Abstract
Study Design
Narrative Review.
Objectives
The objective of this review is to provide a stepwise approach to the assessment of patients with potential symptoms of degenerative cervical myelopathy (DCM).
Methods
N/A
Results
DCM is an overarching term used to describe progressive compression of the cervical spinal cord by age-related changes to the spinal axis. These alterations to normal anatomy narrow the spinal canal, reduce the space available for the spinal cord, and may ultimately compress the ascending and descending neural tracts. Patients with DCM present with a wide range of symptoms that can significantly impact quality of life, including bilateral hand numbness and paresthesia, gait impairment, motor weakness of the upper and lower extremities, and bladder and bowel dysfunction. Unfortunately, DCM is often misdiagnosed, resulting in delayed assessment and management by the appropriate specialist. The proper evaluation of a patient with suspected DCM includes obtaining a detailed patient history, conducting a comprehensive neurological examination, and ordering appropriate tests to rule in or out other diagnoses.
Conclusion
This review summarizes a stepwise approach to the diagnosis of patients with DCM.
Keywords
Introduction
Degenerative cervical myelopathy (DCM) is an overarching term used to describe progressive compression of the cervical spinal cord by age-related changes to the spinal axis. 1 These changes include facet arthropathy, spondylosis, and disc degeneration; subluxation of the vertebral bodies; and hypertrophy, ossification, or calcification of the supporting ligaments. 2 These alterations to the normal anatomy narrow the spinal canal, reduce the space available for the spinal cord, and may ultimately compress the ascending and descending neural tracts. 3 In addition to static spinal cord compression, hypermobility and instability of the spinal column may cause chronic, repetitive injury to the neural elements. Injury to the spinal cord, whether through static or dynamic mechanisms, initiates a series of pathological events, including vascular changes, neuroinflammation, disruption of the brain–spinal cord barrier, and apoptosis.4,5 These cellular events subsequently result in demyelination, neuronal loss, and astrogliosis.
In a systematic review of the literature, Boogaarts and Bartels 6 (2013) were unable to identify studies discussing the incidence or prevalence of DCM. They did, however, estimate that 1.6 per 100, 000 individuals in their area were operated on for symptomatic cervical spondylotic myelopathy. Similarly, in a study by Kokubun et al 7 (1996), the annual rate of surgical intervention for individuals with DCM was 5.7 per 100, 000 residents in a northeastern region of Japan (estimated population 2.6 million). Finally, Nouri et al 3 (2015) estimated the incidence and prevalence of DCM to be at least 41 and 605 per million, respectively, in North America. Although the exact incidence and prevalence of DCM are unknown, it is anticipated that, with an aging population, clinicians worldwide will be expected to manage an increasing number of patients with degenerative spine disease. 8 As a result, there needs to be an improved understanding of the diagnosis, assessment, and monitoring of this condition.
DCM is often misdiagnosed, resulting in delayed assessment and management by the appropriate specialist. In a systematic review by Karadimas et al 9 (2013), moderate evidence suggested that approximately 20 to 62% of patients with DCM exhibit clinical deterioration by 3 to 6 years if not managed surgically. Given the potential for disease progression, a diagnosis must be made promptly and patients should be referred in a timely fashion to an appropriate specialist. In a study by Behrbalk et al 10 (2013), the mean time to diagnosis of DCM was 2.2±2.3 years (range 1.7 months to 8.9 years) after the first physician visit. Furthermore, patients, on average, attended 5.2±3.6 physician visits with DCM-related complaints before obtaining a diagnosis. DCM was commonly mistaken for carpal tunnel syndrome and cervical radiculopathy without neurological deficit. Finally, a study by Radcliff et al 11 (2016) identified a higher incidence of undiagnosed cervical myelopathy among patients with hip fractures compared to a control population.
The objectives of this review are to provide a stepwise approach to the assessment of patients with potential symptoms of DCM.
