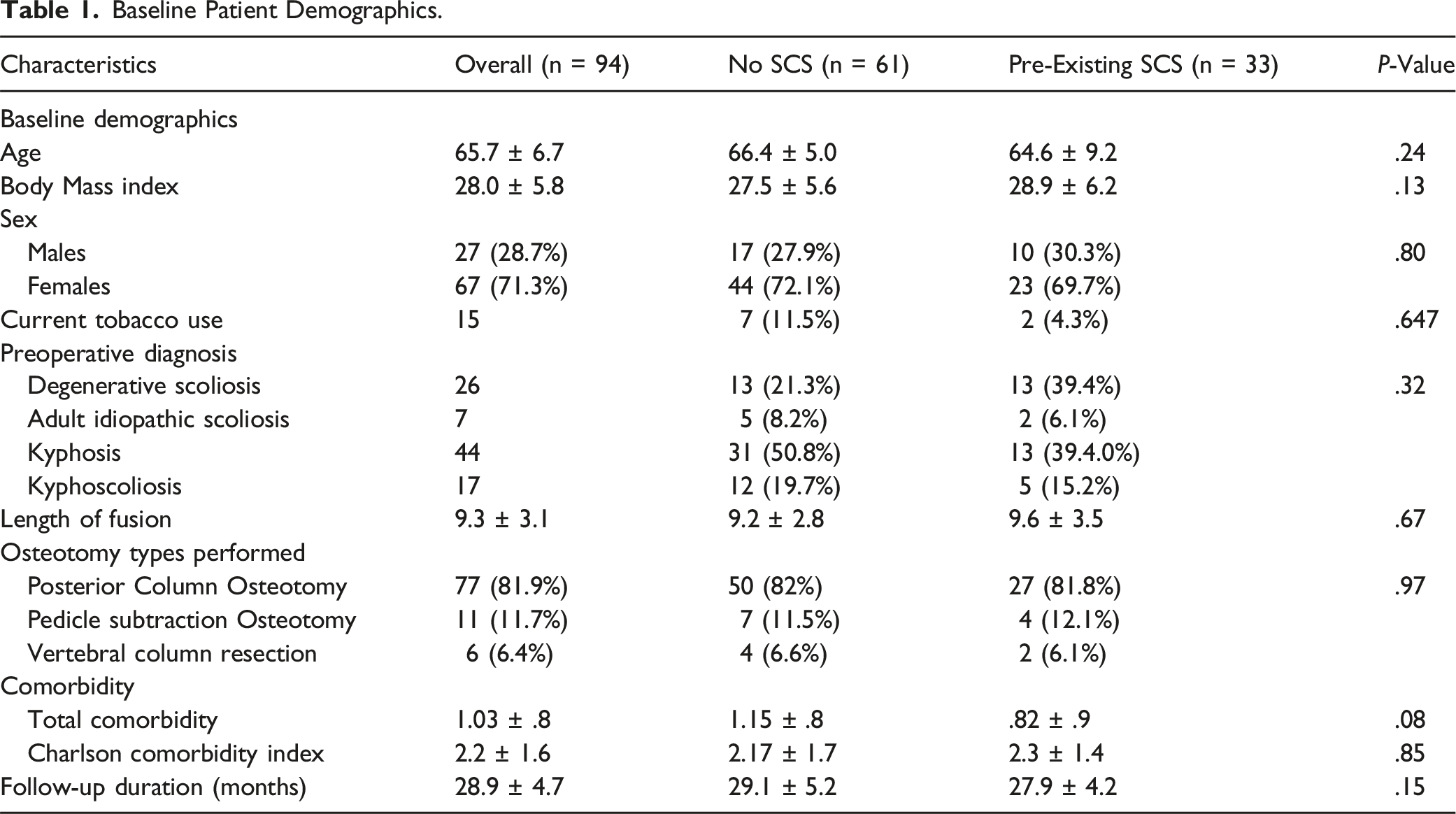
A total of 94 patients with and without a previous history of spinal cord stimulator placement undergoing surgery for ASD with minimum 2-year follow-up. Thirty-three patients with SCS undergoing ASD surgery with minimum 2-year follow-up were compared with a matched cohort of 61 ASD patients without SCS.
Results
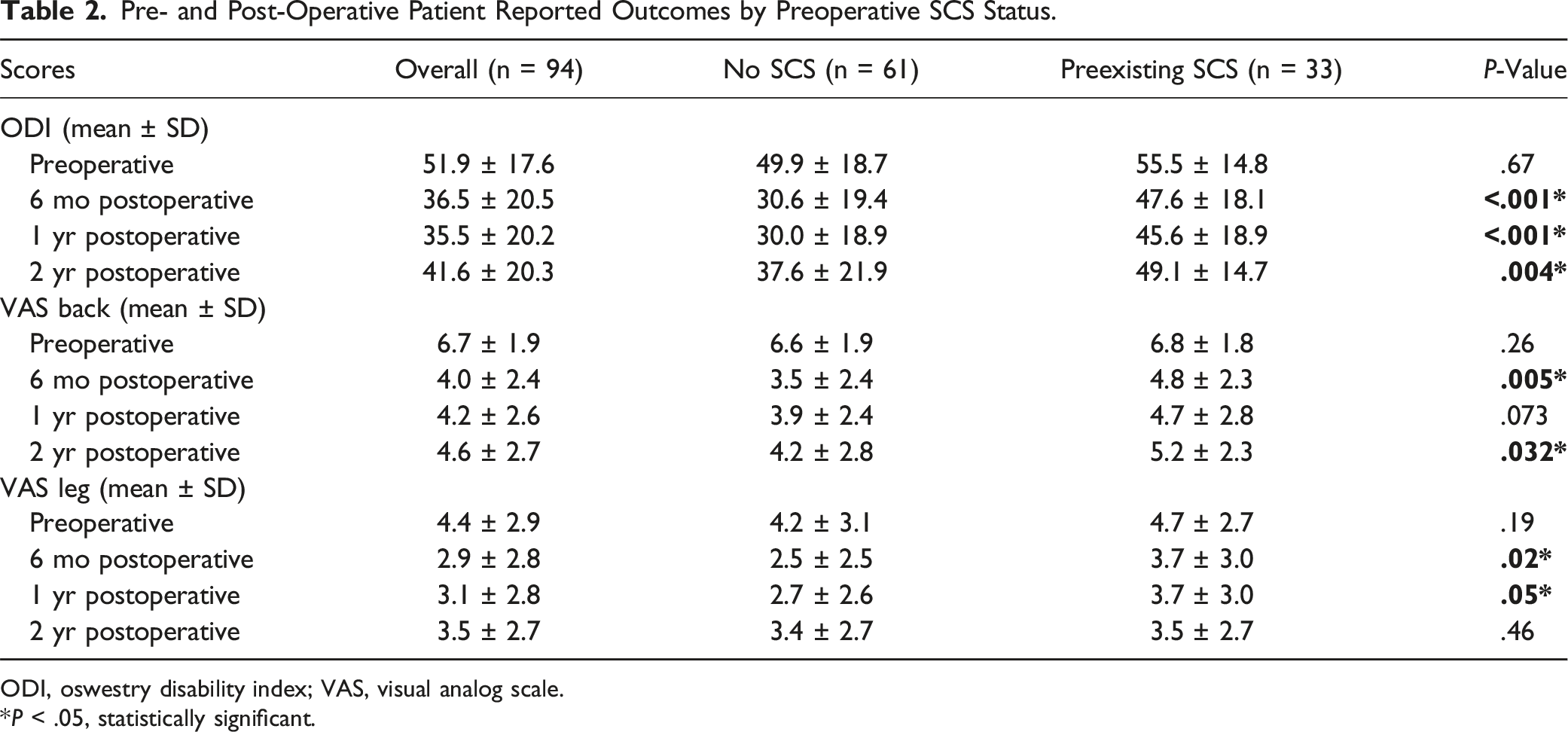
Despite similar baseline ODI (56 vs 50, P = .11) and back VAS (6.8 vs 6.6, P = .52), SCS patients did worse at all post-op time intervals. At 6 months, the SCS cohort had higher ODI (48 vs 31, P < .001) and VAS (4.8 vs 3.5, P = .01). This difference persisted at 1 year for ODI (46 vs 30, P < .001) but not for VAS (4.7 vs 4.0, P = .19). At 2 years, ODI remained significantly worse in the SCS cohort (49 vs 38, P = .004). Both cohorts had significant improvement at 2 years compared to baseline (SCS: −1.6 VAS, P < .001, −7 ODI, P = .03; Control: −2.5 VAS, P < .001, −13 ODI, P < .001). Radiographic parameters such as curve magnitude, curve correction, and balance were similar between the 2 groups.
Conclusion
Despite having substantial improvement after ASD surgery, patients with previous SCS placement did significantly worse in both back VAS and ODI postop compared with controls. They also did not experience a decrease in narcotic use at 2 years despite having similar overall radiographic results and complication rates.
Keywords
Background
Back pain represents a well-recognized contributor of disability in patients with adult spinal deformity. 1 Management of adult spinal deformity (ASD) in symptomatic individuals can be nonoperative or operative. In the absence of progressive neurological deficit or debilitating pain, nonoperative management is typically offered as first-line treatment in symptomatic individuals. 2 Nonoperative treatments typically offered to symptomatic ASD patients may include physical therapy, non-steroidal anti-inflammatory drugs, and steroid injections.3,4 However, more recently, neuromodulation in the form of spinal cord stimulation (SCS) has been implemented in the treatment of chronic back pain, and has been shown to be effective in managing pain from failed back surgery syndrome.5,6 As patients with ASD typically present with back pain, a growing number of these patients are being offered SCS.
Due to the complexity of adult deformity surgery, much work has focused on identifying preoperative factors that could impact clinical and surgical outcomes after operative treatment. Independent factors such as prolonged opioid use, smoking, and depression have all been associated with poor surgical outcomes.7,8 However, little is known on the impact of pre-existing SCS on outcomes of patients undergoing ASD surgery. The purpose of this study is to investigate whether SCS impacts long-term surgical outcomes by comparing ASD patients undergoing deformity surgery with and without preexisting SCS. We hypothesized that patients already treated with SCS at the time of deformity surgery would demonstrate poorer patient reported outcome measures (PROMs) and higher surgical complication rates after ASD surgery.
