Abstract
Study Design:
Systematic review.
Objectives:
The objective of this study was to conduct a systematic review to determine (1) change in function, pain, and quality of life following structured nonoperative treatment for degenerative cervical myelopathy (DCM); (2) variability of change in function, pain, and quality of life following different types of structured nonoperative treatment; (3) differences in outcomes observed between certain subgroups (eg, baseline severity score, duration of symptoms); and (4) negative outcomes and harms resulting from structured nonoperative treatment.
Methods:
A systematic search was conducted in Embase, PubMed, and the Cochrane Collaboration for articles published between January 1, 1950, and February 9, 2015. Studies were included if they evaluated outcomes following structured nonoperative treatment, including therapeutic exercise, manual therapy, cervical bracing, and/or traction. The quality of each study was evaluated using the Newcastle-Ottawa Scale, and strength of the overall body of evidence was rated using guidelines outlined by the Grading of Recommendation Assessment, Development and Evaluation Working Group.
Results:
Of the 570 retrieved citations, 8 met inclusion criteria and were summarized in this review. Based on our results, there is very low evidence to suggest that structured nonoperative treatment for DCM results in either a positive or negative change in function as evaluated by the Japanese Orthopaedic Association score.
Conclusion:
There is a lack of evidence to determine the role of nonoperative treatment in patients with DCM. However, in the majority of studies, patients did not achieve clinically significant gains in function following structured nonoperative treatment. Furthermore, 23% to 54% of patients managed nonoperatively subsequently underwent surgical treatment.
Keywords
Introduction
Degenerative cervical myelopathy (DCM) is a progressive spine disease and the most common cause of spinal cord dysfunction in adults worldwide. 1,2 It is caused by age-related alterations to the spinal axis, including degeneration of the facet joints, intervertebral discs, and/or vertebral bodies; progressive spinal kyphosis; and ossification, calcification, or hypertrophy of the spinal ligaments. 3 These anatomical changes narrow the spinal canal and may result in progressive cord compression, neurological deterioration, and significantly reduced quality of life.
Early reports on the natural history of this disease have defined DCM as a relatively “benign” condition in which patients are often stable for long periods of time following symptom onset. 4,5 However, there is increasing evidence to suggest that DCM is a progressive disorder and that myelopathic individuals may experience a gradual stepwise decline in neurological status. 6 A recent systematic review of the literature reported that 20% to 60% of patients with symptomatic myelopathy deteriorate by at least 1 point on the Japanese Orthopaedic Association (JOA) score 3 to 6 years after initial assessment. 7 It is therefore important to recognize early signs of myelopathy and implement appropriate treatment strategies to minimize pain, disability, and functional impairment.
Surgery is increasingly recommended as the preferred treatment strategy for patients with DCM as decompression not only effectively halts disease progression, but also results in significant gains in function and quality of life.
8
–12
In contrast, the effectiveness of structured nonoperative treatment in stabilizing or improving symptoms is not well defined. This knowledge gap makes it challenging to determine the appropriate role of nonoperative treatment in the management of DCM, particularly in individuals with mild symptoms. As such, the objective of this study was to conduct a systematic review of the literature to address 4 clinical questions. In adult patients with DCM, What is the change in function, pain, and quality of life following nonoperative treatment? Does this change in function, pain, and quality of life vary depending on type of nonoperative treatment? Does the change in function, pain, and quality of life following nonoperative care differ across subgroups (eg, myelopathy severity or duration of myelopathy symptoms)? What are the harms of nonoperative care and what is the percentage of patients who subsequently undergo surgery?
Materials and Methods
Electronic Literature Search
We conducted a systematic search in Embase, PubMed, and the Cochrane Collaboration Library for studies published between January 1, 1950, and February 9, 2015, to identify studies that reported on outcomes of structured nonoperative treatment in patients with DCM. “Structured nonoperative treatment” was defined as any nonsurgical intervention and included therapeutic exercise, manual therapy, bracing, cervical traction, and others. Our search was limited to human studies published in English. Reference lists from the articles produced by the search were reviewed manually to identify additional publications. For clinical questions 1 through 4, we included studies that reported change in function, pain, and/or quality of life following structured nonoperative treatment in adult patients (≥18 years of age) with DCM due to spondylosis, herniated discs, and/or ossification of the posterior longitudinal ligament. We also included studies that reported the percentage of patients who ultimately underwent surgery following a period of structured nonoperative treatment, as well as studies that stratified patients based on baseline myelopathy severity. For clinical question 2, we sought to identify studies that compared the change in function, pain, and quality of life following competing nonoperative interventions.
Studies were excluded if they (1) included patients under 18 years of age or patients with myelopathy due to infection, malignancy, or acute injury from acute disc herniation, inflammatory arthritis, or trauma; (2) only reported outcomes following surgical intervention; (3) did not state what type of structured nonoperative treatment was performed; (4) did not evaluate outcome using at least one primary outcome measure (JOA, Nurick, conversion to surgery following nonoperative treatment); (5) reported on fewer than 10 patients; (6) were related to animals or cadavers; and/or (7) were strictly biomechanical evaluations. Full inclusion and exclusion criteria are provided in Table 1. Two investigators (AHD, IBA) independently reviewed the full texts of potential articles and excluded all studies that did not meet the inclusion criteria (Figure 1). Selection discrepancies were resolved through discussion.
PICO Table.
