Abstract
Study design
Retrospective Multicenter Comparative Study.
Objective
The population’s aging is progressively increasing the number of elderly patients on antiplatelet medications undergoing lumbar fusion procedures. Lateral lumbar interbody fusion (LLIF) has demonstrated valuable clinical and radiological advantages compared to conventional posterior fusion procedures in treating degenerative spine disorders. This study aims to evaluate the safety of continuing antiplatelet drug administration in the perioperative time for patients undergoing single- or multiple-level LLIF for degenerative lumbar disorders.
Methods
This is a multicenter retrospective study on the safety and efficacy of continuing acetyl salicylic acid therapy in patients undergoing single- or multiple-level LLIF. Age, gender, body mass index (BMI), smoking status, presence of diabetes mellitus, and relevant comorbidities were considered as independent variables in influencing clinical-radiological and surgical outcomes and complications.
Results
A total of 421 levels were operated on the 332 patients included in the study. The mean introperative blood loss was 42.3 ± 27.4 cc in the CG, and 47.2 ± 31.8 cc in the TG (P = 0.12). The drainage collected volume in the postoperative 24 h was 21.2 ± 13.9 cc in the CG, and 23.6 ± 11.9 cc in the TG (P = 0.11). No significant differences were reported in clinical, neurological, and radiological outcomes between the 2 groups.
Conclusions
Our results suggest that acetylsalicylic acid discontinuation in patients undergoing single- or multiple-level XLIF for degenerative spine disorders is not supported, according to similar blood loss and complications’ rate between patients discontinuing and those continuing antiplatelet drugs. Further dedicated clinical trials are needed to confirm our results.
Keywords
Introduction
Lumbo-sacral fusion procedures have been progressively increasing over the last 3 decades, according to the population aging and the functional expectations of the elderly. 1 Lateral lumbar interbody fusion (LLIF) has demonstrated valuable clinical and radiological advantages compared to conventional posterior fusion procedures in treating degenerative spine disorders.2,3 The possibility of stabilizing the anterior column and indirectly decompressing the spinal canal, positioning higher and wider cages, represents the main advantages of this technique, aiming to reduce the surgical injury to the spinal posterior elements.4,5 This technique represents a minimally invasive alternative, requiring small incisions (3- to 5 cm) on the flank and passing through the psoas muscle, allowing the surgeon to operate on a single or multiple lumbar levels from L1 to L5 using intraoperative EMG monitoring for reducing lumbar plexus injuries. 6 Positioning the cage with a lateral plating or as a stand-alone implant avoids posterior spinal muscle surgical injury. 7 This also means reducing post-operative pain rates, faster return to work and standard daily activities, and reducing direct and indirect procedure-related social costs.4-7
The population’s average age in Occidental countries is progressively increasing, and the number of elderly patients undergoing lumbar fusion surgeries is increasing. As reported by many authors, this subgroup of patients may benefit the most from the clinical and radiological advantages of LLIF and minimally invasive procedures.1,8 On the other hand, the elderly population is often prescribed antiplatelet medications for primary or secondary prevention of vascular disorders. 9 Different scientific papers have proposed the safety of posterior decompression surgeries, such as in spinal stenosis or disc herniation, without peri-operative discontinuation of antiplatelet drugs. However, there is no data on the safety of continuing these medications in LLIF procedures.10-12
This study aims to evaluate the safety of continuing antiplatelet drug administration in the perioperative time for patients undergoing single- or multiple-level LLIF for degenerative lumbar disorders.
