Abstract
Study Design
Single-center retrospective cohort study.
Objective
To evaluate whether 3D-printed titanium (3DTi) cages reduce the risk of revision surgery compared to polyetheretherketone (PEEK) cages in standalone lateral lumbar interbody fusion (SA-LLIF).
Methods
SA-LLIF procedures performed between 09/2018 and 08/2022 were retrospectively reviewed. Minimum follow-up was 2 years. Baseline characteristics and surgical details were compared between those treated with 3DTi and those with PEEK. Univariable, multivariable Poisson, and hierarchical mixed-effects Poisson regression models were performed to identify independent predictors. Secondary analyses examined differences in timing and types of revision surgeries between the 2 implant types.
Results
A total of 233 patients (43.8% female) were included, with a median age of 65 years (IQR: 56-71), BMI of 28.0 (IQR: 24.7-32.1), and a median of 2 levels fused (range 1-4). Overall, 17.6% underwent revision surgery, with a mean time to revision of 26 ± 16 months. Revisions were more frequent after PEEK (26.3%) vs 3DTi (13.0%; P = 0.017). In multivariable analysis adjusting for follow-up and surgical levels, 3DTi was independently associated with reduced revision risk (RR: 0.52, 95% CI: 0.30-0.90; P = 0.027), which remained significant in a hierarchical mixed-effects model accounting for surgeon-level variation (P = 0.042). Early revisions (<2 years) were more common with PEEK (13.8% vs 4.1%; P < 0.05), while late revisions (>2 years) were similar (11.5% vs 8.9%; P = 0.521).
Conclusions
3D-printed titanium cages in SA-LLIF was associated with lower revision risk compared to PEEK, particularly in the early postoperative period. These findings support the implementation of 3Dti implants to improve surgical outcomes in SA-LLIF.
Introduction
Lateral lumbar interbody fusion (LLIF) is an effective surgical technique for both primary treatment and revision of various spinal conditions, including degenerative disc disease, adult degenerative scoliosis, adjacent segment disease, and pseudarthrosis.1-4 The minimally invasive transpsoas approach facilitates the placement of large interbody cages that span the apophyseal ring, while preserving the posterior annulus and facet joints, which contribute to spinal stability.5,6 In addition, this approach helps maintain the integrity of the posterior paraspinal muscles, including the multifidus, which play a critical role in preventing adjacent segment degeneration (ASD) and the need for subsequent surgery. 7
However, when LLIF is performed as a standalone procedure (SA-LLIF), the surgeon must rely on indirect decompression achieved through segmental height restoration, as well as a construct that generally offers less stability than constructs that include supplemental posterior instrumentation with pedicle screws.8,9 This relative lack of stability can lead to higher rates of residual stenosis, cage subsidence, and lower fusion rates.9-12
In 2017, three-dimensional printed titanium (3DTi) LLIF cages were introduced, offering a potential solution to some of these concerns. Compared to polyetheretherketone (PEEK), titanium cages offer superior osseointegration and osteoconductive properties, which enhance bone-implant contact and may improve fusion outcomes.13,14 Yet, the higher stiffness of conventional titanium cages (∼100 000 MPa) can result in stress shielding and adversely affect bone remodeling. 15 To mitigate this, 3DTi cages were developed with a porous structure that significantly reduces stiffness to approximately 2500 MPa, a value much closer to physiological bone stiffness. 16 This porous design further increases the surface area available for bone growth, improves implant stability through enhanced friction, and promotes osseointegration and load distribution.15,16
Recent studies suggest that 3DTi cages may outperform PEEK cages in the context of SA-LLIF. Subsidence rates as low as 3% have been reported with 3DTi cages, compared to 19% with PEEK cages. 17 Similarly, fusion rates of up to 95% have been observed with 3DTi cages, compared to 80% with PEEK. 18 Despite these promising findings, the impact of 3DTi cages on reducing the need for revision surgery in SA-LLIF remains uncertain. For example, a study by Alan et al. 19 noted a lower reoperation rate due to cage subsidence (1% with 3DTi vs 5% with PEEK), although the difference was not statistically significant.
There is a need for larger studies to identify risk factors for revision surgery and to clarify the role of 3DTi cages in standalone lateral lumbar interbody fusion SA-LLIF. This study aims to evaluate risk factors associated with revision surgery following SA-LLIF, with a specific focus on whether the use of 3DTi cages reduces this risk.
Methods
Patient Population
We retrospectively reviewed all standalone lateral lumbar interbody fusion (SA-LLIF) procedures performed at a high-volume academic center between September 2018 and August 2022. Patients were included if they had a minimum clinical follow-up of 2 years. Procedures involving circumferential constructs, such as pedicle screw instrumentation or interspinous process devices, were excluded. Patient demographics, surgical details, and implant type (PEEK vs 3DTi) were collected. Revision surgeries following SA-LLIF were identified through review of electronic health records (EPIC Systems Corporation), including documentation from external providers via integrated records. For each revision, the indication and procedure type were recorded. Revisions were categorized based on location relative to the original fusion: same segment (segments treated with LLIF), proximal adjacent segment, or distal adjacent segment. This study was approved by the institutional review board (IRB #2020-1877). Written informed consent for study participation and data publication was obtained from all participants. The study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Surgical Technique and Implants
All procedures were performed by 4 senior surgeons with more than 15 years of experience in LLIF. Surgeries were conducted under general anesthesia, with the patient positioned in the lateral decubitus position. A mini-open approach was used in all cases with a skin incision approximately 5 cm in length, oriented diagonally along the direction of the external oblique muscle fibers.20,21 After opening the external oblique fascia, layer by layer dissection is performed through the internal oblique, transverse abdominis, retroperitoneal fat, and psoas muscle. The disc space was accessed under fluoroscopic guidance, followed by discectomy and endplate preparation under direct visualization. Key factors for success include thorough removal of the disc and cartilaginous endplates, contralateral anulus release, which is performed with the cobb and under image control, while preserving the integrity of the endplates, minimizing the risk of postoperative cage subsidence. The interbody cages were then inserted to span the apophyseal ring bilaterally.