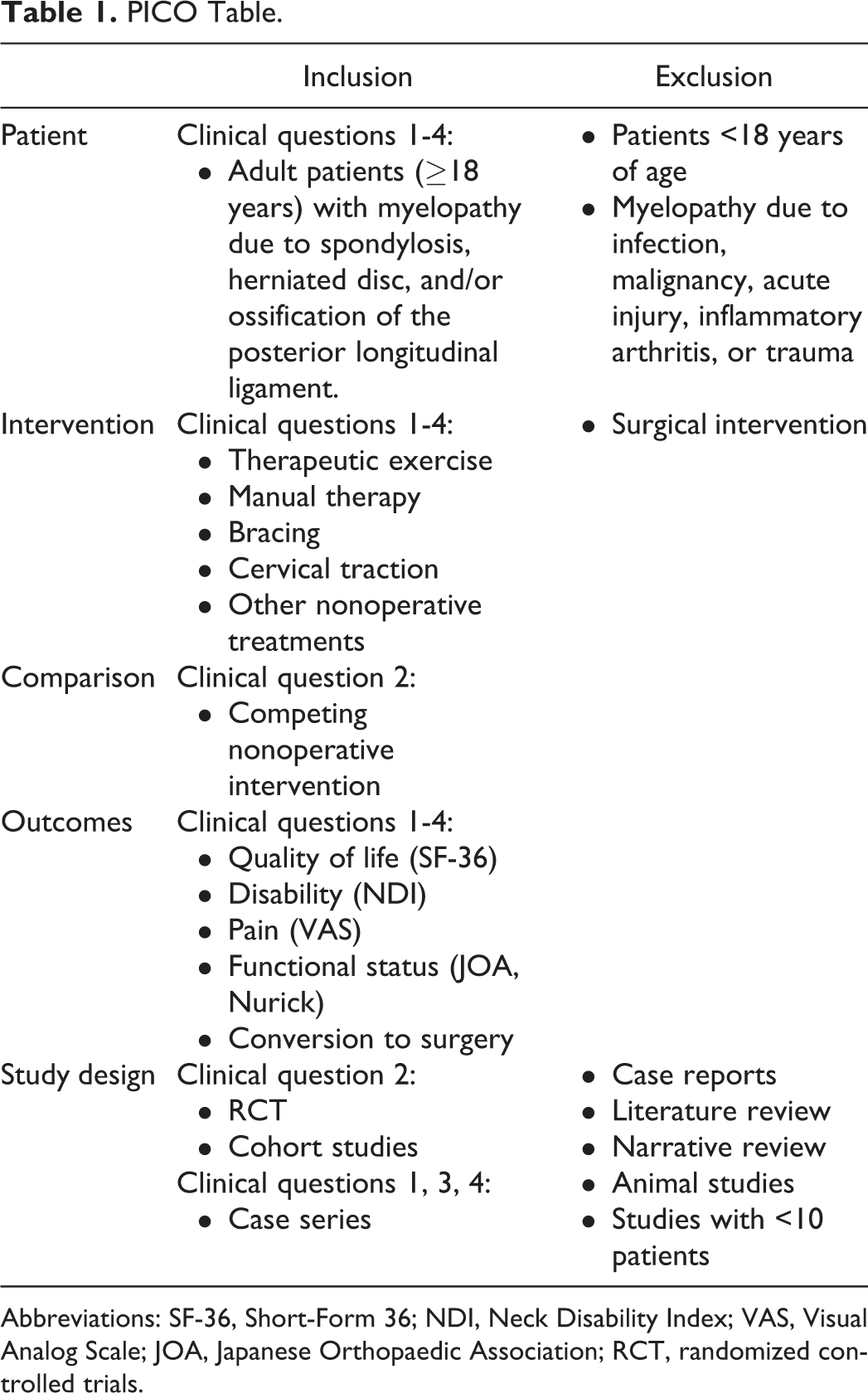
Abbreviations: SF-36, Short-Form 36; NDI, Neck Disability Index; VAS, Visual Analog Scale; JOA, Japanese Orthopaedic Association; RCT, randomized controlled trials.
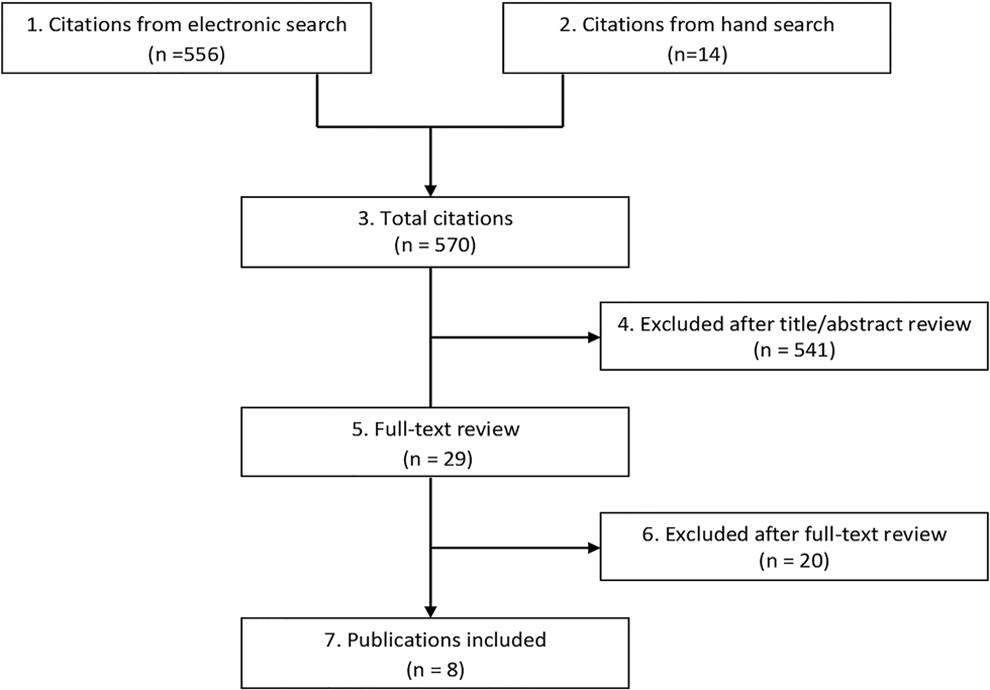
Results of literature search.