Assessment of Patients With Potential Symptoms of DCM
A proper assessment of a patient with suspected DCM includes obtaining a detailed patient history, conducting a comprehensive neurological examination, and ordering appropriate tests to rule in or out other diagnoses. This section will summarize a stepwise approach to the evaluation of patients with potential symptoms of DCM.
Obtain a Detailed Patient History
A Summary of Important Open-Ended and Specific Questions to Ask a Patient with Suspected Degenerative Cervical Myelopathy.

Distinguishing Differential Diagnoses of Degenerative Cervical Myelopathy through Medical History.

Perform a Comprehensive Physical Examination
A physical examination that assesses motor and sensory function, coordination, reflexes, and tone is required to identify and localize problems of the nervous system. A complete neurological examination also consists of assessment of the cranial nerves and mental status; however, these components are not relevant to the evaluation of suspected DCM and will not be discussed in this article. They must be considered in cases where other potential diagnoses should be excluded.
Relevant Components of the Neurological Examination in Patients with Suspected Degenerative Cervical Myelopathy.

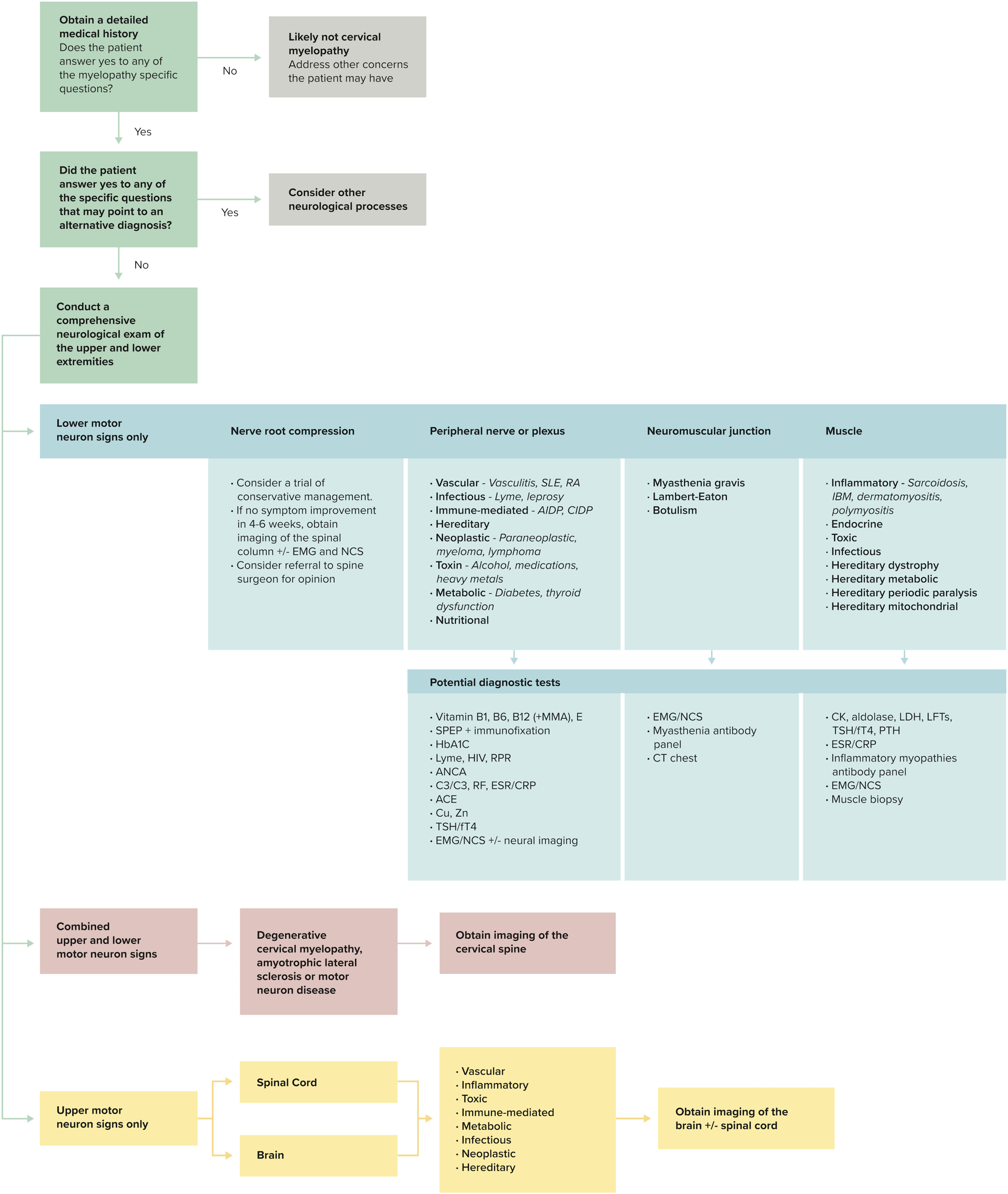
An approach to the diagnosis of degenerative cervical myelopathy. EMG, electromyography; NCS, nerve conduction studies; SLE, systemic lupus erythematosus; AIDP, acute inflammatory demyelinating polyneuropathy; CIDP, chronic inflammatory demyelinating polyneuropathy; IBM, influsion body myositis; SPEP, serum protein electrophoresis; HIV, human immunodeficiency virus; RPR, rapid plasma reagin; ANCA, antineutrophil cyoplasmic antibody; C3/C4, complement 3/4; RF, rheumatoid arthritis; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; ACE, angiotensin-converting enzyme; Cu, copper; Zn, sinc; TSH, thyroid stimulating hormone; fT4, free T4; CT, computed tomography; CK, creatinine kinase; LDH, lactate dehydrogenase; LFTs, liver function tests; PTH, parathyroid hormone.
