Abstract
Study Design
Systematic Review and Meta-Analysis.
Objectives
Degenerative cervical myelopathy (DCM), the leading cause of spinal cord injury globally, may clinically manifest with hand numbness and tingling, loss of hand dexterity and impaired balance and gait. Biomarkers have been identified as disease indicators with recent studies exploring the role of structural and inflammatory biomarkers in diagnostic and treatment outcome prediction. The review aims to identify relevant cerebrospinal fluid (CSF) and blood-based biomarkers associated with DCM diagnosis and prognostication.
Methods
The systematic review was conducted as per PRISMA guidelines. A comprehensive search strategy was devised to identify articles evaluating biomarkers in DCM and comparative control cohorts. The studies were screened by two independent reviewers and discrepancies resolved by a third author. Study characteristics were recorded, and data synthesis was performed to allow for pooled analyses. The quality of included studies and risk of bias were assessed using the QUADAS-2 tool.
Results
A total of 17 studies incorporating 699 individuals with DCM and 475 controls were included. 9 studies evaluated CSF biomarkers while 6 and 2 studies reviewed blood-based and CSF-to-serum quotients, respectively. Pooled analyses revealed significantly higher levels of NF-L and IL-6, albumin-Q, IgA-Q, and IgG-Q in the DCM cohort as compared to the control group.
Conclusion
This systematic review highlights the structural and inflammatory biomarkers that can be potentially used as substitutes for diagnosing and prognosticating outcomes for DCM. The findings further suggest the need for larger prospective studies to assess the clinical relevance of these biomarkers in DCM management.
Keywords
Introduction
Degenerative cervical myelopathy (DCM) is the leading cause of spinal cord impairment worldwide.1,2 DCM is a progressive condition characterized by a wide range of neurological symptoms that develop due to spinal cord compression. Compression may be caused by several pathologies including disc degeneration, spondylosis, and ossification of supporting ligaments. 3 Common symptoms include numbness and tingling of the hands, which can progress to loss of dexterity, making it difficult for patients to perform fine motor tasks such as buttoning clothes or writing. Affected individuals may also experience arm or leg weakness, leading to difficulties with balance and frequent falls. 4 Gait disturbances including an unsteady, wide-based walking pattern are often reported, reflecting compromised coordination and proprioception. 5 Pain in the neck, shoulders, or radiating pain extending into the arms may also occur. In advanced stages, bladder and bowel dysfunction can present due to autonomic pathway involvement. 4 Early detection of these symptoms is critical to minimize irreversible damage, as prompt intervention may prevent the worsening of neurological deficits. Despite the prevalence of DCM, many patients experience a delay of two or more years before receiving an accurate diagnosis, which often results in postponed treatment and worsened outcomes.6,7 Early detection is thus essential, as delayed intervention can lead to irreversible neurological impairment.8,9 DCM is primarily treated with decompression surgery, which in most cases halts disease progression and may even significantly improve symptoms.10-12
A biomarker can be defined as an “objective indication of medical state observed from outside the patient – which can be measured accurately and reproducibly”. 13 Recent advancements in biomarker research have shown great promise in enabling earlier identification of medical conditions, such as cancer, sepsis, and concussion.14-17 Biomarkers also offer the potential for a more personalized approach to treatment, enabling clinicians to offer therapeutic decisions based on unique patient-specific characteristics. Certain unique biomarkers may be recognized in DCM, owing to the underlying pathophysiological mechanisms of the disease. Neuroinflammation induced by the mechanical compression of the tissues and vasculature results in an increase in inflammatory markers, like IL-6 (interleukin-6). 18 Neuronal tissue damage may also result in the release of structural biomarkers in the cerebrospinal fluid (CSF) and peripheral vascular system, such as neuron specific enolase (NSE). Further, the dynamic compression caused by the movement of the head and neck can cause variable spinal cord compression and irritation.3,4 Altogether, these parallel mechanisms can result in a unique DCM biomarker profile that is specific to each patient’s symptomatology. 19
While proposed DCM biomarkers to date are primarily imaging-based,12,20 our goal in this systematic review is to identify fluid-based biomarkers that can also be used as reliable diagnostic and/or prognostic tools. In our analysis, we include all studies that assess the relevance of blood- and CSF-based biomarkers. Our results will help prioritize ongoing biomarker research and thus will potentially accelerate clinical translation by identifying specific candidate biomarkers that require further validation.
Methods
This systematic review and meta-analysis study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. 21 No ethical approvals were required for this study.
Search Strategy
The aim of the study was to identify the complement of relevant fluid-based biomarkers that can be potentially used for DCM diagnosis and outcome prediction. A comprehensive search strategy was developed, incorporating the synonyms of search terms “degenerative cervical myelopathy”, “spondylotic myelopathy”, “spinal cord compression”, “biomarker*”, “serum”, and “cerebrospinal fluid*”. The databases searched included PubMed, Embase, and Web of Science. When available, relevant controlled vocabulary was also implemented, including MeSH (Medical Subject Headings) for PubMed and Emtree terms for Embase. Study language was restricted to English, but publication dates were not limited. The initial literature search was completed on September 30, 2024. Full search strategies for each database are presented in Supplementary Material.
Study Selection
Two independent reviewers (HA and CW) performed initial title and abstract screening, followed by full-text screening based on the study inclusion and exclusion criteria. The inclusion criteria included all human observational studies that evaluated either quantitative serum, blood, or CSF biomarkers in patients with DCM as compared to a control group regardless of the year of publication. Non-human studies, studies focusing on imaging biomarkers, and publications without quantitative data such as case reports, case series, reviews, technical notes, editorials, and correspondence were all excluded. Additionally, references identified from the included studies that met the eligibility criteria were also reviewed and incorporated. Disagreements regarding potential study eligibility were resolved by a third reviewer (HD).
Data Extraction and Outcome Measures
Baseline study data, including author name, year of publication, sample size, study design, and patient demographics were extracted and recorded by the reviewers. In addition, the types of biomarkers assessed, pre-operative biomarker levels in individuals with DCM and in controls, and any reported correlations between pre-operative biomarker levels and clinical outcomes were documented.
Quality Assessment
The quality assessment for the included studies was conducted using the QUADAS-2 tool, 22 which evaluates the risk of bias and applicability concerns across four key domains: patient selection, index measure, reference standard, and flow and timing. The two reviewers independently performed the assessments, with discrepancies resolved through discussion. Each domain was categorized as having a low, high, or unclear risk of bias.
