Abstract
Study Design
Retrospective analysis of prospective data.
Objectives
Neck pain represents a crucial factor underscoring a patient’s decision to receive surgical intervention for degenerative cervical myelopathy (DCM). However, postoperative pain trajectories are poorly defined. This study aimed to employ machine learning-based trajectory modeling to identify patient subpopulations with distinct pain trajectories after surgery.
Methods
We pooled subjects from three major clinical studies on DCM. Group-based multivariate trajectory (GBMT) modeling was used to classify patients into distinct trajectories based on their neck pain score over one year. Outcome differences were examined with univariate analyses. Predictors of group membership were revealed with multinomial logistic regression.
Results
Three distinct trajectories of neck pain were identified from a total of 968 patients with DCM: “slow pain improvement” (n = 239; 25%), “no pain improvement” (n = 537; 55%), and “fast pain improvement” (n = 192; 20%) groups. Each trajectory exhibited a unique baseline pain profile. The “fast pain improvement” group, comprised of patients experiencing profound neck pain, had the best overall outcomes for pain, NDI, SF-36 PCS, and SF-36 MSC postoperatively. On the other hand, the “no pain improvement” group, consisting of patients with pain and multimodal impairment of moderate severity, had residual pain that remained constant and was least likely to experience functional outcome and quality of life improvement after one year.
Conclusions
Unsupervised learning on neck pain identified unique pain recovery trajectories that consist of distinct patient phenotypes. Trajectory grouping offers an important framework to both identify novel DCM subpopulations and predict patterns of pain over time.
Clinical Trials Included
(1) Assessment of Surgical Techniques for Treating Cervical Spondylotic Myelopathy (CSM); https://clinicaltrials.gov/study/NCT00285337; ClinicalTrials.gov ID NCT00285337. (2) Surgical Treatment of Cervical Spondylotic Myelopathy; https://clinicaltrials.gov/study/NCT00565734; ClinicalTrals.gov ID NCT00565734. (3) Efficacy of Riluzole in Surgical Treatment for Cervical Spondylotic Myelopathy (CSM-Protect) (CSM-Protect); https://clinicaltrials.gov/study/NCT01257828; ClinicalTrials.gov ID NCT01257828.
Keywords
Introduction
As the worldwide leading cause of non-traumatic spinal cord disability, degenerative cervical myelopathy (DCM) is a highly prevalent condition that affects roughly 1.6 to 7.88 per 100,000 individuals. 1 It has substantial negative impacts on the physical and functional well-being of middle aged and older persons, frequently outweighing the effects of diseases like diabetes or cancer on overall quality of life. Ultimately, DCM results in significant psychosocial and financial burden on the patient, their families, caregivers, and the healthcare system. 2
DCM represents an overarching term that includes cervical spondylotic myelopathy and ossification of the posterior longitudinal ligament. Pathologically, it affects the cervical spinal cord and is caused by degenerative changes to the spinal discs, joints, and ligaments due to aging, osteoarthritis, intervertebral herniation, spinal stenosis, or other degenerative processes. 3 Clinically, DCM can present with a range of signs and symptoms, including loss of fine motor skills, coordination problems, difficulty maintaining balance, loss of bladder or bowel control, and neck pain. 4 Diagnosis of DCM requires a careful history and physical examination to identify signs and symptoms of myelopathy while ruling out alternative diagnoses, and clinical findings should be correlated with MRI findings. 5 Nevertheless, diagnosing DCM can be challenging due to a combination of its insidious onset, variable course of progression and response to therapy, along with the possibility of concomitant clinical symptoms. DCM has a poorly characterized natural history of progression. While some patients remain clinically stable or experience minimal pain without surgical intervention, others have rapid onset of symptoms. 6 A previous cohort study revealed that neurological deterioration was observed in 40-70% of newly diagnosed DCM patients who were managed nonoperatively over the span of 2.5 years. 7 Evidently, this population is quite heterogenous; it remains difficult to predict prognosis and provide clear predictors for which DCM symptoms patients would experience improvement in following surgery. Unfortunately, this may result in an expectation-actuality discrepancy, which denotes the gap between what patients expect from their surgical outcomes vs the actual results they experience postoperatively. This is a widespread phenomenon in the field of spine surgery that negatively impacts patient satisfaction. 8
The predominant classification system used to classify disease severity for DCM and assess patient outcomes following surgery remains the modified Japanese Orthopedic Association (mJOA) score. 9 DCM can be classified as severe (mJOA≤11), moderate (mJOA = 12-14), or mild (mJOA = 15-17). 10 Although surgery is considered the standard management for patients with moderate-to-severe DCM, 5 certain subpopulations of patients do not find adequate improvement in all their symptoms, including ones that are not assessed by the mJOA, such as neck pain. A report revealed that neck pain was considered a crucial factor for many patients with DCM, 4 and a survey of 659 patients with DCM indicated that neck pain was their number one recovery priority. 11 Despite the importance of neck pain for patients, the mJOA does not assess this aspect of DCM since it focuses solely on motor, sensory, and autonomic dysfunction assessments. 10 Nevertheless, our group employed a cluster analysis technique to demonstrate that there is a distinct patient phenotype of mild DCM that is characterized by neck pain alongside other factors not captured by the mJOA. 12 There is also an increased recognition in the literature that using mJOA alone does not correlate well with the prognosis of DCM. 13 This warrants examination of alternative approaches that consider neck pain, such as modeling the trajectory of pain over time, to supplement the mJOA with the goal of enhancing prognosis prediction and thereby improving patient satisfaction following surgery.