During the study period, PEEK cages were used exclusively by 1 surgeon, while 3DTi cages were used by 3 surgeons, all of whom had previously used the same PEEK cage systems before transitioning to 3DTi. The PEEK cage systems used included the XLIF (NuVasive, Inc., San Diego, CA, USA) and the COUGAR (DePuy Spine Inc., Raynham, MA, USA). 3D-printed titanium cages included the Modulus XLIF (NuVasive, Inc.) and the Lateral Spine Truss System (4WEB Medical, Inc., Frisco, TX, USA). Cage dimensions were determined at the surgeon’s discretion based on preoperative and intraoperative assessments, with all cages selected being lordotic in shape.
Statistical Analysis
Statistical analyses were conducted using STATA (STATA/BE 18.0, Stata Corp LLC, TX, USA). Data distribution was assessed by visual inspections of histograms and with Shapiro-Wilk tests. Associations between implant type (PEEK or 3DTi) and patient characteristics and surgical details were assessed using Wilcoxon rank-sum tests, chi-square tests, and Fisher’s exact tests. Univariate Poisson regression was used to identify potential predictors of revision surgery, with variables having P < 0.2 included in a multivariable Poisson regression model with robust standard errors to determine independent predictors and estimate relative risks (RR). To account for non-random implant assignment and surgeon-level clustering, a hierarchical mixed-effects Poisson regression model was performed as a sensitivity