Statistical Analysis
Descriptive statistics were employed to summarize the study variables; measures of central tendency were used for continuous variables while frequencies and proportions were used for categorical variables. Data synthesis was conducted after extraction of biomarker levels from the included studies. Ratios comparing biomarker concentrations between the DCM and control cohorts were calculated where ratios >1 indicated higher biomarker levels in the DCM cohort and ratios <1 signified higher biomarker levels in the control cohort. For studies stratifying subjects based on symptom severity or duration, DCM subgroup biomarker levels were individually compared with control subjects, generating multiple ratios per study. Meta-analyses were conducted using the DerSimonian and Larid random effects method and forest plots were generated to display effect estimates. Heterogeneity was assessed using I2 statistic, with thresholds of <50%, 51%–75% and >75% indicating low, moderate, and high heterogeneity, respectively.23,24 The alpha level was set at 0.05. All statistical analyses were performed using R Studio 4.2.3. 25
Results
Literature Search
A total of 3420 abstracts were initially retrieved from the database registries. After excluding duplicates (n = 1086), the remaining articles (n = 2334) underwent title and abstract screening. As a result, 2248 studies were deemed ineligible and were excluded. The remaining 86 articles underwent full-text screening, which resulted in further exclusion of 69 articles. Reasons for exclusion are shown in Figure 1. As such, 17 studies were included in the final analysis.18,19,26-40 PRISMA Flowchart Depicting the Abstract and Full-Text Screening Stages and Final Inclusion of 17 Studies From the Initially Identified 3420 Abstracts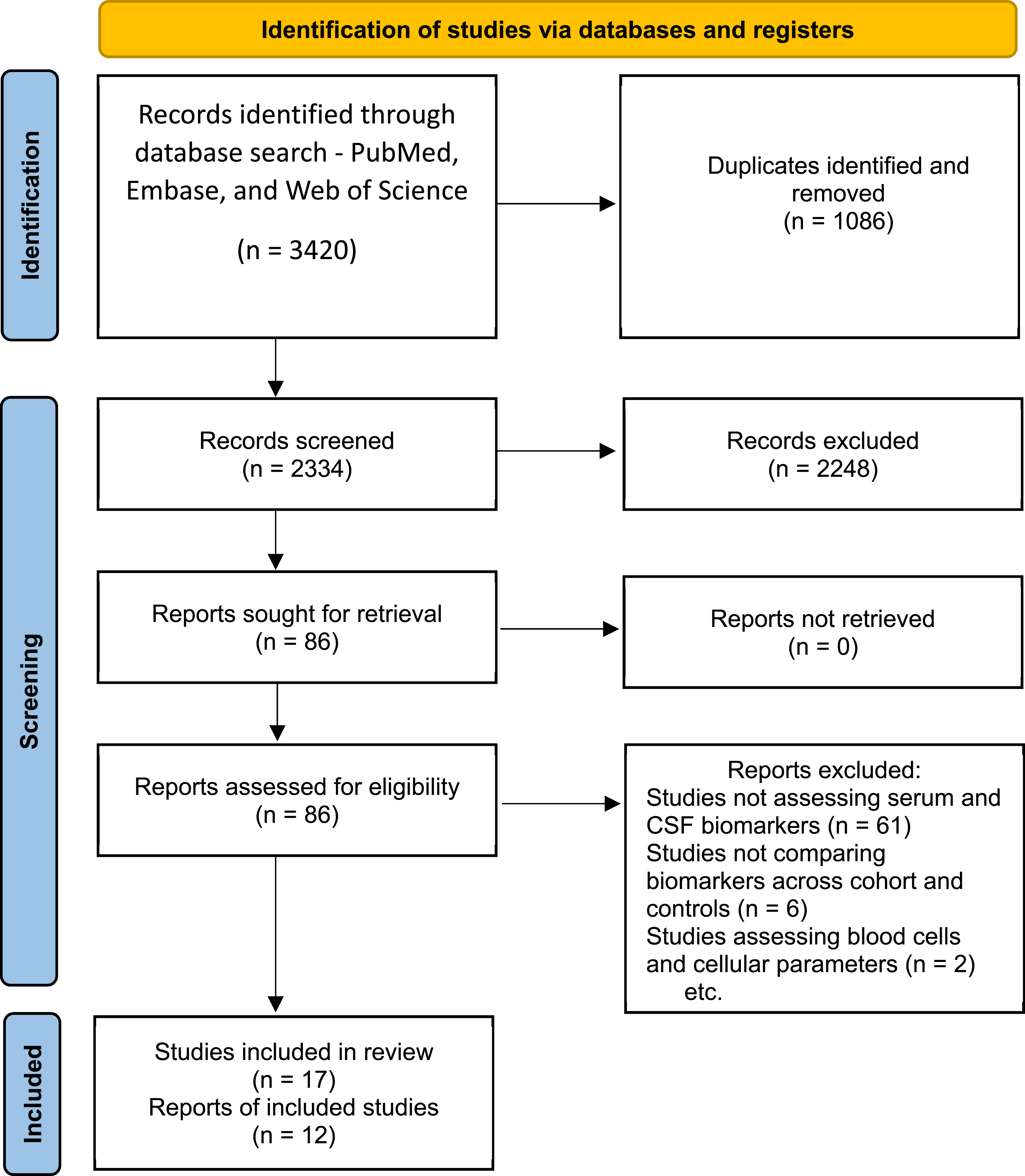
Baseline Study Characteristics
