Abstract
Study Design
A retrospective comparative study.
Objective
This study explored the factors influencing L5-S1 fusion success following posterior lumbar interbody fusion (PLIF) and identified risk factors for symptomatic nonunion.
Methods
The cohort comprised 134 patients who underwent single- or multiple-level lumbar fusion, including PLIF at L5-S1, due to degenerative spine disease. Radiographic fusion was assessed using 1-year postoperative CT scans. Demographic, clinical (VAS, ODI, and EQ-5D), and radiological data were compared based on fusion status using univariate and multivariate logistic regression analyses. Subgroup analysis was performed in patients exhibiting nonunion with or without symptoms.
Results
Fusion was achieved in 98 (73.1%) out of 134 patients, whereas 36 (26.9%) patients exhibited nonunion at the 1-year follow-up. Factors associated with nonunion included a higher body mass index [BMI; P = .020; 95% confidence interval (CI): .702, .971] and longer fusion levels (P = .032; 95% CI: .345, .952). Bilateral bicortical purchase of S1 pedicle screws significantly improved fusion outcomes (P = .014; 95% CI: 1.281, 9.047). Among the 36 nonunion cases, symptomatic patients showed significantly worse clinical assessment in terms of VAS, ODI, and every domain of EQ-5D. However, radiological parameters exhibited no differences based on the presence of symptoms.
Conclusion
The radiological nonunion rate at L5-S1 was 27%. Higher BMI and longer fusion levels were identified as risk factors, whereas bicortical screw placement at S1 emerged as a protective factor against L5-S1 nonunion. Therefore, bilateral anterior cortical purchase of S1 pedicle screws is recommended, particularly in patients undergoing longer fusion procedures.
Introduction
Posterior lumbar interbody fusion (PLIF) is widely used in spinal surgery owing to its high fusion rate, ease of application, and low complication rate.1,2 In recent years, improvements in fusion rates have been attributed to advancements such as pedicle screws and fusion cages. 3 Achieving solid fusion is critical to prevent complications associated with nonunion. Monitoring radiologic fusion and clinical assessment during follow-up is also essential for ensuring surgical success, as both factors play a critical role in determining PLIF outcomes.
Nonunion at the L5-S1 level remains a major challenge for spine surgeons despite advancements in recent years.4,5 Factors contributing to this challenge include poor sacral bone quality, complex anatomy, strong biomechanical forces directed toward the region, and use of multilevel or extended fixation constructs.6,7 Some patients with nonunion remain asymptomatic, whereas others may exhibit persistent or recurrent back or leg pain, instability, and hardware failure. 8 Diagnostic imaging techniques, such as plain radiographs, ultrasound, technetium scans, computed tomography (CT), and magnetic resonance imaging (MRI), facilitate accurate diagnosis when symptoms are present.9,10
The lumbosacral spine reportedly exhibits lower interbody fusion rates compared with other lumbar levels, although few studies have investigated the underlying mechanism. Various factors, such as cancellous bone stock, wide pedicles, greater disc wedge angle, and increased biomechanical stress, potentially contribute to relatively lower fusion rates following PLIF at L5-S1.11,12 Notably, some patients remain asymptomatic despite nonunion at L5-S1. 8 Evaluating fusion status, associated risk factors, and their clinical relevance is crucial; however, definitive conclusions regarding clinical and radiological outcomes post-PLIF at the L5-S1 level remain elusive.
This study aimed to investigate demographic, clinical, and radiological factors, as well as sagittal alignment, influencing fusion at the L5-S1 level post-PLIF. Furthermore, is explored the underlying cause of patients not experiencing symptoms despite exhibiting nonunion at this surgical level.
Material and Methods
Study Design
We performed a retrospective observational study involving 211 consecutive patients who underwent single- or multiple-level PLIF from April 2014 to January 2024 at our institution. The study was reviewed and approved by the Institutional Review Board for Clinical Research Ethics at our institute (IRB number: 2024-0316). Informed consent was exempted for this study based on its retrospective nature, as approved by the Institutional Review Board in line with institutional policies and applicable regulations. Ultimately, 134 patients met the inclusion criteria and were finally enrolled. All surgeries were performed by a single surgeon, and patients were followed up for >1 year.
Inclusion criteria were as follows: (i) patients diagnosed with degenerative lumbar spinal stenosis, degenerative or isthmic lumbar spondylolisthesis, or stenosis due to herniated intervertebral disc; (ii) patients undergoing single- or multiple-level instrumented fusion surgeries, including L5-S1 PLIF or transforaminal lumbar interbody fusion with unilateral or bilateral cages; and (iii) a minimum 12-month follow-up, with flexion and extension lateral radiographs and CT scans. Exclusion criteria included the following: (i) deformity, trauma, infection, tumors, unilateral instrumentation, and prior revision surgeries; (ii) surgeries such as oblique lumbar interbody fusion or posterolateral fusion at L5-S1; and (iii) a lack of clinical and radiographic data.
Baseline data were extracted from medical records, and preoperative radiographs were acquired, including standing anterior/posterior and lateral views of the lumbar spine, as well as flexion and extension views. Radiographic fusion was assessed using flexion and extension lateral studies and CT scans during a minimum 12-month follow-up period.
Clinical assessment scores were obtained preoperatively and at 6 months and 1 year postoperatively using the visual analog scale (VAS) for low-back pain, Oswestry disability index (ODI), and EuroQol-5 dimension (EQ-5D). Symptomatic nonunion was defined as new or recurrent onset low-back pain or radicular pain consistent with foraminal stenosis accompanied by nonunion at the 1-year postoperative period.
