Abstract
Study Design
Online questionnaire following the CHERRIES framework.
Objectives
Degenerative cervical myelopathy (DCM) is a heterogenous condition with a broad clinical presentation profile. Understanding the adoption of clinical signs and symptoms for both diagnostic and surgical decision-making could inform the development of standardised diagnostic criteria for DCM. Therefore, we sought to survey orthopaedic spine surgeons and neurosurgeons in secondary care across Australasia (New Zealand and Australia).
Methods
An online 19-item questionnaire was distributed to orthopaedic spine surgeons and neurosurgeons across Australasia. It examined factors influencing diagnostic and surgical decision-making, including responses to clinical vignettes. Post-stratification weighting was applied and an exploratory post-hoc pattern analysis was performed.
Results
Seventy-two completed responses were analysed. For diagnosis, gait clumsiness (86.1%), hand clumsiness (76.4%) and reduced hand dexterity (75.0%) were the most frequently selected symptoms, and hyperreflexia (83.3%) and ankle clonus sign (68.1%) were the most commonly selected clinical signs. Symptom severity, myelomalacia, and extent of cord compression were ranked as the top 3 variables influencing surgical decision-making for over 70% of surgeons. In clinical vignettes, most (72.2%) chose serial observation for asymptomatic cord compression; however, accompanying myelomalacia was found to increase the chance of offering surgery from 5.6% to 34.7%.
Conclusions
Although several signs and symptoms were commonly used to diagnose DCM, a broad range of features were employed, reflecting substantial variability amongst Australasian surgeons. Surgical decision-making also varied, particularly for asymptomatic cord compression with myelomalacia. These findings reinforce the need for standardized diagnostic criteria and referral pathways.
Keywords
Background
Degenerative cervical myelopathy (DCM) is the most common cause of adult spinal cord dysfunction worldwide with an estimated adult prevalence of 2.3%, rising to 5% in adults over the age of 40 years. 1 With a globally aging population, this prevalence is expected to rise. 2 Early diagnosis and timely surgical intervention are critical to minimize the progressive neurofunctional deterioration associated with chronic spinal cord compression, a hallmark feature of DCM.1,3 However, patients with DCM face significant diagnostic delays, particularly in primary care, often leading to delayed intervention and subsequent chronic disability.3,4 Variability in clinical presentation, low clinician awareness and the absence of standardized diagnostic criteria for DCM are likely to contribute to these diagnostic delays.5,6
In addition, population-based magnetic resonance imaging (MRI) studies report asymptomatic cord compression in up to 24.2% of healthy adults, complicating decision-making around the necessity and timing of surgical intervention given the uncertain disease trajectory. 7 Consensus is lacking on the management of these cases as well as the defining features and surgical thresholds for mild DCM. 8
To inform the development of standardized diagnostic criteria for DCM, it is important to understand the current criteria employed by orthopaedic spine surgeons and neurosurgeons when diagnosing DCM and recommending surgical intervention. The secondary care sector is of particular interest given the likelihood that more cases of milder and earlier stage DCM may be encountered there. In this sector, patients often seek evaluation of axial neck pain or radicular arm pain, where subtle accompanying features of DCM may only be recognised following systematic clinical assessment and correlation with MRI findings. Detecting DCM at this early stage offers an opportunity to consider disease-halting decompressive surgery before the onset of more severe neurological deficits.
As such, this study aimed to identify which features of the clinical presentation influence diagnostic and surgical decision-making for DCM among orthopaedic spine surgeons and neurosurgeons in Australasia (New Zealand and Australia).
Methods
Study Design
An internet-based questionnaire, hosted on Qualtrics, was used to explore diagnostic and surgical decision-making in DCM. The study followed the CHERRIES (Checklist for Reporting Results of Internet E-Surveys) framework, with the completed checklist provided in Supplemental material 1. 9 Ethical approval was obtained from the Auckland University of Technology Ethics Committee (Ref: 23/351).
Survey Development and Piloting
The questionnaire was developed based on evidence from recent systematic reviews examining the clinical signs and symptoms associated with DCM.10,11 Two orthopaedic spine surgeons who routinely diagnose and engage in surgical decision-making for DCM piloted the draft survey for face and content validity, with revisions made based on feedback.
The final questionnaire (Supplemental material 2) included 19 items across 4 domains: (1) respondent characteristics (3 items), (2) current clinical practice (3 items), (3) diagnostic practices for DCM (5 items), and (4) surgical decision-making (8 items), the latter incorporating 5 clinical case vignettes to simulate surgical decision-making scenarios, enabling further exploration of factors influencing surgical decision-making.
Participant Recruitment and Eligibility
Eligible participants were orthopaedic spine surgeons and neurosurgeons based in New Zealand or Australia, registered as current fellows with their respective regulatory bodies and maintaining an active secondary care caseload. Recruitment was conducted through email newsletters distributed by the New Zealand Orthopaedic Spine Society, Neurosurgical Society of Australasia, and Spine Society of Australia. An information sheet and questionnaire link were embedded within the newsletter. Participation was voluntary, non-incentivized, and anonymous, with informed consent obtained via Qualtrics. The survey remained open from February 2024 to June 2024, with one reminder email sent.
Survey Administration
The survey consisted of 7 sequential pages and participants could navigate backward to review or modify responses before submission. Automated checks ensured completion of mandatory questions. Internet Protocol addresses or other identifiable data were not collected.
Duration-based response filtering was applied to ensure data quality and reduce the likelihood of invalid responses. 12 Responses completed in under 4 min were excluded, based on expert statistical guidance and the estimated minimum time required to thoughtfully complete all survey items.
Data Analysis
Descriptive statistics, including percentages, means, medians (M), and interquartile ranges (IQR), were used to summarise responses across all survey domains.