Characteristics of all 17 Studies Included in the Systematic Review With Descriptions of the CSF and Serum Biomarkers
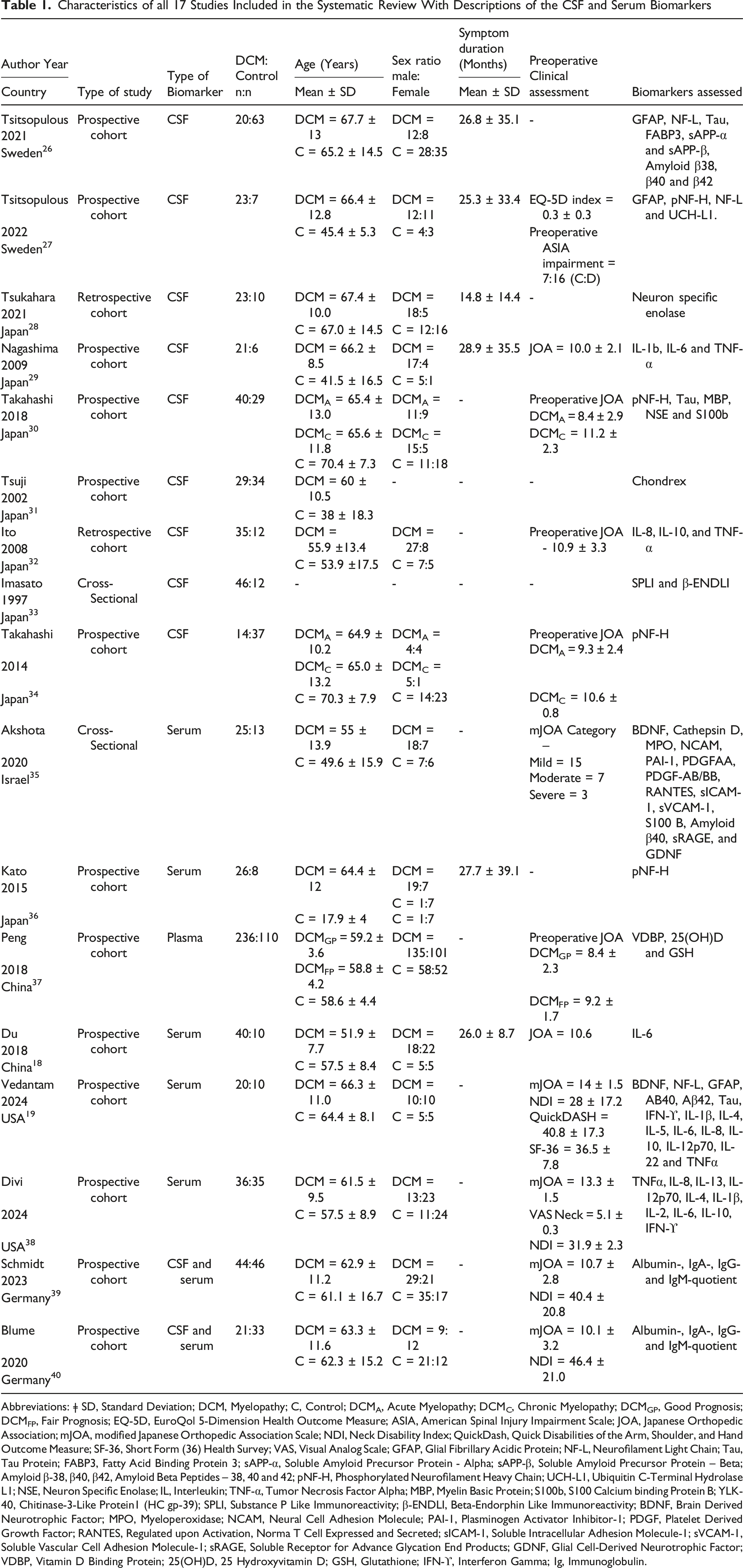
Abbreviations: ǂ SD, Standard Deviation; DCM, Myelopathy; C, Control; DCMA, Acute Myelopathy; DCMC, Chronic Myelopathy; DCMGP, Good Prognosis; DCMFP, Fair Prognosis; EQ-5D, EuroQol 5-Dimension Health Outcome Measure; ASIA, American Spinal Injury Impairment Scale; JOA, Japanese Orthopedic Association; mJOA, modified Japanese Orthopedic Association Scale; NDI, Neck Disability Index; QuickDash, Quick Disabilities of the Arm, Shoulder, and Hand Outcome Measure; SF-36, Short Form (36) Health Survey; VAS, Visual Analog Scale; GFAP, Glial Fibrillary Acidic Protein; NF-L, Neurofilament Light Chain; Tau, Tau Protein; FABP3, Fatty Acid Binding Protein 3; sAPP-α, Soluble Amyloid Precursor Protein - Alpha; sAPP-β, Soluble Amyloid Precursor Protein – Beta; Amyloid β-38, β40, β42, Amyloid Beta Peptides – 38, 40 and 42; pNF-H, Phosphorylated Neurofilament Heavy Chain; UCH-L1, Ubiquitin C-Terminal Hydrolase L1; NSE, Neuron Specific Enolase; IL, Interleukin; TNF-α, Tumor Necrosis Factor Alpha; MBP, Myelin Basic Protein; S100b, S100 Calcium binding Protein B; YLK-40, Chitinase-3-Like Protein1 (HC gp-39); SPLI, Substance P Like Immunoreactivity; β-ENDLI, Beta-Endorphin Like Immunoreactivity; BDNF, Brain Derived Neurotrophic Factor; MPO, Myeloperoxidase; NCAM, Neural Cell Adhesion Molecule; PAI-1, Plasminogen Activator Inhibitor-1; PDGF, Platelet Derived Growth Factor; RANTES, Regulated upon Activation, Norma T Cell Expressed and Secreted; sICAM-1, Soluble Intracellular Adhesion Molecule-1; sVCAM-1, Soluble Vascular Cell Adhesion Molecule-1; sRAGE, Soluble Receptor for Advance Glycation End Products; GDNF, Glial Cell-Derived Neurotrophic Factor; VDBP, Vitamin D Binding Protein; 25(OH)D, 25 Hydroxyvitamin D; GSH, Glutathione; IFN-ϒ, Interferon Gamma; Ig, Immunoglobulin.