Data Extraction
The following data was extracted from each included article: study design; patient characteristics, including mean age, baseline severity score, and type of DCM; length and rate of follow-up; type and duration of nonoperative treatment; outcomes assessed; and associations between nonoperative management and outcomes (function, pain, quality of life, and/or conversion to surgery). We attempted to identify studies with overlapping data to prevent double-counting. In such cases, we selected the study with the most complete data, largest sample size, and greatest follow-up period.
Study Quality and Overall Strength of Body of Literature
Each article was appraised for risk of bias by 2 reviewers (KTP, JRD) using the modified Newcastle-Ottawa Scale (NOS). 13 Strength of the overall body of evidence for each outcome was determined by guidelines outlined by the Grading of Recommendation Assessment, Development and Evaluation (GRADE) Working Group. 14,15 Though the GRADE scale is intended to rate the quality of evidence of comparative studies, we adapted its principles for this systematic review to determine the confidence we have in our conclusions.
The overall body of evidence is considered Low if all studies are observational. The quality of the body of evidence may be upgraded or downgraded depending on a number of factors. Criteria for downgrading 1 or 2 levels include (1) inconsistency of results, (2) indirectness of evidence, or (3) imprecision of the effect estimates (eg, wide variance). Alternately, the body of evidence may be upgraded 1 or 2 levels based on (1) large magnitude of effect or (2) dose-response gradient.
A quality level of High indicates high confidence that the true effect lies close to that of the estimate of effect. A Moderate quality level reflects moderate confidence in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. A Low quality level represents limited confidence in the effect estimate, and that the true effect may be substantially different from the estimate of the effect. 15 Very Low ratings indicate very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect. This rating may be used if there is no evidence or if it is not possible to estimate an effect.
Data Analysis
For clinical questions 1, 2, and 3, JOA or modified JOA (mJOA) scores before and after structured nonoperative treatment were reported and summarized. The minimal clinically important difference (MCID) for the JOA has not been established; however, expert opinion indicates a score change ≥2.0 points is considered clinically significant. 16,17 Furthermore, the MCID of the mJOA has been estimated to be between 1 and 2 points. 18 For clinical question 4, a summary table was used to identify the proportion of patients that received surgical intervention after a period of structured nonoperative treatment.
Results
Study Selection