In accordance with the CHERRIES framework and upon expert statistical consultation (IZ), post-stratification weighting was applied to items most susceptible to sampling bias, including: DCM symptoms and signs used for diagnostic and surgical decision-making, and variables ranked for surgical decision-making. Weights were derived based on known distributions of surgical specialty (orthopaedic vs neurosurgeon) and country of practice (New Zealand vs Australia), using membership data obtained via personal correspondence with relevant societies. A four-strata contingency table was created ((1) New Zealand orthopaedic spine surgeons, (2) New Zealand neurosurgeons, (3) Australian orthopaedic spine surgeons, and (4) Australian neurosurgeons), and weights were calculated by dividing population proportions by corresponding sample proportions. IBM SPSS Statistics Version 30 was used to apply weights, and results were compared to unweighted data to assess discrepancies. Only weighted results are reported below, with unweighted results available in Supplemental material 3.
A post hoc exploratory pattern analysis was conducted to identify frequently co-selected DCM symptoms and signs. This approach analyses patterns identified through textual data to determine potentially unconscious trends and their frequencies. 13 Although participants were not explicitly asked to select combinations of signs and symptoms, this analysis sought to reveal unconscious diagnostic reasoning patterns by examining recurring patterns. To achieve this, each symptom and sign was numerically coded (eg, 1-12) and frequently occurring 3- and 4-item combinations were manually identified within raw exported data. Frequencies were calculated and adjusted using post-stratification weights in SPSS. Combinations selected by ≥ 35% of respondents were considered clinically relevant, accounting for anticipated heterogeneity.
Results
Respondent Characteristics
Over a 4-month timeframe, 96 responses were received, of which 72 complete responses were included in the final analysis. This reflects a response rate of 15.9% from 454 potential participants and a completion rate of 75%. The completion rate provides an attrition percentile of those who consented to participate and those who completed all survey items as per the CHERRIES framework. 9
Respondent Characteristics and Current Practice