Cerebrospinal Fluid Biomarkers
Corresponding Biomarker Levels in the DCM and Control Cohorts
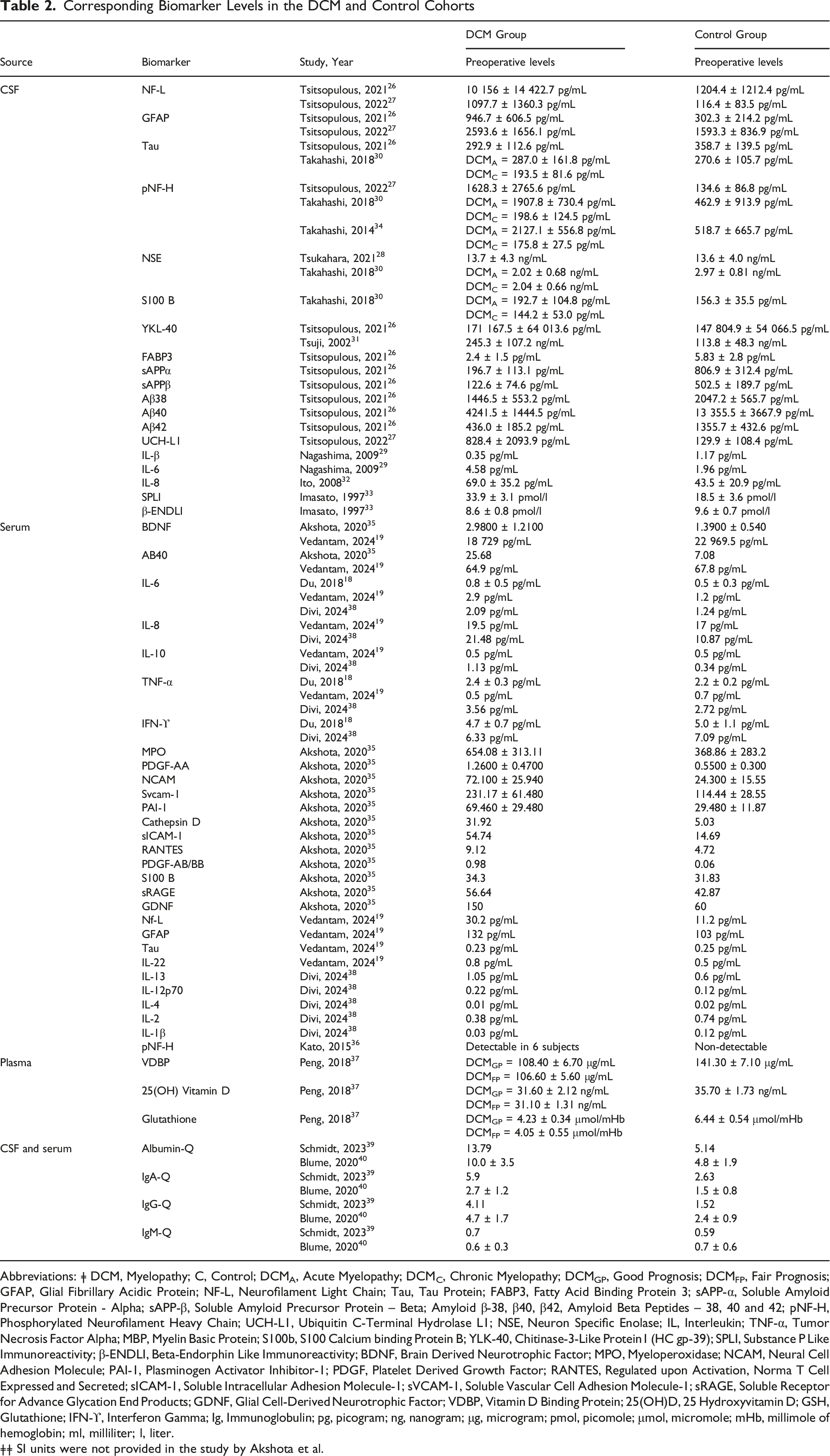
Abbreviations: ǂ DCM, Myelopathy; C, Control; DCMA, Acute Myelopathy; DCMC, Chronic Myelopathy; DCMGP, Good Prognosis; DCMFP, Fair Prognosis; GFAP, Glial Fibrillary Acidic Protein; NF-L, Neurofilament Light Chain; Tau, Tau Protein; FABP3, Fatty Acid Binding Protein 3; sAPP-α, Soluble Amyloid Precursor Protein - Alpha; sAPP-β, Soluble Amyloid Precursor Protein – Beta; Amyloid β-38, β40, β42, Amyloid Beta Peptides – 38, 40 and 42; pNF-H, Phosphorylated Neurofilament Heavy Chain; UCH-L1, Ubiquitin C-Terminal Hydrolase L1; NSE, Neuron Specific Enolase; IL, Interleukin; TNF-α, Tumor Necrosis Factor Alpha; MBP, Myelin Basic Protein; S100b, S100 Calcium binding Protein B; YLK-40, Chitinase-3-Like Protein1 (HC gp-39); SPLI, Substance P Like Immunoreactivity; β-ENDLI, Beta-Endorphin Like Immunoreactivity; BDNF, Brain Derived Neurotrophic Factor; MPO, Myeloperoxidase; NCAM, Neural Cell Adhesion Molecule; PAI-1, Plasminogen Activator Inhibitor-1; PDGF, Platelet Derived Growth Factor; RANTES, Regulated upon Activation, Norma T Cell Expressed and Secreted; sICAM-1, Soluble Intracellular Adhesion Molecule-1; sVCAM-1, Soluble Vascular Cell Adhesion Molecule-1; sRAGE, Soluble Receptor for Advance Glycation End Products; GDNF, Glial Cell-Derived Neurotrophic Factor; VDBP, Vitamin D Binding Protein; 25(OH)D, 25 Hydroxyvitamin D; GSH, Glutathione; IFN-ϒ, Interferon Gamma; Ig, Immunoglobulin; pg, picogram; ng, nanogram; μg, microgram; pmol, picomole; μmol, micromole; mHb, millimole of hemoglobin; ml, milliliter; l, liter.
ǂǂ SI units were not provided in the study by Akshota et al.
Pooled analyses of CSF biomarkers revealed significantly higher levels of NF-L (9.01; 95% CI = 1.03-78.62; P = .047) within the diseased population as compared to the control group. No significant differences were observed for GFAP (2.16; 95% CI = 0.63-7.35; P = .219), NSE (0.78; 95% CI = 0.48-1.26; P = .308), pNF-H (1.76; 95% CI = 0.41-7.67; P = .449), S100B (1.02; 95% CI = 0.51-2.02; P = .955), Tau (0.83; 95% CI = 0.34-1.86 P = .595) or YLK-40 (1.66; 95% CI = 0.15-18.93; P = .684) between the two groups (see Figure 2). Pooled Analyses of the Ratios of the Cerebrospinal Fluid Biomarkers Amongst the DCM and Control Cohorts. (A) Ratio of 1 Indicates No Difference Amongst the DCM and Control Populations, while Higher or Lower Values Indicate Relatively Increased and Decreased Biomarker Concentrations in the DCM Population. (A) Glial Fibrillary Acidic Protein; B. Neuron Specific Enolase; C. NF-L (Neurofilament Light Chain); D. pNF-H (Phosphorylated Neurofilament Heavy Chain); E. S100 B; F. Tau Protein; G. Chondrex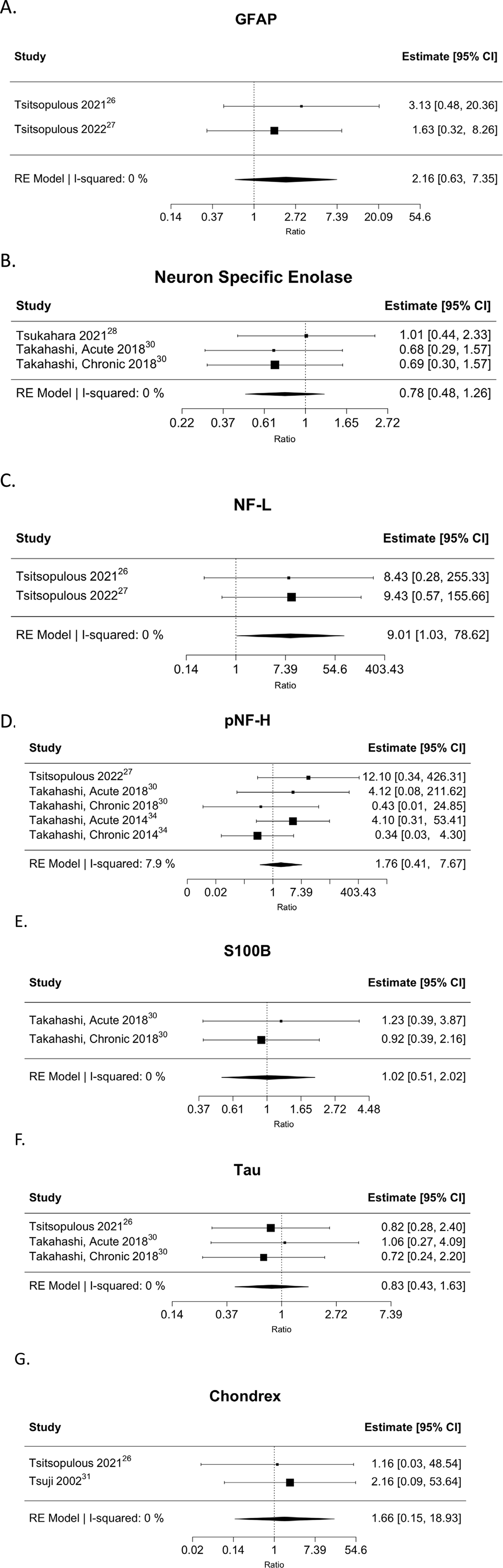
Blood-Based Biomarkers
Thirty-two distinct biomarkers were evaluated across 6 studies in the serum of subjects with DCM and in controls.18,19,35-38 IL-6 and TNF-α (tumor necrosis factor – α) were evaluated in 3 studies each. Similarly, BDNF (brain-derived neurotrophic factor), Aβ40, IL-8, IL-10, and IFN-ϒ (interferon-gamma) were each assessed in 2 studies, while all remaining biomarkers were evaluated in single studies (see Table 2).