Our initial search yielded 570 citations. Following title, abstract, and full-text review, we identified 8 studies that met our inclusion criteria for clinical questions 1, 2, and 3 (Figure 1, Table 2). Five of these studies also addressed clinical question 4 and reported proportions of patients that subsequently underwent surgical intervention after a period of structured nonoperative treatment. Of the remaining 562 studies, 541 were excluded at title and abstract levels as they primarily focused on surgical intervention and did not appropriately evaluate outcome following structured nonoperative treatment. After full-text review, 20 additional studies were excluded for the following reasons: inappropriate study design (n = 5), inclusion of patients with trauma or radiculopathy (n = 3), abstract publication only (n = 2), inappropriate outcome measures (n = 2), duplicate data (n = 2), surgical treatment only (n = 1), noncervical condition (n = 1), non-English publication (n = 1), and no description of structured nonoperative intervention (n = 2). A list of excluded studies and full data abstraction tables can be found in the Supplemental Material (available in the online version of the article).
Characteristics of Studies.
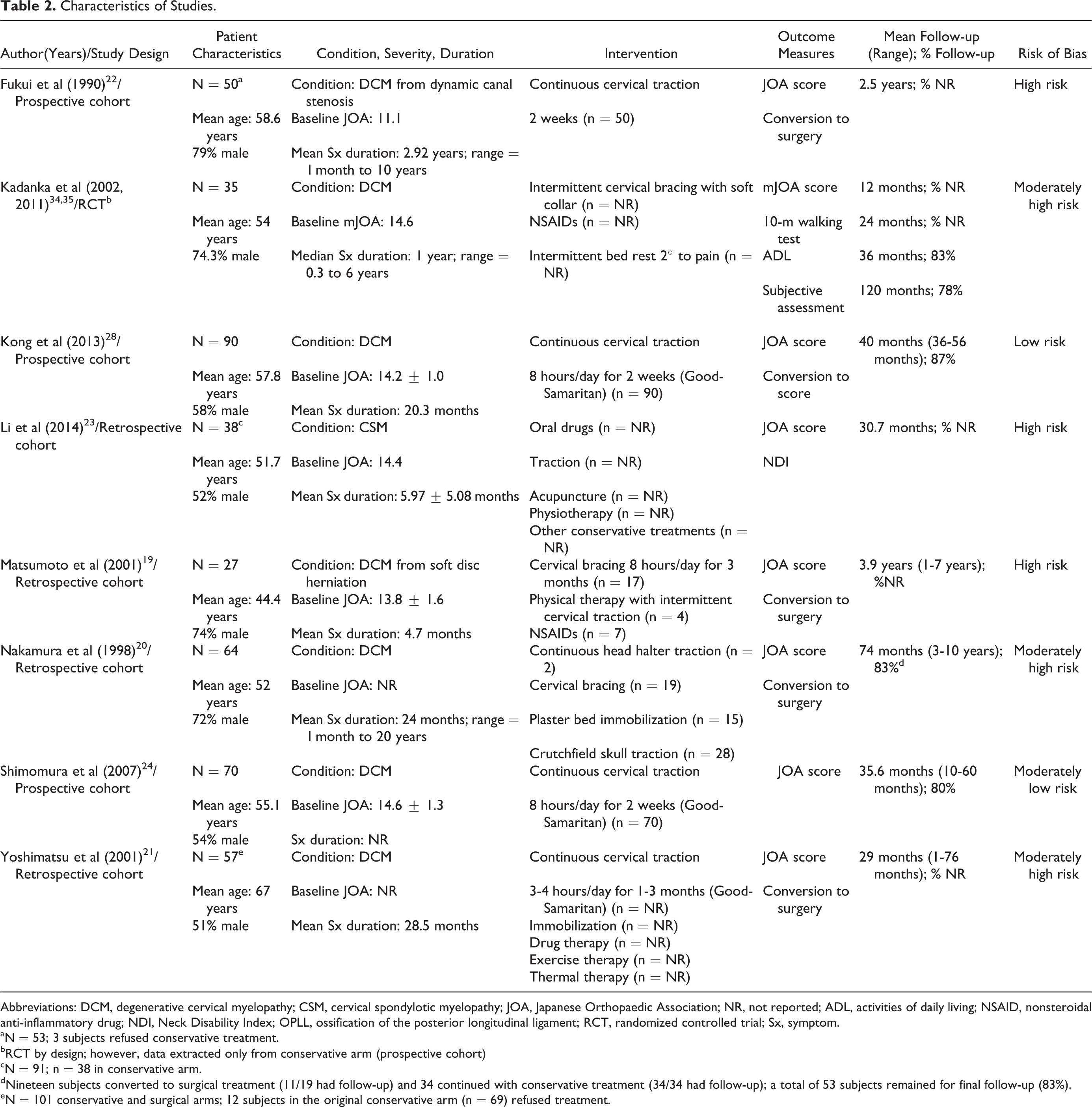
Abbreviations: DCM, degenerative cervical myelopathy; CSM, cervical spondylotic myelopathy; JOA, Japanese Orthopaedic Association; NR, not reported; ADL, activities of daily living; NSAID, nonsteroidal anti-inflammatory drug; NDI, Neck Disability Index; OPLL, ossification of the posterior longitudinal ligament; RCT, randomized controlled trial; Sx, symptom.
aN = 53; 3 subjects refused conservative treatment.
bRCT by design; however, data extracted only from conservative arm (prospective cohort)
cN = 91; n = 38 in conservative arm.
dNineteen subjects converted to surgical treatment (11/19 had follow-up) and 34 continued with conservative treatment (34/34 had follow-up); a total of 53 subjects remained for final follow-up (83%).
eN = 101 conservative and surgical arms; 12 subjects in the original conservative arm (n = 69) refused treatment.
Summary of Studies and Risk of Bias
Types of structured nonoperative treatment varied across studies and were not well-defined. Treatments included bed rest, cervical traction, cervical immobilization or bracing, thermal therapy, physical therapy, and/or nonsteroidal anti-inflammatory drugs. Outcomes were assessed using a variety of measures such as the JOA/mJOA, timed 10-meter walking test, Neck Disability Index, and Activities of Daily Living. Some studies also reported rates of conversion to surgery following an initial trial of conservative management.
Based on the modified NOS, 6 studies had “moderately low risk of bias” and 2 had “moderately high risk of bias.” Significant methodological flaws included high attrition rate (n = 4), selection bias in choosing source population (n = 1), and small sample size (n = 3). A detailed critical appraisal of each study can be found in the Supplemental Material (available in the online version of the article).
What Is the Change in Function, Pain, and Quality of Life Following Structured Nonoperative Treatment for DCM?
Assessment of JOA or mJOA Scores
Six studies reported outcomes of structured nonoperative treatment using change in JOA (n = 5) or mJOA (n = 1) scores from baseline to follow-up (Table 3). Sample sizes ranged from 32 to 90 patients, with mean baseline JOA/mJOA scores ranging from 11.1 to 14.6. Response to treatment was minimal, with change scores ranging from 0 to 2.3. Only a single study by Matsumoto et al 19 reported a mean JOA change score ≥2.0 points at final follow-up (mean = 47 months).
Change in (Modified) Japanese Orthopaedic Association Score Following Structured Nonoperative Treatment in Patients With DCM.