Materials and Methods
The study was conducted in accordance with the Declaration of Helsinki and was approved by the HonnorHealth IRB (HIRB # 22-0029) with the need for written informed consent waived. We retrospectively analyzed a prospectively collected database from a single institution. Patient data was queried from 2012 to 2018. Inclusion criteria were adults aged 18 or older diagnosed with ASD who underwent thoracolumbar deformity correction involving ≥4 vertebral levels and a history of spinal cord stimulator placement without removal prior to ASD surgery. All SCS were removed at the time of the ASD surgery. Minimum 2-year follow-up was also required. Exclusion criteria included patients younger than 18 years old, surgery for trauma or tumor, workers’ compensation, and surgery not specifically to address deformity correction (infection, non- union, adjacent segment degeneration). We identified 33 consecutive ASD patients with pre-existing SCS at the time of surgery and then matched a control cohort of 61 consecutive ASD patients without SCS at the time of surgery, for a total of 94 patients. The SCS cohort had their SCS deactivated at baseline to ensure matched controls were not biased by the effect of an active SCS. Collected data points included patient age, sex, diagnosis, number of spinal levels fused, previous surgeries, BMI, preoperative opioid use, smoking history, total comorbidity burden (computed using the Charlson Comorbidity Index), and complications after spinal arthrodesis. Furthermore, we examined pre-operative, 3 mo, 6 mo, 1 yr, and 2 yr outcomes including Oswestry disability index (ODI), visual analog scale (VAS) for leg and back, radiographic parameters including sagittal balance, coronal balance, pelvic incidence (PI), and lumbar lordosis (LL). The primary outcomes in this study were the Oswestry Disability Index (ODI) and Visual Analog Scale (VAS) for back and leg. We also assessed opioid usage in daily morphine equivalent dosages (MED) at all postoperative time intervals. Based on previous studies, minimal clinically important difference (MCID) was set at 2.1 for VAS (back and leg) and at 12.8 for ODI.9,10
Statistical analysis was performed using the SPSS Statistics for Windows, Version 28 (IBM Armonk, NY, USA). Continuous variables were summarized with means and standard deviations and compared using the two-sample t test. The chi- squared test or Fisher’s Exact test were used to compare the distribution of categorical variables. All tests were two-sided, and a P-value <.05 was considered statistically significant.