Methods
Study Design
This is a multicenter retrospective study conducted at 3 different Italian spinal centers. According to the retrospective and non-interventional design, the present investigation was exempt from the Ethical Committee evaluation. The included patients signed an informed consent for the surgical procedure and the scientific use of the anonymized clinical-radiological data, including publication in scientific journals. All procedures conformed to the ethical standards set by the 1964 Helsinki Declaration and its amendments. This study was conducted according to the STROBE guidelines. 13
Study Population
The institutional databases of 3 different spinal institutions were analyzed to retrieve data on patients who underwent LLIF on a single or multiple levels for degenerative lumbar disorders. The following inclusion criteria were adopted: availability of full clinical-radiological data, degenerative disorder of the lumbar spine, including spinal stenosis, degenerative disc disease, and spondylolisthesis, single or multiple level stand-alone extreme-lateral lumbar interbody fusion (XLIF - Globus Medical, Philadelphia - USA) with or without lateral plating as the unique surgical procedure. We excluded patients with an oncological diagnosis, previous lumbar or retroperitoneal surgeries, radiotherapy, coagulopathies, or age <18. The study period was set from January 2021 until December 2024. The included patients were categorized in the treatment group - acetylsalicylic acid administered for> 6 months - or in the comparison group - no antiplatelet medication - according to the pharmacological history. Antiplatelet prescription was categorized as primary or secondary prevention, while the administration was continued in all the patients.
Outcomes Measurement
Data were meticulously extracted from clinical records and perioperative documentation, encompassing demographic variables such as patient age, gender, body mass index (BMI), smoking status, presence of diabetes mellitus, and relevant comorbidities. Clinical information included the underlying spinal pathology, specific cardiovascular or cerebrovascular conditions requiring antiplatelet therapy, and the indication for acetylsalicylic acid (ASA) administration, distinguishing between primary and secondary prevention strategies. Surgical parameters were also collected, including the number and lumbar levels treated, the side of surgical approach, the volume of estimated intraoperative blood loss, the amount of postoperative drainage collected within the first 24 hours, and the occurrence and nature of any perioperative complications, such as hematoma formation, infection, neurological impairment, or vascular injury. The primary outcomes of the study were the total intraoperative blood loss, the postoperative drainage volume within the first 24 hours, and the incidence of bleeding-related complications. Secondary outcomes included the overall complication rate, the occurrence of intraoperative neurological or vascular events, the need for reoperation or hospital readmission due to hemorrhagic events, and the incidence of postoperative cerebrovascular complications during hospitalization.
Data Collection and Statistical Analysis
All data were collected prospectively and retrospectively reviewed for analysis. Prior to study initiation, a formal sample size calculation was performed to ensure sufficient statistical power. Based on expected effect sizes derived from the literature and a significance level (α) of 0.05 with a desired power (1–β) of 0.80, the minimum required sample size was determined to be 97 patients per group. This threshold was met in both the treatment group (TG) and the comparison group (CG), thereby ensuring adequate power to detect clinically meaningful differences. Descriptive statistics were used to characterize the study population. Continuous variables were reported as mean ± standard deviation (SD), while categorical variables were expressed as frequencies and percentages. The Shapiro–Wilk test was employed to assess the normality of continuous data distributions. For intergroup comparisons, independent samples t-tests were used for normally distributed variables, and the Friedman test was applied for repeated measures of non-normally distributed variables. Where appropriate, Bonferroni correction was applied to control for multiple comparisons.
Categorical variables were analyzed using Fisher’s exact test, which was preferred due to the presence of small subgroup sizes. In addition to P-values, effect sizes were computed using Cohen’s d to provide an estimate of clinical relevance. All tests were two-tailed, and a P-value <0.05 was considered statistically significant. Statistical analyses were performed using StatPlus Pro version 8 (AnalystSoft Inc., Alexandria, VA, USA).
Results
After retrieving data from the clinical dataset, 332 patients met the inclusion criteria and were included in the present investigation. 209 patients did not assume antiplatelet medications and were assigned to the comparison group (CG), while acetylsalicylic acid was administered to 123 assigned to the treatment group (TG).
Demographic Results
The mean age was 66.4 ± 11.7 in the CG and 67.1 ± 13.6 in the TG (P = 0.54), the M:F ratio was 0.86 in the CG and 0.97 in the TG (P = 0.34), the mean BMI was 22.3 ± 3.2 in the CG and 23.4 ± 1.8 in the TG (P = 0.03), smoking/non-smoking ratio was 0.21 in the CG and 0.14 in the TG (P = 0.45), diabetes requiring oral medication or insulin administration was reported in 0.9% patients in the CG and 0.6% in the TG (P = 0.76).