Pooled analyses revealed significantly higher levels of IL-6 (1.85, 95% CI = 1.26-2.72, P = .002) and nearly significantly higher IL-8 levels (1.65, 95% CI = 1.00-2.73, P = .051) in subjects with DCM as compared to controls. Finally, no significant differences were observed in Aβ40 (1.89; 95% CI = 0.51-6.95; P = .339), BDNF (1.15; 95% CI = 0.46-2.83; P = .768), IL-10 (1.90; 95% CI = 0.59-6.13; P = .285), TNF-α (1.10; 95% CI = 0.86-1.39; P = .460) or IFN-γ (0.91; 95% CI = 0.65-1.29; P = .600) between the two cohorts (see Figure 3). Pooled Analyses of the Ratios of Serum Biomarkers Amongst the DCM and Control Cohorts. (A) Ratio of 1 Indicates No Difference Amongst the DCM and Control Populations, while Higher or Lower Values Indicate Relatively Increased or Decreased Biomarker Concentrations. (A) Amyloid Beta 40; B. Brain Derived Neurotrophic Factor; C. Interleukin-6; D. Interleukin-8; E. Interleukin-10; F. Tumor Necrosis Factor Alpha; G. Interferon Gamma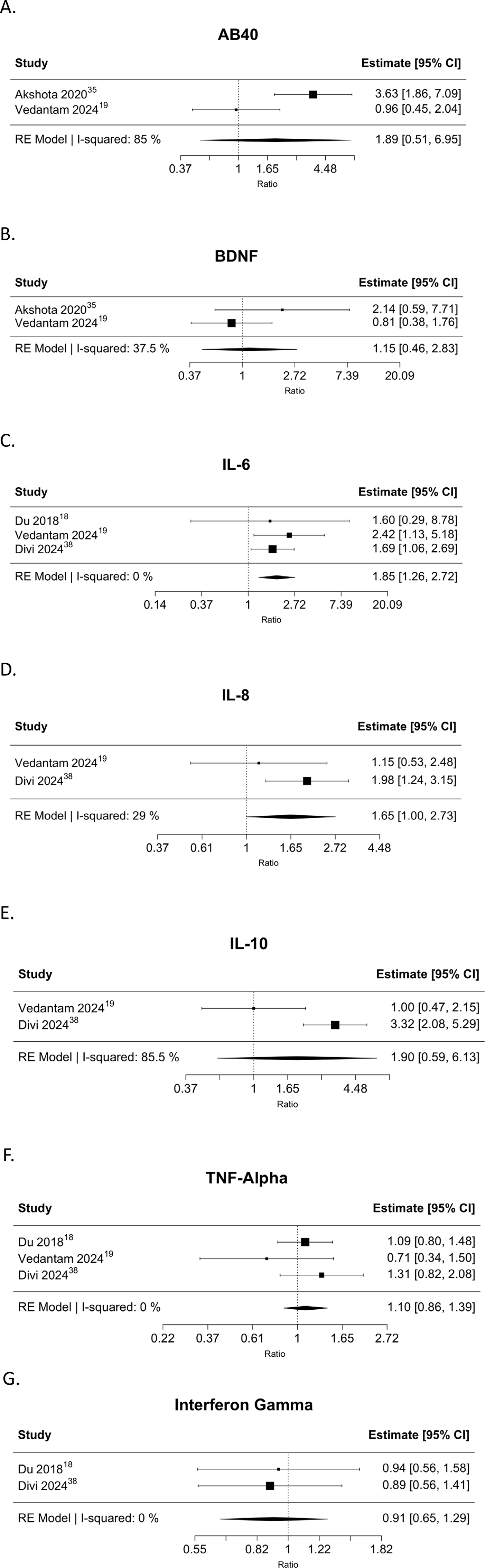
CSF-Serum Quotients
Two studies evaluated the levels of albumin, IgA, IgG, and IgM (immunoglobulin – A, G and M, respectively) in both CSF and serum,39,40 comparing the corresponding quotients to assess BSCB integrity (see Table 2). Pooled analyses demonstrated significantly higher quotients in subjects with DCM as compared to controls – albumin-Q (2.60, 95%CI = 1.81-3.74, P < .001), IgA-Q (2.20, 95%CI = 1.52-3.19, P < .001), and IgG-Q (2.59, 95%CI = 1.81-3.72, P < .001). However, IgM-Q (1.17, 95%CI = 0.80-1.62, P = .240) levels were comparable between DCM cases and controls (See Figure 4). Description of the Pooled Analyses of the Ratios of the Biomarkers Amongst the DCM and Control Cohorts. A Ratio of 1 Indicates No Difference Amongst the DCM and Control Cohorts, while Higher or Lower Levels Indicate Increased or Decreased Biomarker Concentrations. A = Albumin CSF-To-Serum Quotient; B = IgA CSF-To-Serum Quotient; C = IgG CSF-To-Serum Quotient; D = IgM CSF-To-Serum Quotient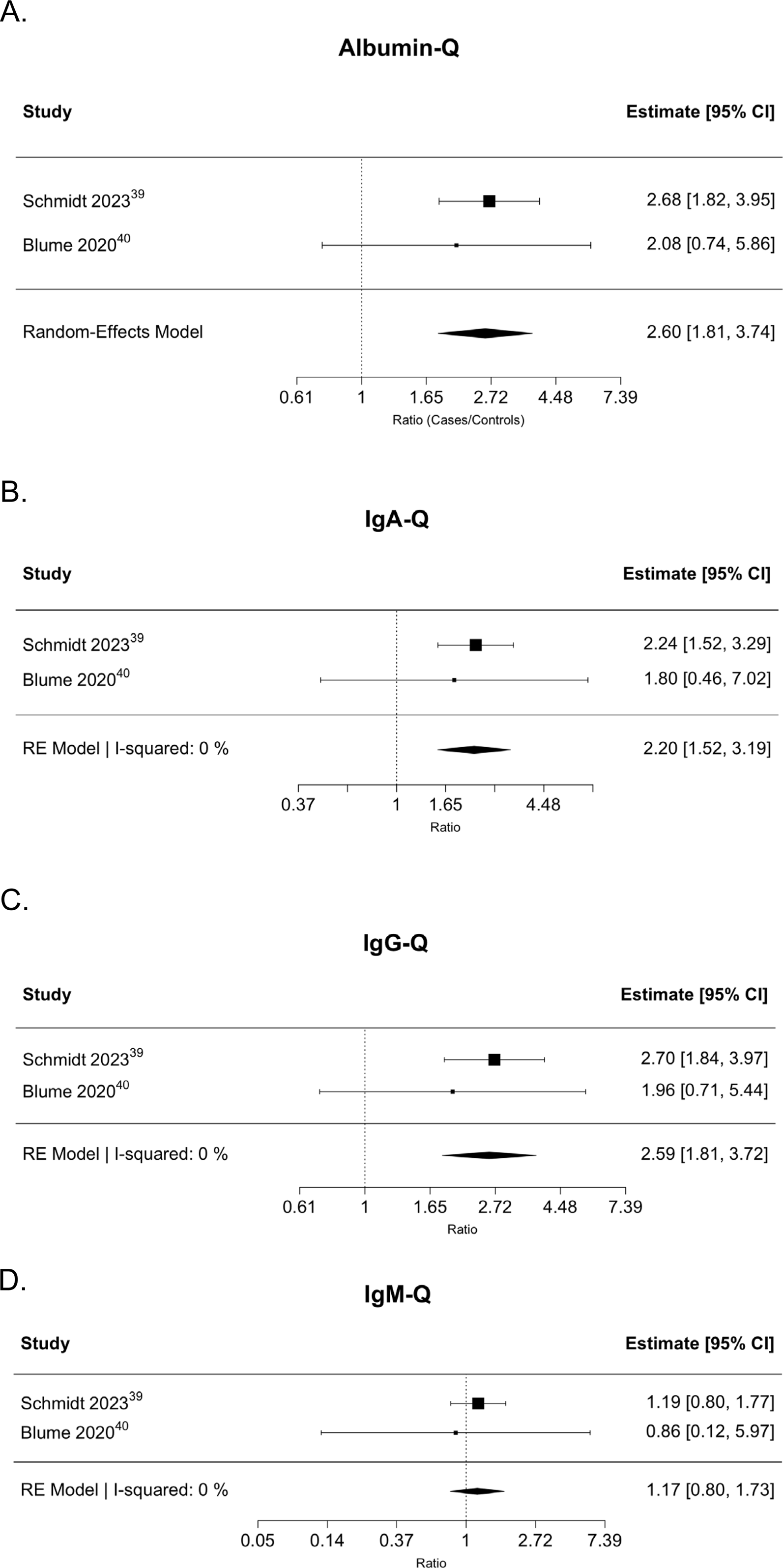
Risk of Bias Assessments
An unclear to high risk of bias was identified in the flow and timing component, primarily due to the general lack of follow-up data. Patient selection was largely representative of the myelopathy population, while the index measures were well defined in the majority of studies, utilizing pre-established biomarker assessment methodologies. The reference standard for diagnosing myelopathy consistently incorporated clinical and radiological criteria. Regarding applicability concerns, the variability in control populations introduced uncertainty in the patient selection domain, leading to unclear applicability in a subset of studies (see Figure 5). Quality Assessment of the Included Studies Using the QUADAS-2 Tool. The Risk of Bias and Applicability were Evaluated Across Four Key Domains: Patient Selection, Index Measure, Reference Standard, and Flow and Timing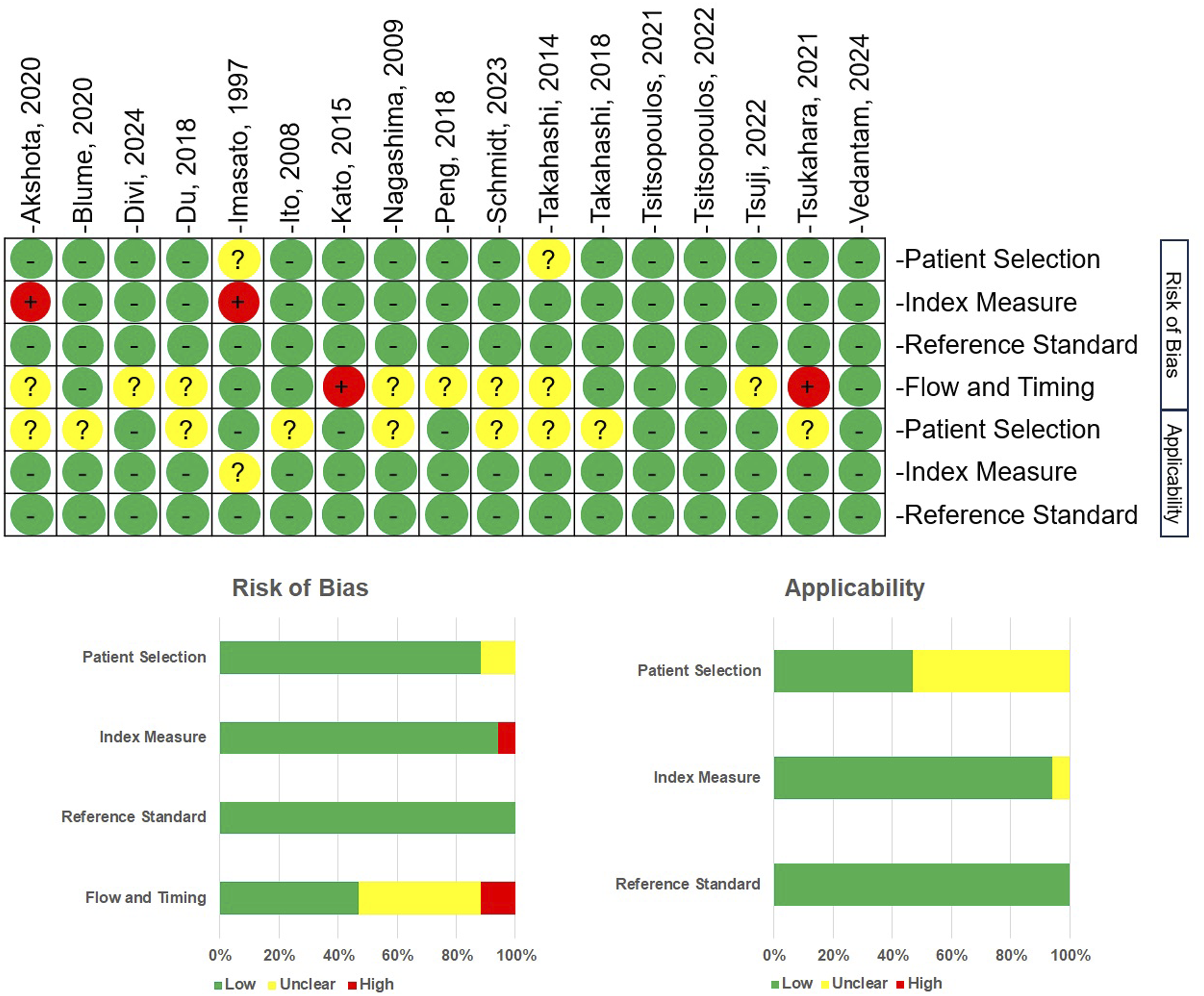