Group-based trajectory modeling was initially developed in the field of criminology to identify different groups of people who displayed similar patterns of criminal activity, which helped reveal factors that influenced criminal behavior. 14 Since then, it has been applied in various fields where longitudinal data analysis is valuable for understanding patterns of change over time, including the social sciences, epidemiology, psychology, and public health. 15 Previously, our group used the original group-based trajectory modeling technique to demonstrate that there are subpopulations of patients with traumatic spinal cord injury who have distinct recovery trajectories for upper limb motor function. 16 In the present paper, we employed a variation of this model, known as group-based multivariate trajectory (GBMT) modeling via an expectation-maximization (EM) algorithm, which is considered a form of unsupervised machine learning.
GBMT is used to identify distinct latent subpopulations that follow similar developmental trajectories across multiple correlated outcomes over time. By extending traditional group-based trajectory modeling to accommodate multivariate data, GBMT provides a more comprehensive framework for analyzing complex longitudinal patterns. 15 The EM algorithm is commonly employed to estimate model parameters, iteratively refining group membership probabilities (E-step) and optimizing trajectory coefficients (M-step) until convergence. This probabilistic approach allows for flexible handling of missing data and uncertain group assignments, enhancing the model’s ability to capture heterogeneous trajectories within a population. 17
Predefined labels or categories are not used in GBMT modeling. Rather, it identifies groups based on the trajectories of variables and finds underlying structures without explicit supervision. 15 Furthermore, the EM algorithm is a common approach used for unsupervised learning tasks; it iteratively determines the latent variables (i.e., group memberships in the case of GBMT modeling) and models parameters that best account for the observed data. 17 The present study aims to leverage the advantages of these approaches to yield a more robust analytical tool when applied to a large trial-based DCM database. By using this “bottom up”, data driven approach, we conducted an observational study to retrospectively analyze prospective data with the goal of elucidating distinct neck pain trajectories and examining how different outcome measures are impacted after surgery. Here, we showed that patients with DCM represent a heterogenous population, where there potentially exist three subpopulations that follow unique neck pain temporal profiles.
Methods
Study Population and Research Design
Subjects were pooled from three prospective, multicenter clinical studies: the Cervical Spondylotic Myelopathy (CSM)-North America (CSM-NA), 18 CSM-International (CSM-I), 19 and CSM-PROTECT (CSM-P; Efficacy of Riluzole in Surgical Treatment of Cervical Spondylotic Myelopathy) trials. 20 Key features are outlined in Supplemental Table 1. Patients with an mJOA score of 17 and under were enrolled over a period of two, four, or 5 years (CSM-NA, CSM-I, or CSM-P, respectively). All three trials had a uniform prospective design, DCM diagnostic criteria, and comparable patient groups. They were rigorously carried out in accordance with established protocols with a high standard of data quality. These clinical studies were originally designed to assess the efficacy of surgical decompression alone (CSM-NA & CSM-I) or in conjunction with the drug riluzole (CSM-P) for managing DCM to improve function and quality of life. Together, they represent one of the largest cumulations of clinical studies for DCM to date, with a total of 1047 patients.
