Abstract
Study Design
Literature review.
Objective
To systematically review blood biomarker profiles in adolescent idiopathic scoliosis (AIS) and evaluate their pathophysiological implications and clinical potential for diagnosis, prognosis, and therapeutic monitoring.
Methods
A comprehensive literature search was conducted to identify studies examining blood-based biomarkers in AIS patients, including alterations in vitamin D, calcium, trace elements, sex hormones, leptin, melatonin, growth hormone, ghrelin, bone metabolism factors, microRNAs, proteomic and metabolomic profiles. Relevant findings were synthesized to elucidate systemic biological alterations associated with AIS.
Results
Consistent vitamin D and calcium deficiencies, hormonal dysregulation (leptin, estrogen, ghrelin), altered trace element levels (selenium), and distinct changes in bone metabolism markers (osteocalcin, P1NP, RANKL) were observed in AIS patients. Emerging evidence supports the involvement of specific microRNAs and proteomic/metabolomic profiles in AIS pathogenesis and curve progression. These blood biomarkers exhibit potential for early diagnosis, risk stratification, and individualized treatment approaches.
Conclusions
Blood biomarkers offer valuable insights into the systemic pathophysiology of AIS and may serve as accessible tools for improving early detection, progression monitoring, and personalized management of AIS. Further longitudinal and multi-omics studies are warranted to validate their clinical utility.
Introduction
Scoliosis is a complex three-dimensional spinal deformity defined by a lateral curvature (Cobb angle >10°) and vertebral rotation, often accompanied by trunk asymmetry and functional impairment.1-3 Adolescent idiopathic scoliosis (AIS), the most prevalent form accounting for 75%-80% of spinal deformities, predominantly affects females during puberty and may progress to severe complications such as cardiopulmonary compromise.2-4 Despite its prevalence of 2%-4% globally, AIS remains etiologically enigmatic. 5 Currently, most research believes it is a multifactorial disease and arises from discordant interactions between genetic susceptibility and environmental interference during growth peak. 6 This gene-environment synergy suggests that AIS is not merely a static genetic disorder but a dynamic process shaped by systemic biological responses.7-10 This underscores the need to integrate dynamic molecular layers, particularly proteomic and epigenetic analyses, to bridge the gap between genetic risk and phenotypic manifestation.11-14
The fundamental research of AIS mainly relies on surgical samples of paraspinal muscle and skeletal bone, which are ethically and practically restricted to a small group of severe cases that progress more than 50°. 6 This skews data toward end-stage pathology, obscuring early drivers of curvature initiation. In contrast, blood—a systemic, minimally invasive sample—reflects the real-time whole-body metabolic states. Its accessibility enables longitudinal sampling, critical for tracking disease progression and disentangling causative pathways from compensatory adaptations.
Over the past decades, numerous studies have focused on changes in the blood of individuals with AIS to identify biomarkers that can reflect both the onset and progression of the disease. Early research primarily investigated variations in levels of specific metabolic products found in blood samples, such as plasma vitamin D and growth hormone levels. With advancements in molecular biology during the 21st century, researchers have increasingly shifted their focus to more detailed investigations at the micro-level, such as transcriptomics, proteomics and metabolomics, which have opened new avenues for studying blood-based biomarkers specific to AIS. This study firstly reviews previous findings related to plasma component disorders in AIS patients in order to identify potential associations among diverse metabolic products. Such discoveries could provide novel directions for diagnosing AIS, predicting prognosis, and identifying therapeutic targets based on metabolic imbalances (Figure 1). The Diagram of Blood Biomarker Profiles in Adolescent Idiopathic Scoliosis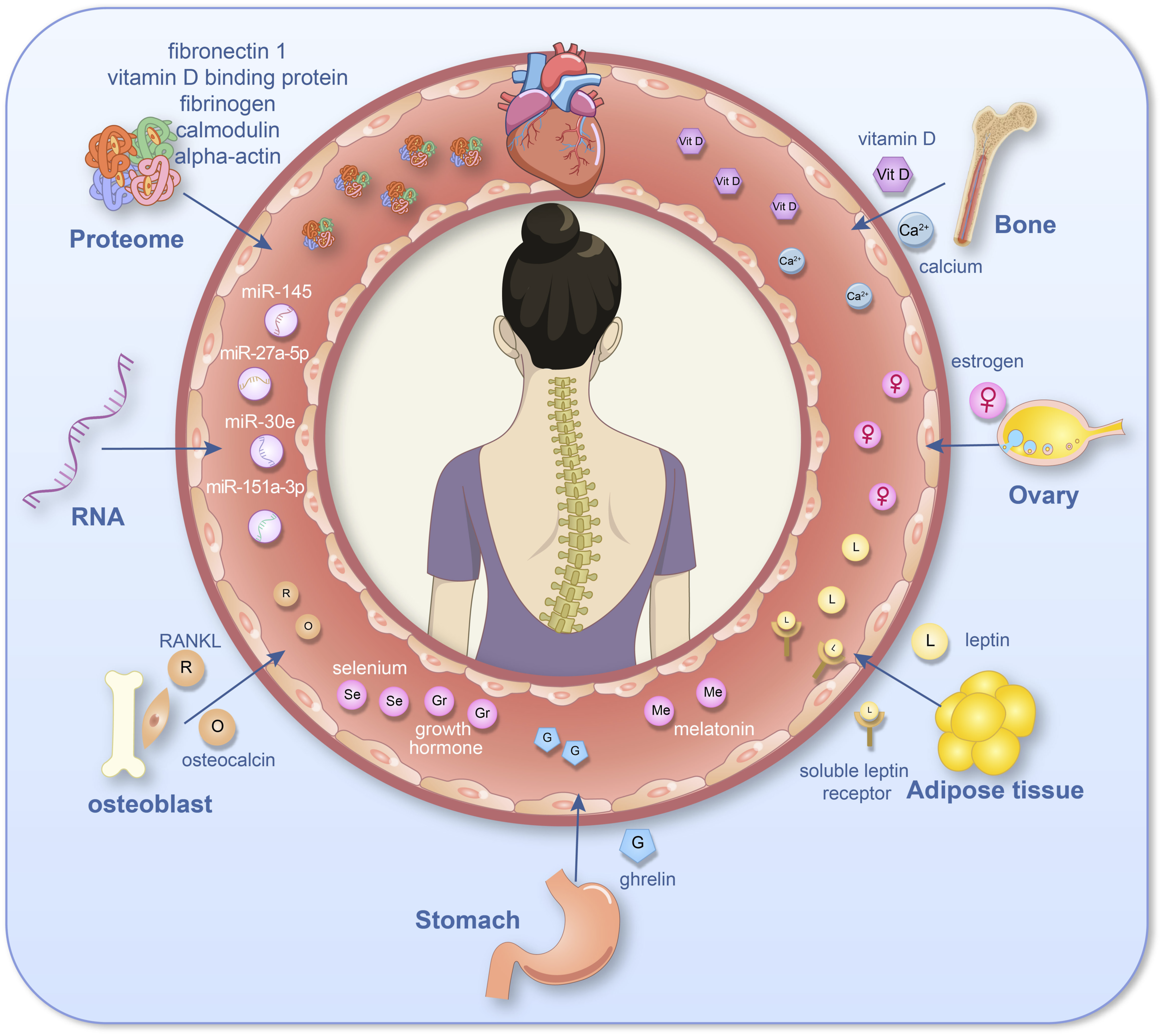
Materials and Methods
This review was conducted in accordance with the general principles of narrative and systematic literature reviews. A comprehensive search strategy was designed to capture relevant studies investigating blood biomarkers in patients with AIS.