analysis, with surgeon ID included as a random intercept. Fixed-effect covariates included implant type, number of levels treated, proximal level involvement, and follow-up duration. Secondary analyses examined differences in the rates, timing, and types of revision surgeries between the 2 implant types. Statistical significance was defined as P < 0.05.
Results
Between September 2018 and August 2022, a total of 703 patients underwent LLIF surgery, of whom 285 (40.5%) underwent SA-LLIF (Figure 1). Among these, 233 patients (81.8%) had a minimum follow-up of 2 years and were included in the final analysis. No patient, who underwent revision surgery within the first 2 years, had a follow-up of less than 2 years. 43.8% (n = 102) of patients were female. Of the included patients, 102 (43.8%) were female. The median age was 65 years (interquartile range [IQR]: 56-71), median BMI was 28.0 kg/m2 (IQR: 24.7-32.1), and the median number of levels fused was 2 (range: 1-4). PEEK cages were used in 87 patients (37.3%), and 3D-printed titanium (3DTi) cages were used in 146 patients (62.7%). Study Flowchart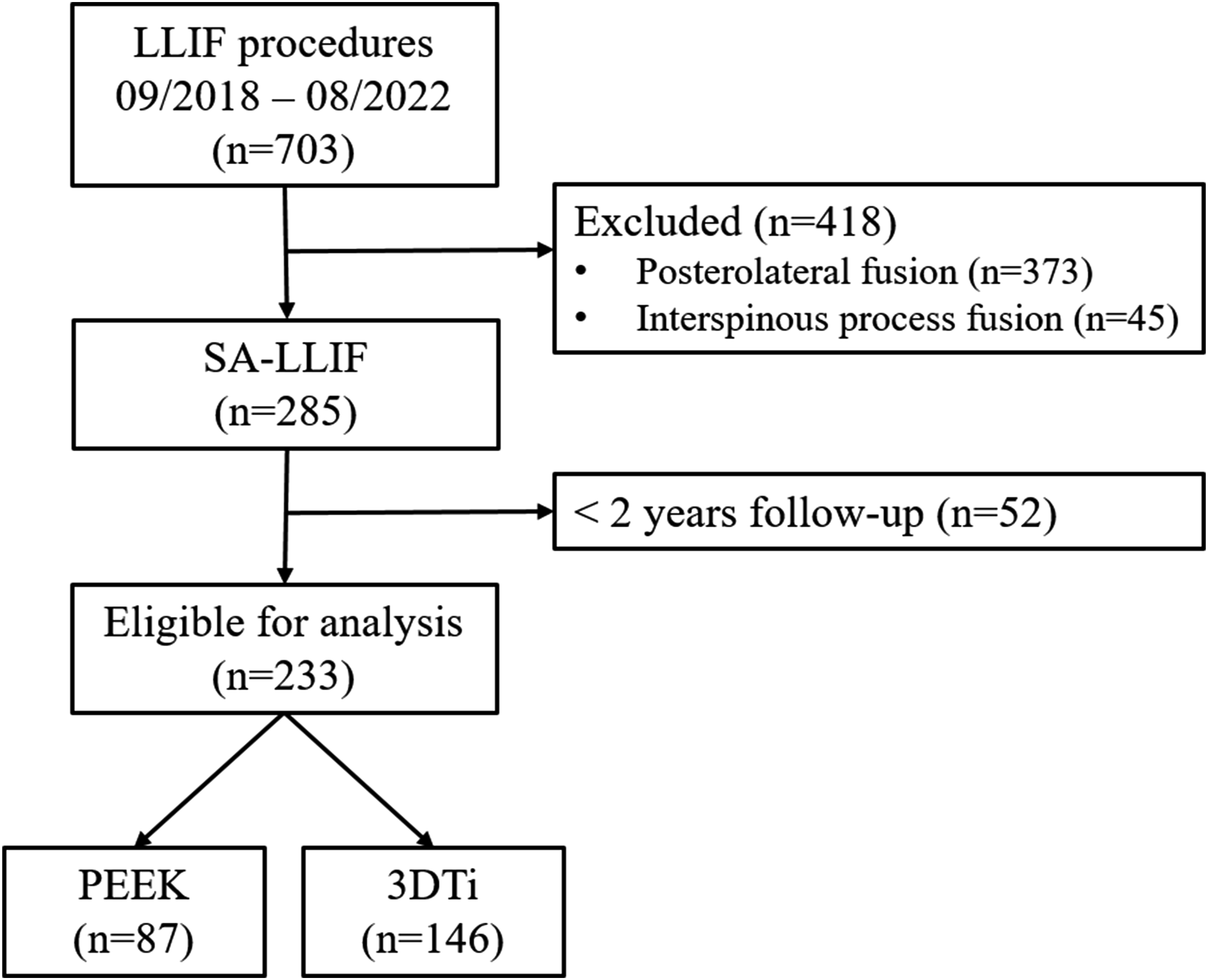
Patient Characteristics and Surgical Details
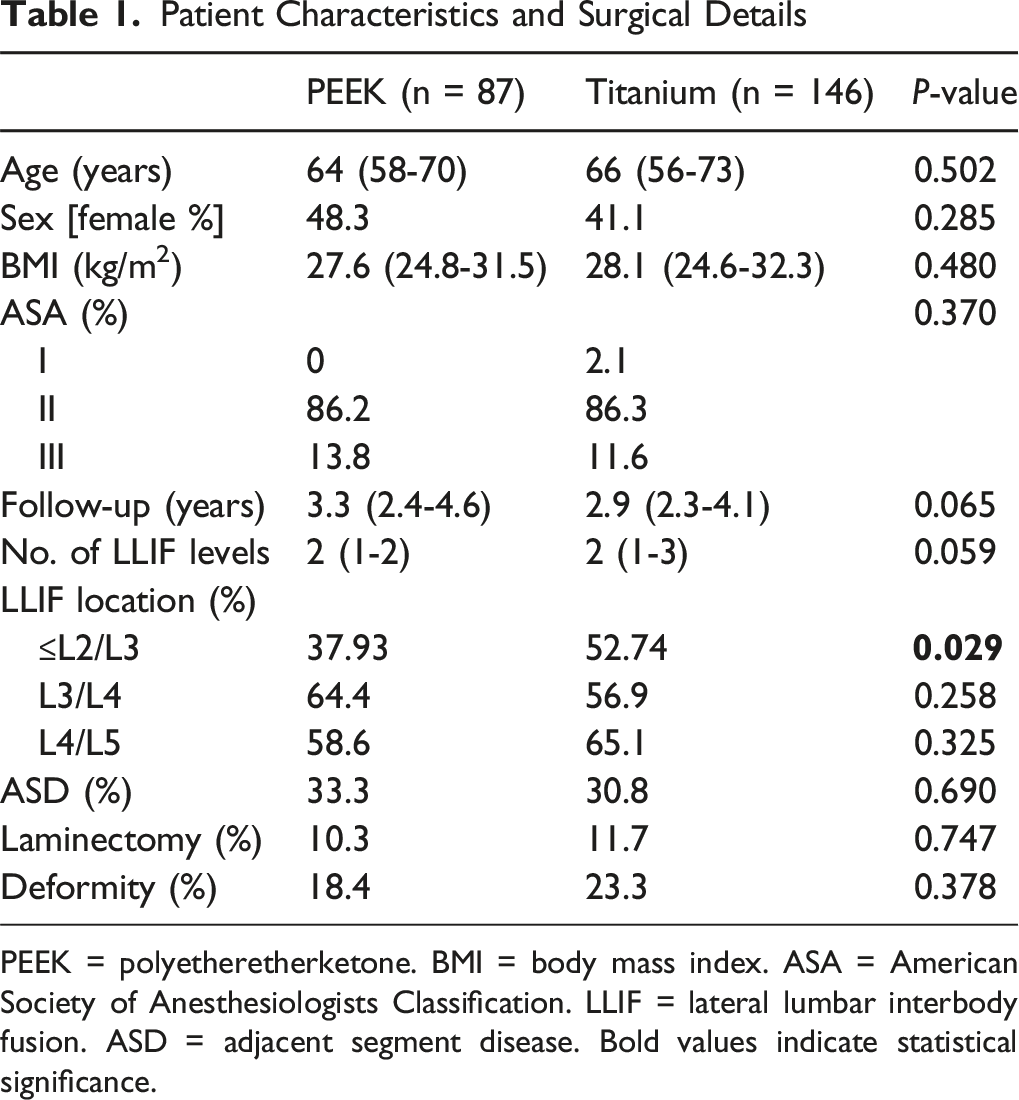
PEEK = polyetheretherketone. BMI = body mass index. ASA = American Society of Anesthesiologists Classification. LLIF = lateral lumbar interbody fusion. ASD = adjacent segment disease. Bold values indicate statistical significance.