Radiographic Assessment
We measured preoperative and postoperative disc segmental angle (DSA), disc height, lumbar lordosis (LL), C7-S1 sagittal vertical axis (SVA), and pelvic parameters. The DSA was defined as the angle between the horizontal lines drawn on the lower endplate of the cranial vertebra and the upper endplate of the caudal vertebra. Disc height was calculated as the mean of the anterior and posterior intervertebral disc height. LL was measured between the superior endplate of L1 and the upper endplate of the sacrum, whereas the C7-S1 SVA was determined as the distance from a C7 vertical plumb line to the superior posterior corner of S1. Pelvic parameters included pelvic incidence (the angle between a perpendicular line to the sacral plate at its midpoint and a line connecting this point to the center of the femoral heads), sacral slope (the angle between the superior plate of S1 and a horizontal line), and pelvic tilt (the angle between the midpoint of the sacral plate to the femoral head axis and a vertical line). For detailed measurements of radiological parameters, refer to Figure 1. Measurements of radiological parameters. (A) PI, pelvic incidence; PT, pelvic tilt; SS, sacral slope; SVA, sagittal vertical axis. (B) LL, lumbar lordosis. (C) DSA, disk space angle. (D) DH, disc height.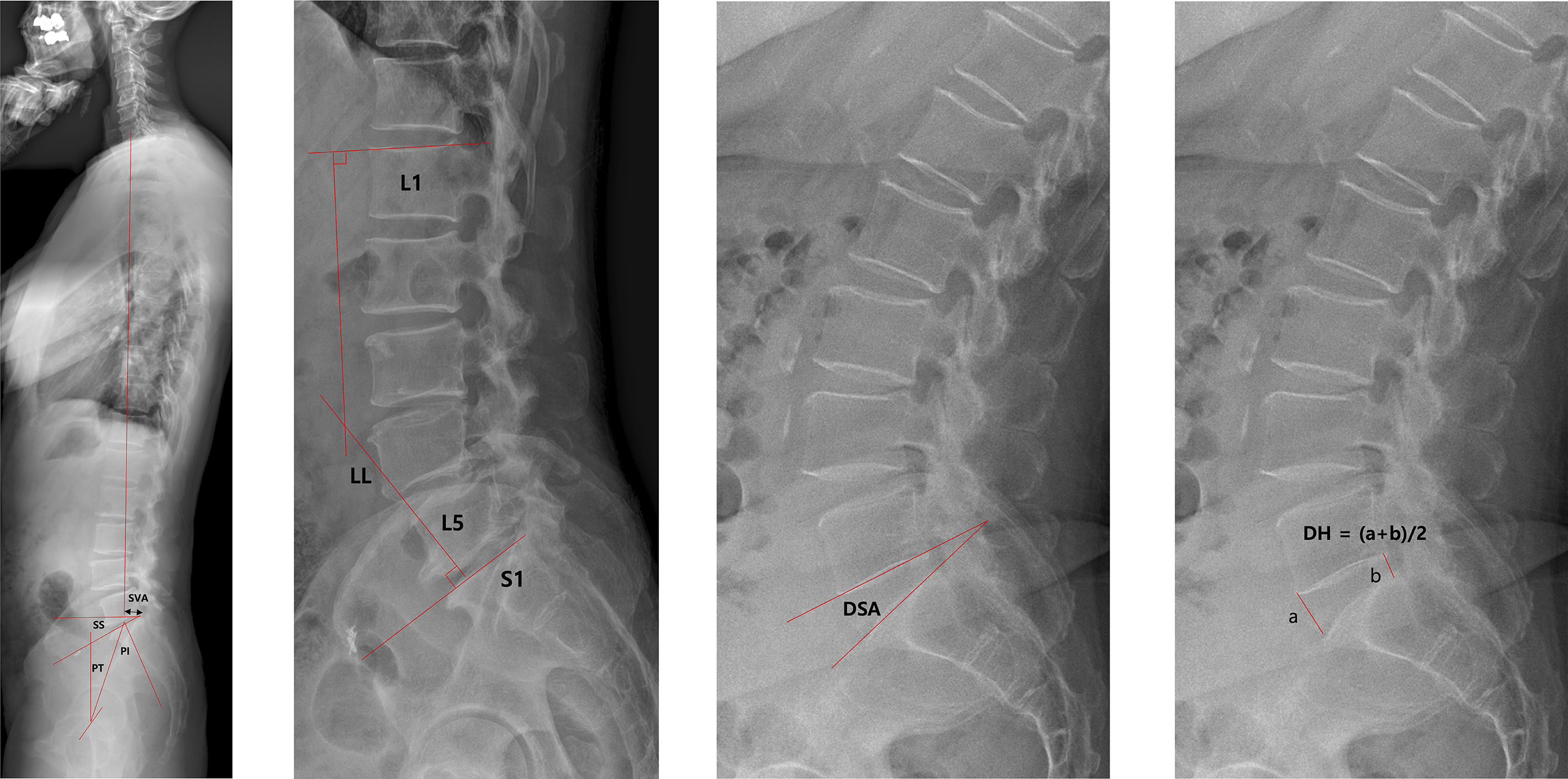
Instability was assessed using flexion and extension radiographs. Instability was defined as >3 mm translation or 5° angular motion. 13 Fusion status at 1 year was evaluated using CT scans, as they provide clear visualization of bridging trabecular bone at the fusion site and can detect radiolucent lines or haloing around implants that may indicate pseudarthrosis. 13 If instability was present on dynamic radiographs or if there was no evidence of fusion on CT scans, the case was classified as non-fusion.