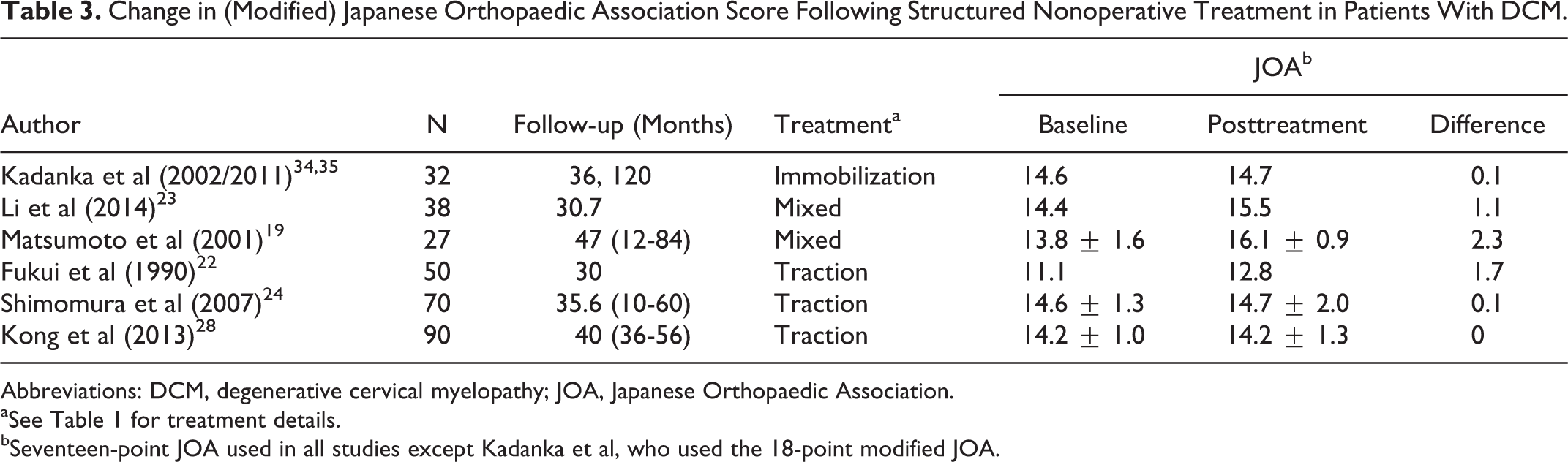
Abbreviations: DCM, degenerative cervical myelopathy; JOA, Japanese Orthopaedic Association.
aSee Table 1 for treatment details.
bSeventeen-point JOA used in all studies except Kadanka et al, who used the 18-point modified JOA.
Two additional studies evaluated outcomes using the JOA but did not report change scores. A retrospective cohort study by Nakamura et al 20 evaluated changes in motor function of the upper and lower extremities following a variety of structured nonoperative treatments: continuous head-halter traction (n = 2), cervical bracing (n = 19), plaster bed immobility (n = 15), or Crutchfield’s skull traction (n = 28). Extremity function was assessed in 64 patients (74% male, mean age = 54 years) using a disability scale from 0 (“severe impairment”) to 4 (“no disability”) based on JOA scores. At final follow-up (mean = 47 months), 27% (15/56) and 26% (16/61) of patients who received structured nonoperative treatment had “no disability” in the upper and lower extremities, respectively.
In a second retrospective study, Yoshimatsu et al 21 investigated symptomatic changes in 69 patients with DCM who elected not to undergo surgery immediately following diagnosis. Myelopathy severity and functional disability were assessed at baseline using the JOA. Of the 69 patients, 12 refused treatment, 37 underwent “rigorous” nonoperative care, and 20 received nonrigorous care. “Rigorous” treatment consisted of 3 to 4 hours of continuous cervical traction per day for 1 to 3 months, combined with immobilization by cervical orthosis, exercise therapy, drug therapy, and thermal therapy. A description of nonrigorous intervention was not provided. To evaluate treatment effects, baseline and posttreatment JOA scores were compared and patients were classified into 3 groups based on whether they exhibited “improvement,” “no change,” or “exacerbation of symptoms” at final follow-up (mean = 29 months). Twenty-six percent (15/57) of the patients who received structured nonoperative treatment demonstrated JOA improvements between baseline and follow-up, whereas only 8% (1/12) of the patients who refused structured nonoperative care exhibited functional gains. In addition, a smaller percentage of patients who received structured nonoperative care experienced “exacerbation of symptoms” based on the JOA (58%; 33/57) than those who refused nonoperative treatment (83%; 10/12). However, the difference in improvement and exacerbation of symptoms between patients receiving structured nonoperative care and those refusing treatment was within the limits of chance.
Does the Change in Function, Pain, and Quality of Life Vary Depending on Treatment Type?
No studies directly compared outcomes between different strategies of structured nonoperative treatment; however, one study evaluated outcomes based on different treatment “intensities.” A retrospective cohort study by Yoshimatsu et al 21 investigated symptomatic changes in 69 patients with DCM who received either rigorous or nonrigorous nonoperative treatment. Thirty-eight percent (14/37) of patients receiving rigorous nonoperative treatment reported some improvement, whereas only 6% (2/32) of patients receiving nonrigorous nonoperative treatment reported improvement. The proportion of patients who experienced worsening of symptoms was 49% (18/37) and 78% (25/32), respectively.
Does the Change in Function, Pain, and Quality of Life With Nonoperative Care Vary According to Subgroups (ie, Myelopathy Severity or Duration of Symptoms)?
Duration of Symptoms
Three studies evaluated the correlation between pretreatment duration of symptoms and posttreatment JOA scores. 20,22,23 Fukui et al 22 evaluated changes in functional impairment on the JOA score following 2 weeks of cervical traction. Pretreatment JOA scores for 53 patients ranged from 6 to 15, with a mean of 11.1 points (3 patients refused structured nonoperative treatment; n = 50). Fifty-six percent (28/50) of the patients demonstrated JOA improvements following treatment. In patients with a duration of symptoms less than 3 months, 80% (12/15) improved by at least 1 point on the JOA; in contrast, only 46% (16/35) of patients with a duration of symptoms greater than 3 months exhibited a ≥1 point JOA improvement (risk ratio = 1.75; 95% confidence interval = 1.13-2.72). Nakamura et al 20 also evaluated whether duration of symptoms is predictive of JOA improvements following structured nonoperative treatment. For patients with a duration less than 6 months, 30% (3/10) had “no disability” in the upper extremity and 36% (5/14) had “no disability” in the lower extremity following treatment. For patients with a symptom duration >6 months, a slightly smaller percentage of patients achieved “no disability” in the upper (26%; 12/46) and lower (23%; 11/47) extremities. Although these differences were not statistically significant, the authors indicated that early intervention could result in improved treatment effects following structured nonoperative treatment.