Surgical Procedure
All patients underwent deformity correction surgery with posterior spinal fusion under general anesthesia in the prone position. In both groups, the procedure included posterior interlaminar fusion and posterolateral intertransverse process fusion. Osteotomies, including posterior, pedicle subtraction, or vertebral column resection, were performed as indicated at the surgeon’s discretion.
Results
Patients’ Demographics
Baseline Patient Demographics.
Patient Reported Outcomes
Pre- and Post-Operative Patient Reported Outcomes by Preoperative SCS Status.
ODI, oswestry disability index; VAS, visual analog scale.
*P < .05, statistically significant.
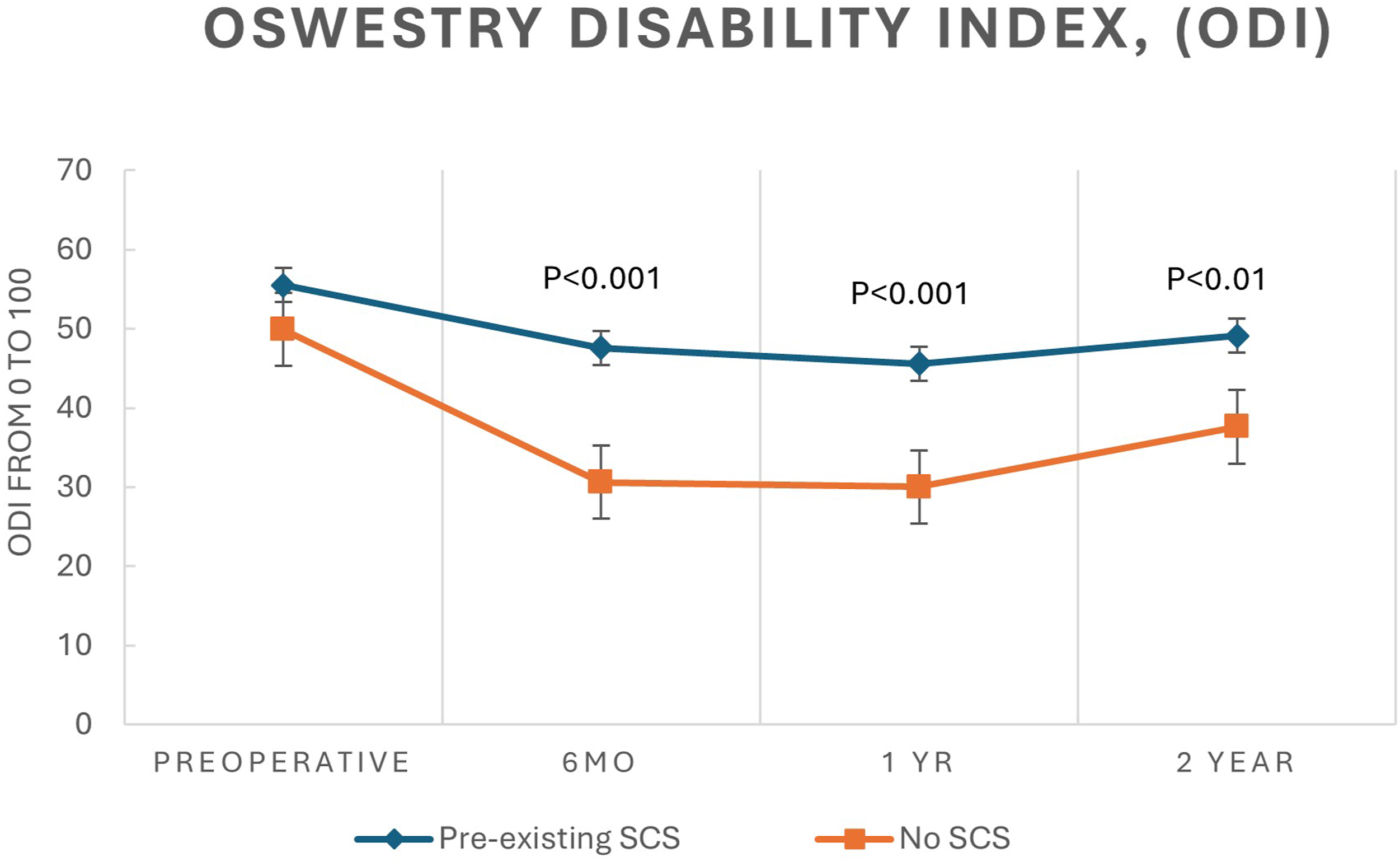
Line graph displaying the mean ± SEM scores for the oswestry disability index for Back preoperatively and up to 2 years postoperatively for patients with and without preexisting SCS.
