Abstract
Study Design
Retrospective cohort
Objective
Lateral lumbar interbody fusion (LLIF) commonly involves a transpsoas approach. Despite the association between LLIF, postoperative iliopsoas weakness, and iatrogenic neuropraxia, no study has yet examined the effect of psoas or multifidus muscle quality on patient-reported outcomes (PROs).
Methods
This study retrospectively reviewed patients who underwent LLIF with 1-year minimum follow-up. Psoas and multifidus muscle qualities were graded on preoperative magnetic resonance imaging using two validated classification systems for muscle atrophy. Average muscle quality was calculated as the mean score from all levels (L1-2 through L5-S1). Univariate and multivariate statistics were utilized to investigate the relationship between psoas/multifidus muscle quality and preoperative, 6-weeks postoperative, and final postoperative PROs.
Results
74 patients (110 levels) with a mean follow-up of 18.71 ± 8.02 months were included for analysis. Greater multifidus atrophy was associated with less improvement on ODI, SF12, and VR12 (
Conclusion
Despite the direct manipulation of the psoas muscle inherent to LLIF, preoperative psoas muscle quality did not affect postoperative outcomes. Rather, the extent of preoperative multifidus fatty infiltration and atrophy was more likely to predict postoperative pain and disability. These findings suggest that multifidus atrophy may be more pertinent than psoas atrophy in its association with patient-reported outcome measures after LLIF.
Keywords
Introduction
Sarcopenia, or the age-related loss of both muscle mass and function (strength or performance) is a condition with worldwide prevalence as high as 10% in both men and women.1,2 More importantly, its association with poor clinical outcomes in a variety of specialties has now been well documented, with studies demonstrating higher mortality in emergency surgery situations, poorer prognosis in oncologic patients, and worse outcomes in elective procedures.3-6 As the population continues to age, and sarcopenia remains a prevalent but under-diagnosed condition, its deleterious effects on patients’ ability to recover will become even more pronounced.
With regards to the spine, sarcopenia has been shown to be a predictor of complications and poor clinical outcomes across a variety of different pathologies in the cervical, thoracic, and lumbar spine.7-10 Currently, there remains some variability between results, with individual studies reporting both significant and non-significant associations with outcomes following lumbar fusion. In addition, large systematic reviews have found an equivocal effect, with heterogeneity in definition and measurement tools being particularly problematic.11-15 However, several studies have reported significant associations between sarcopenia at baseline and adverse postoperative pain, functional disability, balance, and alignment.16-20
Lateral lumbar interbody fusion (LLIF) is a commonly performed less invasive lumbar fusion technique which relies on an access portal that traverses the psoas muscle. Although there is a plethora of data on the impacts of psoas muscle quality on outcomes following a variety of surgical procedures in varying specialties, surprisingly there is a paucity of data on the role psoas muscle quality plays in LLIF. Similarly, there is limited data available on the impact of paraspinal muscle quality on outcomes in LLIF despite recently increasing focus on this parameter in spine literature.
This is study sought to assess the impact of psoas and multifidus muscle quality on patient reported outcomes (PROs) following LLIF. Due to the known association between traditional posterior approach lumbar spinal surgery and paraspinal muscle atrophy, we additionally sought to identify effects of prior lumbar surgery on PROs in LLIF as well as control for this variable in statistical analysis.
Methods
Patient Selection
Institutional Review Board approval was obtained (ORA 20073102) to retrospectively query a prospectively maintained database of all consecutive patients who had undergone LLIF with one of the two senior surgeons between 2014 and 2019. Due to its retrospective nature, this study was exempt from obtaining informed consent. The inclusion criteria were patients over the age of 18 who underwent LLIF for indications of isthmic spondylolisthesis, degenerative spondylolisthesis, severe degenerative disc disease, or foraminal stenosis with a minimum of 1 year follow-up, available preoperative magnetic resonance imaging (MRI) of the lumbar spine, and preoperative, 6-weeks postoperative, and final postoperative (minimum 1 year) PRO surveys. Patients were excluded from the analysis if they had received concomitant treatment for the indications of tumor, trauma, or infection.
Demographic and operative data collected included age, sex, body mass index (BMI), American Society of Anesthesiologists (ASA) physical status, the modified frailty index (mFI), medical comorbidities, operative level(s), operative time, and estimated blood loss (EBL). Procedural characteristics, such as the use of posterior percutaneous instrumentation and past open surgery to the lumbar spine were also collected. In patients with previous lumbar surgery, percutaneous instrumentation was used when there was concern for instability at the operative level. The PROs evaluated included the Oswestry Disability Index (ODI), Visual Analog Scale (VAS) Back, VAS Leg, Short Form 12-item healthy survey physical component score (SF12 PCS), and Veteran’s RAND 12-item health survey (VR12) PCS. PRO scores were collected from the preoperative, 6 weeks, and final postoperative (minimum 1 year) time points.