Univariable Poisson Regression Analysis for Outcome Revision Surgery
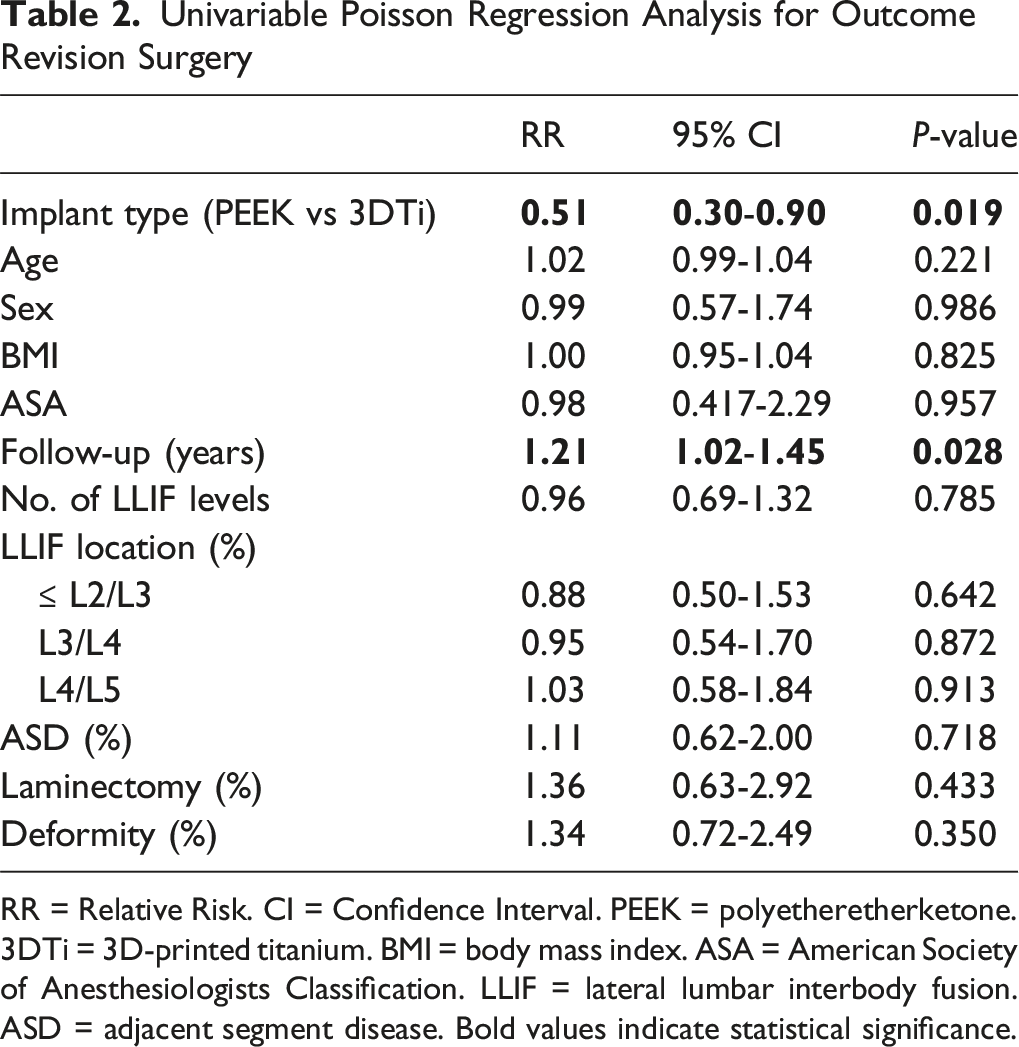
RR = Relative Risk. CI = Confidence Interval. PEEK = polyetheretherketone. 3DTi = 3D-printed titanium. BMI = body mass index. ASA = American Society of Anesthesiologists Classification. LLIF = lateral lumbar interbody fusion. ASD = adjacent segment disease. Bold values indicate statistical significance.
Multivariable Poisson Regression Model Predicting Revision Surgery
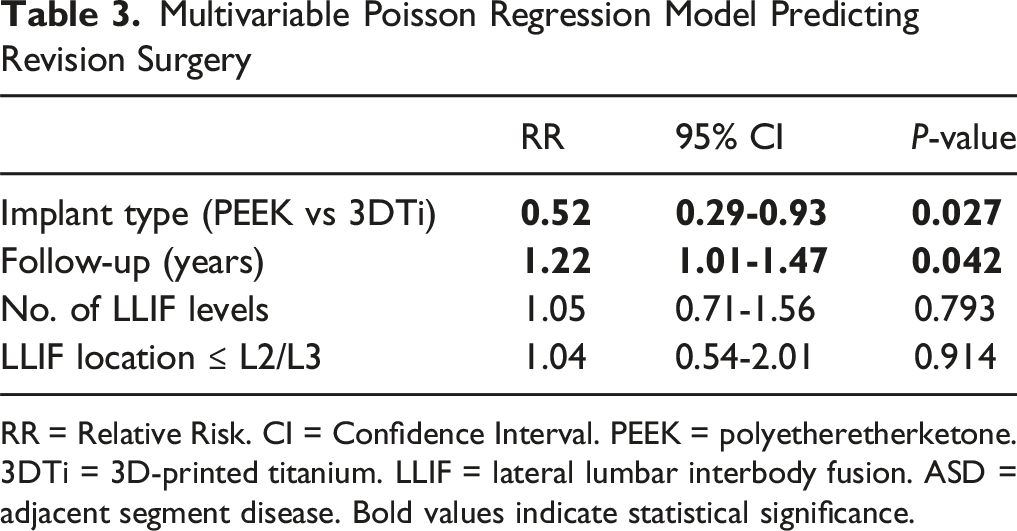
RR = Relative Risk. CI = Confidence Interval. PEEK = polyetheretherketone. 3DTi = 3D-printed titanium. LLIF = lateral lumbar interbody fusion. ASD = adjacent segment disease. Bold values indicate statistical significance.