In a retrospective study, Li et al
23
reported a significant correlation between JOA recovery ratios and disease durations (
Baseline Severity Score
There were no studies that stratified their sample based on pretreatment myelopathy severity.
Other Subgroups
A retrospective cohort study by Matsumoto et al 19 evaluated outcomes following structured nonoperative treatment in patients with myelopathy secondary to cervical soft disc herniation. This study analyzed data from 27 patients with moderate myelopathy (mean baseline JOA 13.8) who underwent cervical bracing, traction, and nonsteroidal anti-inflammatory drug therapy for 6 months, with a mean follow-up time of 3.9 years. Sixty-three percent (17/27) of the patients demonstrated improvement or stability on the JOA at final follow-up and 59% (10/17) experienced spontaneous regression of their disc herniation and a reduction in myelopathy symptoms. The authors concluded that structured nonoperative treatment may improve neurological symptoms in patients with myelopathy secondary to cervical disc herniation.
What Are the Harms of Nonoperative Care and What Is the Percentage of Patients Who Convert to Surgery?
No studies reported direct harms of structured nonoperative treatment. Based on 5 studies, the proportion of patients who underwent surgical intervention following a period of structured nonoperative treatment ranged from 23% to 54% (mean follow-up = 27-74 months; Table 4). In patients with baseline JOA scores ≥13.0, 23% to 38% of patients ultimately received surgery. In patients with more severe myelopathy (JOA < 13.0 [11.1]), Fukui et al 22 reported a rate of conversion of 54% (27/50) following a period of structured nonoperative treatment. Nakamura et al 20 did not specify baseline JOA scores, but did indicate that 30% (19/64) eventually received surgical intervention at a follow-up period ranging from 36 to 129 months.
The Proportion of Patients With DCM Who Converted to Surgery Following Structured Nonoperative Treatment.
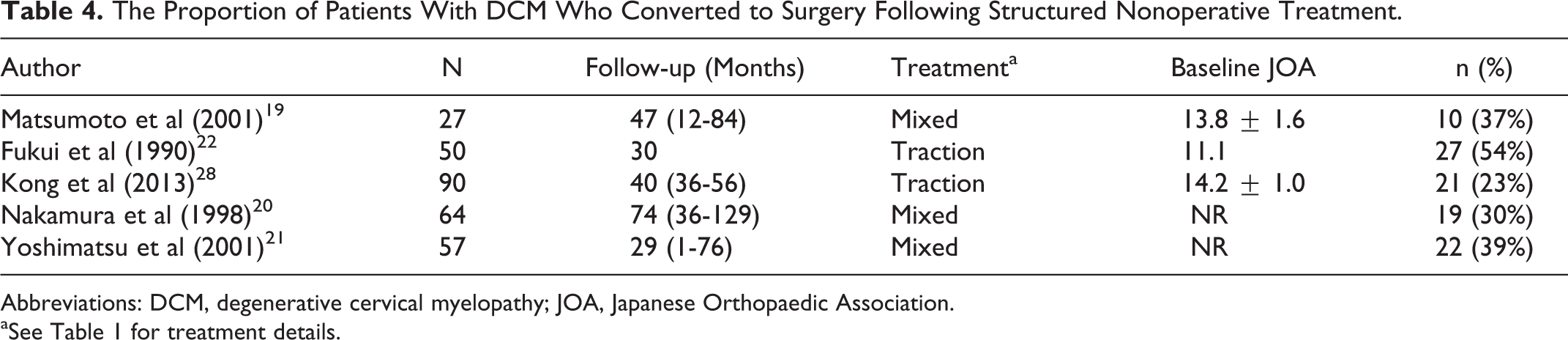
Abbreviations: DCM, degenerative cervical myelopathy; JOA, Japanese Orthopaedic Association.
aSee Table 1 for treatment details.
Evidence Summary
Eight small studies, ranging in size from 27 to 90 patients, evaluated outcomes following structured nonoperative treatment in patients presenting with mostly mild to moderate DCM (mean baseline mJOA score ≥12). mJOA or JOA improvement from baseline was generally below the MCID, with mean change scores ranging from 0 to 1 in most studies. One subgroup of patients with DCM from soft disc herniation reported improved JOA scores in 63% (17/27) of patients at a mean follow-up of 4 years. The proportion of patients receiving surgery following nonoperative care ranged from 23% to 54% across 5 small studies. The quality of evidence for these findings is Very Low.
Discussion
There is increasing evidence to support that surgery results in significant and clinically meaningful improvements in functional status and quality of life in patients with varying degrees of myelopathy severity. 8 –12 In contrast, the role of nonoperative treatment in these patients has not been well defined. It is therefore the objective of this review to evaluate change in function, pain, and quality of life following structured nonoperative treatment in patients with DCM.
Based on our results, nonoperative treatment does not result in clinically meaningful or statistically significant gains in function. Across 6 studies, improvements on the JOA/mJOA ranged from 0 to 2.3. Interestingly, the greatest reported improvements following nonoperative care was observed in patients with myelopathy due to soft disc herniation (Matsumoto, difference 2.3) and dynamic cervical myelopathy (Fukui, difference 1.7). These etiologies might be expected a priori to respond better to nonoperative care, since soft disc herniations may spontaneously regress, and immobilization may at least temporarily decrease cord irritation if the primary mechanism of compression is dynamic rather than static. In contrast, nonoperative treatment was less effective in patients with DCM due to static spinal cord compression, or etiologies that do not tend to regress spontaneously over time (Table 3; difference in mJOA/JOA for these studies was 0 to 1.1). Therefore, based on the evidence in this review, nonoperative care may be reserved for patients with milder myelopathy secondary to soft disc herniations or dynamic stenosis.