Muscle Grading
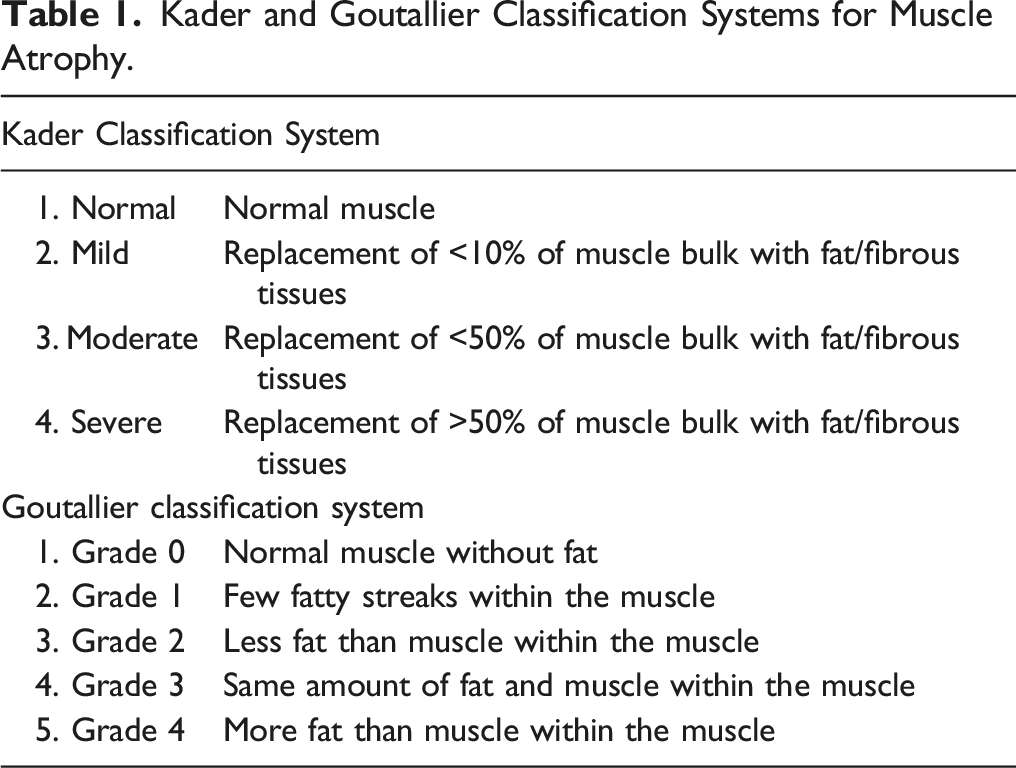
Kader and Goutallier Classification Systems for Muscle Atrophy.
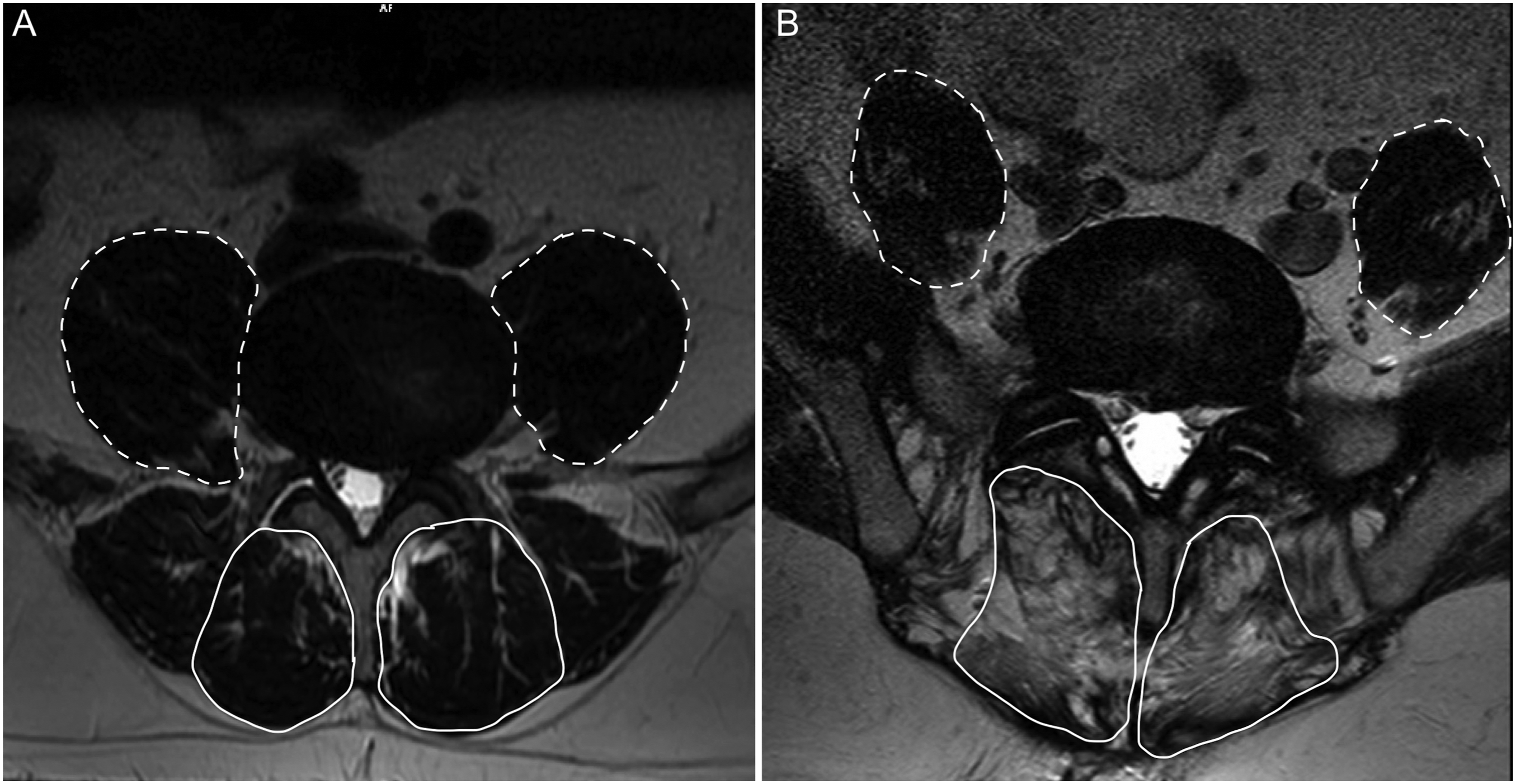
Representative axial magnetic resonance imaging demonstrating (A) normal and (B) atrophic psoas (dashed line) and multifidus (solid line) musculature.
Statistical Analysis
All statistical methods were performed using SPSS version 26.0 (IBM Corporation, Armonk, NY). Patient demographics were established using descriptive