Surgical Approach
We used a Jackson spine table equipped with supportive cushions on the chest and pelvic areas to minimize abdominal pressure during surgery, and the following operative procedure was performed. A midline incision was made on the back, and subperiosteal dissection was performed until complete bone exposure was achieved. Pedicle screws were inserted under checking intraoperative X-ray to check entry point by marking needle. Fluoroscopic guidance was not used. For S1 pedicle screw fixation, 7.5-mm width screws were used. Intraoperative X-ray imaging was also utilized to adjust the trajectory toward the sacral promontory with maximal convergence. However, in some cases, when the trajectory risked contacting the ilium, achieving sufficient convergence was challenging. The screw length of S1 was determined intraoperatively using a ball-tipped probe to palpate the sacral anterior cortex. Screws approximately 2-3 mm longer than the measured depth were selected to achieve bicortical fixation and were inserted after confirming cortical bone penetration. All screws were from the same manufacturer (Medyssey Co., Ltd, Korea). In the early phase of the series, no specific consideration was given to uni- vs bicortical fixation. However, as the importance of bicortical fixation became more apparent, we made a consistent effort to achieve bicortical purchase whenever feasible.
After screw insertions, the disk space was distracted with temporary rod fixation. Then, total laminectomy with complete total facetectomy was conducted, and bone grafts of adequate quantity and quality were obtained from the spinous process, lamina, and enlarged superior and inferior articular processes bilaterally. Subsequently, the ligamentum flavum was resected, and the nerve roots were retracted medially. Disc material was removed through serial dilation, and endplate preparation was performed via curettage using a ring curette. Central and/or foraminal decompression was performed prior to the cage insertions. PEEK cages were used in all cases, inserted either bilaterally (straight-type) or unilaterally (straight-type or banana-shaped). Morcellized local bone fragments mixed with demineralized bone matrix (DBM) were placed inside the cages. Additional morselized local bone chips mixed with DBM were also packed into the prepared disc space before cage insertion. We inserted the largest possible cage size without causing excessive distraction. Following the placement of interbody cages, pedicle screw-rod instrumentation was performed using conventional techniques. Sequentially, meticulous hemostasis was achieved, a drain was placed, and the wound was closed in layers. 1
Statistical Analyses
Demographic data, operation-related data, clinical outcomes, and radiological results were compared between groups using Student’s t-test or the chi-square test. Subgroup analyses were conducted based on symptomatic and asymptomatic nonfusion groups. Statistical analyses were performed using the Statistical Package for Social Sciences software, version 21.0 (SPSS, Chicago, IL). P values <.05 were considered statistically significant.
Results
Demographic and Preoperative Comparisons Between Fusion and Nonunion Groups.
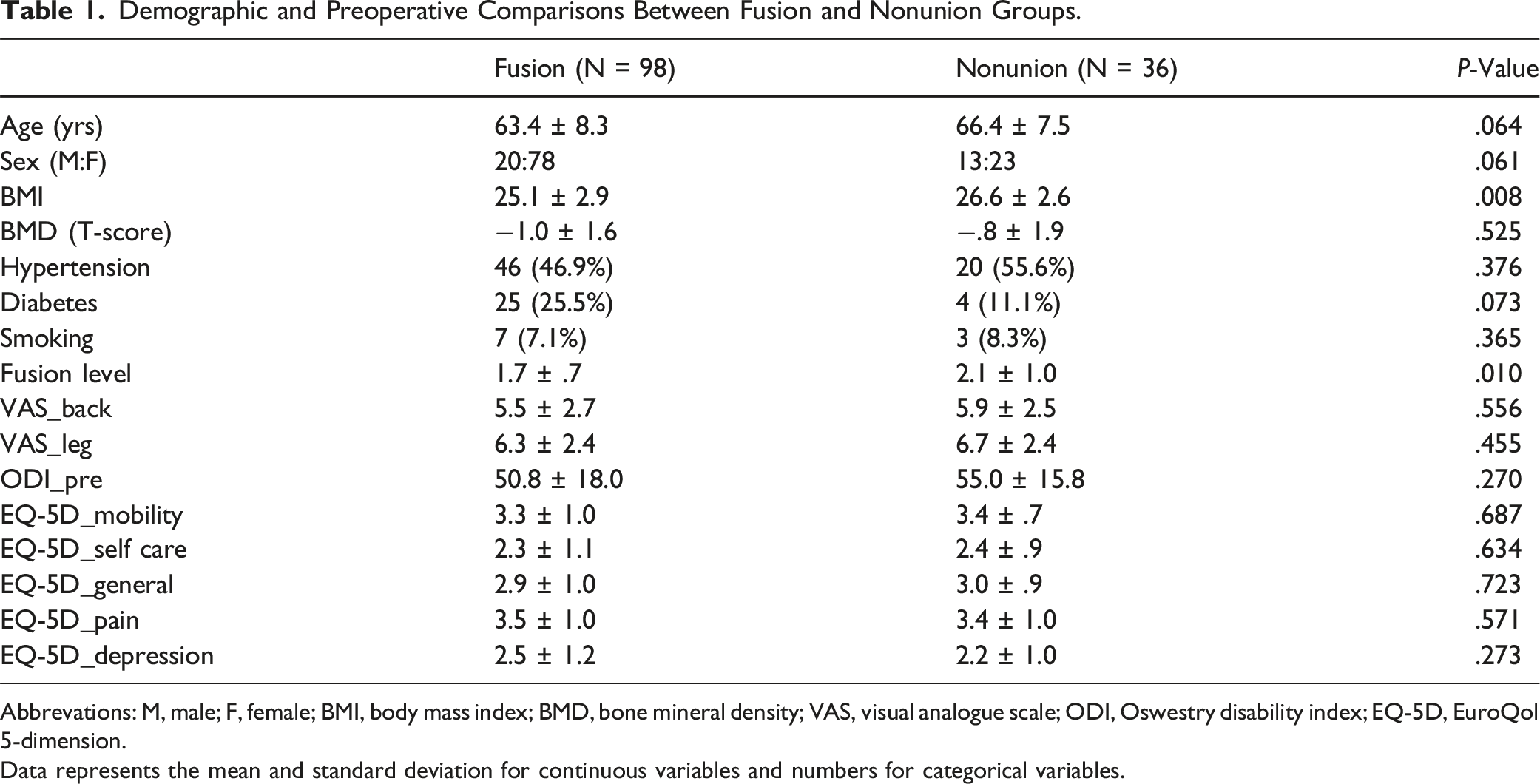
Abbrevations: M, male; F, female; BMI, body mass index; BMD, bone mineral density; VAS, visual analogue scale; ODI, Oswestry disability index; EQ-5D, EuroQol 5-dimension.