Databases including PubMed, Embase, Web of Science, and Cochrane Library were systematically searched from database inception to February 2025. The search terms combined Medical Subject Headings (MeSH) and free-text keywords related to AIS and blood-based biomarkers, including but not limited to “adolescent idiopathic scoliosis”. “blood”, “serum”, “plasma”, “biomarker”, “Vitamin D”, “calcium”, “trace elements”, “hormones”, “leptin”, “ghrelin”, “microRNA”, “proteomics” and “metabolomics”. Reference lists of relevant reviews and included articles were manually screened to ensure literature saturation.
Studies were included if they met the following criteria, (1) Investigated AIS patients; (2) Reported blood-, plasma-, or serum-derived biomarkers associated with AIS onset, progression, or clinical features; (3) Published as original articles, case-control studies, cohort studies, meta-analyses, or systematic reviews. Exclusion criteria included: (1) Case reports, conference abstracts, or studies without accessible full text; (2) Studies focusing exclusively on tissue-based biomarkers (eg, bone or paraspinal muscle) without corresponding blood-based analysis.
Two authors independently screened titles and abstracts for relevance, followed by full-text review of potentially eligible studies. Given the heterogeneity in study designs, patient cohorts, and biomarker detection methods, a qualitative synthesis was performed. Studies were grouped according to biomarker categories (Figure 2). Research Hotspots Across Decades (Heatmap)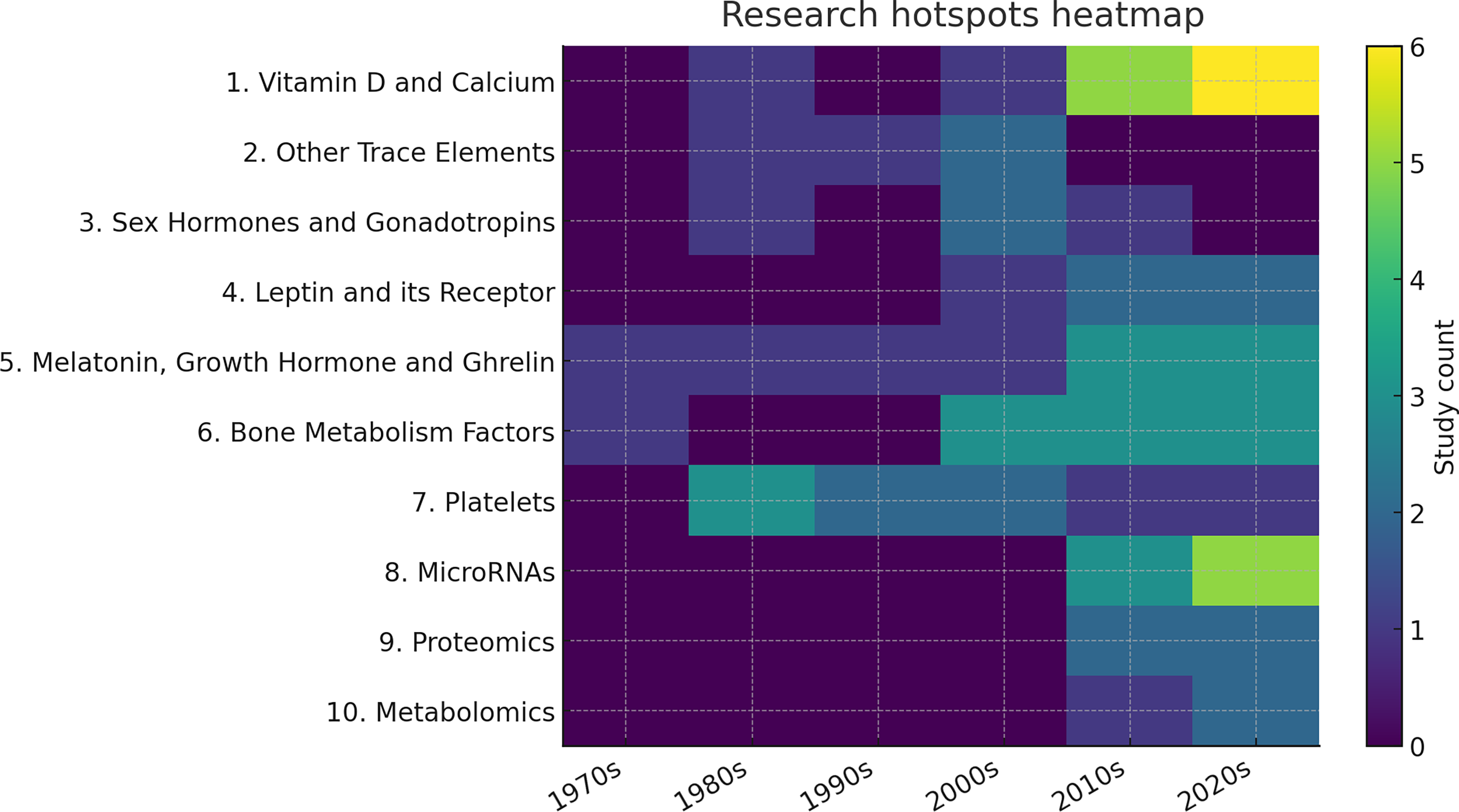
Results
Vitamin D and Calcium
Vitamin D plays a crucial role in bone development by influencing calcium absorption, maintaining calcium-phosphorus homeostasis, and promoting the mineralization of bone matrix. In the early years of life, a deficiency in vitamin D can lead to poor bone quality, resulting in reduced bone mass and various skeletal deformities. 15 Since AIS patients generally exhibit reduced bone mineral density, researchers have long focused on the vitamin D and calcium levels. 16 a meta-analysis by Llopis-Ibor et al revealed that 36.19% of AIS patients had insufficient vitamin D levels, and 41.43% were vitamin D deficient. However, they found no significant correlation between vitamin D deficiency and the severity of spinal curvature or gender. 17 Another study by González-Ruiz et al included 53 AIS patients and measured their plasma vitamin D levels along with Cobb angles, finding a significant negative correlation between vitamin D levels and Cobb angles only in the prepubertal group. 18 Several other studies also found that AIS patients generally had lower serum vitamin D levels18-20 In contrast, Kim et al's meta-analysis found no significant difference in plasma vitamin D levels between AIS patients and a control group, but noted that blood calcium levels in AIS patients were significantly lower than those in the control group. 21 Hampton et al 22 similarly reported that vitamin D deficiency was common among AIS patients but also prevalent among healthy adolescents.