statistics. Continuous variables were reported as means and standard deviations, and categorical variables were reported as proportions and absolute counts, unless otherwise specified. Postoperative improvement on PROs was evaluated with dependent
Results
Patient Demographics
Demographics and Operative Characteristics.
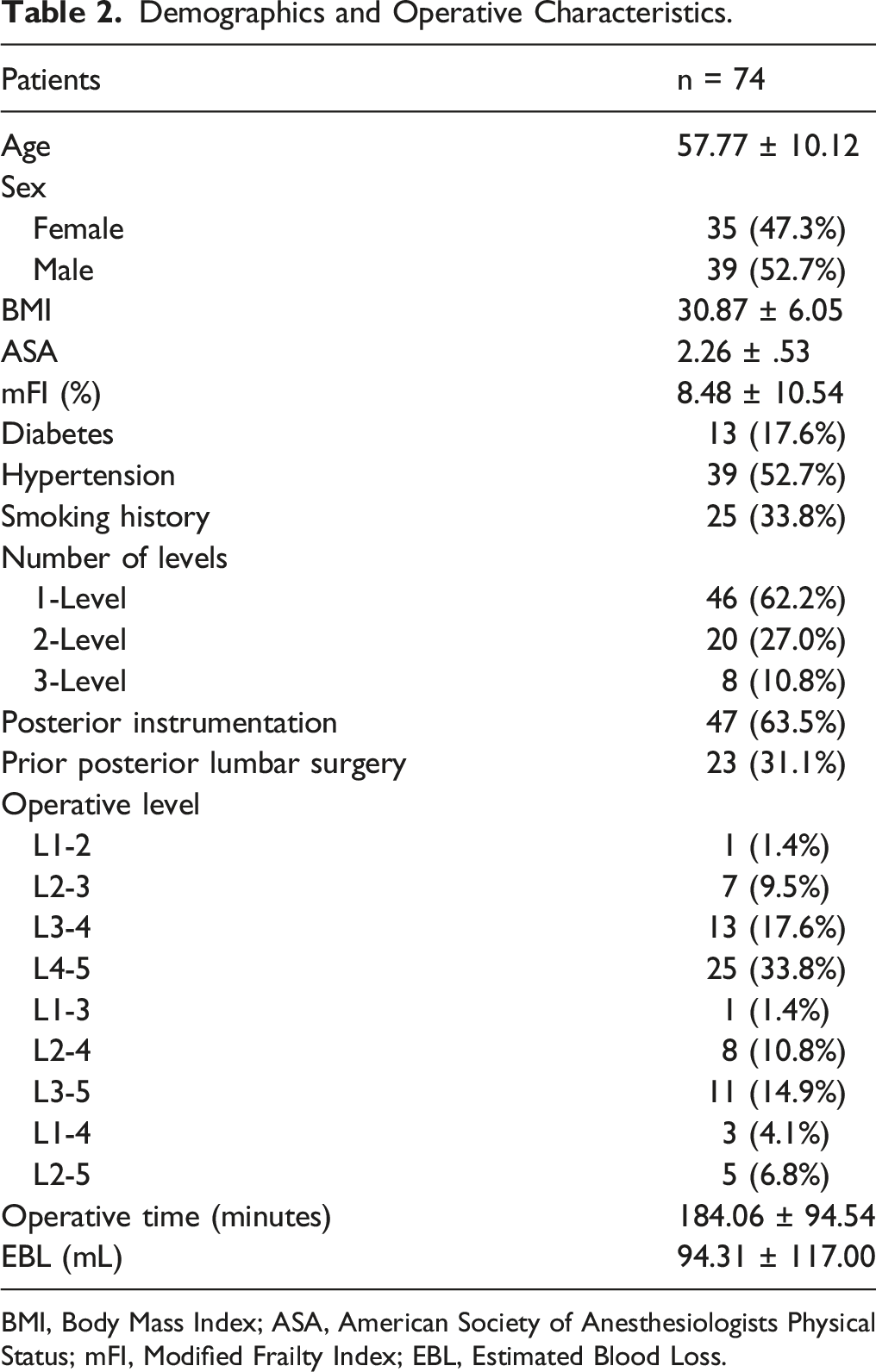
BMI, Body Mass Index; ASA, American Society of Anesthesiologists Physical Status; mFI, Modified Frailty Index; EBL, Estimated Blood Loss.