All eligible patients with DCM provided written informed consent and fulfilled the following criteria: i) over 18 years of age; ii) never had previous cervical spine surgery and had at least one clinical sign of myelopathy; and iii) imaging evidence of cervical cord compression. Patients with concomitant lumbar stenosis, ankylosing spondylitis, rheumatoid arthritis, neoplasms, and active infection were excluded. Ethics approval was obtained through the internal review board of each respective study site. The University Health Network (UHN) Research Ethics Board granted approval for the combined de-identified datasets to be analyzed.
Data Collection
Age, sex, race, symptom duration, surgical approach, number of levels operated, fusion techniques, presence of ossified posterior longitudinal ligament, and medical frailty based on calculation via the modified frailty index-5 (mFI-5) 21 were among the surgical, clinical, and sociodemographic variables taken into consideration in this study.
At the time of trial enrollment, the severity of the disease was assessed using a multimodal clinical assessment that was conducted using 5 measurement tools consistent across the three studies: Nurick grade (gait impairment assessment), Neck Disability Index (NDI; disability status assessment); neck pain based on the NDI pain subscore (somatic pain assessment), and the combined upper & lower extremity motor subscores as well as the sensory subscore derived from the modified Japanese Orthopedic Association score (mJOA; physical disability assessment). Together, these 5 scores indicated pain, disability, and functional impairment. Review of the literature often demonstrates that patients with DCM see notable improvements in several areas after undergoing surgery. 22
Each patient’s outcome was assessed at 6 months and one year postoperatively using the change in score of mJOA, NDI, and SF-36. The mental component score (MCS) and physical component score (PCS) of SF-36 were used to evaluate mental and physical health, respectively, which provided specific details about the quality of life after surgery. We used a multiple imputation approach to deal with missing follow-up data. Since there were no discernible patterns for the missing data in relation to the research variables, we deemed the missing at random assumption likely.
Statistical Analysis
To determine subgroups of patients following distinct trajectories of neck pain over one year following surgery, we employed a type of finite mixture modeling known as group-based multivariate trajectory (GBMT) modeling that applies the expectation-maximization (EM) algorithm. 15 This enables maximum likelihood estimation from incomplete data of neck pain without the need for explicit imputation. Specifically, an expectation (E) step computes the expected value of the latent variables given the parameter estimates, and a maximization (M) step updates the parameter estimates to maximize the likelihood of the observed data. 17 Use of the EM algorithm reduced the likelihood of bias introduced via imputation of neck pain data and helped deal with potential multiple local maxima. 15 The model assigned each patient to the trajectory with the highest probability after determining the probability of fit. Based on the criteria outlined by the original creators of group-based trajectory modeling, 14 model of best fit consists of the following characteristics: i) close correspondence between the expected probability of group membership and the proportion of patients assigned to that group; ii) each group has an average posterior probability of greater than 70%; iii) odds of correct classification should exceed a minimum threshold (i.e., 5) for each group; and iv) least Bayesian information criterion (BIC).
Next, clinical variables and outcomes were compared across groups following trajectory identification. Categorical variables were reported as counts and percentages, whereas continuous data were reported as means and standard deviation (SD). Differences between neck pain trajectory groups were assessed using univariate approaches such as analysis of variance (ANOVA) for continuous variables and 3-way chi-square for categorical data. To test distribution normality, the Shapiro-Wilk test was used. Post hoc Tukey’s HSD test was used to further assess significant ANOVA results while correcting for pairwise family error rate.
Lastly, we employed multinomial logistic regression to identify significant predictors of neck pain trajectory groups. This type of regression analysis was chosen due to the presence of multiple dependent variables and their categorical nature in the current data structure.
23
This multivariate approach was repeated to assess each of the pain trajectories as the baseline reference group. The variables were chosen, with modification, based on previously published research that made use of these datasets. Coefficient estimates, odds ratios (OR) with 95% confidence intervals (CI), and
Results
Study Participants
A total of 968 patients who had neck pain data for at least one of the three time points (i.e., baseline, 6 months, one year) were identified from the pooled DCM dataset spanning the years 2005 to 2017. Baseline measures that were examined included demographic information (Supplemental Table 2), comorbidities and frailty (Supplemental Table 3), and clinical factors (Supplemental Table 4). One-year outcomes are shown in Supplemental Table 5. When looking at the total patient population, the mean (SD) baseline preoperative neck pain was 1.87 (0.74), which decreased to 1.21 (0.63) and 1.13 (0.66) at 6 months and one year after surgery, respectively.