Regarding calcium levels, a meta-analysis by Zhu et al found that AIS patients had lower levels of calcium ions and calcitonin compared to the control group, while no significant differences were observed in phosphate and parathyroid hormone levels between the 2 groups. 23 However, this finding contradicted the results of Brzęczek et al and Willner et al,24,25 which showed elevated Calcium and 25-OH-D3 levels in AIS patients. Subsequently, Lam et al conducted a 6-year longitudinal study involving 330 AIS patients, which showed significant improvements in bone health after 2 years of calcium and vitamin D supplementation. 26 This suggests that supplementing with vitamin D and calcium may help slow the progression of scoliosis.4,26 A literature review by Ng et al, which synthesized numerous theories exploring the relationship between vitamin D and the etiology of AIS, suggests that vitamin D deficiency or insufficiency may contribute to the development of AIS by affecting bone mineral density, posture control, and fibrosis regulation. However, further research is still needed. 27
The Expression Levels and Clinical Implications of Common Blood Biomarkers in AIS Patients
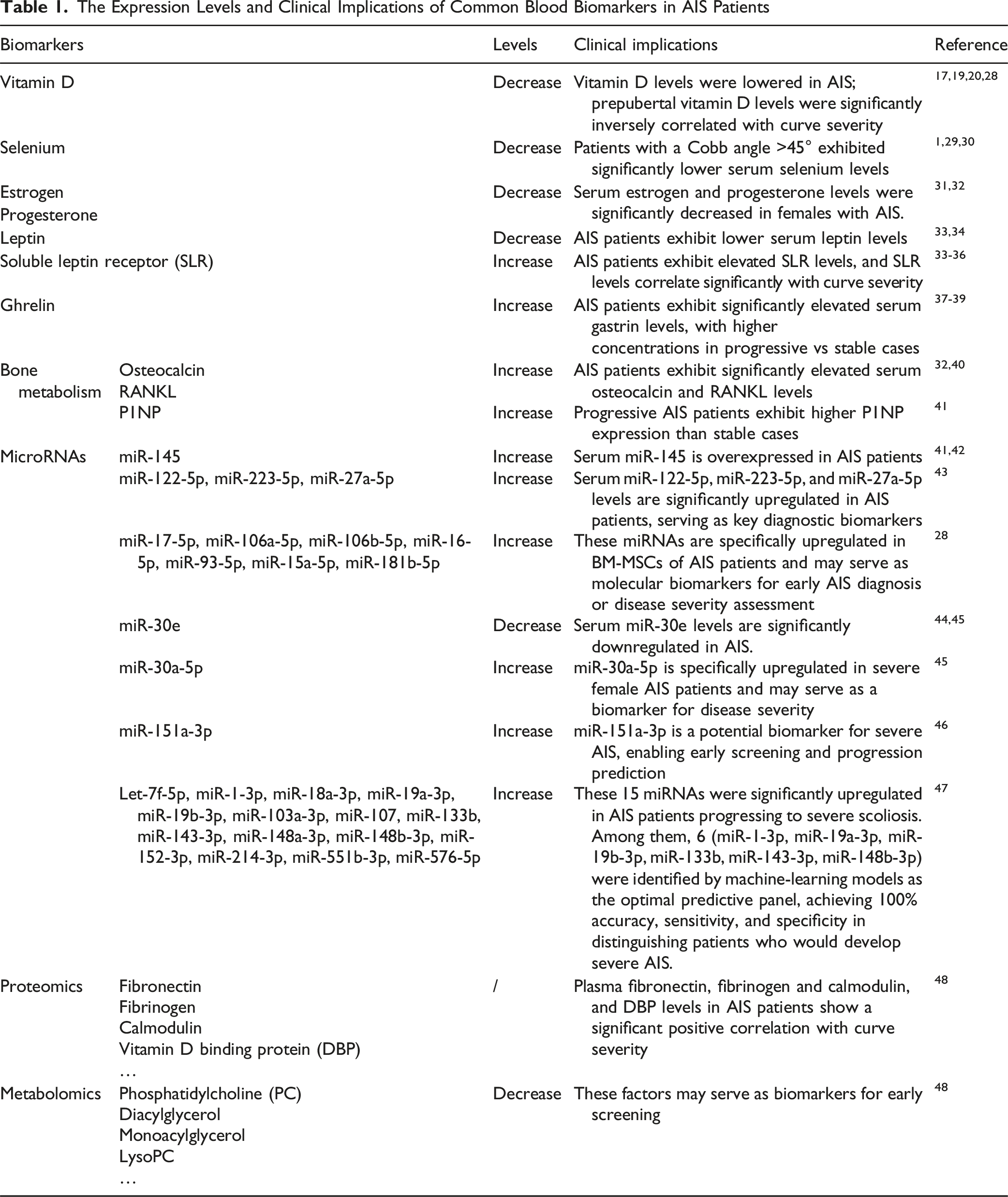
Other Trace Elements
Researchers have also explored the relationship between other trace elements, such as copper, zinc, and selenium, and AIS. The earliest studies in this area were conducted by Pratt et al, who examined the hair of 74 AIS patients and found significantly higher copper levels compared to healthy controls. They hypothesized that this might be because copper is a crucial component of lysyl oxidase, a metalloenzyme involved in the cross-linking of collagen and elastin. However, the study also noted that there was no direct relationship between copper levels and the severity of scoliosis. 49 Dastych et al conducted several studies investigating the levels of zinc in the sacrospinal muscles, hair, peripheral blood leukocytes, and serum of AIS patients, as well as the concentrations of zinc, copper, selenium, albumin, and ceruloplasmin in plasma.29,30,50 Their research found that plasma selenium levels were significantly lower in AIS patients compared to the control group, and that the average plasma selenium concentration was significantly reduced in patients with a Cobb angle>45° compared to those with a Cobb angle<45° who were treated conservatively.29,30 However, other indicators such as zinc and copper did not show significant differences. In a subsequent study, the same research team observed increased selenium levels in the hair of another AIS patient cohort, along with elevated levels of copper and zinc. 50 In summary, current studies show lower plasma selenium levels in AIS patients.29,30,50 Selenium may serve as a novel biomarker for hematological testing in AIS.