Muscle Quality
Grading of Psoas and Multifidus Muscle Atrophy.
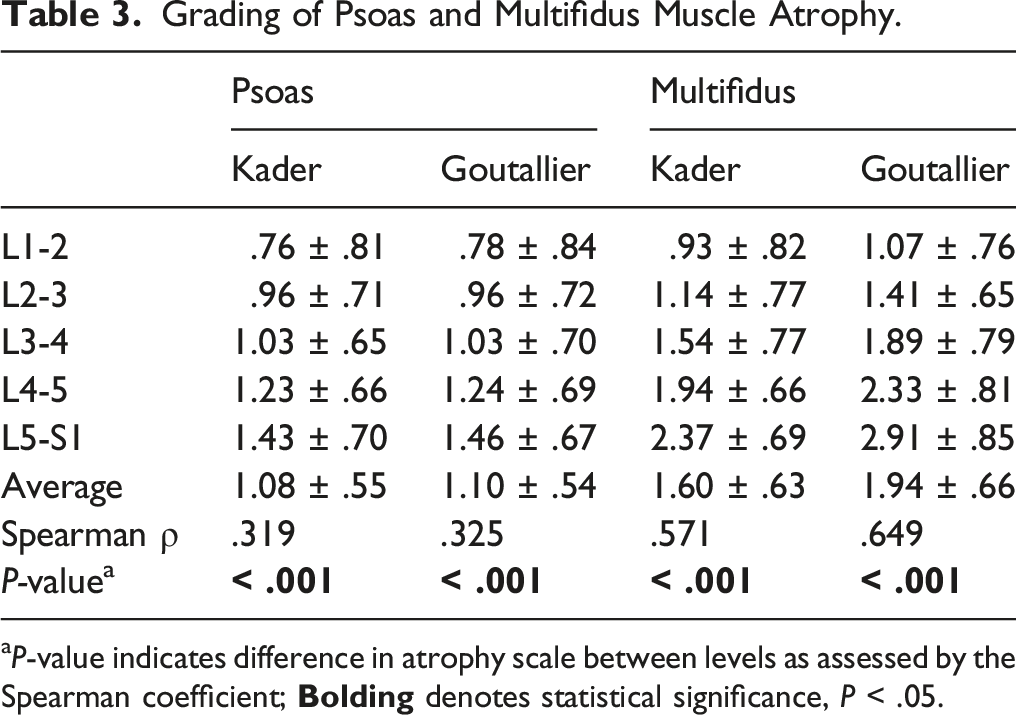
a
Psoas atrophy significantly correlated with increased age (KCS:
Multifidus atrophy was significantly worse among patients with a history of prior posterior lumbar surgery (KCS: 1.44 ± .63 vs 1.94 ± .51,
Patient-Reported Outcomes: Primary Versus Prior Lumbar Surgery
Both patients with and without prior lumbar surgery experienced improvement from the preoperative to 6-weeks and final postoperative time points on ODI, VAS Leg, and VAS Back (
Patient Reported Outcomes in Patients Undergoing Primary Lateral Lumbar Interbody Fusion Compared to Those With a History of Prior Lumbar Surgery.