Discussion
SCI is characterized by initial structural trauma followed by several secondary injury processes including progressive hemorrhagic necrosis, programmed cell death, and inflammation. DCM represents a non-traumatic form of SCI involving multiple parallel and chronic pathophysiological processes, including neuronal/axonal degeneration and neuroinflammation, often leading to progressive neurological deterioration.41,42 Two major categories of biomarkers – structural and inflammatory - have been identified in both traumatic and non-traumatic forms of SCI43-46 and in other spinal conditions such as in degenerative disc disease.47-49 While the underlying pathophysiological mechanisms between these conditions are distinct, there is considerable overlap at the cellular and molecular levels in terms of cellular damage.
Our study represents the first comprehensive effort to identify clinically relevant CSF and blood-based biomarkers with potential utility in the diagnosis and severity stratification of DCM. Among the 17 included studies assessing structural and inflammatory biomarkers in CSF or blood, we identified significantly higher levels of NF-L and IL-6, and IL-8 level increases also reached near significance in DCM patients as compared to controls. Additionally, the CSF-to-serum quotients of albumin, IgA, and IgG, were all significantly increased in subjects with DCM, consistent with potential disruption of the BSCB in this condition.39,40
As an extension of the blood brain barrier, the BSCB is primarily comprised of capillary endothelium, a supporting basement membrane, pericytes, and astrocytes. 50 Following traumatic SCI, disruption of the BSCB leads to increased permeability,51,52 initiating a secondary cascade of biochemical reactions leading to spinal cord edema and further disruption of BSCB integrity.2,42 In DCM, chronic BSCB disruption similarly allows for protein leakage into CSF and serum indicative of neuronal and astrocytic injury and demyelination.26,53,54
The initial insult to neuronal and glial cells also triggers the release of proinflammatory cytokines that in turn promote substantial immune cell infiltration. In a rat model of traumatic SCI, increased levels of IL-1, IL-6, IL-10, and TNF-α were initially observed within 6 hours of compression injury while only IL-6 and TNF-α showed elevated levels persisting even after a week.55-57 Elevated serum IL-6 is thought to be indicative of ongoing neural and/or surrounding disco-ligamentous element degeneration, which are also common findings in DCM. 19