Clinical Results
Baseline Demographic and Clinical Characteristics of the Study Groups
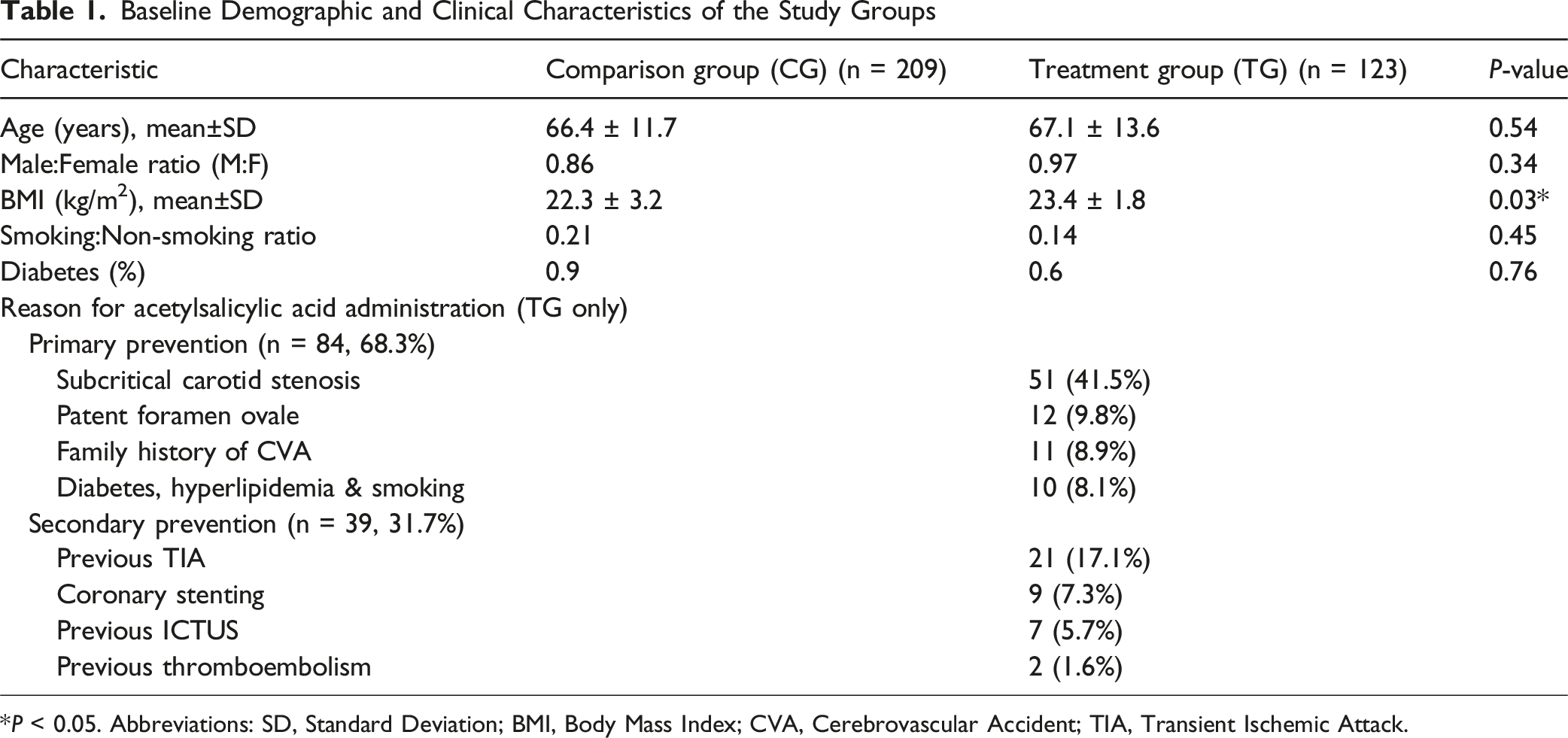
*P < 0.05. Abbreviations: SD, Standard Deviation; BMI, Body Mass Index; CVA, Cerebrovascular Accident; TIA, Transient Ischemic Attack.