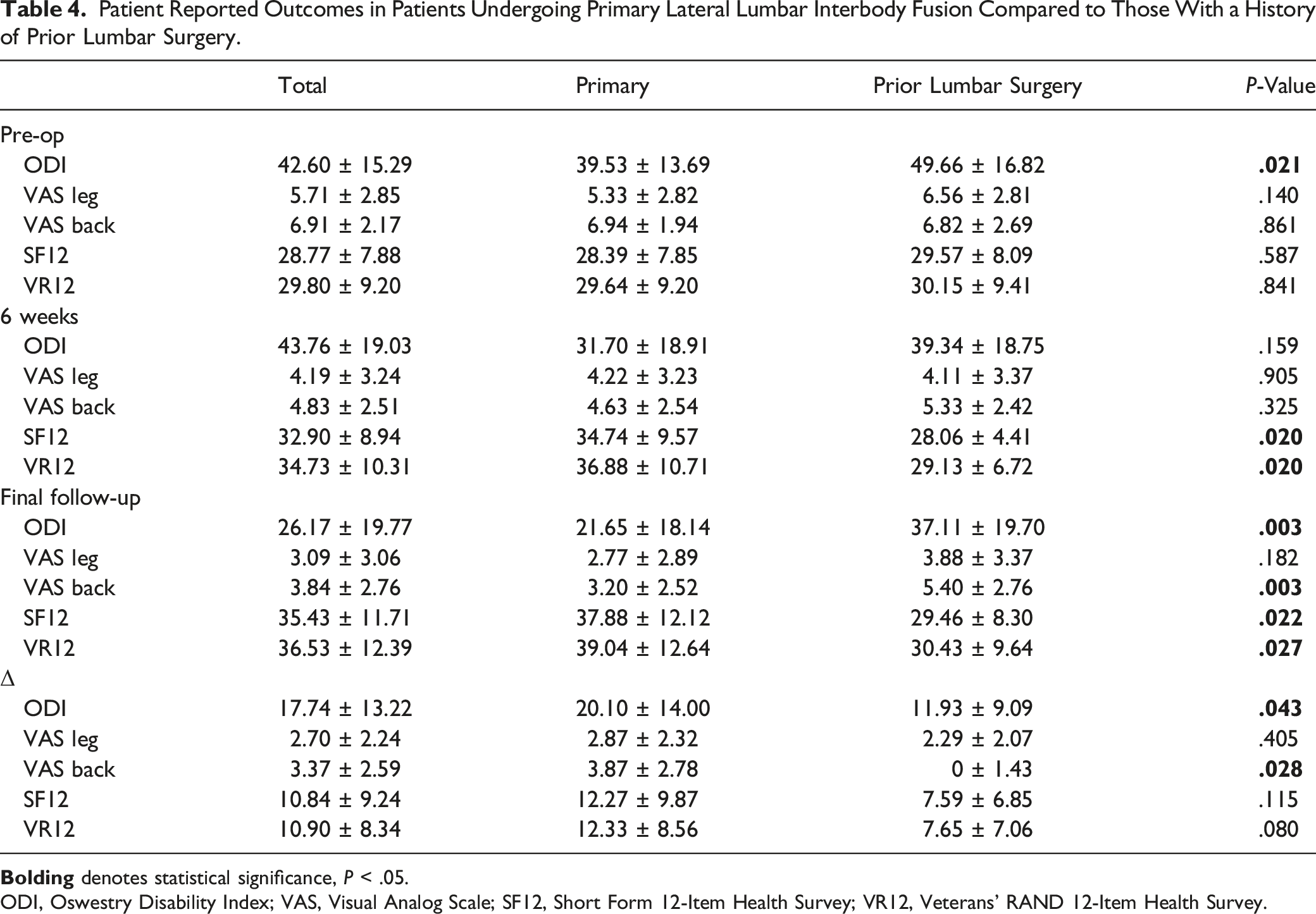
ODI, Oswestry Disability Index; VAS, Visual Analog Scale; SF12, Short Form 12-Item Health Survey; VR12, Veterans’ RAND 12-Item Health Survey.
Patient-Reported Outcomes: Effect of Psoas Quality
Preoperative Psoas Atrophy and Patient-Reported Outcomes.
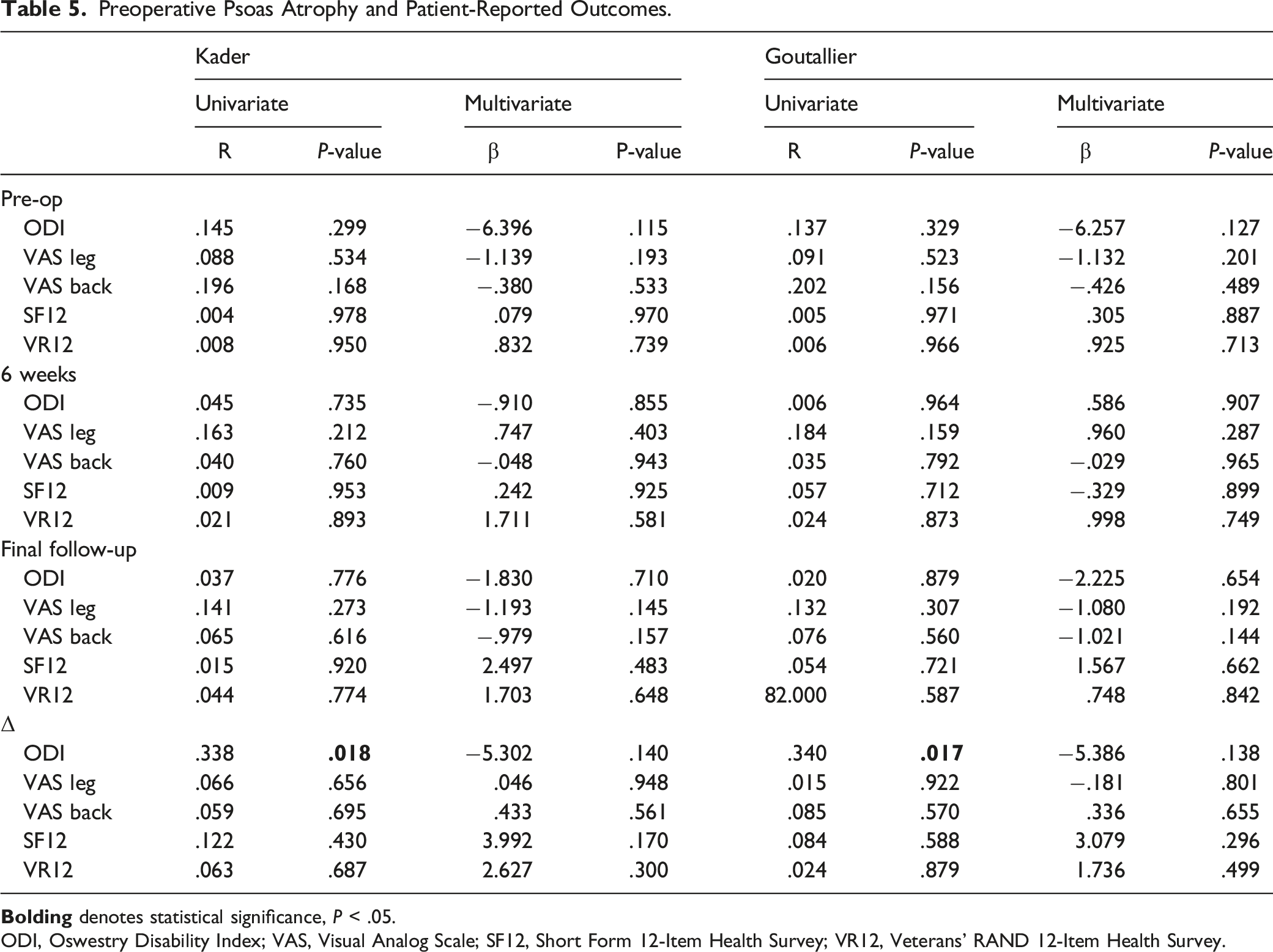
ODI, Oswestry Disability Index; VAS, Visual Analog Scale; SF12, Short Form 12-Item Health Survey; VR12, Veterans’ RAND 12-Item Health Survey.
On multivariate regression adjusting for age, sex, BMI, smoking history, diabetes, posterior percutaneous instrumentation, and history of prior lumbar surgery, this relationship was no longer significant.
Patient-Reported Outcomes: Effect of Multifidus Quality
Preoperative Multifidus Atrophy and Patient-Reported Outcomes.
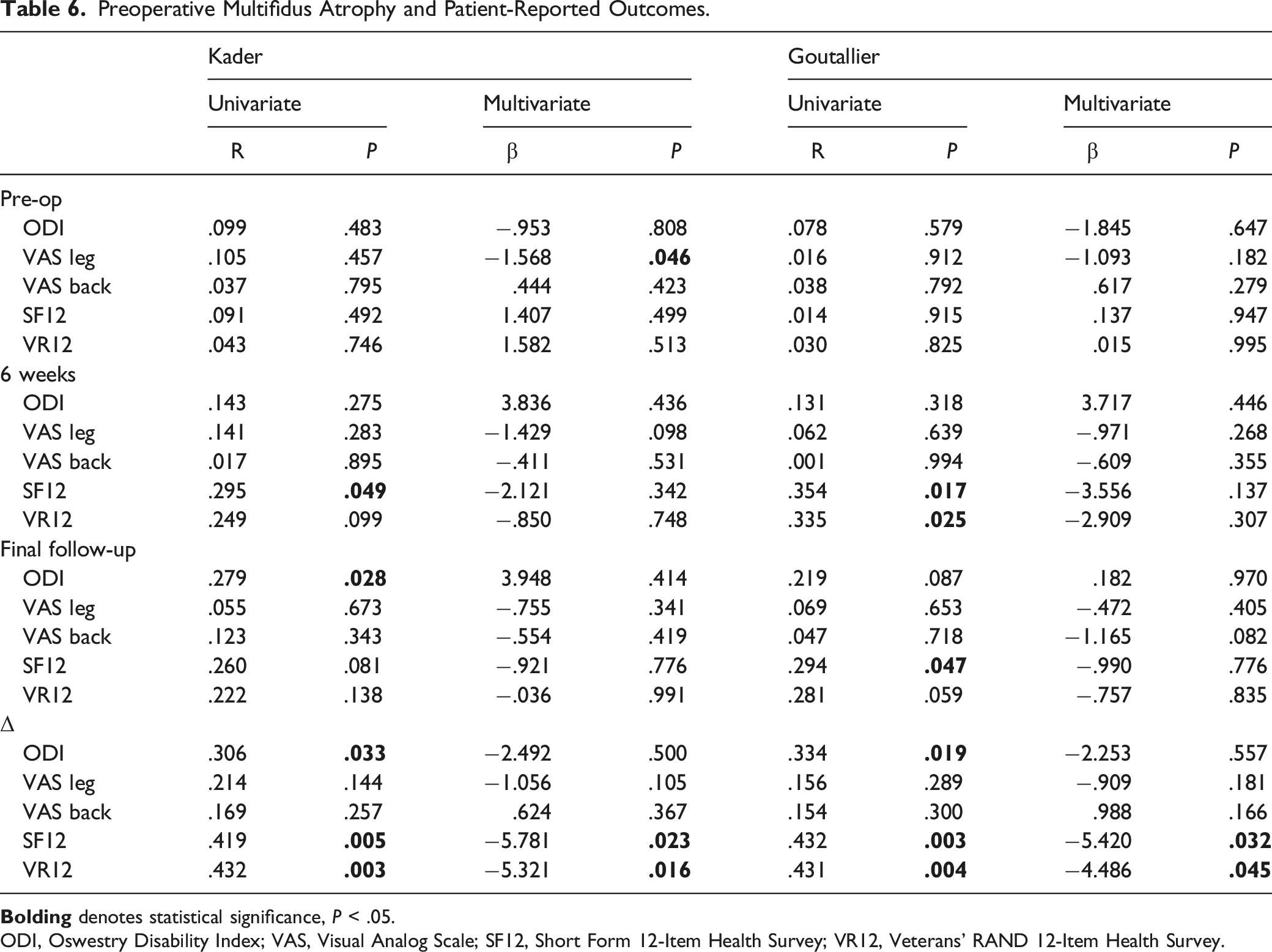
ODI, Oswestry Disability Index; VAS, Visual Analog Scale; SF12, Short Form 12-Item Health Survey; VR12, Veterans’ RAND 12-Item Health Survey.
On multivariate analysis controlling for age, sex, BMI, smoking history, diabetes, posterior percutaneous instrumentation, and history of prior lumbar surgery, a higher degree of preoperative multifidus atrophy independently predicted worse preoperative VAS Leg scores (KCS: β = −1.568,
Discussion
This study used two validated qualitative grading systems to assess preoperative psoas and multifidus fatty infiltration in patients undergoing LLIF. The analysis found significant correlations between advanced age and greater atrophic changes in both the psoas and multifidus muscles, while elevated