Note. This table includes demographic and clinical practice characteristics of participating surgeons. The survey focussed on ‘secondary care,’ representing specialist medical services provided in the private sector, upon referral from primary care. ‘Surgical candidates at the point of DCM diagnosis’ refers to surgeon’s approximation of the percentage of cases which they would consider offering surgery upon initial diagnosis of DCM. DCM: Degenerative Cervical Myelopathy.
Current Clinical Practice for DCM in Secondary Care
Respondents had a median secondary care DCM case load of 2.0 cases per month (IQR = 0). There was high variability in the proportion of patients considered to be surgical candidates at the point of diagnosis, with 19 surgeons (26.4%) responding that 1%-25% were surgical candidates and 18 (25.0%) reporting 51%-75% (Table 1). Seven surgeons (9.7%) reported that they don’t see patients with DCM in their secondary care clinics (but do assess and manage DCM in their tertiary care roles).
With regard to the use of cervical MRI in usual practice, 30.6% of respondents ordered MRI pre-consultation, compared to 18.1% only ordering post-consultation and 51.4% ordering MRI pre-consultation on a case-by-case basis. Computerised tomography was the next most employed investigation in the diagnostic workup for DCM (68.06%), followed closely by plain radiographs (61.11%). Nuclear medicine bone scans (SPECT-CT) and EMG/NCS were not utilised as often (11.11% and 9.7% respectively).
The use of whole spine sagittal MRI (WSSMRI) sequence in assessing cervical spinal cord compression was asked of respondents. The majority (59.2%, n = 42) reported they did not receive WSSMRI sequences in their usual spinal MRI protocol. Of the remaining 40.8%, 86.2% (n = 25) actively reviewed this sequence to assess compression, with the majority (84.0%, n = 21) receiving T2-weighted WSSMRI sequences.
Symptoms Influencing Diagnostic and Surgical Decision-Making for DCM
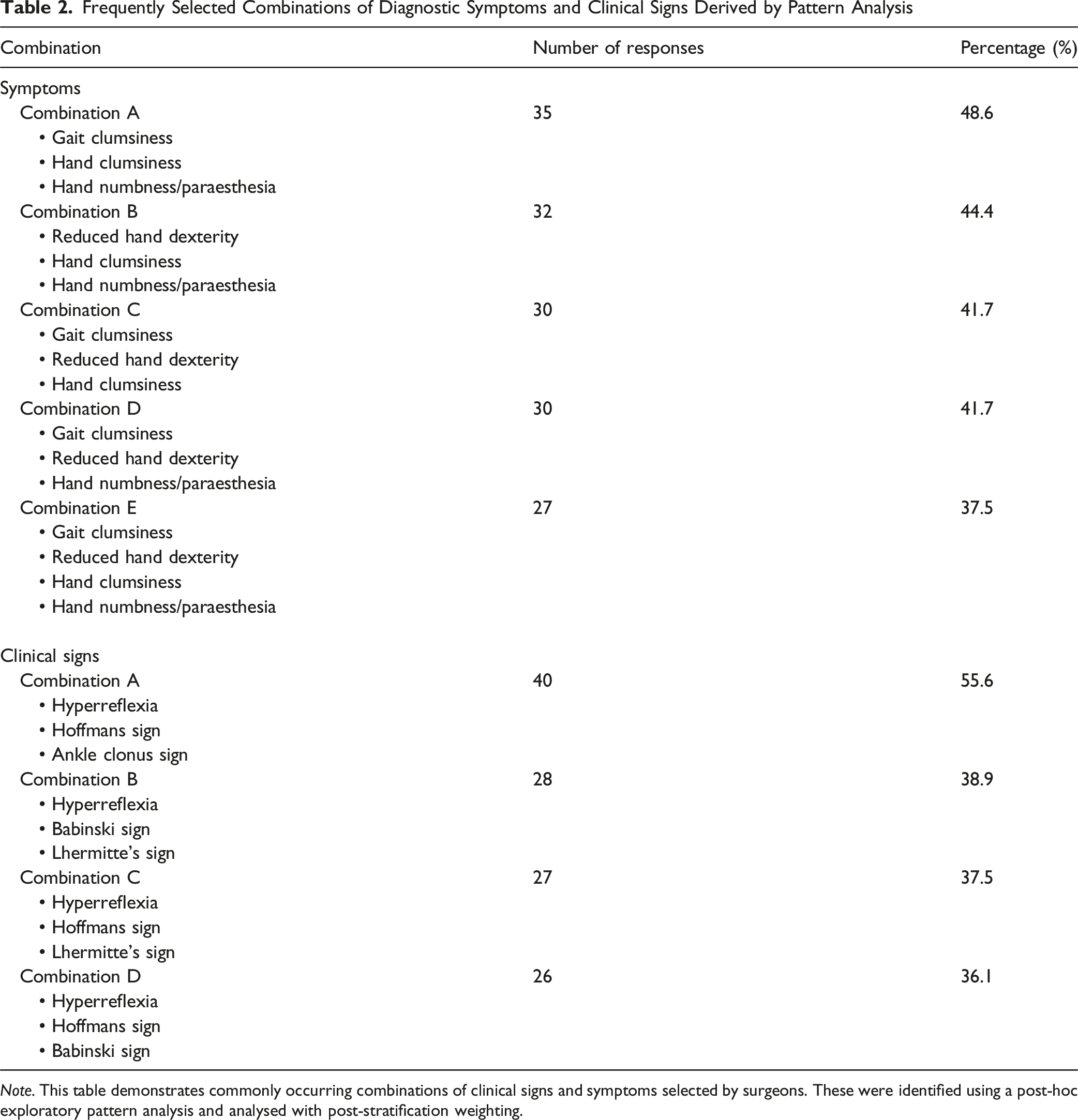
Figure 1 displays the frequency of responses for symptoms influencing both diagnosis and surgical decision-making for DCM. Gait clumsiness (selected by 86.1%) was the most frequently selected symptom informing the diagnosis of DCM, followed by hand clumsiness (76.4%), reduced hand dexterity (75.0%), hand numbness/paraesthesia (70.8%), and a history of falls or trips (37.5%). Pattern analysis identified 5 recurring symptom combinations; the most common, including gait clumsiness, hand clumsiness, and hand numbness/paraesthesia, was selected by 48.6% of respondents (Table 2). Most Selected Symptoms for the Diagnosis and