Data represents the mean and standard deviation for continuous variables and numbers for categorical variables.
Pre- and Postoperative Radiological Parameters Between Fusion and Nonunion Groups.
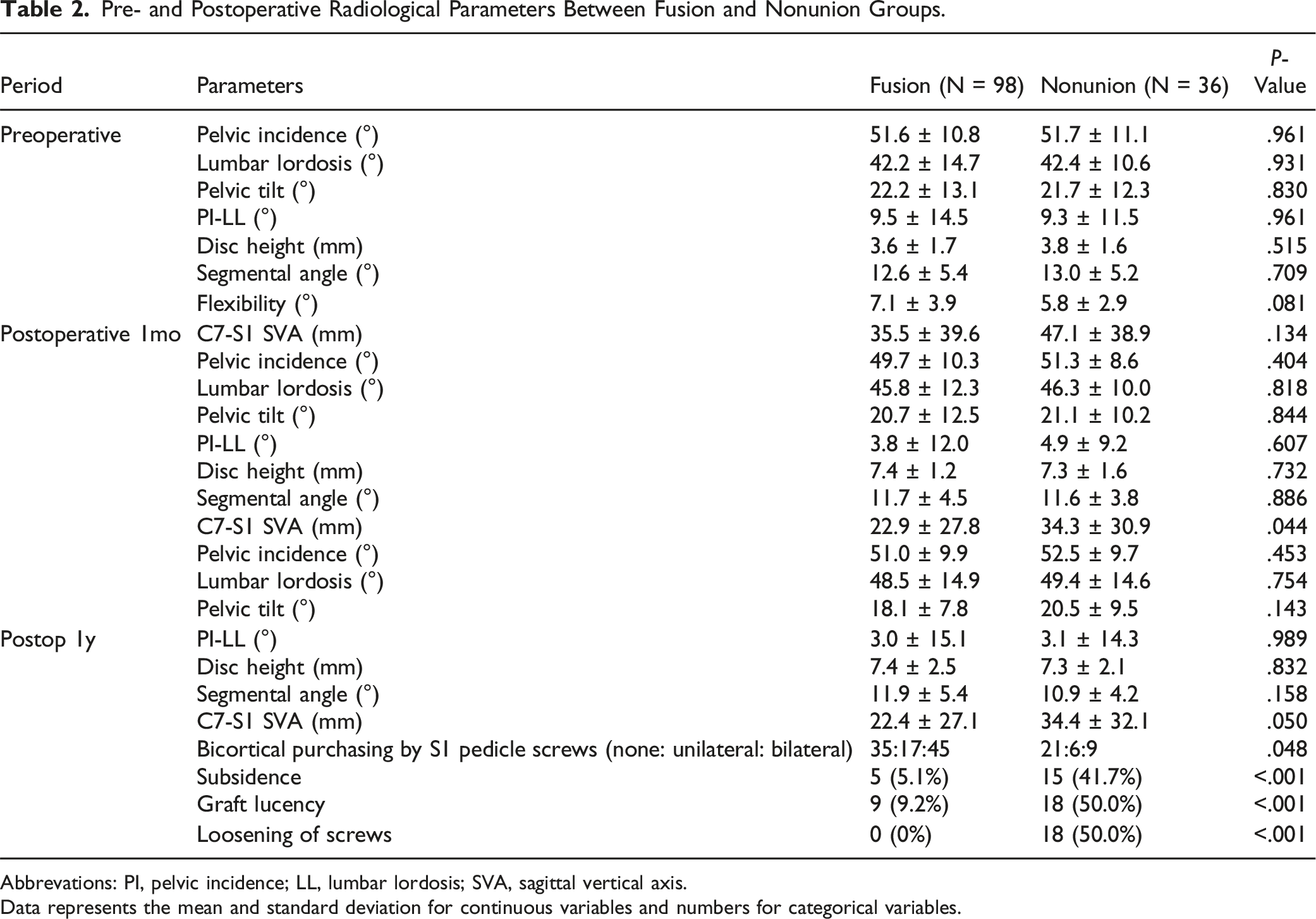
Abbrevations: PI, pelvic incidence; LL, lumbar lordosis; SVA, sagittal vertical axis.
Data represents the mean and standard deviation for continuous variables and numbers for categorical variables.
Multivariate Logistic Regression Analyses.