BMI, diabetes, and history of prior posterior lumbar surgery were significantly associated with worse multifidus muscle quality. Patients with a history of prior lumbar spine surgery reported worse pain and disability on several PROs postoperatively and experienced less symptomatic resolution. Furthermore, worse psoas muscle quality significantly predicted less postoperative improvement in ODI on univariate analysis, although it was not an independent predictor in a multivariate model. Worse preoperative multifidus atrophy predicted worse ODI and SF12 at final follow-up, in addition to a less improvement in ODI, SF12, and VR12 via univariate statistics. On multivariate analysis, multifidus quality independently predicted worse baseline VAS Leg and a lower magnitude of improvement in SF12 and VR12, even after controlling for the effect of prior lumbar surgery.
Sarcopenia is a known risk factor contributing to poor outcomes in various orthopedic settings.4,12,24-26 In the context of lumbar surgery procedures, preoperative multifidus atrophy and fatty infiltration has been associated with worse baseline disability status but may also predict poorer postoperative outcomes.9,15,27 In addition to disuse atrophy, these atrophic changes are contributed to by denervation secondary to spinal stenosis resulting from stretching of the posterior primary ramus.28,29 In the traditional posterior approaches to lumbar fusion, including posterolateral lumbar fusion (PLF) and transforaminal lumbar interbody fusion (TLIF), the multifidus is dissected and retracted laterally to