This review also reported that 23% to 54% of patients convert to surgery following an initial period of conservative treatment. Given the large variability in estimates, it is important to determine which patients are at a higher risk of disease progression. Important predictors of neurological deterioration and ultimate conversion to surgery include (1) circumferential cord compression on an axial magnetic resonance image; 24 (2) an “angular-edged” spinal cord, defined as an acute angled or lateral corner at one or both sides; 25 (3) greater range of preoperative neck and head motion; 26 (4) lower segmental lordotic angle and greater percentage of vertebral slip; 27 and (5) segmental instability and reduced diameter of the cerebrospinal fluid column. 28 For patients who are in these high-risk groups, surgical intervention should be recommended regardless of myelopathy severity. This is especially critical given recent reports that a longer duration of preoperative symptoms is predictive of a worse surgical outcome. 29,30
To better define the role on nonoperative treatment, outcomes should be separately evaluated in patients with mild, moderate, and severe myelopathy. In a recent systematic review of the literature, Rhee et al investigated the comparative effectiveness and safety of surgery versus nonoperative management. 31 This review reported that there is little evidence to suggest that nonoperative treatment halts or reverses the progression of myelopathy and that nonoperative care should not be the primary treatment modality in patients with moderate to severe disease. Surgery should be recommended in these patients without significant delay, as further disease progression could result in considerable harm, reduced quality of life, significant functional disability, and decreased responsiveness to surgery. In addition, Wu et al reported that myelopathic patients may be at a higher risk of spinal cord injury or central cord syndrome, both of which are associated with debilitating neurologic impairment and increased economic burden. 32
In contrast, there may be a role for nonoperative management in patients with milder and stable myelopathy. In these cases, patient preferences must be strongly considered as patients may be reluctant to undergo surgery for mild symptoms, especially if they have not deteriorated over time. Furthermore, factors that influence the risk-benefit ratio of either operative or nonoperative management must be weighed when determining the optimal treatment strategy in these patients; these include age, comorbidities, duration of symptoms, and smoking status. Since no studies stratified their samples based on preoperative severity, we are unable to determine whether patients with mild myelopathy (mJOA ≥15) improve by the MCID on the mJOA/JOA following structured nonoperative treatment.
The comparative cost-effectiveness of nonoperative strategies and of nonoperative versus surgical management may also be considered when developing treatment protocols. Unfortunately, no studies were identified that compared the cost-effectiveness of various nonoperative strategies. A recent study by Witiw et al, however, aimed to evaluate the value (incremental cost-utility) of surgery versus nonoperative management using data from 171 Canadian patients enrolled in either the AOSpine North America or International study. 33 Aggregate costs, from a hospital payer perspective (ie, costs incurred during index admission, readmission, or returns to the emergency department due to a related complication) were collected for the duration of the 24-month study period. These were combined with health-related quality of life (HRQoL) data to create a 2-dimensional vector of cost and change in quality of life associated with surgical intervention for each individual patient. This data was incorporated into a 2-arm, Markov State Transition model where these values for patients undergoing surgery were compared with estimated counterfactual outcomes of initial nonsurgical management. Initial costs from the hospital payer perspective for conservative management were zero and HRQoL outcomes were based on data available on the natural history of DCM. In their primary model, the lifetime incremental cost to utility ratio (ICUR) was determined to be $11 496 per quality adjusted life year (QALY) gained for surgical intervention. This point estimate falls well within the criteria defined by the World Health Organization as very cost-effective. Further testing using a Monte Carlo probabilistic sensitivity analysis revealed that 97.9% of estimates fell within this threshold, suggesting robustness to variability in the parameter estimates. To supplement this testing, a highly conservative assumption that individuals undergoing initial nonoperative management would not experience any neurologic decline over their lifetime was added to the model. In this scenario, the ICUR was calculated as $20 548/QALY gained with 94.7% of estimates falling within the World Health Organization threshold; this finding further supports the cost-effectiveness of surgical intervention.
Limitations
Clinicians who treat myelopathic patients may ask the question, “Is it reasonable to prescribe an initial trial of nonoperative care for patients with DCM?” This systematic review reveals significant flaws in the literature and cannot provide a strong evidence-based answer to this question. The major limitation in the body of evidence is that “structured nonoperative care” is often poorly defined and consists of a myriad of treatments, including traction, bracing, massage, exercise, and drug administration. The variability of treatment modalities across studies makes it challenging to derive conclusions regarding the effectiveness and safety of nonoperative care for DCM. As presented in Table 5, the level of evidence for each question was deemed “Very Low,” which means we have little confidence that the estimate of the treatment effect reflects the true effect.
GRADE Summary Table.