Hierarchical Mixed-Effects Poisson Regression Model for Predicting Revision Surgery
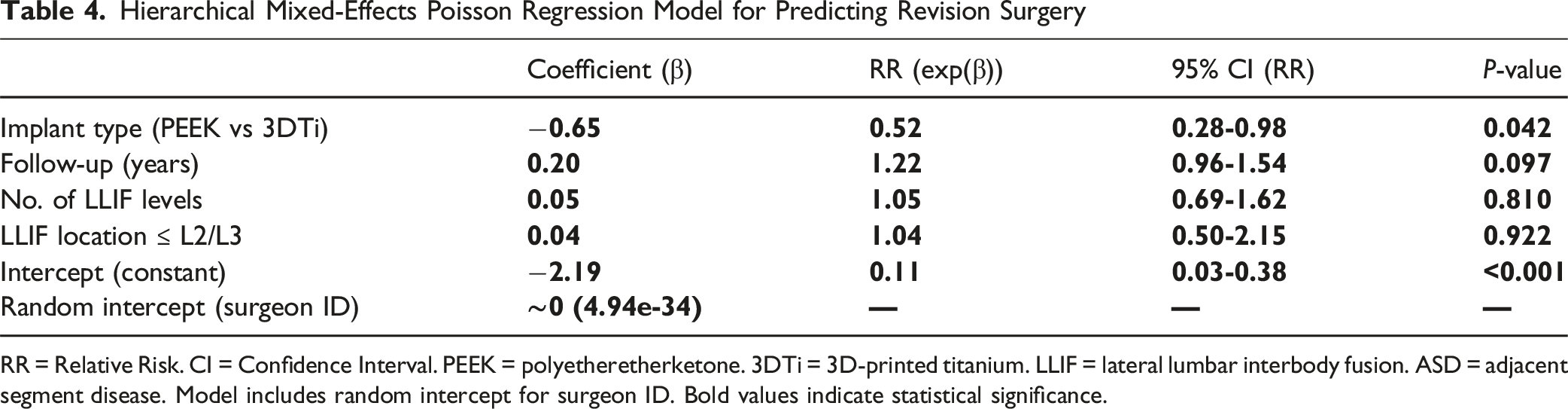
RR = Relative Risk. CI = Confidence Interval. PEEK = polyetheretherketone. 3DTi = 3D-printed titanium. LLIF = lateral lumbar interbody fusion. ASD = adjacent segment disease. Model includes random intercept for surgeon ID. Bold values indicate statistical significance.
Rates, Timing and Types of Revision Surgery
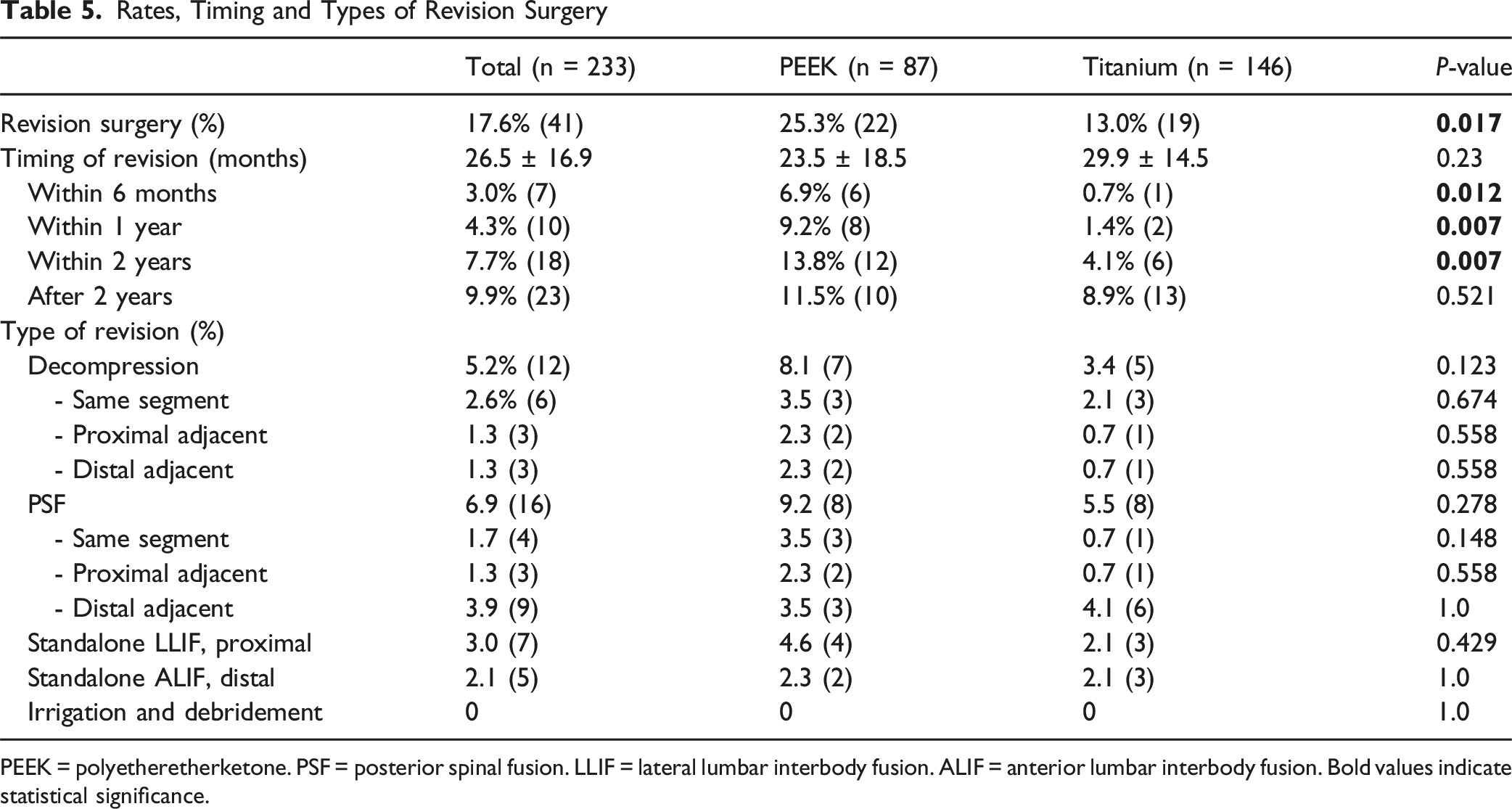
PEEK = polyetheretherketone. PSF = posterior spinal fusion. LLIF = lateral lumbar interbody fusion. ALIF = anterior lumbar interbody fusion. Bold values indicate statistical significance.