gain adequate exposure. This can lead to acceleration and further progression of sarcopenia, ultimately limiting the extent of postoperative recovery for the patient.30-32 This was also seen among our patient cohort as the 23 patients with a history of prior posterior lumbar surgery had a greater extent of multifidus atrophy prior to undergoing LLIF, though psoas atrophy was comparable to patients undergoing primary surgery. LLIF is a less invasive procedure that spares disruption of the posterior paraspinal muscles while still achieving interbody fusion and indirect decompression, though this is accomplished through a transpsoas approach. In order to avoid the technical challenges and morbidity associated with operating through scar tissue and prior instrumentation, the LLIF procedure is becoming an increasingly popular option for revision surgery and secondary intervention for adjacent segment pathology, with studies reporting low complication rates, excellent correction of spinopelvic alignment, and improvement in PROs.33-36 However, to our knowledge, no study has compared outcomes between patients undergoing LLIF as a primary surgery to those undergoing LLIF as a secondary intervention. The results of the analysis demonstrated worse baseline disability among patients with prior lumbar surgery, as well as worse pain and disability on several PRO metrics at 6 weeks and 1 year. Moreover, while these patients did report improvement on ODI, VAS Leg, and VAS Back, no significant improvement was noted in any of the general health metrics at 6 weeks or 1 year.