Surgical Decision-Making for DCM. Note: The vertical bars in this figure represent the post-stratified percentages of surgeons who selected each symptom for informing diagnostic (blue bars) and surgical decision-making (orange bars). *Note: ‘Tremors’ was selected by one respondent (0.6%) for DCM diagnosis and is therefore not well-appreciated on the bar graph. ‘Gait clumsiness,’ ‘sexual dysfunction,’ ‘tremors’ and ‘cervical vertigo’ were not included as options for the question regarding surgical decision-making. DCM: degenerative cervical myelopathy Frequently Selected Combinations of Diagnostic Symptoms and Clinical Signs Derived by Pattern Analysis Note. This table demonstrates commonly occurring combinations of clinical signs and symptoms selected by surgeons. These were identified using a post-hoc exploratory pattern analysis and analysed with post-stratification weighting.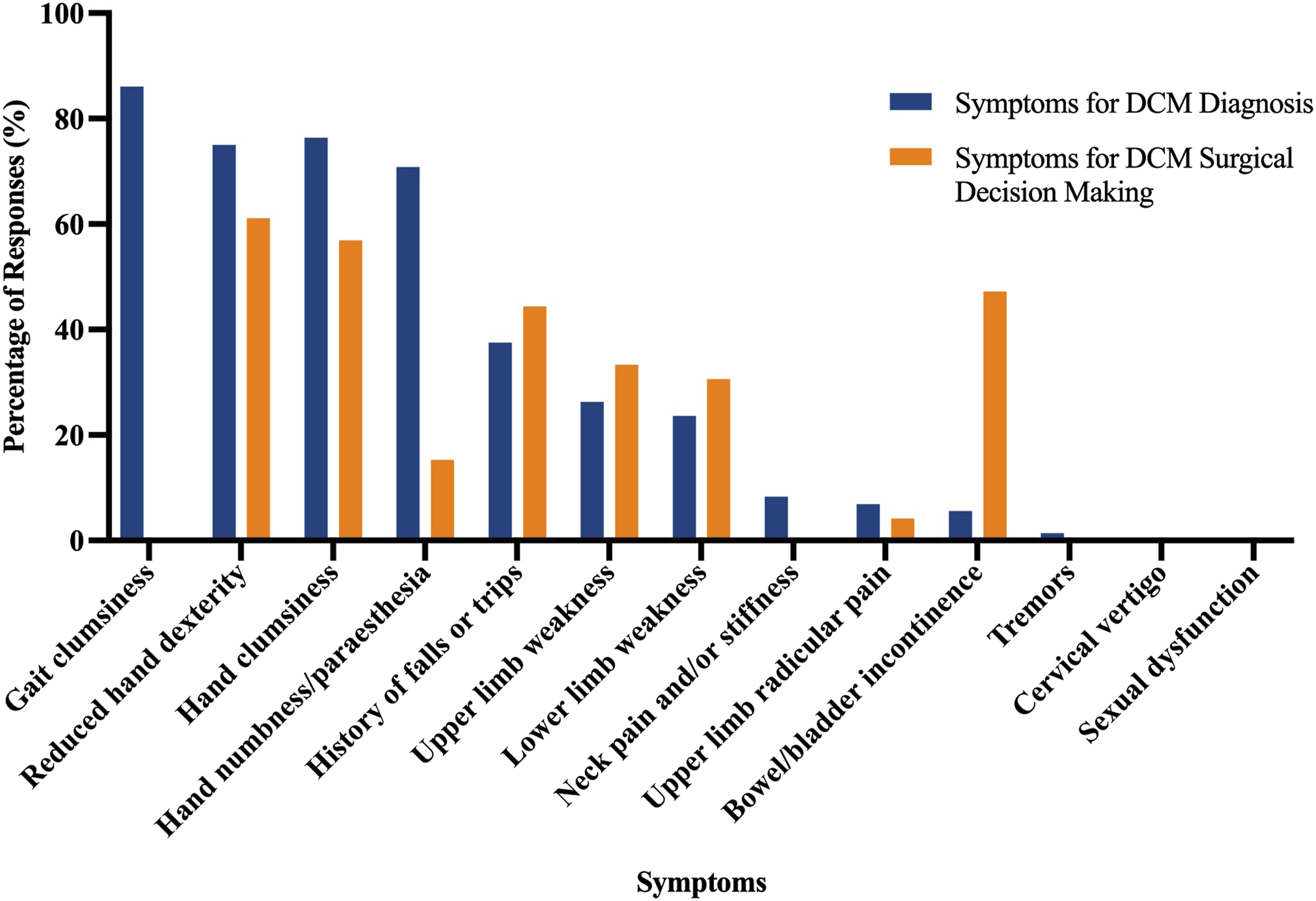
The symptoms most influential for surgical decision-making included hand dexterity decline (61.1%), hand clumsiness (56.9%), bowel or bladder incontinence (47.2%) and history of falls or trips (44.4%).
Clinical Signs Influencing Diagnostic and Surgical Decision-Making for DCM
Figure 2 shows the frequency of clinical sign selection for both diagnosis and surgical decision-making in DCM. Hyperreflexia (selected by 83.3%) was the most frequently selected clinical sign for diagnostic decision-making, followed by ankle clonus sign (68.1%), Hoffmann’s sign (66.7%), Babinski sign (61.1%), Lhermitte’s sign (48.6%), and inability to tandem gait (38.9%). Pattern analysis identified 4 recurring clinical sign combinations; the most common, including hyperreflexia, Hoffmann’s sign, and ankle clonus sign, was selected by 55.6% of respondents (Table 2). Most Selected Clinical Signs for the Diagnosis and Surgical Decision-Making for DCM. Note: The vertical bars in this figure represent the post-stratified percentages of surgeons who selected each clinical sign for informing diagnostic (blue bars) and surgical decision-making (orange bars) for DCM. *Note: Neck range of motion deficit’ was selected by 0.0% of respondents, reflected by the absence of a bar. DCM: degenerative cervical myelopathy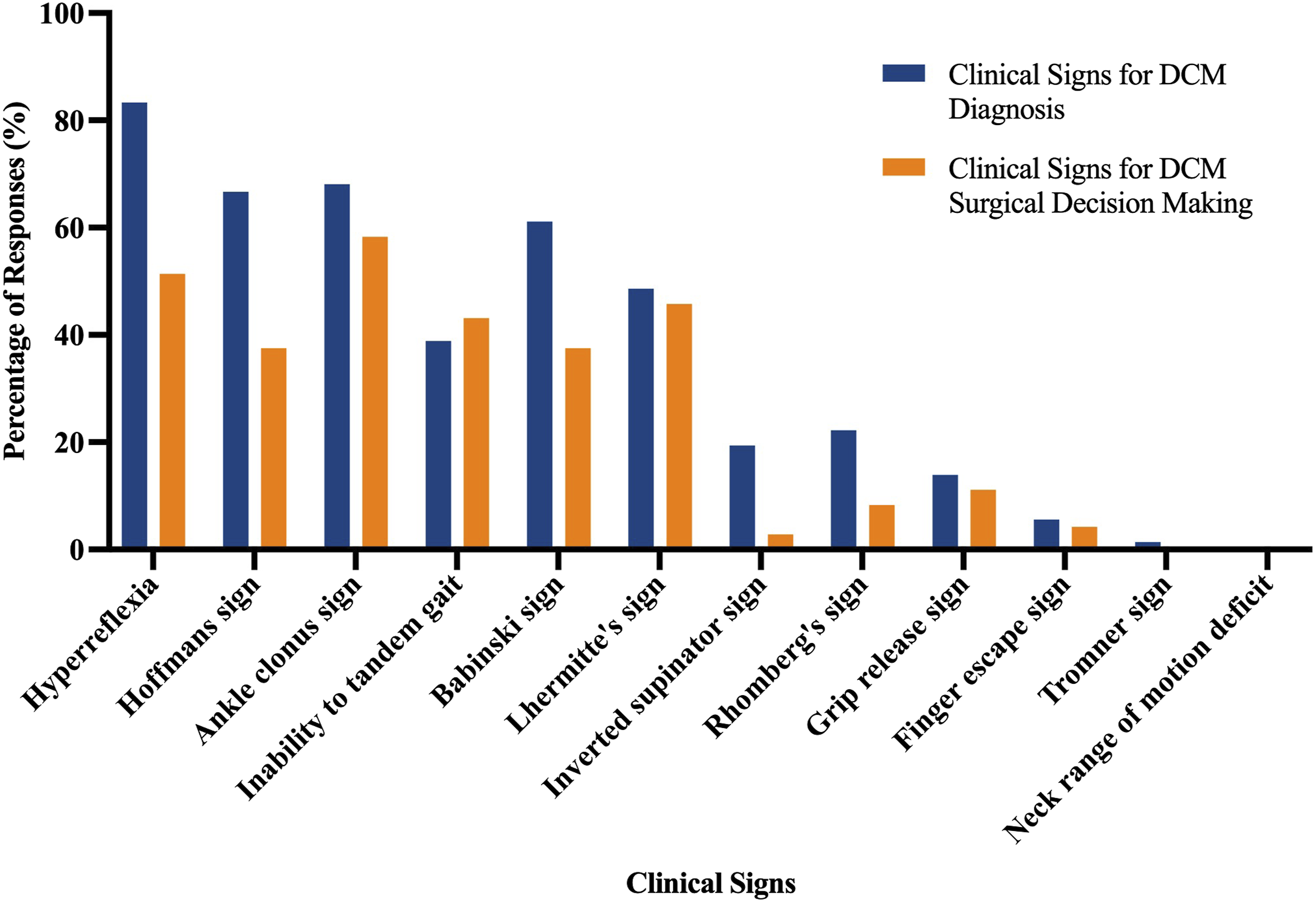
The clinical signs considered most influential for surgical decision-making included ankle clonus sign (58.3%), hyperreflexia (51.4%), Lhermitte’s sign (45.8%), inability to tandem gait (43.1%), Babinski sign (37.5%) and Hoffmann sign (37.5%).