We observed significant cross-study variability in CSF and blood-based biomarker concentrations, which is likely attributable to both heterogeneity of disease severity as well as duration. In 2 studies by Takahashi and colleagues, DCM was classified into acute and chronic groups, based on the duration of symptoms. Significantly higher CSF levels of pNF-H and tau proteins were observed in the acute as compared to the chronic DCM groups.30,34 Similarly, Tsitsopulous and colleagues reported elevated CSF levels of NF-L in patients in the acute phase as compared to patients with chronic symptoms, indicating the potential role of NF-L in reflecting axonal damage as well as neuroplasticity. 26 Kato and colleagues observed a similar trend with pNF-H being more elevated in the acute group. 36 Taken together, these observations may indicate that chronic compression may render neural tissue nonviable over time, making it increasingly difficult to detect biomarker traces in the CSF and serum. As such, given the heterogenous findings across the pooled studies and the limited ability of CSF and blood-based biomarkers to consistently reflect overall disease severity, our findings underscore the need for subject stratification by symptom severity and duration when assessing biomarker levels.
Recent studies have evaluated the potential role of RNA and microRNA levels in the diagnosis of DCM. Laliberte and colleagues identified an association between plasma MIR21-5p levels, symptom severity, and disease progression in patients with DCM; elevated levels correlated with moderate-severe symptoms and worse prognoses at 1-year follow-up. 58 Divi and colleagues identified altered expression in 12 of 15 (80%) analyzed serum microRNAs with specific upregulation of miR-223-3p and downregulation of miR-451a, as reported in various neurodegenerative conditions. 38 In a separate study, Zheng and colleagues developed a predictive model for diagnosing DCM using a five-gene model that included blood-based levels of TBCD, TPM2, PNKD, EIF4G2, and AP5Z1, achieving an accuracy of 93.5%. 59 Single-gene models utilizing TCAP or SDHA genes demonstrated accuracies of 83.3% and 76.7%, respectively. Additionally, recent studies have explored the role of ApoE in DCM prognosis. Desmione and colleagues reported poorer postsurgical neurological recovery in carriers of the ApoE4 allele. 60 Similarly, in another study, Chintapalli and colleagues demonstrated higher odds of DCM prevalence in individuals homozygous for ε4 allele, using a large biobank analysis. 61 These findings provide a foundation for future studies assessing putative DCM molecular markers.