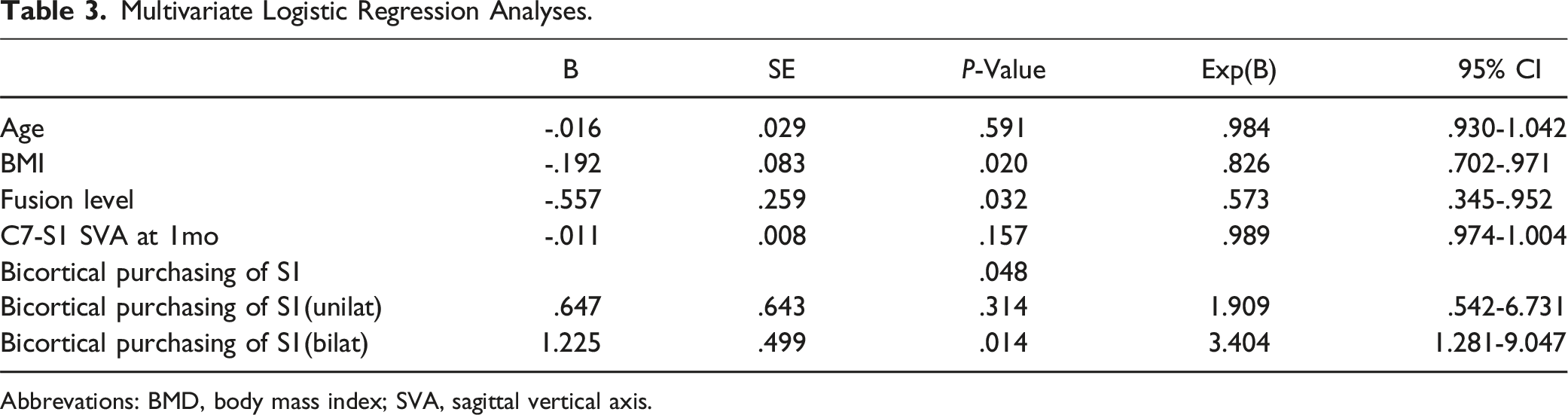
Abbrevations: BMD, body mass index; SVA, sagittal vertical axis.
Subgroup Analysis in Patients With Nonunion: Comparisons of Demographic and Clinical Parameters According to the Presence of Symptom.
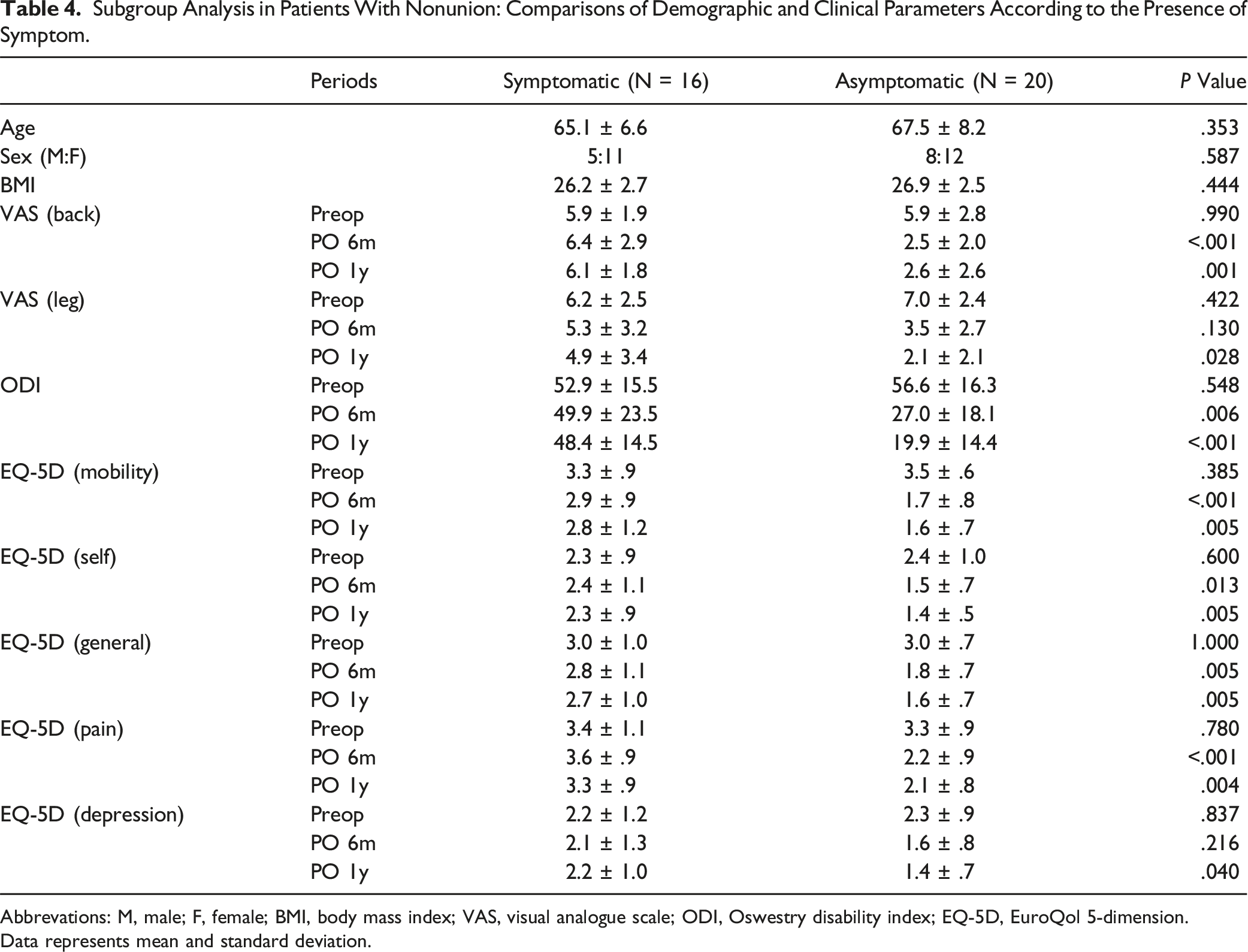
Abbrevations: M, male; F, female; BMI, body mass index; VAS, visual analogue scale; ODI, Oswestry disability index; EQ-5D, EuroQol 5-dimension.