Ranking of Variables Influencing Surgical Decision-Making for DCM
Ranked Importance of Variables Influencing Surgical Decision-Making for Degenerative Cervical Myelopathy
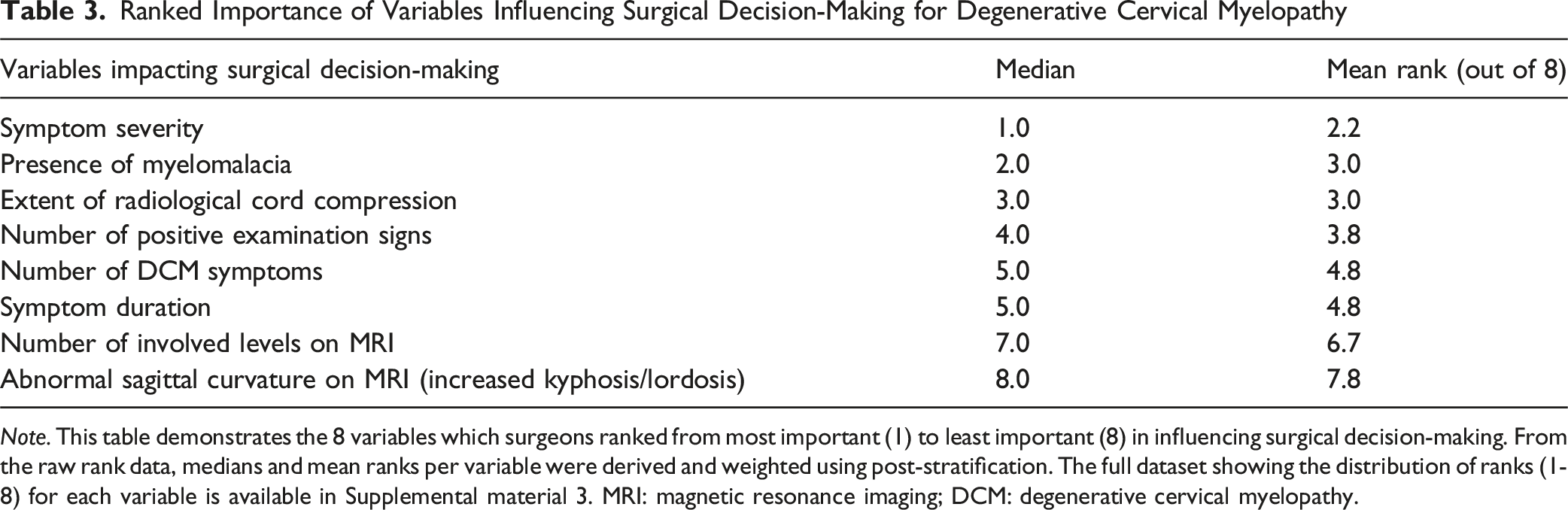
Note. This table demonstrates the 8 variables which surgeons ranked from most important (1) to least important (8) in influencing surgical decision-making. From the raw rank data, medians and mean ranks per variable were derived and weighted using post-stratification. The full dataset showing the distribution of ranks (1-8) for each variable is available in Supplemental material 3. MRI: magnetic resonance imaging; DCM: degenerative cervical myelopathy.