Beyond its function as a hip flexor, the psoas contributes to the dynamic stabilization of the vertebral column, functioning in synergy with the posterior paraspinal muscles. 37 The psoas also harbors the lumbosacral plexus as its nerves exit and travel distally. The transpsoas approach exposes the psoas and the lumbosacral plexus to iatrogenic injury. In mild cases, this can result in transient hip flexion weakness and in severe cases can result in permanent injury to the lumbosacral plexus. 38 Despite these known risks of transpsoas LLIF, little data is available regarding the impact of psoas muscle quality on PROs in patients undergoing this procedure. Similarly, there is a paucity of data regarding the effects of paraspinal quality on PROs following LLIF.
Analysis of psoas muscle quality did not identify significant relationships between psoas atrophy and pre- or post-operative patient reported outcomes. In contrast, analysis of multifidus muscle quality did identify a relationship between multifidus atrophy and worse postoperative outcomes. These findings remained significant on multivariate analysis, even when controlling for confounding variables including prior posterior lumbar surgery, age, and diabetes, among others. This suggests that the level of paraspinal atrophy prior to surgery may predict the magnitude of postoperative improvement in pain and disability, while other factors may also contribute. Interestingly, pre-operative PROs were not correlated with multifidus atrophy.
When comparing the results of this analysis to the limited data reported in the literature, our findings seem to contrast with those reported by Wagner et al 15 in their investigation of the effect of psoas and multifidus cross sectional area (CSA) on preoperative ODI in patients undergoing single level PLF for degenerative spondylolisthesis. 15 The authors concluded that patients with severe disability had a significantly smaller psoas CSA than those with moderate disability but found no differences in posterior paraspinal CSA. However, they did not investigate the relationship between CSA and postoperative pain and disability outcomes. Furthermore, several studies have suggested that qualitative grading of fatty infiltration may be a better tool for assessing the effect of sarcopenia on disability than quantitative measures such as CSA, since quantitative measures do not account for tissue architecture and myosteatosis.39,40 Additionally, although both sarcopenia and frailty have each been independently associated with a higher incidence of adverse events following spine surgery,8,41,42 the results of this analysis did not identify a significant correlation between frailty as assessed by the mFI and sarcopenia as judged by psoas and multifidus atrophy. In a study investigating the impact of frailty on PROs in a population of patients who underwent surgery to the thoracolumbar spine, Beauchamp-Chalifour et al 43 found no significant relationship between mFI scores and postoperative PROs, despite a higher incidence of adverse events in the frail patient population. 43 Therefore, while both frailty and sarcopenia may help predict the incidence of adverse postoperative events, sarcopenia may be a better predictor for patient-reported outcomes following lumbar spine surgery in the adult patient population. Future investigations could also evaluate whether sarcopenia alone is a factor that may lead surgeons to considering supplemental stability with percutaneous instrumentation.
Results of this analysis suggest that multifidus muscle quality is a better predictor for postoperative outcomes than psoas muscle quality. Findings also indicate that despite transpsoas approach in LLIF, poor psoas muscle quality does not portend increased risk of post-operative disability or negatively impact outcomes up to a 1-year time point. Like the psoas, the multifidus is easily visualized on axial cut MRI and CT and can therefore help surgeons assess patients’ recovery potential preoperatively. Moreover, these results further support the utilization of the LLIF procedure as a posterior paraspinal muscle sparing approach, particularly among sarcopenic patients. Future areas for investigation could include directly comparing the predictive value of psoas and multifidus muscle quality on patient outcomes in cohorts undergoing LLIF or lumbar fusion via a posterior approach. Additionally, while a study by Inoue
There are several limitations to this study which must be considered. First, there are inherent limitations to the retrospective study design, such as the potential for observer and reporting bias. Additionally, all procedures were performed by one of two surgeons, thus introducing the potential for heterogeneity in procedural methods and postoperative protocols. Furthermore, multifidus and psoas muscle quality were evaluated using two subjective, qualitative grading systems. However, to mitigate the potential for bias, imaging evaluation was performed by a single reviewer who was blinded to the patients’ identity and outcomes.
Conclusion
Despite the transpsoas approach inherent to the LLIF procedure, preoperative psoas muscle quality did not affect postoperative outcomes. Rather, the extent of preoperative multifidus fatty infiltration and atrophy was more likely to predict postoperative pain and disability. Additionally, analysis identified that a history of prior posterior spinal surgery resulted in worse PROs at all time points following LLIF as well as less postoperative symptomatic improvement. Surgeons should consider these findings during preoperative counseling and planning to better predict patient outcomes. Future longitudinal studies are needed to qualitatively analyze the postoperative changes in psoas and paraspinal muscle architecture following LLIF.
Footnotes
Authors’ Note
The Manuscript submitted does not contain information about medical device(s)/drug(s). This work was performed at Rush University Medical Center, Chicago, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Each author certifies that his or her institution waived approval for the reporting of this investigation and that all investigations were conducted in conformity with ethical principles of research.