Acquire Imaging of the Cervical Spinal Axis and Cord
If DCM is suspected from a patient’s history and physical examination, the next best step is to acquire imaging of the cervical spinal axis and cord (Figure 2). The role of various imaging modalities in the diagnosis and management of degenerative cervical myelopathy. (i) Lateral radiograph for the assessment of cervical alignment; (ii) flexion-extension radiographs for evaluation of spondylolisthesis and cervical instability; (iii) post-operative radiographs for assessment of fusion status and the position of instrumentation; (iv) computed tomography scans for the detection of bony abnormalities and ossification of the posterior longitudinal ligament; and (v) magnetic resonance imaging for the evaluation of the extent of spinal canal stenosis, degree of cord compression, and presence of intramedullary signal change.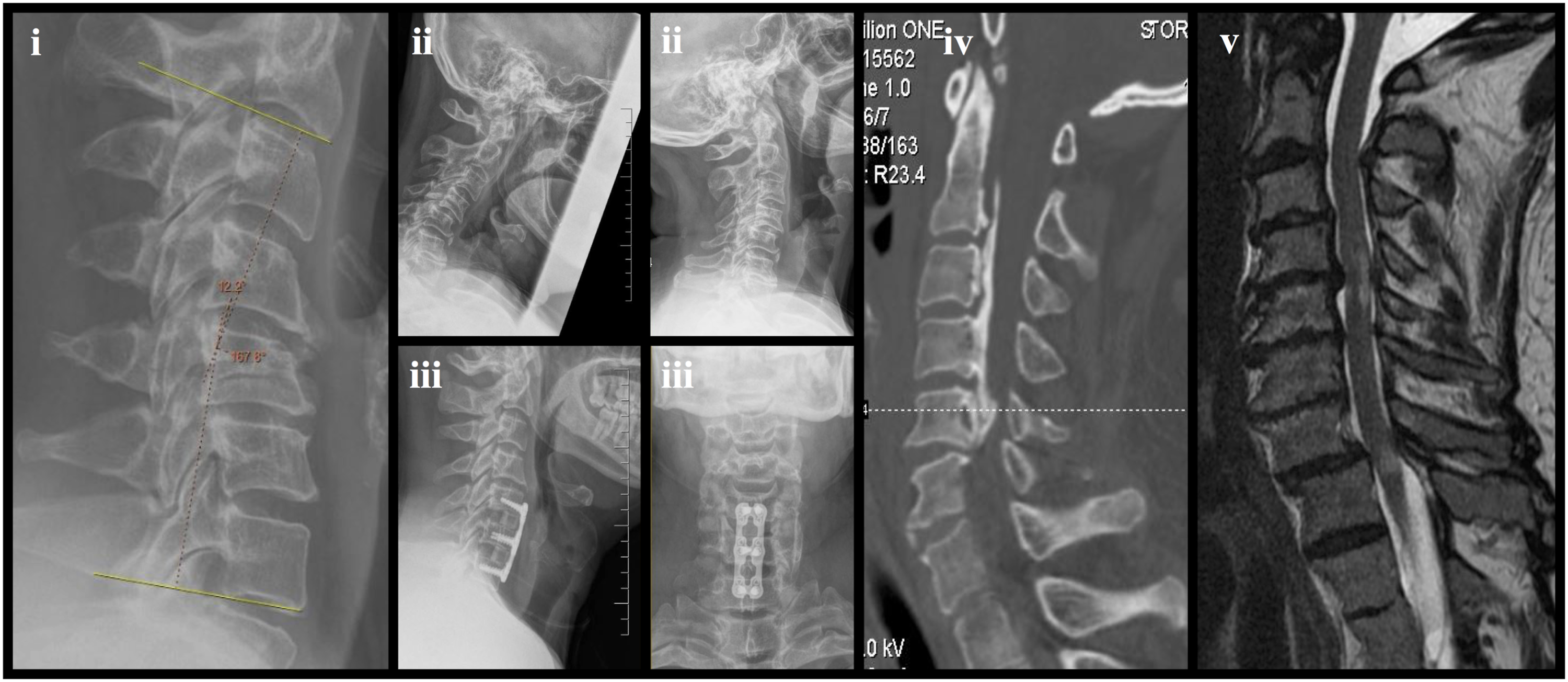
Plain Radiographs
Plain radiographs are generally the initial imaging modality for evaluating a patient with suspected DCM. 1 Although radiographs cannot visualize the spinal cord, they provide versatile two-dimensional views of the spinal axis that can depict various sources of pathology. 14 For example, lateral radiographs are useful for evaluating spinal alignment and disc spaces. The swimmer’s view can be obtained if C7 to T1 cannot be visualized on a normal lateral image. 15 Anterior–posterior radiographs are useful for identifying uncovertebral joint spurs and assessing the vertebral bodies, intervertebral spaces, and pedicles. Finally, right and left oblique views display the neural foramina as well as uncovertebral and facet joints.
Radiographs provide useful information on bone quality, cervical alignment, and can identify osteophytes, spondylosis as well as ossification of the posterior longitudinal ligament. 14 Spinal alignment is best evaluated under load-bearing conditions (eg, while sitting or standing), which cannot be achieved in traditional computed tomography or magnetic resonance imaging (MRI) scanners. 