Discussion
This study represents the largest investigation to date on risk factors for revision surgery following SA-LLIF, specifically examining the impact of novel 3DTi cages. The key findings are as follows: (1) 3DTi cages significantly reduce the risk of revision surgery after SA-LLIF, with a 50% risk reduction compared to PEEK cages. (2) The risk reduction with 3DTi is most evident within the first 2 postoperative years. (3) In a mean follow-up of 3 years, the risk of revision surgery after SA-LLIF with 3DTi is 13%, with 2.7%, 3.4% and 6.9% of the revisions performed at the same, proximal, and distal adjacent segments, respectively.
PEEK vs 3D-Printed Titanium
Our findings are consistent with prior studies demonstrating clinical advantages of 3DTi cages over PEEK cages in SA-LLIF. In a propensity-matched analysis with 1 year follow-up, Alan et al. 19 found a significantly improved rate of cage subsidence of 8% with 3DTi vs 27% with PEEK. Similar findings were reported by Adl Amini et al., 17 who observed high-grade cage subsidence in 3% of patients with 3DTi vs 19% with PEEK. In a subsequent study, Adl Amini et al. 18 reported a fusion rate of 95% with 3DTi compared to 80% with PEEK, based on postoperative computed tomography performed more than 1 year after surgery. This fusion rate also exceeds the pooled fusion rate of 88% for SA-LLIF reported in a recent systematic review, 11 and falls within the range of fusion rates reported for circumferential fusion procedures. 22
The study by Alan et al. 19 on a mixed cohort of SA-LLIF and circumferential LLIF additionally reported a reoperation rate due to cage subsidence of 1% with 3DTi vs 5% with PEEK, though the difference was not statistically significant. Based on this difference, the authors estimated that 3DTi cages could be considered economically superior to PEEK at a differential cost threshold of approximately $1185. However, the analysis did not account for other causes of revision surgery or the impact of longer-term follow-up, both of which are likely to influence overall cost-effectiveness and clinical decision-making. 23
In contrast, our study exclusively investigated SA-LLIF, eliminating confounding from posterior instrumentation and providing a clearer picture of implant performance in this muscle-preserving approach. With a larger sample and longer follow-up than prior work, we found a more pronounced effect of 3DTi use on revision risk. The observed 50% reduction in revision rates has important implications not only for improving patient outcomes but also for optimizing healthcare resource utilization. This association remained significant after adjusting for follow-up duration, number of surgical levels, and clustering by surgeon using a hierarchical mixed-effects model. Notably, the random intercept variance for surgeon was negligible, indicating minimal variability in revision risk attributable to differences in surgical technique or clinical decision-making within our institution. Previous cost-effectiveness analyses have demonstrated comparable economic profiles between SA-LLIF and posterior instrumentation using transforaminal lumbar interbody fusion. 24 Improved outcomes associated with 3DTi may make SA-LLIF an increasingly attractive option for a broad range of patients. Nevertheless, this interpretation remains speculative and requires further investigation.