Surgical Results
Surgical Characteristics by Group
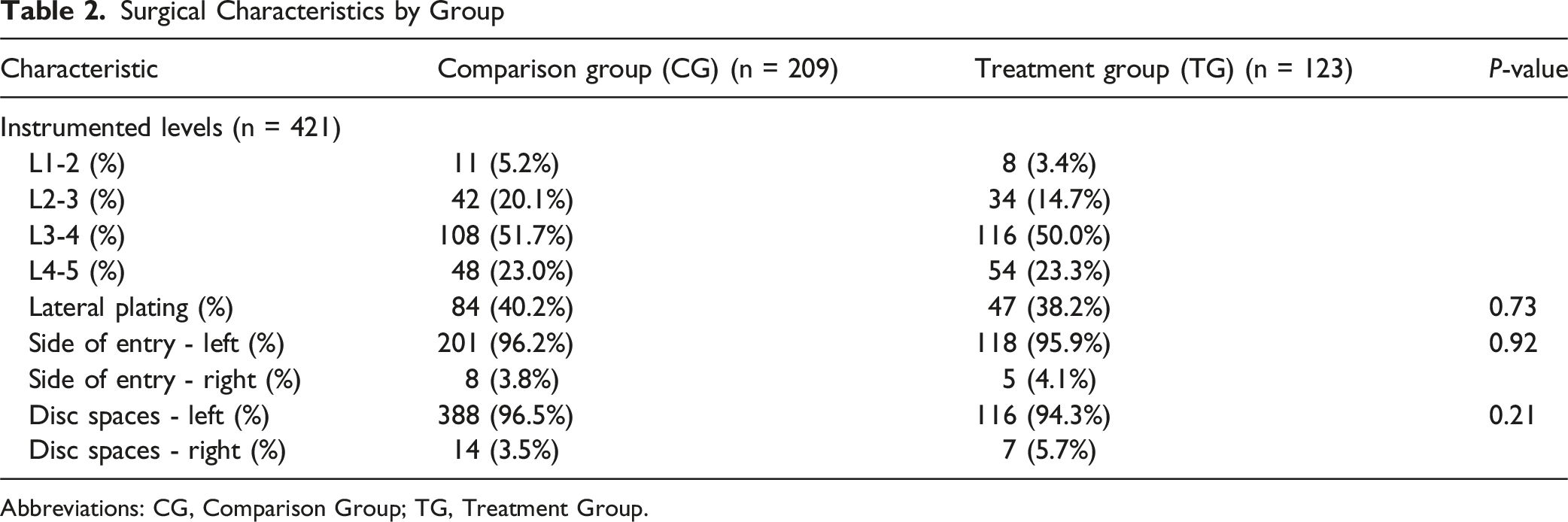
Abbreviations: CG, Comparison Group; TG, Treatment Group.
Intra- and Postoperative Outcomes by Group
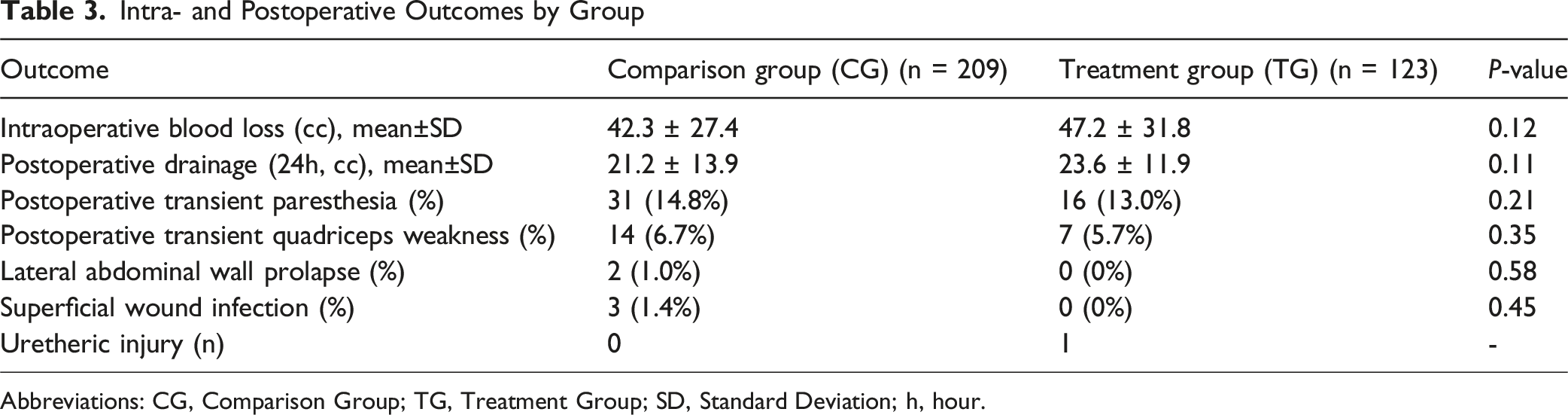
Abbreviations: CG, Comparison Group; TG, Treatment Group; SD, Standard Deviation; h, hour.