In patients with traumatic brain injury, the concept of a “liquid brain biopsy” has been proposed, wherein protein biomarkers are utilized for stratification with both therapeutic and prognostic implications. 62 Several studies have also demonstrated the prognostic value of specific CSF and blood-based biomarkers in determining neurologic outcomes after traumatic SCI.63-66 Similarly, the studies included in this systematic review have also examined the relationship between biomarker levels and outcomes. Tsitsopulous and colleagues observed a negative correlation between CSF GFAP and clinical outcomes, while NF-L and FABP3 (Fatty Acid Binding Protein 3) showed positive correlations with clinical outcomes at 3-months post-operatively.26,27 Further, elevated pre-operative levels of pNF-H were associated with poorer surgical outcomes at follow-up.30,35 Peng and colleagues reported favorable post-operative outcomes in DCM patients with either elevated baseline plasma VDBP (vitamin D binding protein), 25(OH)D (25-hydroxyvitamin D), or GSH (glutathione) levels. 37 Future research should explore the development of a biomarker panel that not only aids in the diagnosis of DCM but also serves as a prognostication tool of post-operative outcomes.
Various Control Groups Identified Across the Included Studies
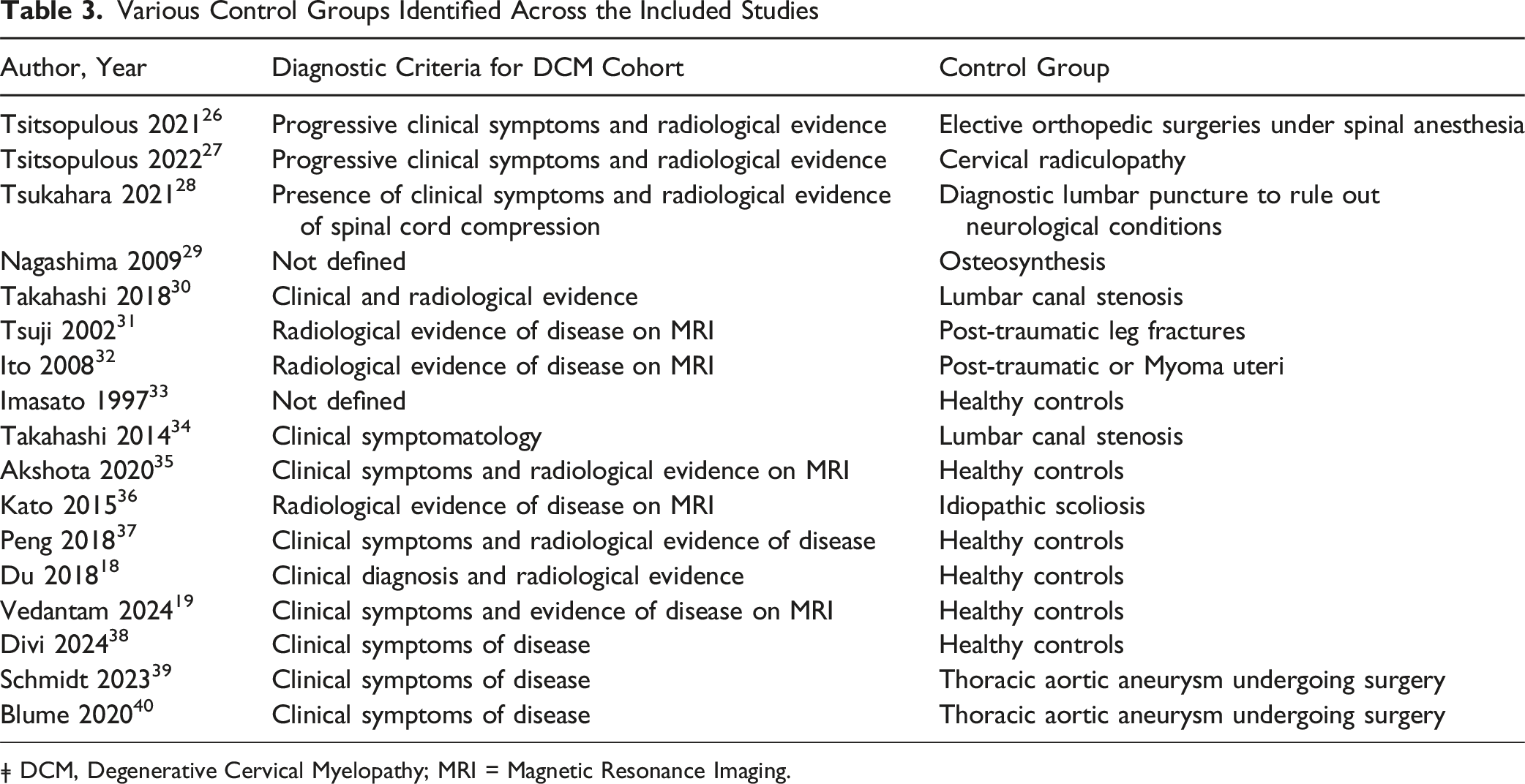
ǂ DCM, Degenerative Cervical Myelopathy; MRI = Magnetic Resonance Imaging.
Several study strengths are noteworthy, including the comprehensive narrative provided by our analysis on the potential utility of various biomarkers in DCM diagnosis, stratification, and prognostication. The meta-analysis further compared relative biomarker levels between DCM and control subjects, offering insights that could enhance the diagnostic accuracy. However, the study was limited by relatively small sample sizes, variable diagnostic criteria for DCM, and the absence of standardized biomarker measurement protocols across studies, which likely further contributed to variability beyond study-specific differences in disease severity and duration. Future larger studies, potentially incorporating randomized designs and stratification by DCM disease duration and severity, could clarify the clinical relevance of CSF and serum-based biomarkers in DCM.
Conclusion
DCM is associated with injury to the spinal cord via chronic compression. Levels of several structural and inflammatory biomarkers have been shown to be significantly altered across studies including NF-L, IL-6, albumin-Q, IgA-Q, and IgG-Q. Nevertheless, marked variability in symptom duration and severity across individuals and studies has thus far limited the ability of these biomarkers to serve as reliable surrogate measures of disease severity and prognosis. By incorporating prospective and unbiased methodologies, we anticipate that future larger studies will identify sensitive and specific biomarker panels with diagnostic and prognostic utility.
Supplemental Material
Supplemental Material - Assessment of Cerebrospinal Fluid and Blood-Based Biomarkers in Degenerative Cervical Myelopathy: Systematic Review and Meta-Analysis
Supplemental Material for Assessment of Cerebrospinal Fluid and Blood-Based Biomarkers in Degenerative Cervical Myelopathy: Systematic Review and Meta-Analysis by Harshit Arora, Hassan Darabi, Cassandra C Wolsh, Kelsey Karnik, John C. Gensel, Francis Farhadi in Systematic Review
Footnotes
Author Contributions
HA, HD and CW conceptualized and designed the study and were involved in data acquisition. Statistical analyses were conducted by HA, HD and KK and analysis and interpretation of data was done by HA. Supervision across all stages of the study and critical revision of the manuscript was done by JCG and FF.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data for the current study are made available from the corresponding author on a reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