Evaluation of Success with Standalone LLIF
Cage subsidence may not necessarily indicate treatment failure, particularly if solid fusion is ultimately achieved. Nonetheless, subsidence results in a loss of disc height and segmental lordosis, which can contribute to recurrent stenosis and may increase biomechanical loading on adjacent segments. This could, in part, explain the observed trend toward lower rates of both adjacent segment and same-segment revision surgeries in the 3DTi group in the present study, although these differences did not reach statistical significance. Evidence suggests that the indirect decompression achieved through LLIF can improve over time, as hypertrophied ligamentum flavum and residual disc protrusions gradually regress once the segment is stabilized.8,25 Therefore, cage subsidence may not preclude favorable clinical outcomes, provided that segmental stability is maintained, and neural elements remain adequately decompressed.
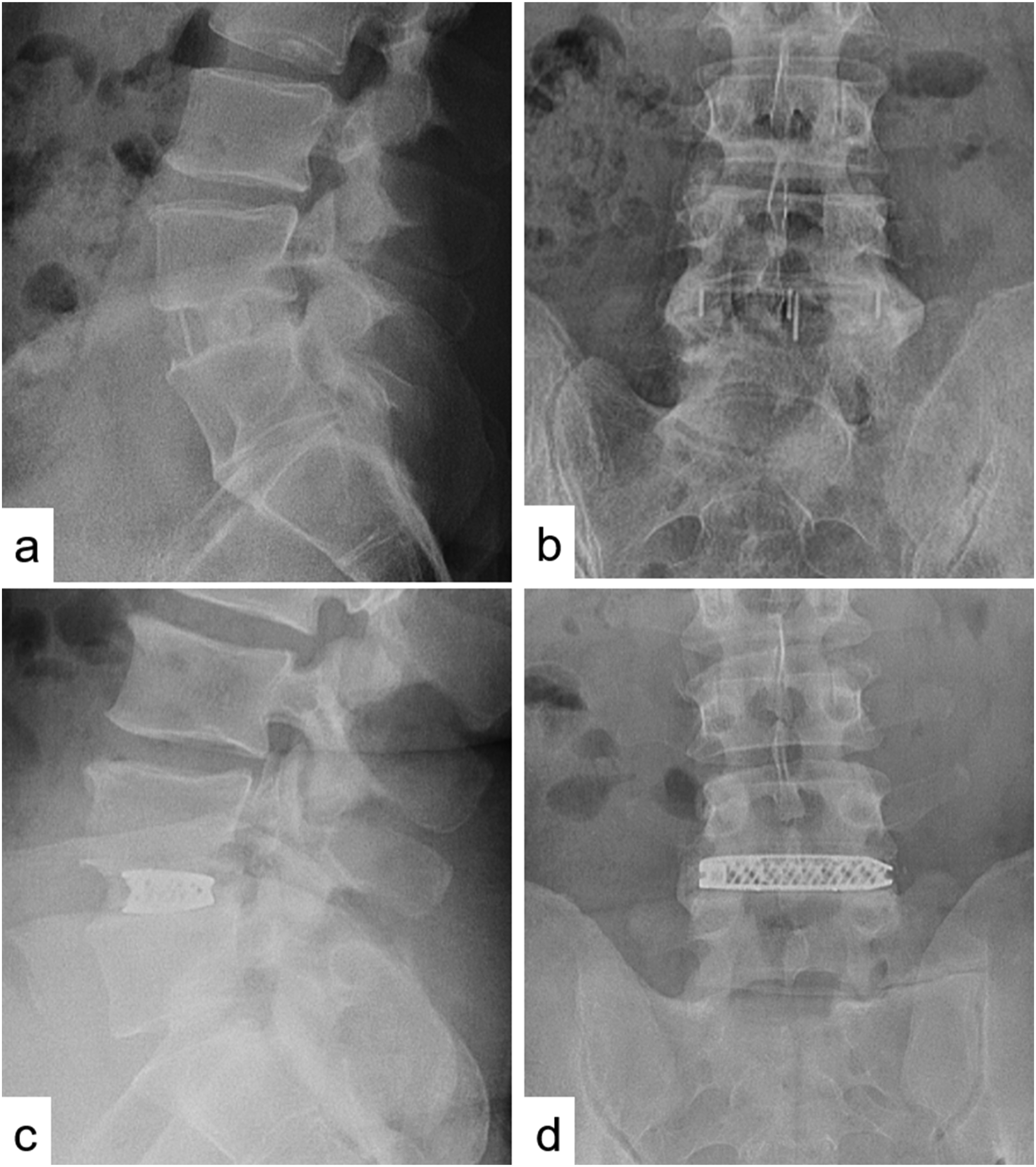
Although higher fusion rates have been reported with 3DTi cages, it is worth questioning whether complete bony bridging across the entire disc space is essential for achieving successful clinical outcomes. Titanium implants, which promote osseointegration, may offer sufficient segmental stability through localized integration into the cranial and caudal endplates, even in the absence of continuous fusion across the intervertebral space. Traditional radiographic criteria for defining fusion success may therefore need to be reconsidered, particularly in the context of 3DTi implants, which provide distinct biomechanical and osseointegrative advantages (Figures 2 and 3). However, this interpretation remains speculative and warrants further investigation. SA-LLIF With PEEK and 3DTi. First Postoperative Lateral and Anteroposterior Radiographs of SA-LLIF with PEEK Cage (A & B) and With a 3DTi Cage (C & D) 2-Year Postoperative Radiographs. Lateral and Anteroposterior Views of SA-LLIF With a PEEK Cage (A & B) and a 3DTi Cage (C & D), Showcasing the Same Patients as in Figure 2. Defining Radiographic Failure or Success Remains Challenging, Particularly With the Unique Properties of 3DTi Implants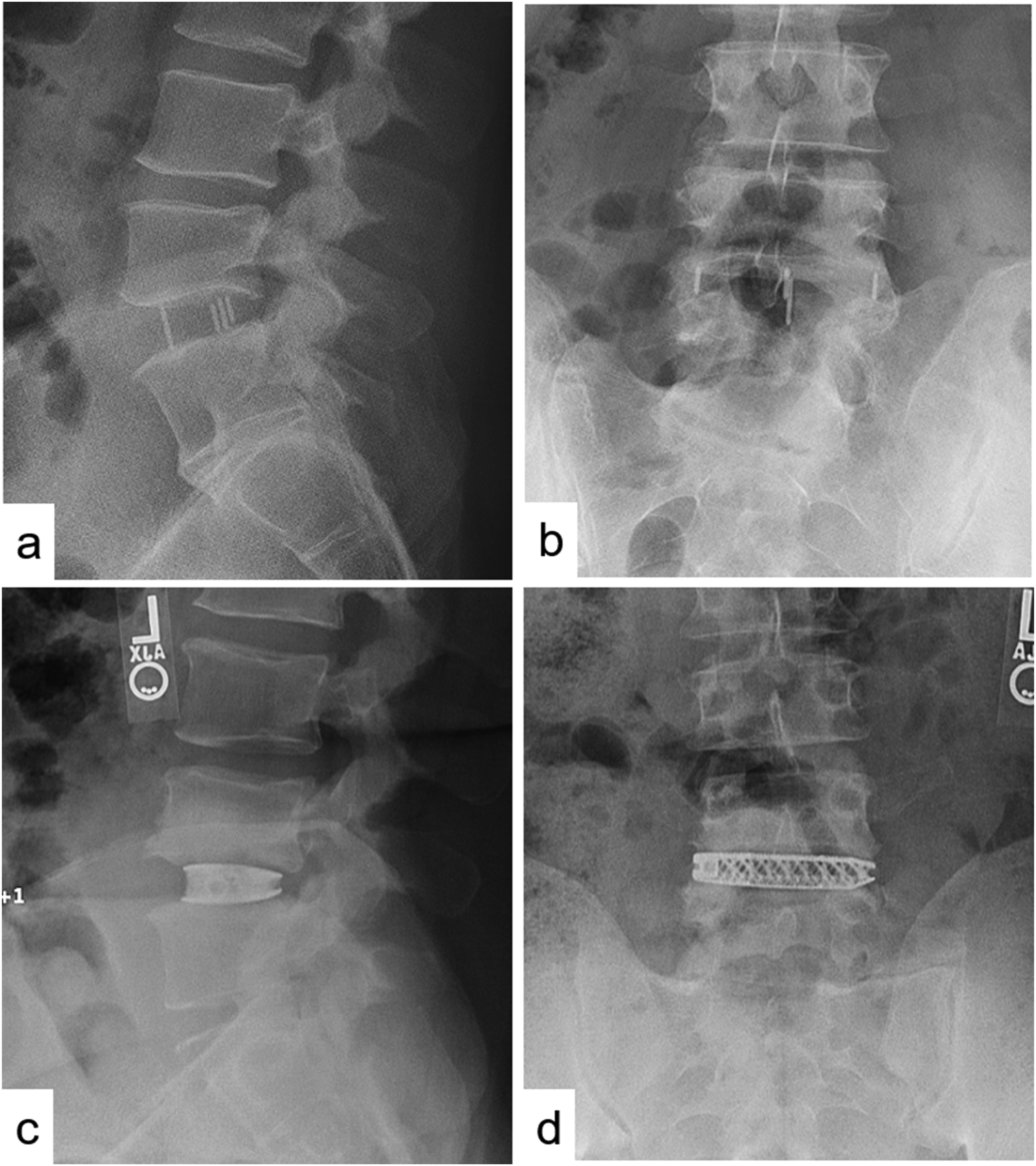
Standalone LLIF vs Posterior/Circumferential Fusion
Our focus on SA-LLIF provides clinically actionable insights that are particularly relevant in the current era, where preserving the paraspinal musculature, particularly the multifidus muscle, is increasingly recognized as critical for achieving favorable outcomes and minimizing the risk of adjacent segment disease. 7 As a muscle-sparing alternative to posterior approaches, SA-LLIF is gaining traction due to its favorable perioperative profile, including reduced intraoperative loss, lower complication rates, and shorter hospital stays. 26 SA-LLIF is also associated with lower rates of surgical site infection, with none occurring in our study cohort, an observation that aligns with other studies. 27 In addition, SA-LLIF has been shown to reduce the risk of adjacent segment surgery compared to circumferential LLIF with pedicle screw instrumentation. 28 However, further research is needed to assess additional long-term outcome measures and investigate which patient may benefit more from 1 procedure over the other.