Sex Hormones and Gonadotropins
The onset and phenotype of AIS are closely related to the patient’s age and gender. In the early stages, the ratio of affected girls to boys is approximately 1:1, but during puberty, this ratio can widen to 8.4:1 (girls to boys), indicating a strong gender predisposition for AIS.1,4 Estrogen, a key regulator of human growth, plays a significant role in this disparity. Previous studies have shown an association between estrogen receptor polymorphisms and susceptibility to AIS as well as its severity, suggesting that the levels and characteristics of estrogen as a ligand may also be involved in the pathogenesis of AIS.11,51 Shao et al identified an asymmetrical expression of estrogen receptor 1 (ESR1) in the concave and convex side muscles of scoliosis patients. They further demonstrated that this asymmetric ESR1 expression leads to differentiation defects in muscle stem cells and progenitor cells, thereby influencing the development and progression of scoliosis. 52 In an earlier study, Skogland et al found that in female AIS patients with a bone age between 9 and 12, the average serum testosterone level was significantly higher. However, no significant differences were observed in other sex hormone-related levels, including estradiol, sex hormone-binding globulin, prolactin, follicle-stimulating hormone, and luteinizing hormone. 53 Esposito et al compared 174 female AIS patients with 104 healthy girls and found that AIS patients had lower levels of estrogen, progesterone, and androgens in their blood compared to their healthy peers. 31 Kulis et al 32 further divided female AIS patients into 2 groups: those who had not yet reached menarche and those who had. They discovered that in the premenarcheal group, only estradiol levels were significantly lower in AIS patients, while in the postmenarcheal group, levels of estradiol, progesterone, follicle-stimulating hormone (FSH), and luteinizing hormone (LH) were all markedly reduced. 32 Raczkowski et al focused on androgens, measuring serum estrogen and androgen levels in 27 right thoracic AIS patients and 7 patients with mild functional left lumbar curves. They found that testosterone levels were slightly higher than normal reference values in the girls with AIS in both groups. However, there were no significant differences in estrogen levels between the 2 AIS groups. 54
To summarize, most literatures demonstrate decreased serum progesterone and estrogen levels in patients with scoliosis31,32 suggesting that these hormonal biomarkers may serve as potential diagnostic and prognostic predictors for AIS. Discrepancies likely arise from heterogeneous patient stratification (pre-/post-menarche, bone age), small sample sizes. Though estrogen signaling dysfunction is mechanistically plausible, inconsistent hormonal profiles underscore the need for standardized longitudinal studies.