Clinical Vignettes on Surgical Decision-Making for DCM
Figure 3 presents responses to 5 clinical vignettes involving hypothetical patients aged >45 years with MRI-confirmed cervical cord compression. For Case 1 (asymptomatic cord compression, normal examination), 72.2% of respondents favoured serial observation, while only 5.6% recommended surgery. In Case 2 (asymptomatic cord compression with myelomalacia), the proportion recommending surgery increased markedly to 34.7%, with 48.6% opting for serial observation and 15.3% reporting insufficient information to decide. All other cases are presented in Figure 3 below. Responses to Clinical Case Vignettes on Surgical Decision-Making in the Presence of MRI-Confirmed Cervical Cord Compression (Unweighted). Note: The vertical bars represent the percentages of surgeons selecting one of the four management decision-making options, including: surgery (red), serial observation (orange), no surgery without follow up (yellow), insufficient information (grey). The clinical contexts for each case vignette are described as follows: case 1: asymptomatic cord compression (normal exam); case 2: asymptomatic cord compression with myelomalacia (normal exam); case 3: asymptomatic cord compression with hyperreflexia; case 4: asymptomatic cord compression with hoffmann’s sign; case 5: cord compression with bilateral hand paraesthesia (normal exam). Note: The vertically-oriented numbers above the bars represent the percentage of responses corresponding to the respective bar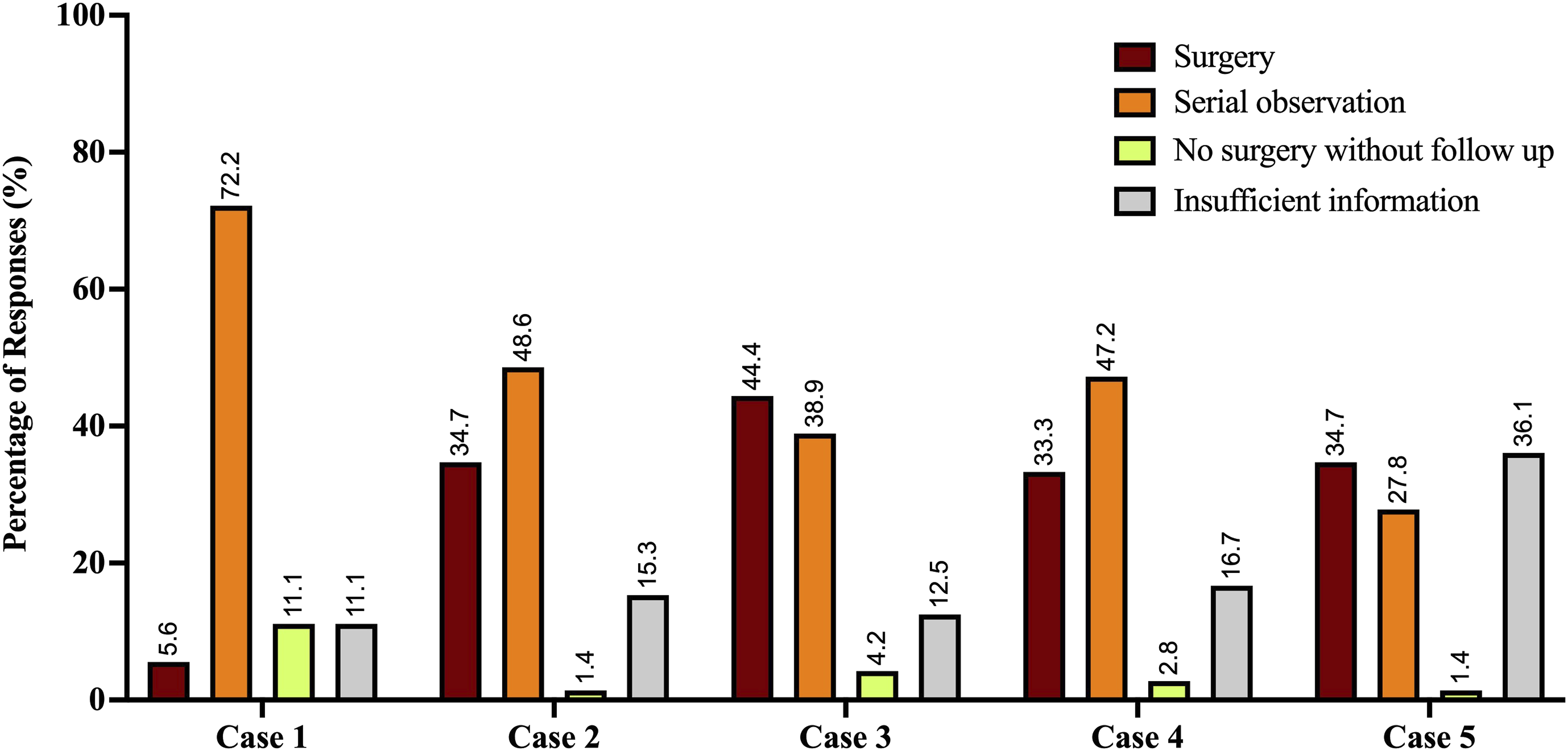
Discussion
This study provides detailed insights into the clinical features and variables guiding the diagnostic and surgical decision-making for DCM among Australasian orthopaedic spine surgeons and neurosurgeons. DCM, as a progressive and heterogeneous disease, poses diagnostic challenges, particularly in its milder and early stages.14,15 Given the emphasis on early identification and intervention, 5 it is crucial to identify the most important clinical features as they present to surgeons in secondary care settings, where DCM presentations are thought to be milder than tertiary settings.
Diagnostic Variability and Symptom Combinations
A key finding was the considerable variability in the selection of clinical signs, and to a lesser extent, symptoms, used by surgeons to inform the diagnosis of DCM. This heterogeneity was an expected finding given that DCM diagnosis rarely hinges on isolated findings but rather on a constellation of signs and symptoms interpreted in the context of MRI-confirmed spinal cord compression. 5
In our study, the most frequently selected symptoms for diagnostic decision-making were gait clumsiness, hand clumsiness, and reduced hand dexterity, each individually selected by over 75% of surgeons. The most frequently selected signs were hyperreflexia (selected by over 80%), ankle clonus and Hoffmann’s sign (both selected by over 60%). While no prior survey to our knowledge has directly assessed clinicians’ selection of these clinical features for DCM, a recent scoping review found substantial variability in the reported prevalence of gait dysfunction (10%-100%; based on 23 studies), hand clumsiness (26%-90%; 10 studies), and reduced hand dexterity (22%-71%; 5 studies) in individuals with DCM. 10 This variation likely reflects not only the heterogeneous presentation of DCM but may also reflect the ambiguity in how surgeons interpret these symptoms clinically.