14 Specific measurements of cervical alignment include cervical lordosis, sagittal plane translation, and horizontal gaze. Cervical lordosis is assessed by computing the C1 or C2 to C7 Cobb angle; this is done by measuring the angle between two lines parallel to the C1 or C2 and the C7 inferior plate. 16 According to Batzdorf and Batzdorff, a patient with ideal cervical alignment will have no portion of the C3 to C6 vertebra cross a line drawn from the posteroinferior aspect of the C2 vertebral body to the posterioinferior aspect of the C7 vertebral body. 16 Sagittal plane translation is evaluated by computing the distance between the C2 or C7 plumb line and the posterior superior corner of the sacrum. 16 Regional cervical sagittal alignment is often computed by drawing a plumb line from the center of C2 to the posterior superior aspect of C7. Horizontal gaze is also a useful measure of cervical lordosis and is assessed by the chin-brow vertical angle. 17 This angle is measured between a vertical line from the forehead and a line drawn between the eyebrow and chin. Finally, T1 slope is the angle between a line along the superior endplate of T1 and a horizontal reference line. 16 Long cassette or three-feet standing radiographs may also be used to assess global spinal alignment as pathology below the cervical region often impacts cervical alignment. 18
Radiographic views in flexion and extension postures can help depict spondylolisthesis as well as cervical instability and be used to calculate sagittal range of motion. 14 This information is essential for surgical planning, can influence the surgical approach, and help decide whether fixation is necessary. Finally, post-operative radiographs are useful for assessing fusion status and the position of bone grafts, pedicle screws, cages, and plates. 14 Postoperative films can also be compared to preoperative images to evaluate correction of cervical alignment and issues with instability.