Data represents mean and standard deviation.
Subgroup Analysis in Patients With Nonunion: Comparisons of Radiological Parameters According to the Presence of Symptom.
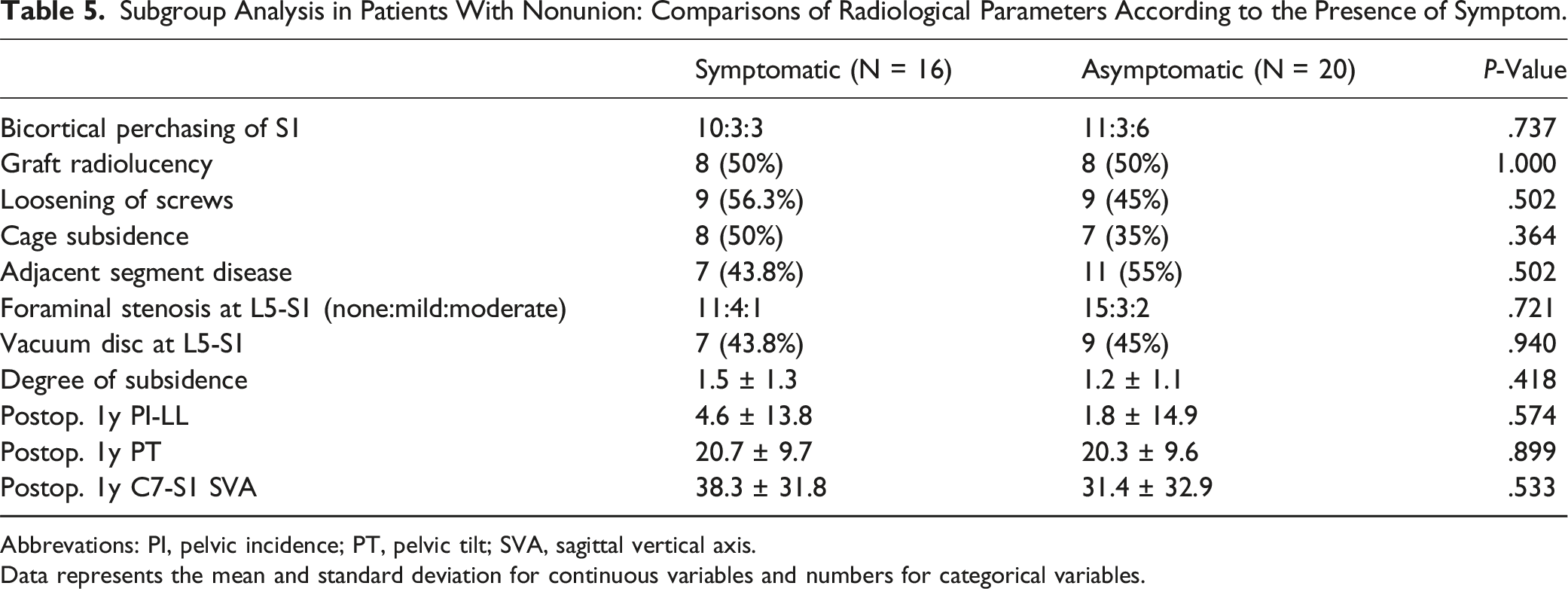
Abbrevations: PI, pelvic incidence; PT, pelvic tilt; SVA, sagittal vertical axis.
Data represents the mean and standard deviation for continuous variables and numbers for categorical variables.