Leptin and Its Receptor
Adolescents with scoliosis typically have lower body weight, lean muscle mass, and bone density. 1 The normal growth and development of muscles and bones are influenced by various hormones, in addition to sex hormones. Among these, leptin, melatonin, growth hormone, and ghrelin have been extensively studied. Leptin, a key peptide hormone, plays a crucial role in regulating energy balance and bone metabolism. It is primarily secreted by adipocytes, and soluble leptin receptor (SLR) can be detected in chondrocytes and osteoblasts. Leptin regulates the differentiation of bone marrow stem cells into osteoblasts and the function of chondrocytes by binding directly to SLR.33,35,37 Man et al exposed osteoblasts from severe AIS patients and a control group to varying concentrations of leptin in vitro. They found that leptin significantly stimulated the proliferation, differentiation, and mineralization of osteoblasts in the non-AIS group. However, these effects were not observed in the AIS group. 37 Qiu et al initially recruited 120 female AIS patients and 80 controls and found that AIS patients had higher adjusted height, lower weight, and lower BMI, along with significantly reduced circulating leptin levels, which were positively correlated with bone mineral content/density (BMC/BMD). 34 Liu et al further measured serum leptin and SLR levels in female AIS patients and healthy controls, discovering that AIS girls had significantly higher SLR levels and significantly lower free leptin index (FLI). They also found a significant correlation between SLR, FLI, and the severity of scoliosis in AIS girls, consistent with the findings of Tam’s team in 2 separate studies. 36 A meta-analysis by Bae et al revealed no statistically significant difference in serum leptin levels between female AIS patients and healthy controls. However, the level of soluble leptin receptor SLR was significantly higher in female AIS patients compared to the control group. 35 Wang Q et al conducted a meta-analysis, which included ghrelin levels, found that AIS patients had significantly lower serum leptin levels compared to the control group, while serum SLR and ghrelin levels were significantly higher in AIS patients. 33
In summary, the majority of studies indicate that AIS patients exhibit lower serum leptin levels33,34 and elevated SLR levels.33-35 Notably, emerging evidence demonstrates a significant correlation between SLR and curve severity, 36 collectively suggesting that the leptin and SLR levels holds promising potential for both diagnostic screening and prognostic evaluation in AIS.
Melatonin, Growth Hormone and Ghrelin
Melatonin is primarily secreted by the pineal gland at night and plays a role in regulating sleep and other cyclical bodily activities. It may also contribute to the biological regulation of mood, emotional disorders, cardiovascular systems, reproduction, and aging. 55 The initial hypothesis that melatonin deficiency might lead to scoliosis was inspired by observations of scoliosis-like deformities in chickens following pinealectomy, a condition resembling AIS in humans. Brodner et al measured the diurnal and nocturnal serum melatonin levels in AIS patients and controls but found no statistically significant differences between the 2 groups. 38 In a study with an expanded sample size, Goultidis et al observed that conservatively treated AIS patients had higher melatonin levels at the initial measurement. However, they found no association between melatonin deficiency and the progression of AIS. Notably, a year later, the serum melatonin levels in both AIS study group and normal control group showed no statistically significant differences. 39 Similarly, Bagnall et al concluded that there was no significant statistical difference between the 2 groups. They did, however, suggest that this finding might be due to the fact that the patients in their sample were 2-3 years beyond the age at which scoliosis is most likely to worsen. Further research is needed on patients who are in the peak growth phase. 56 As a result, current mainstream research does not provide sufficient evidence to support the idea that melatonin deficiency is a causative factor in the development and progression of scoliosis. The biological relevance of melatonin in AIS remains controversial.55,57 It is worth noting that some scholars believe that melatonin supplementation might help slow the progression of mild AIS. 58
The relationship between growth hormone and AIS has been less studied, with most research focusing on the side effects of growth hormone therapy for other conditions, which can lead to the development and progression of scoliosis.59,60 Ahl et al, in an earlier study, found that only in the early stages of puberty did female AIS patients have higher levels of growth hormone compared to healthy girls. 61 Willner et al 62 measured plasma growth hormone levels in AIS patients and controls under 3 different conditions, and the results indicated that female AIS patients had higher growth hormone secretion than healthy girls of the same age. However, this study does not provide an explanation for this phenomenon. 62 In a subsequent study, willer et al also found significantly higher serum levels of morning fast growth hormone in patients with scoliosis. 63
Ghrelin, a hunger-stimulating hormone secreted by the stomach, reflects changes in body weight and stimulates the secretion of growth hormone. 64 Several studies have found that serum ghrelin levels in AIS patients are significantly higher compared to controls,37,64,65 and that circulating ghrelin levels gradually increase with age. Patients with progressive scoliosis had higher ghrelin levels than those with stable scoliosis. Therefore, elevated ghrelin levels may serve as a new quantitative indicator for predicting curve progression in girls with AIS.
Bone Metabolism Factors
The body contains various molecules that are closely related to bone metabolism, and many scholars have studied their behavior in the blood circulation of AIS patients. Among these, osteocalcin is a non-collagen protein secreted by osteoblasts that plays a role in bone mineralization and may influence insulin sensitivity and testosterone levels. Chiru et al explored the relationship between AIS and bone loss, finding that osteocalcin and the receptor activator of nuclear factor-kappa-B ligand (RANKL) levels were significantly elevated in adolescent AIS patients compared to controls, while osteoprotegerin (OPG) levels showed no significant difference. 40 The RANKL/OPG ratio in the AIS group was significantly higher than in the control group. The increase in RANKL levels in the presence of elevated osteocalcin may lead to altered bone remodeling due to an imbalance in the RANKL/OPG system. There is an important link between osteoporosis and AIS, consistent with the findings of Suh et al.40,66 Kulis et al also found that serum osteocalcin levels in AIS patients were significantly increased, along with RANKL and alkaline phosphatase levels. 32 Some scholars, however, believe that serum osteocalcin levels are significantly negatively correlated with Cobb angles in AIS patients.67,68 Sanders et al observed that osteocalcin levels vary with skeletal maturity, but their correlation with the accelerated phase of scoliosis progression is weak. In contrast, alkaline phosphatase peaks during the scoliosis acceleration phase, but its wide reference range limits its clinical application. 69 Danielewicz et al conducted a more comprehensive measurement of various bone metabolic factors in the serum of AIS patients and healthy controls, finding no statistically significant differences in the levels of Klotho protein, osteocalcin, OPG, C-terminal telopeptide of type 1 collagen (CTX), sclerostin, and alkaline phosphatase. 70 Zhang et al compared the serum levels of 2 other bone turnover markers, finding that the expression of N-terminal procollagen type 1 N-terminal propeptide (P1NP) was significantly higher in progressive AIS patients than in non-progressive AIS patients, while C-terminal telopeptide of type I collagen (CTX) showed no difference. 41 Another research team focused on adiponectin, discovering that AIS patients with reduced bone mass had high circulating adiponectin levels, which they speculated might affect bone metabolism by regulating the RANKL/OPG and IL-6 pathways. 71 An Asian research initiative investigating the interplay between bone mass, physical parameters, bone turnover markers, and calcium intake involved 623 female AIS patients and 300 healthy female controls. The comparative analysis revealed that while both groups showed similar developmental patterns before age 13, post-13 AIS patients demonstrated significantly greater longitudinal skeletal growth parameters compared to their healthy counterparts. Furthermore, serum analysis identified markedly elevated levels of bone-specific alkaline phosphatase (bALP) in AIS patients after age 13, with this elevation inversely correlated to bone mineral density measurements across the study population. 72 Besides, there were no significant changes in serum insulin-like growth factor levels in patients with AIS, nor were there any significant gender differences, according a former study. 73