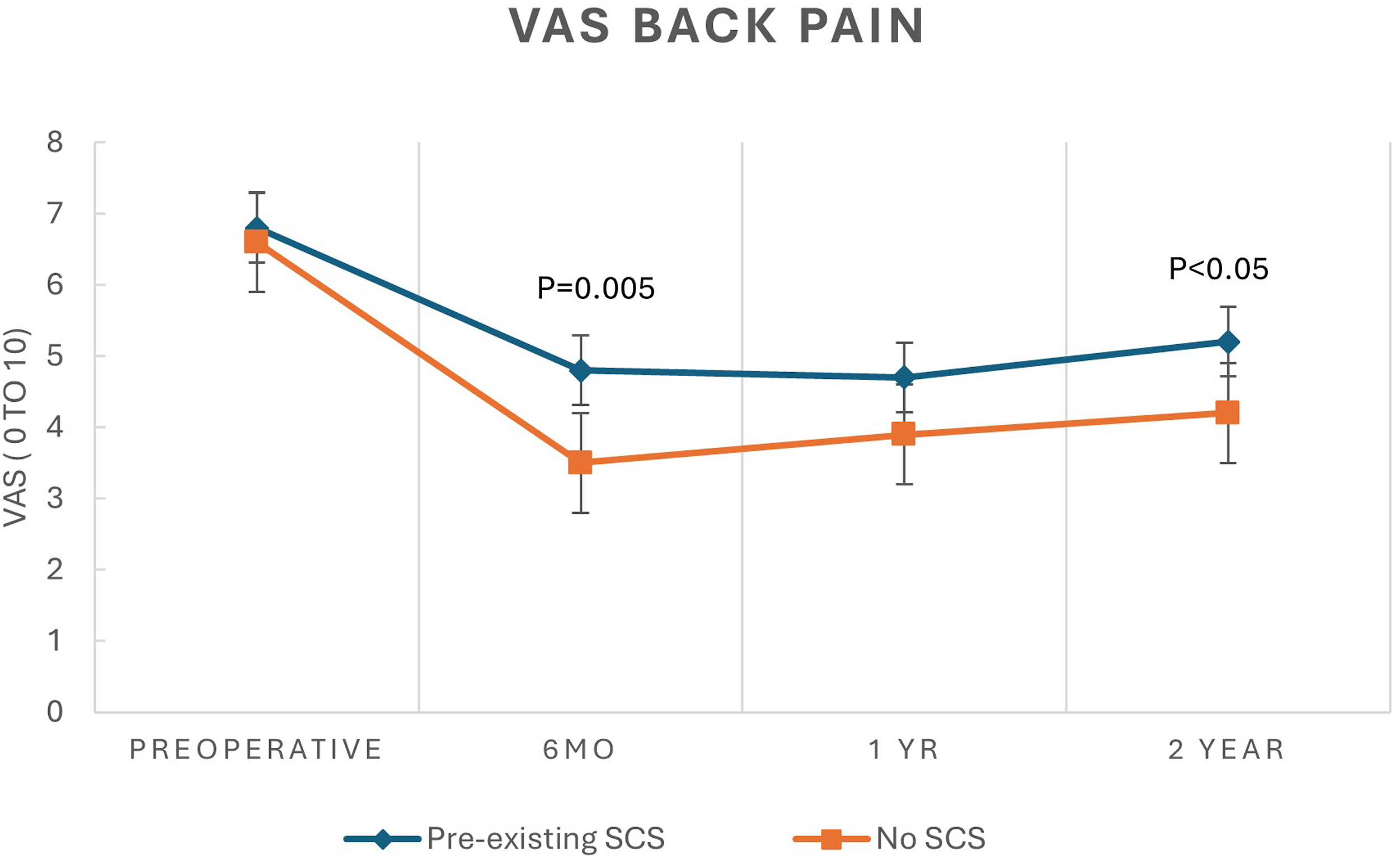
Line graph displaying mean ± SEM scores for visual analogue scale for back pain preoperatively and up to 2 years postoperatively for patients with and without preexisting SCS.
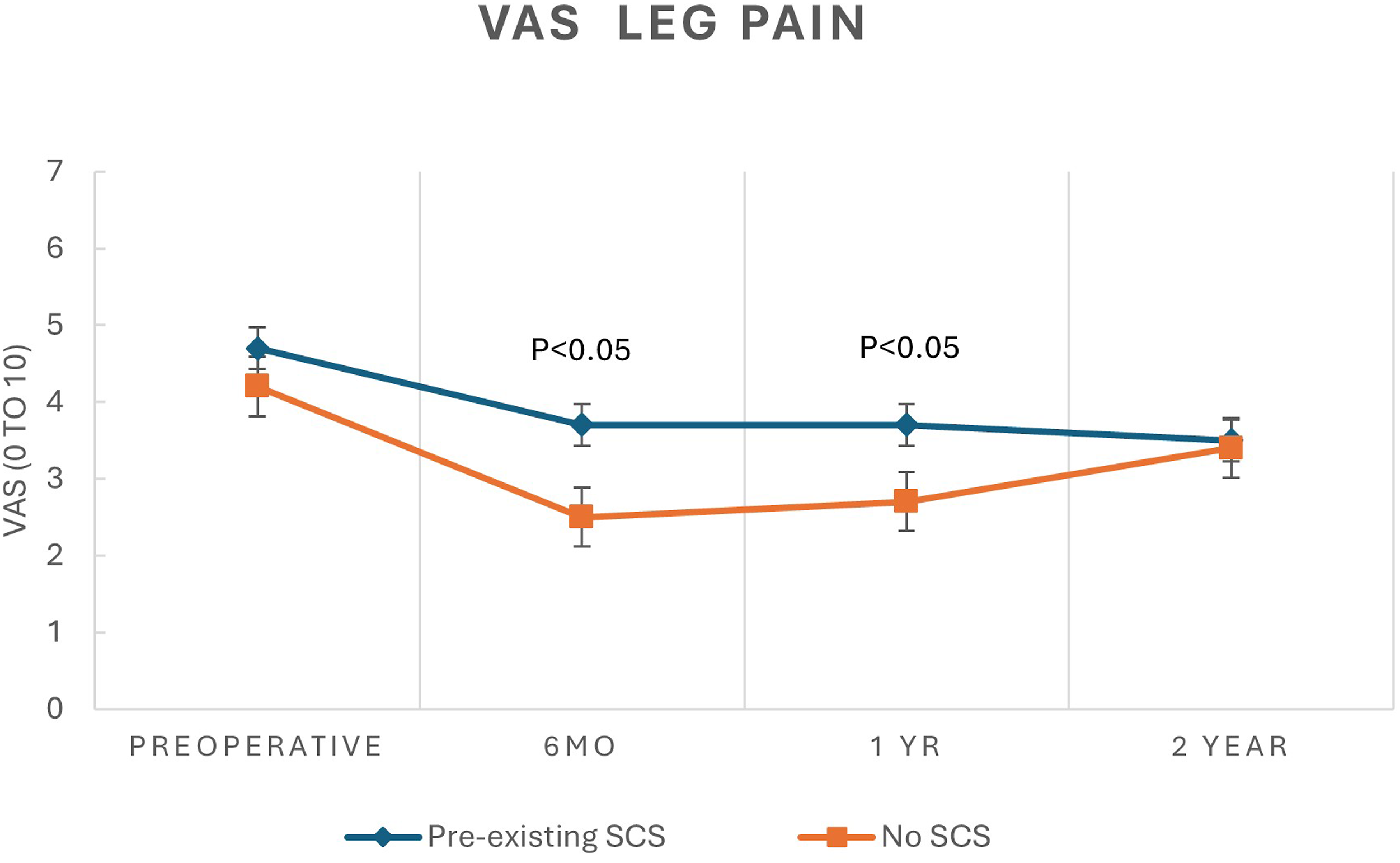
Line graph displaying mean ± SEM scores for visual analogue scale for leg pain preoperatively and up to 2 years postoperatively for patients with and without preexisting SCS.