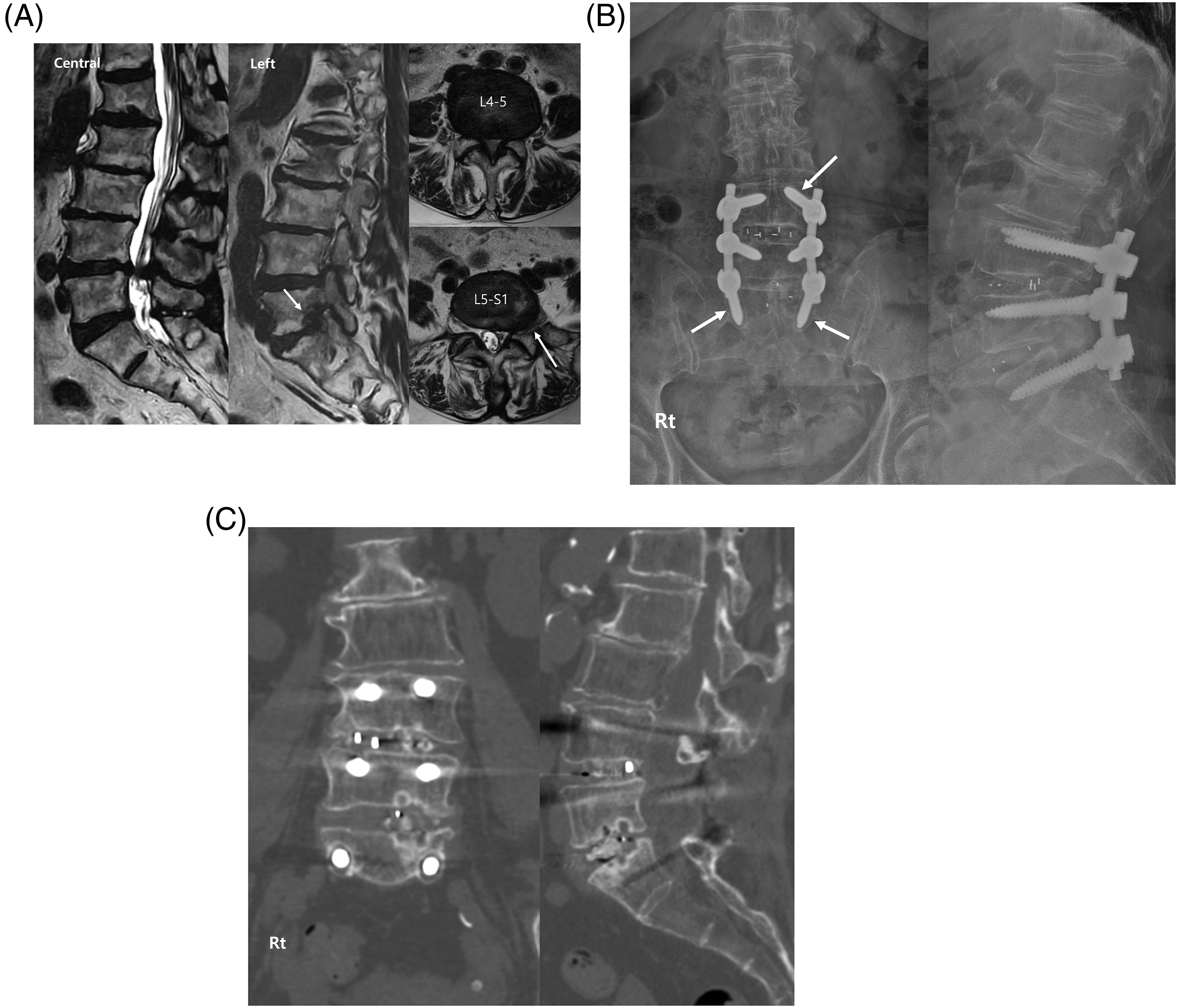
A 72-year-old female patient who complained of low-back pain and left leg sciatica (A) preoperative MRI showed severe central stenosis at L4-5 and severe left foraminal stenosis at L5-S1 (arrows); (B) screw loosening at L4 and S1 (arrows) was observed 1-year postoperatively in radiographs; (C) nonunion was confirmed 1-year postoperatively in CT scans.
Discussion
PLIF is the most commonly performed procedure for stabilization following decompression. It offers several advantages, such as a higher fusion rate, maintenance of proper sagittal alignment, and comparable clinical outcomes. 14 Although PLIF achieves fusion rates of 90%-95% at most levels, 15 the fusion rate at L5-S1 is notably lower, influenced by various factors.
In a study comparing fusion rates, two-level PLIF achieved an 85% fusion rate, whereas fusion involving L5-S1 showed significantly lower rates compared with lumbolumbar fusion (60% vs 97%, respectively). This disparity was attributed to anatomical and biomechanical differences compared with cephalad-level fusion, as well as the higher range of motion at the lumbosacral junction. 11 Similar results were reported in another study, indicating lower fusion rates for lumbosacral fusion compared with lumbolumbar fusion in two-level fusion cases (64% and 96%, respectively). 12 In our study, the fusion rate was 73.1% at L5-S1, consistent with previous findings.
Nonunion, a common complication defined as the absence of solid fusion 1 year postsurgery, was evaluated in the present study using CT scans at 1-year follow-up, with such scans considered superior to X-rays in detecting trabecular bridging as per the Brantigan–Steffee–Fraser classification of interbody fusion success. 16 Nonunion was identified by the absence of bone bridge formation, lucency around the cage, and screw loosening. Notably, a significant proportion of cases remained asymptomatic in our study [20/36 (55.5%)], suggesting underreporting relative to the actual occurrence rates. Reports on symptomatic nonunion following PLIF are limited, complicating its definition in patients with radiographic nonunion.
Low-back pain, whether new or recurrent, is a common occurrence post-PLIF, with previous studies showing that it may be unrelated to nonunion. Owing to the many risk factors associated with back pain, it is difficult to confirm that nonunion is the underlying cause. For example, persistent pain postsurgery may be due to neuropathic components. 17 Nonetheless, studies suggest poorer clinical outcomes in patients with nonunion.18,19 Thus, in our study, we defined symptomatic nonunion as new or recurrent low-back or radicular pain, consistent with foraminal stenosis accompanied by nonfusion.