Computed Tomography
Computed Tomography (CT) provides three-dimensional images of the spinal axis and can help detect bony abnormalities and ossification of ligamentous structures. Unfortunately, in CT images, there is usually little contrast between the cervical spinal cord and subarachnoid space, preventing visualization of intradural processes, intramedullary lesions, and cord compression. CT scans, however, can be used to classify the type of OPLL, determine the extent of the ossification, and assess bone quality. 19 They are also used to measure screw trajectories if instrumentation is required, navigate intraoperatively, and assess the location of metallic hardware such as pedicle screws and plates. 14 CT is the imaging modality of choice in patients who cannot undergo MRI due to ocular metallic foreign bodies, pacemakers, stimulators, embedded wires, aneurysm clips, nitroglycerin patches, or severe claustrophobia. 20 Finally, although not routine practice, CT angiography can visualize anomalies of the vertebral arteries and identify cases with a higher risk of injury. 21
Magnetic Resonance Imaging
MRI is the preferred modality for assessing patients with DCM as it provides high resolution, multiplanar images of the neural, soft-tissue, and bony structures. 22 Furthermore, MRI can clearly depict the extent of spinal canal stenosis and the presence of cord compression, intramedullary signal changes, and bony constriction of the neural foramina. 23 Finally, MRI is useful to exclude other disease processes of the cervical spine that may clinically present like DCM, such as neoplastic infiltration, demyelinating plaques, and syringomyelia.
A Summary of Classification Systems Used to Qualify and Quantify the Extent of Degenerative Changes on Magnetic Resonance Imaging in Patients with Degenerative Cervical Myelopathy.

SI, signal intensity; CSF, cerebrospinal fluid; MCC, maximum canal compromise; MSCC, maximum spinal cord compression; T2WI, type 2-weighted images; T1WI, type 1-weighted images.
Advanced Imaging Techniques
Although conventional MRI has revolutionized the ability to visualize the neural elements, it still has its limitations. Specifically, certain MRI findings often do not correlate with a patient’s clinical presentation or disease severity. Several advanced imaging techniques are available that quantify physical properties of neural tissues and better detect injury to the axons and myelin of the white matter tracts. 14 These include diffusion tensor imaging (DTI), magnetization transfer (MT), myelin water fraction (MWF), and MR spectroscopy. Metrics derived from these modalities are more sensitive at detecting subclinical tissue injury and myelopathy progression than conventional MRI and may be more accurate at predicting surgical outcomes.