In summary, current studies demonstrates elevated serum osteocalcin and RANKL levels in AIS patients,32,40 along with the novel biomarker P1NP may can be used to predict the progression of AIS. 41
Platelets
The number, structure and function of platelets in the blood and their relationship with AIS had been the focus of previous researchers. Because platelets and muscles have similar myofilament gliding structures, some researchers have suggested that they may share some common abnormalities in AIS. In fact, several previous studies have identified platelet abnormalities in patients with AIS,74,75 including abnormalities in the structure of the platelet myosin peptide chain and reduced platelet cytoplasmic myosin ATPase activity, 76 abnormal distribution of total proteins and contractile proteins, 77 the decreased platelet aggregation function mediated by the ADP signaling pathway. 78 Research has also focused on the calcium ion homeostasis of platelets in AIS patients. Studies have found that in AIS patients with thoracic curve, the content of SERCA3a (sarco/endoplasmic reticulum Ca2+-ATPase 3a) in platelets is lower, while the expression level of PMCA4b (plasma membrane Ca2+-ATPase 4b) is higher. However, in AIS patients with thoracolumbar curve, both SERCA3a and PMCA4b levels are higher. 79 However, Kahmann et al argued that there were no significant differences in platelet parameters between AIS patients and the control group, nor were there any significant differences in drug response or ultrastructure. 80 Similarly, Suk et al's study also indicated that the morphology and function of platelets in AIS patients were normal. 81 Another related study also yielded negative results. Acaroglu et al found that there were no significant differences in the ratio of melatonin to calmodulin in the muscles on the concave and convex sides of scoliotic patients compared to the control group, and these ratios could not represent the values of related proteins in the muscle. 16 However, a recent study published in 2023 pointed out that the mean platelet volume (MPV) in AIS patients was significantly higher compared to the control group. 82
In summary, current studies on platelet abnormalities in AIS show significant controversy, limiting clinical utility. Future research should prioritize standardized multimodal analyses in large, well-characterized AIS cohorts to resolve platelet-pathophysiology links and identify actionable biomarkers for risk stratification or treatment monitoring.
MicroRNAs
MicroRNAs (miRNAs) are a class of small non-coding RNAs that regulate gene expression by binding to mRNA. They can convert environmental stimuli into biological signals, thereby modulating sporadic and phenotypic variations.83,84 Due to their presence in various body fluids, miRNAs have gained increasing attention for their clinical applications in molecular medicine. 41 Zhang et al were among the first to identify significant overexpression of miR-145 through sequencing of bone tissues from AIS patients, and they further confirmed the abnormal elevation of miR-145 in the blood.41,42 García-Giménez et al collected blood samples from 20 AIS patients and 10 healthy controls, finding significant differences in the expression of 6 miRNAs closely related to bone metabolism between the AIS and control groups (miR-122-5p, miR-671-5p, miR-223-5p, miR-1226-5p, miR-27a-5p, and miR-1306-3p). Furthermore, they identified that miR-122-5p, miR-223-5p, and miR-27a-5p exhibit consistent upregulation across multiple cohorts and serve as critical diagnostic biomarkers for acute ischemic stroke (AIS).They hypothesized that these miRNAs might participate in the onset and progression of AIS by regulating osteoblast and osteoclast differentiation mechanisms, thereby influencing the balance between bone resorption and formation through epigenetic expression of different signaling pathways. 43 Hui et al conducted an interaction network analysis and identified 7 key central miRNAs, including miR-17-5p, miR-106a-5p, miR-106b-5p, miR-16-5p, miR-93-5p, miR-15a-5p, and miR-181b-5p. These miRNAs may play a critical role in the pathogenesis of AIS and the associated osteoporosis by regulating the osteogenic or adipogenic differentiation of mesenchymal stem cells. 28 Additionally, 2 researchers observed that the expression of circulating miR-30e was significantly downregulated in patients with scoliosis,44,45Raimondi et al have also discovered that Serum miR-30a-5p levels are significantly elevated in AIS patients. This miRNA modulates Wnt/β-catenin and PI3K/AKT signaling pathways to suppress osteogenic differentiation, and its specific overexpression in severe female AIS cases suggests potential utility as a disease severity biomarker. 45 Wang et al identified the overexpression of miR-151a-3p in patients with severe AIS, suggesting that it may directly inhibit the expression of GREM1 in primitive osteoblasts, thereby disrupting bone homeostasis. 46 Yuan et al, after screening a large sample, selected individuals with severe AIS and normal controls, confirming the differential expression of miR-27a-5p in AIS patients and proposing that miR-539-5p and miR-1246 may also have potential diagnostic value. 85 In a 2025 study, Khatami et al compared blood samples from 116 AIS patients and 30 healthy controls and found 15 miRNAs, such as let-7f-5p, miR-1-3p, miR-19a/b-3p, miR-133b, miR-143-3p, and miR-148b-3p, that were significantly increased and related to severe AIS progression. Using random forest models, they built a “6-miRNA” prediction model including miR-1-3p, miR-19a/b-3p, miR-133b, miR-143-3p, and miR-148b-3p, which showed 100% accuracy, sensitivity, and specificity in predicting surgical risk at the first visit. Mechanistic analysis suggested that these miRNAs may drive disease progression by regulating muscle development (miR-1-3p, miR-133b), bone metabolism (miR-19a/b-3p via PITX1), and signaling pathways (miR-143-3p via MAPK7). In addition, sex-specific biomarkers, such as miR-18a-3p in females with severe curves, were also identified, which may help early screening and timely intervention. 47
In summary, studies on AIS highlight the central role of miRNAs in regulating bone metabolism, cellular differentiation, and signaling pathways, positioning them as promising biomarkers or therapeutic targets. Many miRNAs show positive/differential expression in aforementioned studies. miR-145, miR-122-5p, miR-223-5p, miR-27a-5p,miR-17-5p, miR-106a-5p, miR-106b-5p, miR-16-5p, miR-93-5p, miR-15a-5p, miR-181b-5p are all over-expressed,28,41-43 while miR-30e, miR-539-5p and miR-1246 are significantly downregulated in AIS patients,44,45,85 these findings suggest that miRNAs may serve as promising diagnostic and screening biomarkers in AIS. Notably, miR-30a-5p and miR-151a-3p demonstrates significant overexpression specifically in patients with severe AIS, which may represent a novel biomarker for assessing disease severity.45,46 Furthermore, the 6-miRNA model developed by Khatami et al suggest that miRNAs can serve as predictive tools for clinical decision-making in AIS, 47 demonstrating broad prospects for further exploration in this filed.