Discussion
Background and Rationale
The demographic shift toward an aging population has been accompanied by a parallel increase in the prevalence of degenerative spinal disorders, resulting in heightened demand for surgical interventions.1,8 In response, spinal surgery has undergone significant evolution, with a clear trend toward minimally invasive approaches that aim to reduce surgical morbidity, shorten hospital stays, and preserve musculoskeletal integrity. One such technique, extreme lateral interbody fusion (XLIF), has gained prominence for its ability to achieve interbody fusion and indirect neural decompression while minimizing disruption to posterior anatomical structures. 5
XLIF, as originally described by Pimenta et al, 14 employs a lateral transpsoas corridor via a retroperitoneal approach, accessing the lumbar spine through a small flank incision. The use of electromyographic (EMG) monitoring allows for precise navigation through the psoas muscle, reducing the risk of iatrogenic nerve injury. 6 The approach enables the implantation of large, 3D-printed porous titanium cages, which offer biomechanical advantages such as restoration of disc height and sagittal alignment, and distraction of posterior ligamentous structures.4,15 These effects contribute to widening of the neural foramina and vertebral canal, providing indirect decompression without the need for direct manipulation of neural elements or resection of posterior structures. 5
In parallel with the development of minimally invasive techniques, perioperative management of patients—especially the elderly—has become increasingly complex.16,17 Many patients in this demographic are prescribed acetylsalicylic acid (ASA) for primary or secondary prevention of cardiovascular or cerebrovascular events. 9 The traditional practice of discontinuing ASA before elective surgery has come under scrutiny, particularly given the transient prothrombotic risk associated with its interruption. 18 While recent literature has suggested that continuation of ASA is safe in posterior lumbar surgeries that do not involve dural exposure,10-12,17,19 evidence remains scarce regarding its safety profile in lateral and anterior fusion procedures such as XLIF. This retrospective multicenter study was designed to address this knowledge gap by evaluating whether continuation of ASA influences intraoperative bleeding or postoperative complications in patients undergoing stand-alone XLIF procedures.
Analysis of Results and Clinical Implications
Our analysis demonstrated no statistically significant differences in intraoperative blood loss or 24-hour postoperative drainage volumes between patients who continued ASA therapy and those who discontinued it 7 days before surgery. These findings were consistent across both single-level and multilevel XLIF procedures. Importantly, the absence of clinically significant hemorrhagic complications in the ASA group suggests that the lateral, extradural nature of the XLIF approach inherently limits the risk of bleeding, even in the context of ongoing antiplatelet therapy.
The study also observed no significant increase in postoperative complications in the ASA continuation group. Of particular note, there were no reported cases of hematoma formation or reoperation for bleeding-related issues in either cohort. Additionally, the fact that several patients did not require postoperative drainage and yet experienced no adverse outcomes further supports the hemostatic safety of XLIF in the setting of continued ASA use. These results challenge the long-standing paradigm of routine ASA discontinuation prior to spine surgery and suggest that the benefits of uninterrupted therapy may outweigh the theoretical bleeding risks in properly selected patients.18,19
Furthermore, our demographic data confirmed a significantly higher mean body mass index (BMI) in the ASA group, aligning with known associations between obesity, cardiovascular disease, and antiplatelet therapy prescriptions. 19 Despite this increased vascular risk profile, the ASA group did not exhibit a higher rate of surgical or medical complications, reinforcing the overall safety of maintaining ASA in this context.