Electrophysiology
Electrophysiological tests are not routinely used to diagnose patients with DCM. 1 However, they can assess the functional involvement of the spinal cord and help bridge the gap between MRI findings and a patient’s clinical picture. Furthermore, neurophysiological tests are valuable for excluding other mimicking diagnoses, identifying the presence of concomitant radiculopathy, and monitoring disease progression. 25
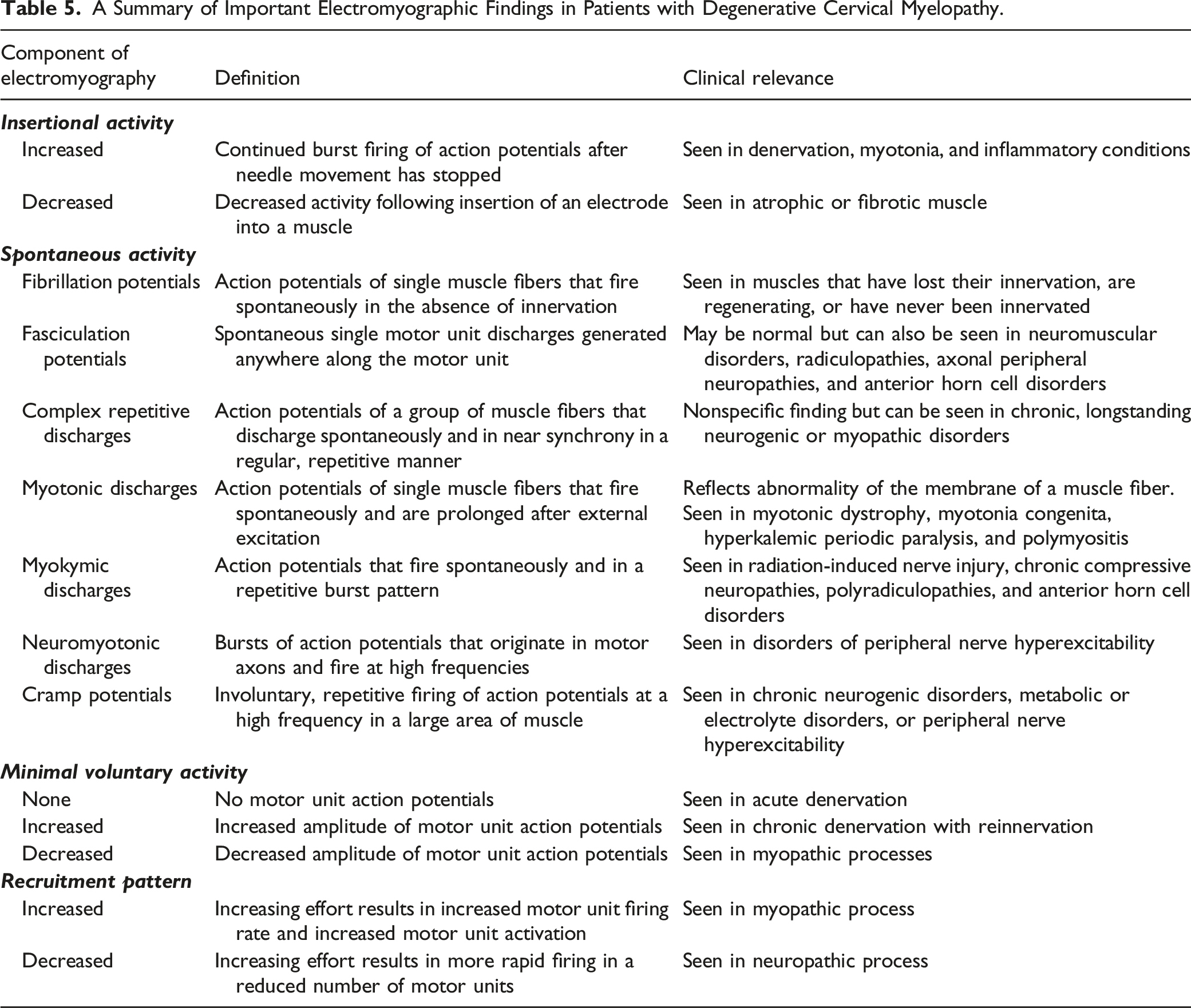
Electromyography (EMG) is the study of the electrical activity of muscles and is used to evaluate the integrity of the motor unit. EMG assesses insertional activity, spontaneous activity of the muscle at rest, the motor unit action potential (MUAP) during minimal voluntary contraction, and the recruitment of motor units during increasing voluntary contraction. Increased electrical activity following insertion of an electrode into a muscle is abnormal and is commonly seen in denervation, myotonia, and inflammatory conditions. 26 In contrast, a decrease in insertional activity may reflect atrophic and/or fibrotic muscle. In terms of spontaneous activity at rest, fibrillations and positive sharp waves occur in cases of denervation, typically 10 to 14 days after acute nerve injury. 26 Fasciculations can be a normal finding but can also be seen in motor neuron disease, radiculopathies, entrapment neuropathies, and metabolic disorders. 26 Myokymia or bursts of MUAPs can occur normally in the orbicularis oculi or oris muscles but may also be present in radiculopathies, radiation plexopathies, and anterior horn cell syndromes. 27 Voluntary activity can be used to distinguish acute and chronic patterns of denervation as well as neuropathies from myopathies. 26 Individuals with complete denervation will have no MUAP, whereas those with chronic denervation will have increased MUAP amplitude. This increased amplitude is because reinnervation following nerve damage results in an increased number of muscle fibers controlled by a single motor unit. 26 Myopathic MUAPs, in contrast, have decreased amplitude which helps distinguish myopathic from neuropathic processes. Finally, a pattern of reduced recruitment is observed in neuropathic disease. In contrast, the force generated by individual motor units is reduced in myopathic disease; a greater number of units must therefore be recruited in order to produce a certain level of force.