Proteomics
Proteomics focuses on systematically identifying, quantifying, and analyzing the characteristics and variations of all proteins within an organism, aiming to reveal the fundamental principles of life and the mechanisms of disease development. This field encompasses not only the static composition of proteins but also a comprehensive description of the dynamic changes in proteins under different physiological and pathological conditions, thereby enhancing our understanding of diseases.86,87 In the context of AIS, Shen et al were among the first to utilize tandem mass tags (TMT) combined with liquid chromatography-mass spectrometry (LC-MS) technology to detect changes in the plasma proteome of AIS patients. They identified key differential plasma proteins associated with AIS, including fibronectin 1 (FN1), voltage-dependent anion channel 1 (VDAC1), Rho family GTPase A (RHOA), and AHNAK nucleoprotein (AHNAK). 88 Makino et al applied two-dimensional fluorescence difference gel electrophoresis (2D-DIGE) to identify proteomes associated with the pathogenesis of thoracolumbar/lumbar AIS, discovering that 9 proteins related to blood coagulation significantly differed from those in the control group. Among these, vitamin D binding protein (DBP) was significantly correlated with the Cobb angle of thoracolumbar/lumbar curvature, suggesting that DBP could serve as a blood biomarker for the severity of thoracolumbar/lumbar AIS. 89 Wang et al included 56 scoliosis patients and 28 healthy controls in their study, identifying 55 proteins with significant concentration differences between the AIS and control groups using a quadrupole orbitrap mass spectrometer. Further mass spectrometry analysis revealed that fibronectin, fibrinogen, and calmodulin were positively correlated with the Cobb angle. 48 Another research group divided AIS patients into non-progressive and progressive groups, along with matched healthy controls. They used isobaric tags for relative and absolute quantitation (iTRAQ) technology for a comprehensive comparison of untargeted proteins, discovering 71 significantly upregulated differential expression proteins in the progressive AIS group. Among these, 18 proteins were expressed in muscles and played crucial roles in muscle physiological activities, with alpha-actin showing the most significant changes. This finding suggests that there may be microstructural damage in the muscles of AIS patients. 63
Among these proteins, DBP, fibronectin, fibrinogen and calmodulin show unique features in positively correlated with Curve severity,48,89 suggesting their potential as biomarkers for AIS progression monitoring. However, there remains a need for further investigations with cross-validation across diverse methodologies and population cohorts to substantiate these findings.
Metabolomics
Metabolomics focuses on the comprehensive quantification of small molecule metabolites in organisms to understand the metabolic dynamics of biological systems under different physiological or pathological conditions. Sun et al were among the first to employ ultra-high-performance LC coupled with quadrupole time-of-flight MS to explore serum metabolic profiles in 30 AIS patients compared to 31 healthy controls. They identified several differential metabolites, including phosphatidylcholine, 2-hexenoylcarnitine, β-D-glucuronic acid, diacylglycerol, monoacylglycerol, and LysoPC, suggesting that AIS patients exhibit lipid metabolism disorders. 90 Xiao et al subsequently analyzed and compared plasma metabolomics differences between 16 AIS patients and 12 healthy controls. They identified 72 differential metabolites in the plasma of AIS patients, primarily including organooxygen compounds, carboxylic acids and derivatives, fatty acyls, steroids and steroid derivatives, and keto acids and derivatives. Among these, α-ketoglutaric acid, arginine, N-acetylaspartate, and citric acid were significantly correlated with clinical indicators such as Cobb angle and bone mineral density. 91 Wang et al combined proteomics and metabolomics approaches to compare differential expression proteins and metabolites in plasma, identifying 13 proteins and 7 metabolites related to galactose metabolism and glycolysis pathways associated with the progression of AIS angle. Additionally, their research team further clarified that some differentially expressed proteins in the paraspinal muscle tissues on the concave and convex sides of AIS patients showed significant alterations compared to non-scoliotic controls. 92
Metabolomics studies in AIS have uncovered significant disruptions in lipid metabolism, energy pathways, and amino acid homeostasis, with key metabolites demonstrating strong correlations with clinical severity. These findings highlight the potential of metabolomics to elucidate metabolic dysregulation underlying spinal deformities and identify biomarkers for diagnosis or progression monitoring.