Change in Patient Reported Outcomes by Preoperative SCS Status.
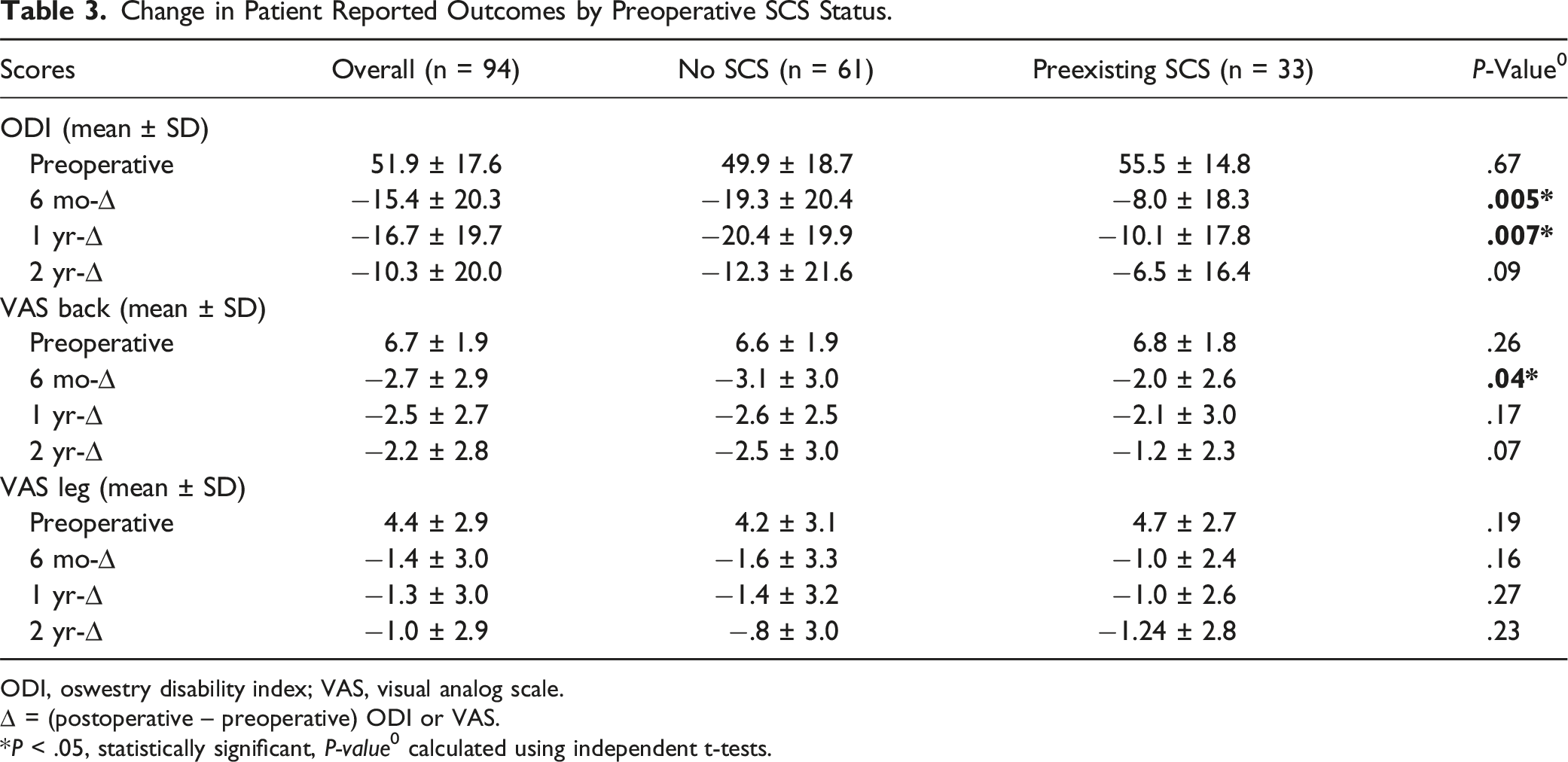
ODI, oswestry disability index; VAS, visual analog scale.
∆ = (postoperative – preoperative) ODI or VAS.
*P < .05, statistically significant, P-value0 calculated using independent t-tests.
While both groups had substantial and significant improvement from baseline, patients without preexisting SCS reported significantly more improvement in ODI at 6 months (−19.3 ± 20.4 vs −8.0 ± 18.3 P = .005), and 1 year (−20.4 ± 19.9 vs −10.1 ± 17.8, P = .007) but this difference in magnitude of improvement diminished by 2 years (−12.3 ± 21.6 vs −6.5 ± 16.4 P = .09) (Table 3). Patients without pre-existing SCS demonstrated significant improvement on the VAS back at 6 months compared with the SCS group (−3.1 ± 3.0 vs −2.0 ± 2.6 P = .04), but this was not maintained at subsequent time points, despite a trend towards greater improvement. At all-time points, both groups exhibited similar magnitude of improvements of the VAS leg, despite the significantly better overall scores of the non-SCS group.