In our opinion, true muscle-preserving fusion techniques include SA-LLIF and standalone anterior lumbar interbody fusion (ALIF). In contrast, minimally invasive transforaminal lumbar interbody fusion (MIS-TLIF) may still result in injury to the multifidus and erector spinae muscles due to posterolateral dissection and disruption of the medial branch of the dorsal rami, which innervates the multifidus and courses directly over the pedicle screw insertion site.28-30 Although this topic falls outside the scope of the present study, it warrants further investigation. Ideally, future research should be conducted in a prospective, multicenter setting with the routine use of advanced postoperative imaging to assess muscle preservation and its association with long-term outcomes.
Limitations
This retrospective, single-center study has several limitations. First, the non-randomized design introduces potential bias, particularly given that PEEK cages were used exclusively by 1 surgeon, while 3 other surgeons used 3D-printed titanium (3DTi) cages. Differences in surgical technique, implant selection preferences, and decision-making for revision surgery may have influenced the observed outcomes. To address this, a hierarchical mixed-effects Poisson regression was performed with surgeon ID included as a random intercept to account for clustering by surgeon. Second, factors such as patient demographics, case complexity, and the availability of advanced technologies specific to our institution may restrict the generalizability of the findings to other settings. However, all 4 surgeons in this study are senior surgeons with more than 15 years of experience in LLIF surgery and routinely perform both SA-LLIF and circumferential LLIF. Importantly, all 4 surgeons were using PEEK cages in all patients before 3DTi cages became available. Third, while revision surgery is an objective and clinically meaningful endpoint, our study did not include data on patient-reported outcomes or specific postoperative complications. Future prospective studies incorporating these measures, as well as direct comparisons across fusion strategies and implant materials, are warranted. Despite these limitations, the promising outcomes observed with SA-LLIF using 3DTi cages support the need for further investigation and may inform surgical decision-making in select patient populations.
Conclusions
3D-printed Titanium cages significantly reduced the revision rate in SA-LLIF by half compared to PEEK, particularly in the early postoperative period. These findings support the implementation of 3DTi implants to improve surgical outcomes in SA-LLIF. Further research is needed to explore the long-term benefits of 3DTi and SA- LLIF over other fusion techniques in various clinical settings.
Footnotes
ORCID iDs
Ethical Considerations
The hospital’s institutional review board (IRB) approved the conduct of the study (#2022-0108).
Consent to Participate
Written and informed consent to participate in this study and to publish their data was obtained from all individual participants included in the study.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Marco D. Burkhard, Ali E. Guven, Bryce Demopoulos, Simon Ortiz and Torben Stepan. The first draft of the manuscript was written by Marco D. Burkhard and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FPG reports royalties from Lanx, Inc. and Ortho Development Corp.; ownership interest in Centinel Spine, BICMD; consulting fees from Lanx, Inc, Ortho Development Corp, and Sea Spine; and stock ownership in Centinel Spine, Healthpoint Capital Partners, LP; membership of scientific advisory board/other office of Healthpoint Capital Partners. FPC reports research support from Camber Spine, Centirnel Spine, Choice Spine, Depuy Syntes and Royal Biologic; ownership interest for 4WEB Medical/4WEB, Inc.; Healthpoint Capital Partners, LP, ISPH II, ISPB III hodlings, VBVP VI LLC, VBVP X LLC, Medical Device Partners II and III, Orthobond Corporation; Spine Biopharma, LLC; Tissue Connect Systems, Inc.; Woven Orthopedics Technologies. AAS reports royalties from Ortho Development, Corp. DePuy Spine Products/Medical Device Business Services, Clariance, Inc.; private investments for Vestia Ventures MiRUS Investment, LLC, ISPH II, LLC, ISPH 3, LLC, and Centinel Spine (Vbros Venture Partners V); consulting fees from Clariance, Inc., Kuros Biosciences AG; and research support from Spinal Kinetics, Inc. and Orthofix, Inc. APH reports research support from Kuros Biosciences AG and Expanding Innovations, Inc.; private investments in Tissue Connect Systems, Inc.; and fellowship support from NuVasive and ATec; outside the submitted work. For the remaining authors none were declared.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