A Summary of Important Electromyographic Findings in Patients with Degenerative Cervical Myelopathy.
Nerve conduction studies are performed to quantify the motor and sensory velocities of peripheral nerves. Two electrodes are first placed at two different points along a peripheral nerve. Conduction velocities are then computed by dividing the time from the onset of stimulus to the recorded response by the distance between the two electrodes. 30 Nerve conduction studies are typically normal in patients with DCM and compressive radiculopathies but are often slowed in peripheral nerve entrapment, peripheral neuropathies, and brachial plexopathies. 31 As such, nerve conduction studies may be useful in distinguishing DCM from common mimickers, especially carpal tunnel syndrome.
Somatosensory evoked potentials (SEPs) can identify injury anywhere along the dorsal column-medial lemniscal pathway. 32 This pathway is initiated by stimulating the large myelinated afferent fibers in a peripheral nerve. These nerves carry information about proprioception, vibration, and fine touch into the dorsal root ganglion where the cell bodies are located. The axons then travel ipsilaterally through the dorsal columns and synapse on second order neurons in the dorsal column nuclei in the medulla oblongata. Axons of the second sensory neuron desiccate as internal arcuate fibers and form the medial lemniscus which ascends to the thalamus. A third and final sensory neuron travels from the thalamus to the somatosensory cortex. The spinal potential N13 reflects the activity of the dorsal horn neurons that receive their inputs from the afferent sensory fibers. 31 The loss of spinal N13 is found in patients with DCM and is considered a reliable diagnostic tool. Specifically, in a study by Restuccia et al 33 (1992), abnormalities in the N13 potential were found in 95% of radial, 90% of medial, and 54% of ulnar nerve SEPs in 11 patients with image-evidence of DCM.
Motor-evoked potentials (MEPs) can detect injury to the descending motor pathways following transcranial magnetic stimulation of the motor cortex. In patients with DCM, MEPs can help localize the levels of motor dysfunction and detect subclinical involvement of central motor pathways. Furthermore, MEPs can help distinguish DCM from ALS. Specifically, patients with DCM tend to have slower central motor conduction times in myelomeres distal to the level of spinal cord compression, whereas patients with ALS can have normal conduction times in caudal myelomeres. 31 SEPs and MEPs may also be useful in predicting disease development in individuals with image evidence of spinal cord compression or canal stenosis but no symptoms of myelopathy. Specifically, according to Bednarik et al 34 (2008), nonmyelopathic patients with evidence of spondylosis or disc compression of the cervical spinal cord were more likely to develop myelopathy if they had abnormal MEPs and SEPs. Finally, SEPs and MEPs can be used to quantify sensory and motor improvements following surgery and are useful for detecting neurological injury intraoperatively. 35
Footnotes
Acknowledgments
MGF would like to acknowledge support from the Gerry and Tootsie Halbert Chair in Neural Repair and Regeneration.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