Radiographic Parameters
All patients underwent full-length standing posteroanterior (PA) and lateral scoliosis radiographs before surgery and at each follow-up visit. Lumbar lordosis (LL) was measured using the Cobb method, from the superior endplate of L1 to the superior endplate of S1. Pelvic incidence (PI) was defined as the angle between a line perpendicular to the midpoint of the S1 superior endplate and the middle axis of the femoral heads. Coronal malalignment was assessed by measuring the deviation from the C7 plumb line to the center sacral vertical line. Sagittal balance was assessed using the sagittal vertical axis (SVA), defined as the distance from the C7 plumb line to the posterior-superior corner of the S1 endplate.
PI-LL mismatch refers to the concept that LL should be proportional to PI to maintain harmonious alignment between the lumbar spine and pelvis. A difference between the PI and LL greater than 10° is associated with increased functional loss and disability.
Pre- and Postoperative Radiographic Parameters by Preoperative SCS Status.
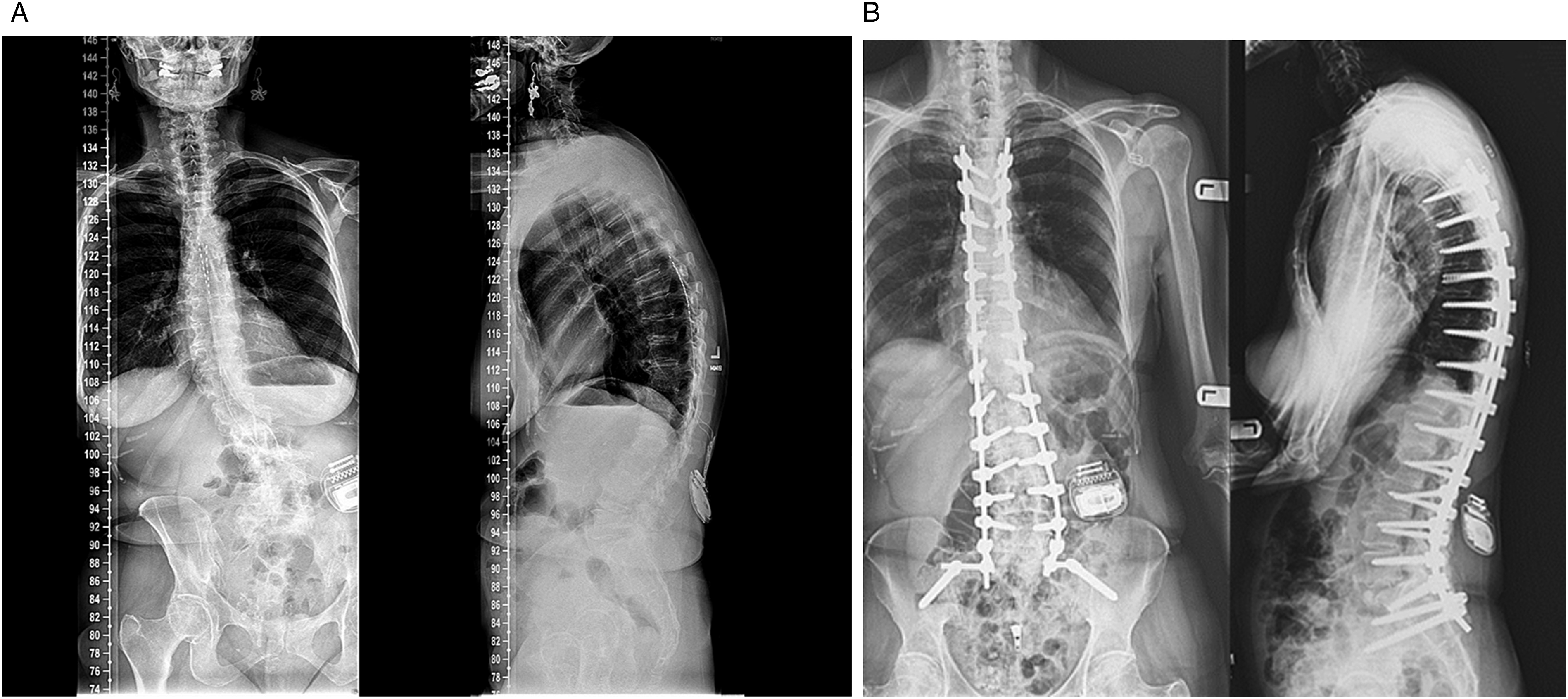
(a) Preoperative antero-posterior and lateral X-ray images of the spine of a patient with previous SCS implant: ODI 46, VAS back 5, Vas leg 3. (b) 2 years postoperative X-rays of the same patient from Figure 4(a). ODI 6, VAS back 0, VAS leg 1.