To better understand how clinicians utilise clinical features in practice, we performed a post-hoc pattern analysis. This identified combinations of symptoms and signs that may represent unconscious clinical patterns used to inform DCM diagnosis. The most frequent combination of symptoms was gait clumsiness, hand clumsiness, and reduced hand dexterity. Based on pattern analysis of individual responses, nearly 50% of surgeons reported these 3 symptoms. The most frequent combination of clinical signs, selected by over 55% included, hyperreflexia, Hoffmann’s sign, and ankle clonus. While these combinations lack formal diagnostic validation, their identification reveals a shared clinical intuition among expert surgeons regarding hallmark features of DCM, particularly involving gait and hand function.
Notably, these findings reflect an emphasis on functional impairment in informing DCM diagnosis, which are presentations more characteristic of moderate-to-severe DCM as per the modified Japanese Orthopaedic Association score. This is surprising, given that the survey aimed to draw responses with relevance to secondary care practice, where DCM is presumed to be less advanced. A recent survey of 171 individuals living with DCM identified hand dexterity decline and clumsiness as the first presenting symptoms in only 3% and 5% of cases, respectively. 16 This discordance highlights the potential lag between early subjective symptom onset and the clinical features that trigger referral and diagnosis in specialist settings.
The Interplay Between Diagnosis and Surgical Decision-Making
Another key focus of this survey was the relationship between diagnostic assessment and surgical decision-making in DCM. This was of interest due to the universally accepted need for timely surgical intervention to prevent progressive neurological decline. 17 Based on this understanding, we expected that the clinical signs and symptoms used to diagnose DCM would largely overlap with those influencing the decision to proceed with surgery. While our findings partially support this (as shown in Figures 1 and 2), we also observed important differences. These differences highlight that the factors determining DCM diagnosis are not always the same as those that make a patient a suitable candidate for surgery, suggesting a more complex decision-making process than a simple overlap.
For instance, hand numbness/paraesthesia were identified as diagnostic symptoms by nearly 70.8% of surgeons, yet influenced surgical decision-making for only 15.3% of surgeons. In contrast, bowel and bladder dysfunction had a markedly greater influence on surgical intervention (47.2%) than on diagnosis (5.6%), reflecting its strong prognostic association with greater DCM severity, the risk of permanent autonomic dysfunction, and neurological compromise.10,18
A similar pattern was observed in the application of clinical signs. The Babinski sign was selected by 61.1% of respondents as a diagnostic clinical sign, yet it influenced surgical decision-making for only 37.5% of surgeons. Despite its high specificity (90%) for corticospinal tract dysfunction, its lower weighting in surgical decision-making is unexpected, particularly considering its established association with greater disease severity and worse postoperative outcomes.19,20
Additionally, despite a recent meta-analysis identifying the Tromner sign as having the highest diagnostic accuracy for DCM (sensitivity 94%, specificity 93%), 11 it was selected by fewer than 2% of respondents for diagnosis and not at all for surgical decision-making. This contrasts with the Hoffmann’s sign (66.7% diagnostic, 37.5% surgical), a neuro-mechanistically similar, yet more frequently recognised reflex abnormality, and highlights how surgeon’s examination practices remain influenced more by clinical familiarity than emerging diagnostic evidence.
Surgical Decision-Making for Asymptomatic Spinal Cord Compression – Clinical Vignettes
Deciding whether to perform surgery for asymptomatic spinal cord compression is a complex clinical challenge. 21 This decision requires a nuanced consideration of operative risk, the likelihood of neurological progression, long-term functional prognosis, unpredictability of the disease trajectory and the broader impact on patient quality of life.22,23
Findings from this study suggest a lack of consensus on the optimal management of asymptomatic cord compression, particularly in the context of isolated clinical signs. In clinical vignettes describing asymptomatic cord compression without myelopathic signs, over 70% of surgeons opted for serial observation, an approach consistent with current clinical practice guidelines. 17 However, decision-making became more divided in the presence of asymptomatic cord compression with accompanying myelomalacia, resulting in a near 30-percent point increase for offering surgical intervention (5.6% to 34.7%). Myelomalacia is a well-established radiological marker of chronic cord compression, ischemia, and apoptotic cell death, and is associated with poorer neurological outcomes.24,25 Yet in case 2 (asymptomatic cord compression with myelomalacia), nearly 50% of surgeons opted for serial observation, suggesting a reluctance to operate in the absence of overt clinical symptomology or impairment.
While these case vignettes were deliberately designed to pair radiological cord compression with individual clinical items that may be unreliable in isolation (eg, hyperreflexia, Hoffmann’s sign, bilateral hand paraesthesia), the wide variation in responses was striking. Such heterogeneity was unexpected given our survey sampled from a relatively small and regionally homogenous cohort of Australasian surgeons, who frequently share a common training background. As such, it is very possible that similar variability may exist and, in fact, be more profound if this study were replicated globally.
Implications for Early Detection and Referral Pathways
This study did not directly explore the reasons underlying variability in diagnostic and surgical decision-making. However, such variation is likely influenced by differences in clinical settings. 26 Tertiary centres typically manage advanced presentations of DCM, while secondary and primary care clinicians are more likely to encounter early-stage disease, where symptoms may be subtle or non-specific.2,27,28 Although survey responses were framed within a secondary care context, substantial heterogeneity in diagnostic criteria was still observed.