Although no cerebrovascular events were documented during hospitalization among patients who discontinued ASA, our study was not powered to detect late-onset complications related to temporary cessation. Nevertheless, the absence of any demonstrated advantage in bleeding control—coupled with the well-documented thrombotic risks associated with ASA withdrawal—raises important clinical and ethical considerations. The decision to interrupt antiplatelet therapy should be reserved for specific, high-risk surgical scenarios or patients with elevated hemorrhagic risk, rather than applied as a routine precaution. 20
From a medico-legal perspective, these findings are especially relevant. In an era where the legal burden associated with adverse surgical outcomes continues to rise, especially in spine surgery, adherence to evidence-based practices is critical. Our results suggest that continued ASA therapy in XLIF does not increase perioperative risk and may, in fact, reduce the medico-legal liability associated with thrombotic complications resulting from unnecessary drug discontinuation.20,21
This study contributes meaningfully to the growing body of literature advocating for individualized perioperative antiplatelet management. It also highlights the specific advantages of XLIF—not only in terms of spinal decompression and stabilization but also in enabling safer surgical planning for patients on chronic antiplatelet therapy. While our findings do not eliminate the need for careful risk stratification, they support a more permissive approach to ASA continuation in minimally invasive lateral lumbar procedures. On the other hand, our results do not apply to different lumbar procedures. Anterior-oblique approaches, posterior invasive surgeries including osteotomies, or even lateral approaches for performing corpectomies or specific maneuvers potentially increasing the hemorragic risk represent different surgical scenarios, requiring a proper case-by-case risk stratification, for the patients’ safety in a personalized treatment algorithm definition.
Limitations
Despite the encouraging results, this study has several limitations that must be acknowledged. First and foremost, the retrospective design introduces potential selection bias and limits the ability to control for confounding variables. Although multicenter data collection enhances generalizability, differences in surgical technique, perioperative management protocols, and institutional practices could have influenced the outcomes.
Second, the sample size may not have been sufficiently large to detect rare but potentially serious complications, such as delayed epidural hematomas or thromboembolic events occurring beyond the immediate postoperative period. Our inability to capture long-term follow-up data, including delayed cerebrovascular incidents, restricts the conclusiveness of our findings regarding ASA safety beyond hospitalization. The use of lateral plating was limited to single-level procedures; then, the impact of ASA continuation on multilevel plating was not detectable in the present series.
Third, this study focused exclusively on XLIF, which, while increasingly utilized, is only one of several approaches to lateral lumbar interbody fusion. Variations such as oblique lumbar interbody fusion (OLIF) or anterior lumbar interbody fusion (ALIF) may carry different bleeding and risk profiles, limiting the applicability of our conclusions to these techniques. Furthermore, our analysis did not include patients on dual antiplatelet therapy or those taking other anticoagulants, which represent common clinical scenarios in this population.
Finally, subgroup analyses based on comorbidities, ASA dosage, or use of intraoperative adjuncts (eg, tranexamic acid) were not feasible due to sample size limitations. Future studies should incorporate these variables into their design to better delineate risk profiles and refine perioperative management strategies.
Conclusions
In the present multicentric case series, acetylsalicylic acid continuation in patients undergoing single- or multiple-level XLIF for degenerative spine disorders was not associated with a significant increase in bleeding or complications.According to the potential risk of significant neurological consequences in case of acetylsalicylic interruption in patients at risk of cerebrovascular accidents, the management of these drugs should be carefully evaluated, and their interruption should be reserved for a properly stratified subgroup of patients. Properly designed prospective studies and clinical trials are needed to confirm our data.
Footnotes
Ethical Consideration
According to the retrospective and non-interventional design, the present investigation was exempt from the IRB evaluation.
Consent to Participate
The included patients signed an informed consent for the surgical procedure and the scientific use of their anonymized clinical-radiological data, including publication in scientific journals.
Author Contributions
Idealization and Conceptualization - L.R., S.T., S.F., A.P., M.M.; Data collection: L.R., S.T., A.P., A.S., M.M.; Data analysis: S.T., A.P., A.S., G.L.; - Drafting: A.P., A.S., M.M.; - Supervision: L.R., G.L., A.R.; Final paper revision: L.R., A.R.; Final version acceptance: all the authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