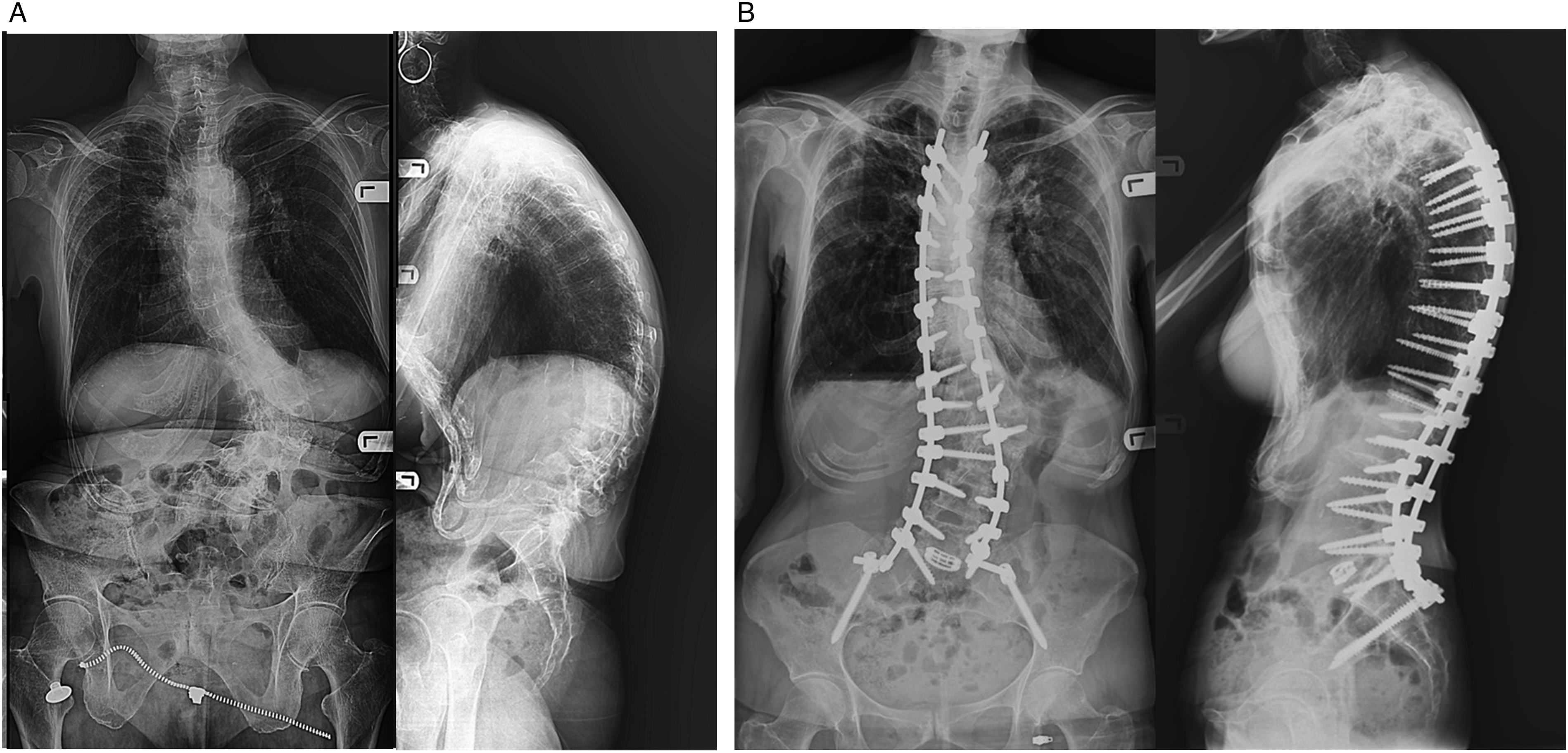
(a) Preoperative X-ray images of a patient without preexisting SCS. ODI 48, VAS Back 6, VAS Leg 6. (b) Two-year postoperative X-ray images of the same patient in Figure 5(a). ODI 8, VAS back 0, Vas leg 0.
Complications, MCID Achievement, and Opioid Usage
Complications Observed by Preoperative SCS Status.
SCS, spinal cord stimulator.
There were no medical complications observed in either cohort after surgery. Surgically related complications recorded included infections, adjacent segment degeneration, nonunion, instrumentation failure and proximal junctional kyphosis (PJK) however, no significant differences were observed between the groups. Revisions were also tracked and trended towards being greater in the SCS group. In the non-SCS cohort, revision etiology included 3 nonunion, 2 proximal junction kyphosis, and 1 instrumentation failure. In the SCS cohort, revisions were performed for 2 instrumentation failures, 2 non-unions, 3 proximal junctional kyphosis, and 1 infection. Additionally, 6 patients had their SCS removed within the 2-year follow-up period (1 for infection and 5 for device malfunction).
Opioid Medication Use and MCID Achievement by Preoperative SCS Status.
ODI, oswestry disability index; VAS, visual analog scale.
*P < .05, statistically significant.
Despite having similar pre-operative opioid consumption levels, at all postoperative time intervals opioid consumption was significantly higher in the preexisting SCS cohort (6 mo P < .001, 1 yr P = .003, 2 yr P = .005) when compared with patients without SCS. Furthermore, we observed decreased opioid consumption post-operatively in the control group while this was not the case in patients with preexisting SCS (Table 6).
Discussion
We set out to evaluate whether preexisting SCS predicts a worse outcome in patients undergoing ASD surgery. Our study confirms that pre-existing SCS placement predicted poorer outcomes of ODI and VAS at all follow-up time intervals. SCS cohorts also had a higher percentage of opioid utilization at all time intervals. Despite these generally worse outcomes, patients with preexisting SCS at 2 years still demonstrated a significant amount of improvement from baseline and had a similar magnitude of improvement when compared to patients without SCS. Thus, surgery for ASD is still beneficial to patients with SCS at 2 years of follow up. This is consistent with previous studies that have demonstrated better outcomes of ASD with operative treatment when compared to non-operative treatment.11-13
The use of SCS for chronic back pain and its long-term clinical benefits have been a major topic of controversy in the medical community over the last few years mostly due to the mixed results in studies that have evaluated the clinical efficacy of this device. While some studies claim that SCS alleviates pain in up to 50 percent of patients, other studies have suggested that physical therapy and SCS both provide similar benefits. 14 Additionally, a placebo-controlled trial study showed no significant difference in disability from baseline after spinal cord burst stimulation. 15 Despite some trials that support the use of SCS for failed back surgery syndrome (FBSS) and complex regional pain syndrome, its long-term clinical benefit in specifically ASD patients is poorly studied in the literature. Segupta et al reported a few case studies of kyphosis previously treated with SCS only having significant symptom relief after deformity correction. 16 This supports the findings from our study as we also observed significant improvement in the SCS cohort after surgical correction.