This is particularly concerning given that orthopaedic spine surgeons and neurosurgeons in New Zealand and Australia serve as the primary referral point for suspected DCM. 6 As such, one might anticipate greater consistency amongst these surgical specialists, given their greater exposure to DCM cases and research. If variability exists at the specialist level, it is likely to be more pronounced in primary care, where timely diagnosis is further compounded by low awareness and confidence.6,29 This raises concerns that early-stage cases may be missed or referred for diagnostic and surgical workup late, potentially delaying timely surgical intervention.2,3 Developing structured, consensus-based diagnostic and referral criteria is therefore an urgent priority to support early diagnosis and referral for timely intervention.5,27
Limitations
While this study benefited from the application of post-stratification weighting to mitigate potential sampling bias, several limitations are acknowledged. First, findings are based on self-reported practices, which are susceptible to recall bias and may not necessarily reflect real-world clinical reasoning, particularly given the dynamic nature of decision-making.
Second, some symptom terminology (eg, gait clumsiness) was undefined, potentially leading to inconsistent interpretation. While terms like “hand clumsiness” and “reduced hand dexterity” were clarified, ambiguity remains in how these are interpreted among surgeons. Notably, gait clumsiness was inadvertently omitted from the surgical decision-making survey item.
Third, the pattern analysis used to identify frequently co-selected features was descriptive and not based on algorithmic clustering methods; thus, observed groupings are hypothesis-generating and lack inferential validation.
Finally, clinical vignettes were deliberately simplified for feasibility, but therefore may not capture the full complexity of surgical decision-making.
Implications for Future Research
Future research should build on both diagnostic accuracy findings from prior systematic reviews and the practice trends identified in the present survey to develop tools that support timely recognition and referral of suspected DCM in primary care. Having a clearer understanding of the thresholds surgeons deem appropriate for diagnostic and/or surgical referral, while considering at-risk clinical groups (eg, age, ethnicity, co-existing lumbar spinal stenosis etc) to enhance the estimation of pre-test diagnostic probability, could facilitate appropriate primary care referrals and positively impact care trajectories for individuals with DCM.
Replicating this survey internationally would be valuable to clarify if the observed variability in surgical decision-making is consistent or perhaps even amplified in other geographic regions. Better understanding of regional surgical thresholds is important to inform the development of region-specific referral criteria and standardized care pathways.
In addition, adoption of clinical case vignettes with enhanced granularity could clarify the specific sources of variability informing surgical decision-making and guide consensus development around severity classification, surgical criteria and surgical timing, as well as helping to standardize approaches for serial observation in asymptomatic cord compression with and without myelomalacia.
Conclusion
This study identified considerable variability in how Australasian orthopaedic spine surgeons and neurosurgeons approach the diagnosis and surgical decision-making for degenerative cervical myelopathy, particularly in the use of clinical examination signs. While some common patterns emerged, the absence of clear consensus on key diagnostic features and the divergence between diagnostic reasoning and surgical thresholds highlight ongoing uncertainty and the need for standardization efforts. To optimize early diagnosis and referral for timely surgical assessment, consensus-driven diagnostic frameworks and referral pathways are urgently needed.
Supplemental Material
Supplemental Material - Diagnostic and Surgical Decision-Making for Degenerative Cervical Myelopathy: A Survey With Post-Stratification Analysis
Supplemental Material for Diagnostic and Surgical Decision-Making for Degenerative Cervical Myelopathy: A Survey With Post-Stratification Analysis by Rohil V. Chauhan, Anand H. Segar, Ashish D. Diwan, Nashwa Najib, Irene Zeng, David Rice, and Steven G. White in Global Spine Journal.
Supplemental Material
Supplemental Material - Diagnostic and Surgical Decision-Making for Degenerative Cervical Myelopathy: A Survey With Post-Stratification Analysis
Supplemental Material for Diagnostic and Surgical Decision-Making for Degenerative Cervical Myelopathy: A Survey With Post-Stratification Analysis by Rohil V. Chauhan, Anand H. Segar, Ashish D. Diwan, Nashwa Najib, Irene Zeng, David Rice, and Steven G. White in Global Spine Journal.
Supplemental Material
Supplemental Material - Diagnostic and Surgical Decision-Making for Degenerative Cervical Myelopathy: A Survey With Post-Stratification Analysis
Supplemental Material for Diagnostic and Surgical Decision-Making for Degenerative Cervical Myelopathy: A Survey With Post-Stratification Analysis by Rohil V. Chauhan, Anand H. Segar, Ashish D. Diwan, Nashwa Najib, Irene Zeng, David Rice, and Steven G. White in Global Spine Journal.
Footnotes
Acknowledgements
The authors would like to sincerely thank the surgeons who responded to the survey for their time and contributions to this study. We also acknowledge the support of the New Zealand Orthopaedic Spine Society, the Neurosurgical Society of Australasia, and the Spine Society of Australia for their assistance in survey dissemination.
Ethical Considerations
Ethical approval for the questionnaire was obtained from the Auckland University of Technology Ethics Committee (23/351).
Author Contributions
RVC – Conceptualisation, Project administration, Methodology, Data curation, Formal analysis, Writing – original draft preparation.
AHS – Conceptualisation, Resources, Project administration, Supervision, Writing – reviewing and editing.
IZ – Methodology, Formal analysis, Writing – reviewing and editing.
NN – Participant recruitment, Writing – reviewing and editing.
ADD – Participant recruitment, Writing – reviewing and editing.
DR – Visualisation, Writing – reviewing and editing.
SGW– Conceptualisation, Resources, Supervision, Visualisation, Writing – reviewing and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported, in part, by a contract from the Health Research Council of New Zealand.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Co-authors RVC, ADD, NN, IZ, DR and SGW have no conflicting interests to disclose. Co-author AHS is a consultant for, and receives unrelated research funding from ATEC Spine.
Data Availability Statement
Raw weighted and unweighted data are available in Supplemental material 3.
Supplemental Material
Supplemental material for this article is available online.