Previous studies have proposed several risk factors for nonunion, including old age, poor surgical technique, excessive motion, multilevel surgery, smoking, high BMI, cage contact area, graft materials, and paraspinal muscle fat infiltration.20,21 Despite its clinical importance, few studies have focused specifically on L5-S1 segments. Our study identified higher BMI and longer fusion levels as significant risk factors for nonunion at L5-S1, consistent with the existing literature. Longer construct surgery, including PLIF, is associated with a 2-3-fold higher risk of nonunion. 4 In our study, multivariate logistic regression analysis indicated a fusion probability of 57.3% in two-level fusion compared with single-level fusion [Exp(B) = .573]. High BMI is associated adverse surgical outcomes, such as longer operative times, increased estimated blood loss, deep infection, deep vein thrombosis, and higher reoperation rates, although its direct impact on lumbar spine fusion remains unclear. 22 In a meta-analysis, only one study reported BMI’s effect on fusion rate: patients with BMIs >, 25-30, and <25 kg/m2 had fusion rates at 12 months of 60%, 76%, and 88%, respectively. 23 This is likely because mechanical stress to the L5-S1 segments is higher in patients with high BMIs, potentially leading to pedicle screw loosening and eventual nonunion. Despite our study also suggesting a correlation between higher BMI and nonfusion, further studies are warranted to obtain conclusive evidence.
Additionally, hip pathology may influence L5-S1 fusion outcomes. Severe hip osteoarthritis has been reported to limit pelvic retroversion, which may increase stress at the L5-S1 segment and potentially contribute to nonunion. 24 Although we did not collect data on hip pathology in this study, it could be an important factor to consider in the future.
Preoperative and postoperative radiological parameters did not significantly differ between the fusion and nonfusion groups, except for higher incidence of bicortical S1 pedicle screw purchase in the fusion group. Using bicortical S1 pedicle screws may enhance the fusion process. That was the main reason why bicortical insertion was not initially performed intentionally. However, with accumulated surgical experience, we came to recognize the potential importance of bicortical fixation in improving screw stability, and we gradually made greater efforts to achieve bicortical screw placement whenever possible. Although the most significant differences between the fusion and non-fusion groups appeared at approximately 1.7 (fusion) and 2.1 (non-fusion) levels, it is well known that even at the single-level L5–S1 fusion, the nonunion rate tends to be relatively high compared to other levels. 5 Therefore, we believe that bicortical screw insertion at S1 should be considered even in single-level fusions. The S1 pedicle portion is more cancellous, and the L5-S1 segment has higher angular motion compared with other segments; therefore, anterior cortical purchase can increase stability and pull-out strength, which is particularly beneficial for patients with osteoporosis. Surgeons may encounter challenges such as sacral screw fixation loss due to various factors, including smaller S1 body size, incorrect screw direction or insertion depth, osteoporotic sacral bone stock, or substantial bending loads applied to the distal instrumentation. Some surgeons advocate supplementation with anterior support, iliac fixation, or even S2 screw placement to enhance lumbosacral fixation stability. 25 Although anterior cortical purchase theoretically improves stability, potential risks include injuries to internal iliac vessels and the lumbosacral trunk. 26 However, such complications were not observed in the present study. Multiple anatomical studies have consistently shown that anteromedial screw placement offers a larger safe zone compared with anterolateral placement, reducing the risk of neurovascular injury with bicortical fixation, thereby enhancing safety and efficacy. 26 Luk et al. found that the bicortical S1 endplate sacral pedicle screw fixation technique, which leverages the density of the upper sacrum and the thick cortical endplate of the S1, offered better screw insertion torque and stronger fixation after cyclic loading compared with the conventional bicortical anterior cortex fixation technique. 27 In our study, only bilateral cortical purchase of S1 screws showed significant potential to reduce nonunion rates, suggesting that surgeons should prioritize achieving bilateral cortical purchase of S1 to minimize nonunion at L5-S1.
Unfortunately, we did not identify any relevant sagittal parameters as risk factors for nonunion. However, caution is advised when performing PLIF at L5-S1 in cases of existing sagittal malalignment, as PLIF may not reliably restore ideal sagittal balance at this level, potentially leading to poorer clinical outcomes.28,29 The relationship between sagittal alignment and fusion outcomes warrants further investigation. 30
Initially we aimed to identify factors related to symptomatic nonunion at L5-S1. However, we did not find any significant associations with relevant radiological parameters, likely due to the small number of patients and heterogeneity of symptoms among nonunion cases. Several factors may explain the presence of asymptomatic nonunion. For instance, in cases of so-called “locked nonunion,” where gross instability is absent despite a lack of solid bony fusion, patients may remain symptom-free. Furthermore, a recent study reported that symptomatic nonunion is more common at the L5-S1 level in cases of PLF compared to PLIF, which may be meaningful, since PLIF can achieve stability through cage insertion even in the absence of radiographic fusion. 4 Additionally, the presence of fibrous rather than bony union, as well as individual variability in pain sensitivity, could contribute to the absence of symptoms.
Our study had some limitations. For instance, its retrospective design potentially introduces selection bias and unconsidered confounding factors. As with many registry-based studies, lack of randomization may also have introduced biases. The heterogeneity present in the graft types used, including allografts and bone morphogenic protein, further complicates data interpretation. Moreover, the relatively small number of nonunion cases limits robust conclusions regarding risk factors for symptomatic nonunion. Nevertheless, our findings provide valuable insights into the risk factors for nonunion at L5-S1 post-PLIF, a challenging scenario frequently encountered by spine surgeons.
Conclusions
In the present study, the radiological nonunion rate at L5-S1 was 27%. Higher BMI and longer fusion levels were identified as risk factors, whereas bicortical screw placement at S1 emerged as a protective factor against L5-S1 nonunion. Therefore, we recommend bilateral anterior cortical purchase of S1 pedicle screws, especially in patients undergoing extended levels of fusion. Although symptomatic nonunion cases exhibited worse clinical outcomes across all assessments, no radiological parameters were associated with symptom presence. Thus, further research is necessary to elucidate the biological and mechanical influences on nonunion and symptomatology post-PLIF at L5-S1.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