A few other studies have specifically evaluated the efficacy of SCS on ASD. However, none of these studies have been able to demonstrate long term benefit in ASD patients. Additionally, these studies were generally severely limited by small sample sizes and very short follow-up duration making it difficult to evaluate the effectiveness of SCS in ASD management. Cheng et al reported improvement in disability and pain in a single patient with severe scoliosis after a permanent SCS was implanted. However, the follow-up was limited to 5 months and this case study did not report improvement in disability scores nor MCID. 17 Similarly, Atallah et al reported an improvement in ODI from 48% to 16% in a patient with severe kyphoscoliosis implanted with SCS. However, follow-up was limited to 14 weeks making it difficult to assess long-term outcome. 18 In another related study, Lucia et al demonstrated a mean improvement of 9.8 on the ODI scale after 6 months of SCS therapy for symptomatic ASD patients not eligible to undergo extensive corrective surgery. 19 The study was limited by a small sample of 18 in which only 8 patients were available for analysis at 12 months. It also did not specify the selection criteria for determining those not eligible for corrective surgery. Additionally, the study did not report on MCID, nor the mean improvement in ODI at 12 months. In contrast, our study demonstrates at 6 months and 12 months, respectively, a mean improvement of 19.3 and 20.4 on the ODI in the non-SCS cohort, with only 8.0 and 10.1 improvement in patients with SCS.
The inferior clinical outcome scores achieved by SCS patients compared with non-SCS patients in our study may suggest that SCS placement caps efficacy of pain relief in ASD patients. Since the underlying pathology in ASD is from structural elements of the spine, it is unsurprising that SCS frequently fails in this patient population because the stimulator does not address the underlying structural problem.8,20 Intuitively, one could assume patients previously treated with SCS are those with higher disability and pain; however, our study consisted of cohorts with similar baseline ODI, VAS and preoperative opioid consumption. Furthermore, there were no differences in preoperative and postoperative radiographic parameters as well as postoperative complication rates. Additionally, preoperative clinical factors demonstrated to adversely affect outcomes in spinal surgeries such as smoking and opioid use were not statistically different between the groups.21,22 In a study similar to ours, Daniel et al, examined 30 patients with preoperative SCS or intrathecal pump (ITP) undergoing ASD surgery and found that at all postoperative intervals these patients demonstrated worse outcomes and had higher consumption of opioid medication when compared to the control group. 23 They also found in their study that despite having worse outcomes, these patients still benefited significantly from surgery. These findings by Daniel et al are also consistent with results from our study that previous SCS placement predicts a poorer outcome. However, in contrast to the study by Daniels et al, in which SCS cohorts had a significantly higher baseline ODI and opioid usage, our matched control had similar baseline ODI, VAS and opioid utilization. Additionally, our study consisted of patients treated with SCS only and excluded patients with ITP to avoid any confounding effect that could result from prolonged intrathecal opioid use.
Relating to opioid use after SCS implantation, Hwang et al in a retrospective study consisting of 45 patients to characterize long-term patterns of opioid use after SCS implantation demonstrated that daily opioid use did not decrease in these patients after 1 year. 24 Pollard et al conducted a meta-analysis of 5 randomized comparative studies of SCS vs conventional medical therapy and found that the individual studies did not reach statistical significance for reduced opioid use. 25 Similarly, Vu et al, in a large cohort study involving 552,937 patients with FBSS in which 26,179 were treated with SCS concluded that although those treated with SCS resulted in statistically significant reductions in the number of opioid prescriptions, the reduction was so small that its clinical relevance may be questionable. 23 Although these studies were not specifically designed to assess the pattern of post SCS opioid consumption in ASD patients treated with SCS, it does support the findings of our study, in which we did not observe a significant reduction in opioid consumption in the SCS cohort as compared with the control group.
There are several potential explanations as to why patients with previous SCS treatment exhibited worse outcomes. Major concerns over the years have been the development of fibrosis in the epidural space due to inflammatory response after placement of SCS electrodes.12,13 Richardson et al described dense fibrous encapsulation around stimulator cables in pathology specimens excised from 1 week to 2 years after implantation. 26 Multiple reports have associated epidural fibrosis from previous spinal surgeries to poorer outcomes in subsequent spinal surgeries, and this may suggest that the inferior outcomes observed in our SCS cohort could have resulted from epidural fibrosis during SCS paddle placement. 27
Another possible explanation for the inferior outcomes in the SCS cohort is altered pain perception. Kumar et al. described a tolerance phenomenon, in which patients experience a gradual decline in satisfactory pain relief with prolonged SCS use. 28 Spinal cord stimulators modulate pain signals transmitted to the brain based on the “gate control theory,” where electrical impulses from the SCS interfere with pain transmission in the spinal cord. 29 Over time this modulation may induce changes in both the sensory and emotional components of pain, potentially contributing to diminished long-term efficacy. Further research is needed to explore the long-term effects of SCS on pain pathways to enhance our understanding of this device.
There are also economic concerns with regards to SCS placement in ASD patients. With an increasing frequency of ASD patients with preexisting SCS eventually opting to have surgical correction, prior SCS placement would add to healthcare costs while increasing adverse outcomes for an already expensive ASD surgery in these patients. Since SCS has neither been shown to offer long-term benefit to ASD patients nor proven to offer a superior benefit to surgery, it should be restricted to use only when there is no surgical treatment available to patients.
Our study has several limitations. Our sample size is relatively small in our SCS cohort; however, power analysis suggests the likelihood of a type II error is small. Furthermore, our cohort included patients that did not get SCS implants at our center, meaning we could not ascertain if the patient initially benefited from SCS placement. Finally, opioid usage was evaluated from filled prescriptions converted to morphine equivalent doses; it may not accurately depict actual opioid utilization if patients do not take their prescribed dosage or if they obtain additional opioids through non-prescription methods. The strengths of our study include a large, matched control group with similar baseline patient reported outcome metrics and a follow-up of 2 years with which we were able to effectively evaluate longer term outcomes in both cohorts.
Conclusion
Our study suggests that SCS placement negatively impacts outcomes in spinal deformity surgery for ASD, with significantly worse VAS and ODI scores at 2 years. Additionally, patients previously treated with SCS are more likely to utilize higher level of opioids postoperatively. Despite these challenges, surgical intervention remains beneficial for patients with prior SCS placement, as they showed significant improvement from baseline at all postoperative time points.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MSC serves as a consultant to Stryker and MiRus and receives speaking or teaching arrangements fees from BK Meditech USA, and sits on their Advisory Board. DGC sits on the Board of Directors of Handel, LLC. BA has no conflict to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
Deidentified data could be made available upon reasonable request from the corresponding author.