Limitations and Future Directions
Conflict Outcomes of Blood Biomarkers in AIS Patients From Different Studies
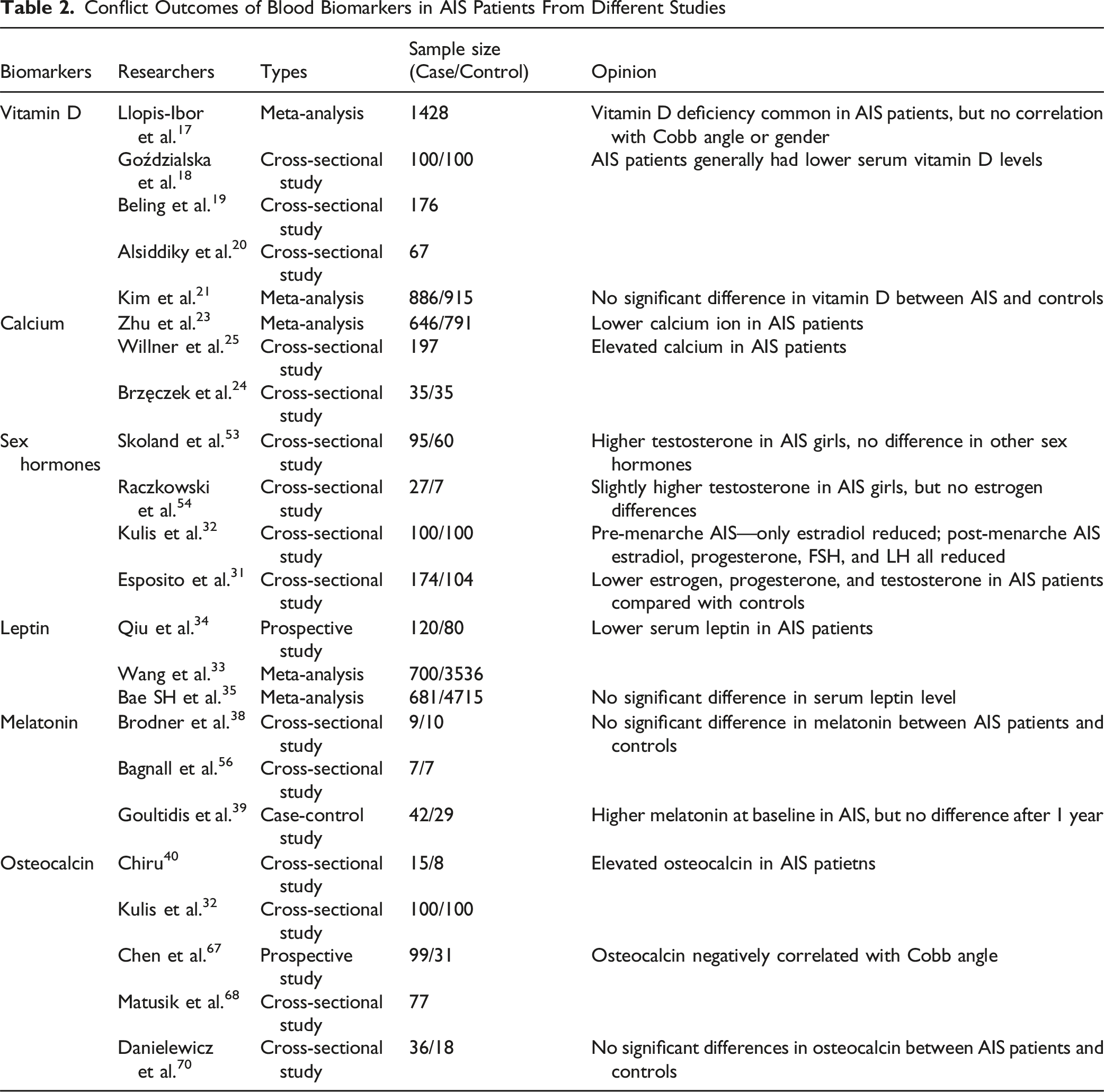
Meanwhile, several limitations should be acknowledged. Most included studies are limited by small sample sizes, cross-sectional designs, and heterogeneous patient cohorts. Inconsistencies in age stratification, pubertal status, and biomarker detection techniques further hinder comparability across studies. Moreover, geographic and environmental factors such as diet and sunlight exposure are rarely controlled for, restricting the generalizability of conclusions.
Future research should prioritize rigorous, standardized, and longitudinal designs, ideally incorporating multi-center cohorts to validate findings across diverse populations. Special attention should be given biomarkers with clinical promise, such as leptin signaling, specific miRNAs and predictive panels. Focused validation studies may enable the translation of single biomarkers or composite panels into tools for early diagnosis, risk stratification, and treatment monitoring in AIS.
Conclusion
In summary, this review mainly investigates 2 aspects of blood components research in AIS. Firstly, blood components serve as a comprehensive reflection of the current biological characteristics of AIS, helping to reveal its underlying pathogenic mechanisms. Secondly, blood biomarkers are closely correlated with curve progression of AIS, offering potential indicators for clinical outcomes and translational applications. This review highlights several blood biomarkers, such as leptin, ghrelin, bone metabolism-related factors, and others, providing valuable information for early diagnosis, disease progression, and treatment prognosis. Monitoring these biomarkers can assist in evaluating disease progression and offer evidence-based support for clinical decision-making, particularly in the non-surgical management of AIS.
Supplemental Material
Supplemental Material - Blood Biomarker Profiles in Adolescent Idiopathic Scoliosis: A Literature Review of Pathophysiological Insights and Clinical Implications
Supplemental Material for Blood Biomarker Profiles in Adolescent Idiopathic Scoliosis: A Literature Review of Pathophysiological Insights and Clinical Implications by Yuheng Zhang, Yaolong Deng, Wenyuan Sui, Tianyuan Zhang, and Junlin Yang in Global Spine Journal.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (Research on prevention and treatment of common frequently-occurring diseases/2023YFC2507702) and the National Nature Science Foundation of China (82472789).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