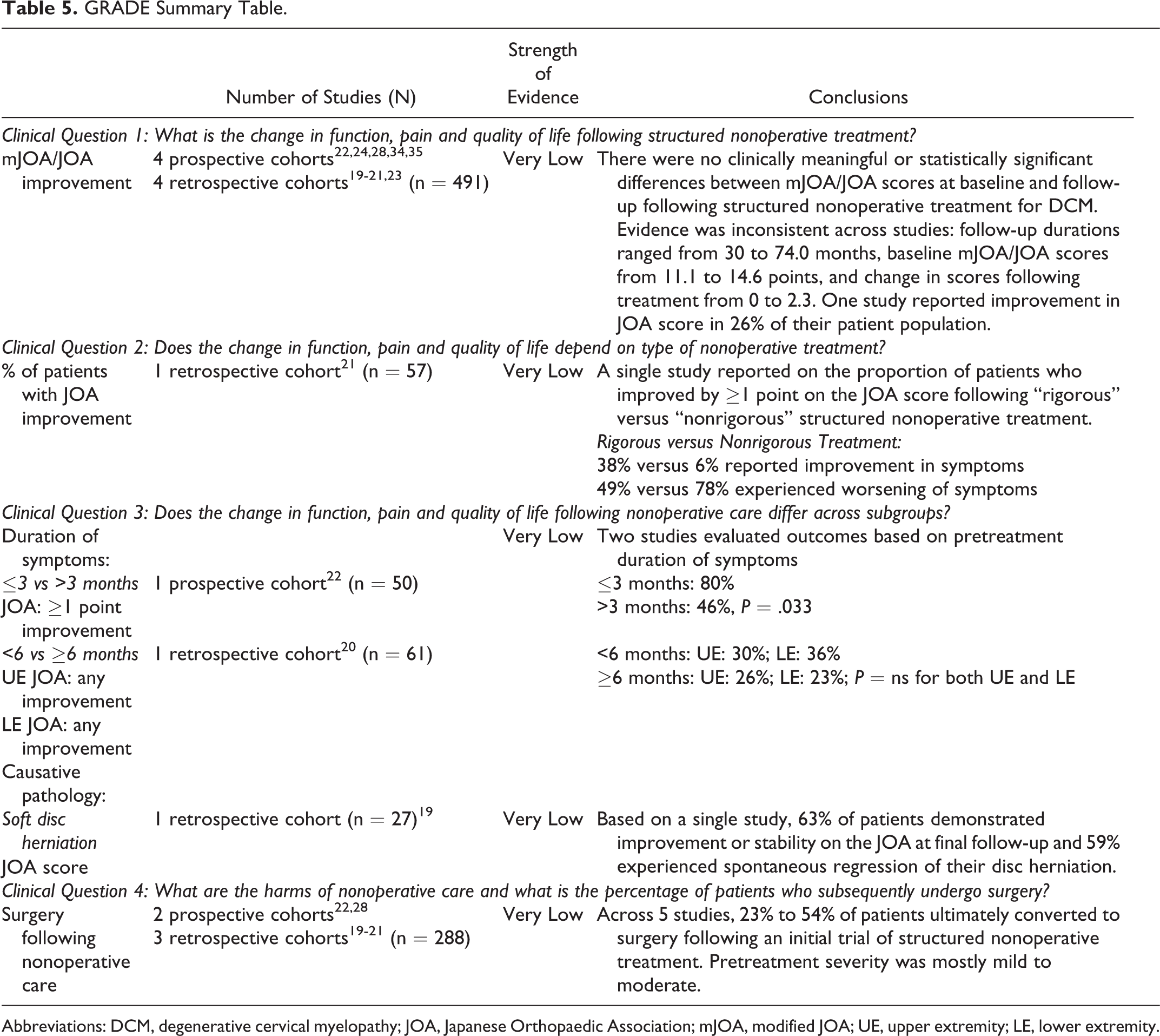
Abbreviations: DCM, degenerative cervical myelopathy; JOA, Japanese Orthopaedic Association; mJOA, modified JOA; UE, upper extremity; LE, lower extremity.
There are additional limitations in the body of the evidence. Studies included in this review poorly defined treatment parameters. For example, 4 studies reported that drug therapy was used as a form of structured nonoperative care. 19,21,23,34 However, none of these studies defined the types of drugs, dosing instructions, or duration of use. Additionally, 3 studies discussed other forms of treatment including exercise, thermal therapy, or physical therapy 19,21,23 but did not provide further description of these treatments, whether they overlapped, the intensity of administration, and how compliant individuals were. As a result, we are unable to draw concrete conclusions about the superiority of various nonoperative treatment modalities over other strategies.
Second, although most studies evaluated functional status using the JOA, one study used the mJOA, a scale developed to account for cultural differences between Eastern and Western societies. 34,35 A recent study by Kato et al 16 compared the original JOA with the mJOA and determined that, although the 2 scales are highly correlated (Spearman’s ρ = 0.87), it is not ideal to use them interchangeably. Consequently, the ability to generalize mJOA data with JOA data is limited. Furthermore, 2 studies used different methods to assess functional status that could not be fully compared to change in JOA or mJOA scores.
Third, the MCID of the mJOA has been shown to vary depending on myelopathy severity: 1 in mild patients (mJOA ≥ 15), 2 in moderate patients (mJOA = 12-14), and 3 in severe patients (mJOA < 12). 18 However, the studies included in this review did not stratify their sample based on preoperative severity scores. There may be a role for nonoperative treatment in mild patients (mJOA ≥ 15) if they could demonstrate improvements on the mJOA by 1 or more points.
Finally, there is a wide range of follow-up duration among the included studies, which makes it difficult to distinguish between changes from intervention and changes from natural disease progression.
Conclusion
There is a lack of evidence to concretely define the role of nonoperative treatment in patients with DCM. However, in the majority of studies, patients did not achieve clinically significant gains in function following structured nonoperative treatment. Furthermore, 23% to 54% of patients initially managed nonoperatively subsequently underwent surgical treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by AOSpine and also received funding from the Cervical Spine Research Society (CSRS). Dr Fehlings wishes to acknowledge support from the Gerald and Tootsie Halbert Chair in Neural Repair and Regeneration and the DeZwirek Family Foundation. Dr Tetreault acknowledges support from a Krembil Postdoctoral Fellowship Award.
Supplemental Material
The supplemental materials are available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