Average Number of EM Iterations Across 100 Random Restarts, Log Likelihood, Number of Free Parameters, and Values of Information Criteria for One to Four Groups With Polynomial Degree (
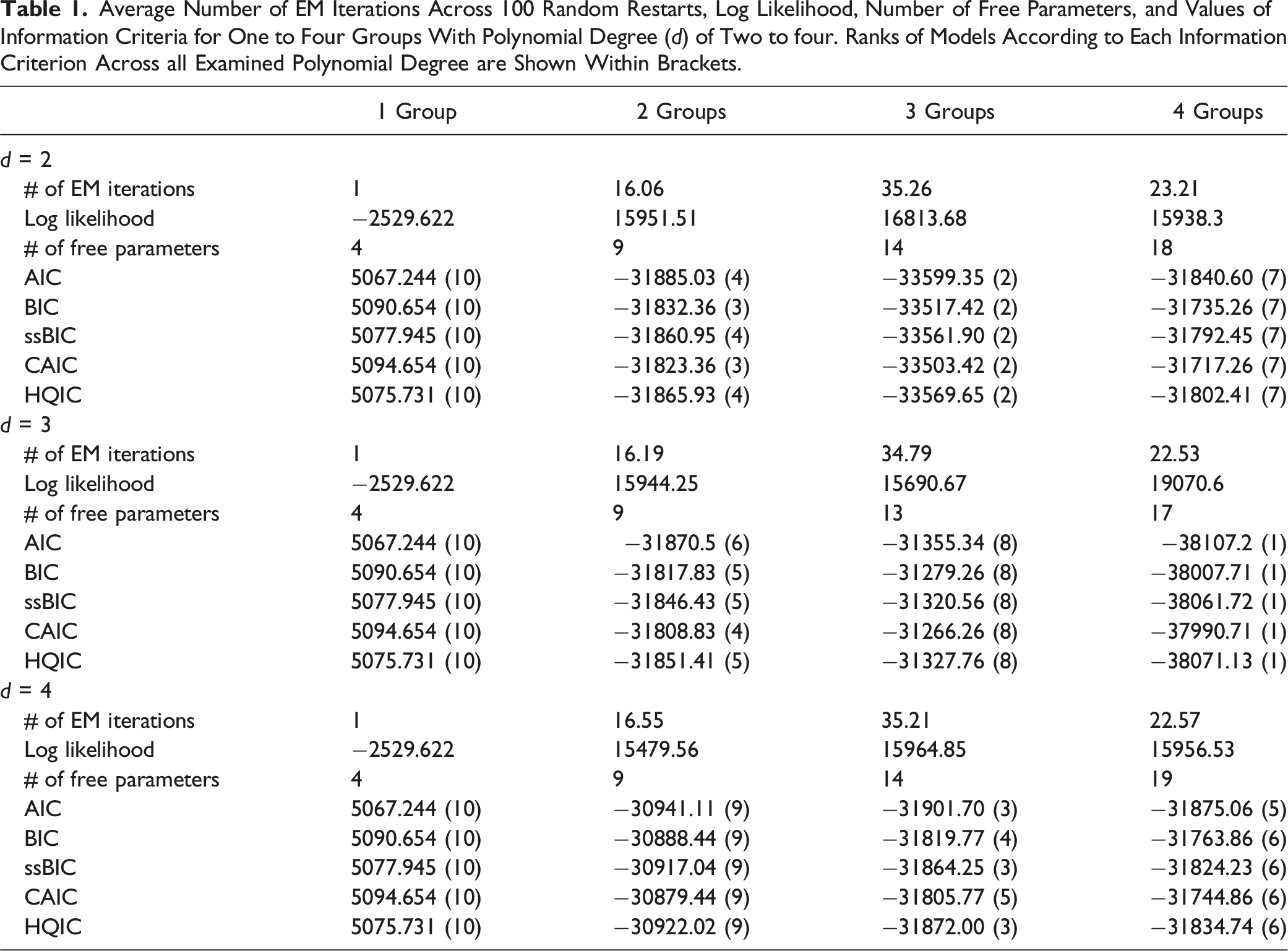
Model Selection for Neck Pain Trajectory
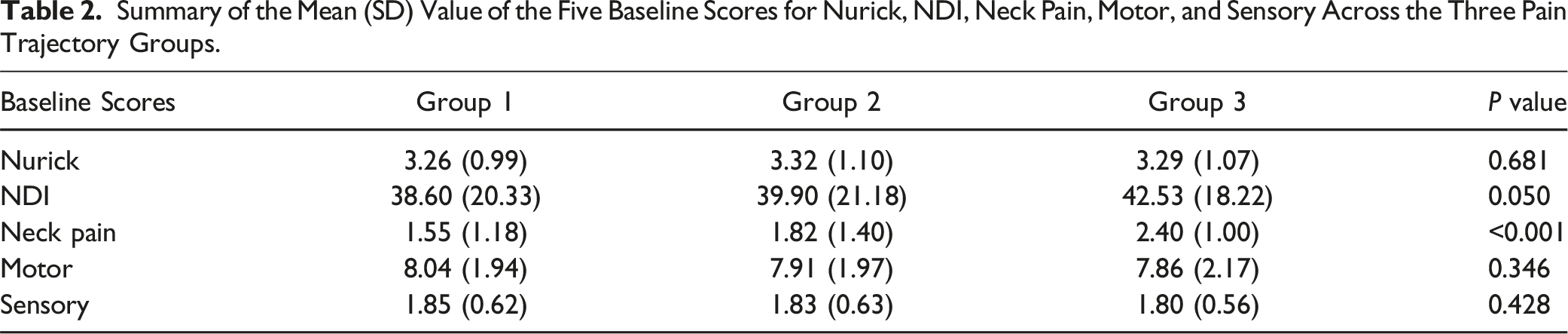
Summary of the Mean (SD) Value of the Five Baseline Scores for Nurick, NDI, Neck Pain, Motor, and Sensory Across the Three Pain Trajectory Groups.
Out of the 12 GBMT models, we found that four groups with a polynomial degree of three (4G3D) ranked first by all criteria, and three groups with a polynomial degree of two (3G2D) ranked second. However, 4G3D did not satisfy the posterior probability criterion of greater than 70% for each group, whereas 3G2D did (i.e., 88.6%, 75.1%, and 93.1% for each of the three groups). 3G2D also fulfilled the other criteria of group-based trajectory modeling. Therefore, the optimal trajectory number and shape for modeling neck pain in our dataset were three groups and quadratic, respectively.
Neck Pain Trajectory Group Characteristics
As shown in Figure 1 and Supplemental Table 6, the three identified trajectories were classified as: Group 1) slow pain improvement (n = 239; 25%), Group 2) no pain improvement (n = 537; 55%), and Group 3) fast pain improvement (n = 192; 20%). The distribution of age, sex, race, ethnicity, and educational level did not differ significantly between the three neck pain trajectory groups (Supplemental Table 2). Three different pain trajectories identified by GBMT modeling. Group 1 (n = 239, 25%), red; Group 2 (n = 537, 55%), green; Group 3 (n = 192, 20%), blue. [Top] Spaghetti graphs showing individual patient trajectories. Note that multiple patients can follow the same trajectory. [Bottom] Column charts showing the progression of neck pain over one year. * Reached estimated MCID compared to baseline; ‡ reached estimated MCID compared to 6 months.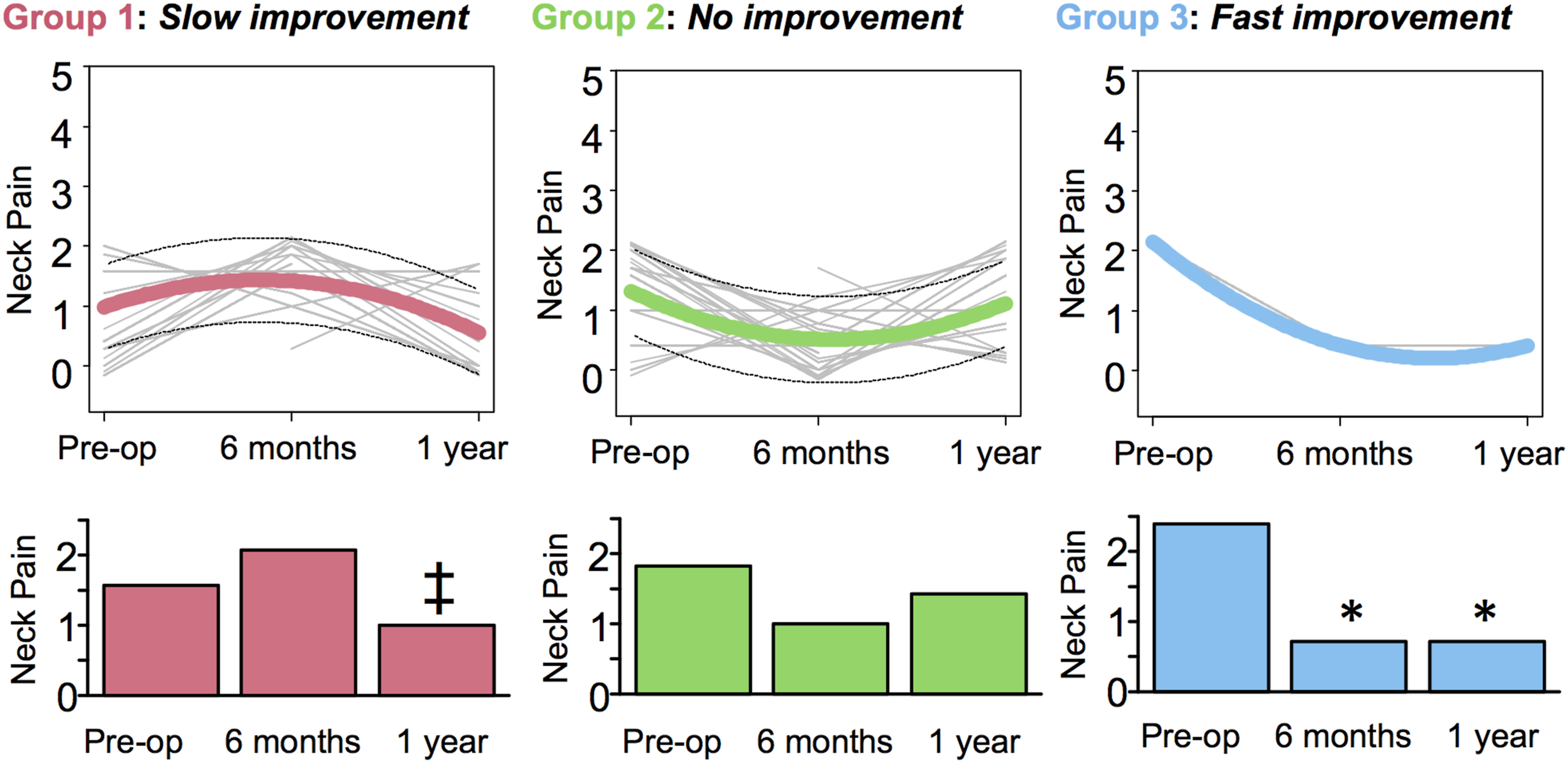
Slow Pain Improvement
At baseline, Group 1 trajectory was characterized by the lowest mean neck pain at 1.55. This increased slightly after surgery at 6 months to 2.08, which did not reach the estimated minimal clinically important difference (MCID) threshold for neck pain (i.e., 1 on a 0-5 scale; summary of literature review shown in Supplemental Table 7). At one year, neck pain was reduced to 1.01, which was clinically significant when compared to 6 months.
No Pain Improvement
Group 2 trajectory at baseline was characterized by a medium level of neck pain at 1.82, which was greater than Group 1 but less than Group 3. Postoperatively, there was a decrease to 0.99 at six months followed by an upwards rebound at one year to 1.41. However, these fluctuations were not clinically significant.
Fast Pain Improvement
Group 3 was characterized by the highest neck pain of 2.40 at baseline. After surgery, we observed a drastic clinically significant reduction in neck pain to 0.70 starting as early as six months and remaining at this level at the one-year timepoint.
DCM Patient Phenotype Identification of Neck Pain Trajectory Groups
Figure 2 and Table 2 illustrate a profile plot and summary, respectively, characterizing the three trajectory groups based on 5 key symptom assessment scales for DCM (i.e., Nurick, NDI, neck pain, motor, and sensory scores) at baseline. The most important differentiating factor was the neck pain score, as demonstrated by the significant ANOVA and corresponding post hoc tests that showed significance between every pairing ( Profile plot of neck pain trajectory groups using GBMT modeling. The x-axis indicates the 5 symptom assessment scales while the y-axis represents the mean standardized score for each of the 5 scales at baseline. The red, green, and blue lines represent the three identified pain trajectories (i.e., Groups 1, 2, and 3, respectively). For Nurick, NDI, and neck pain, higher value is worse. For motor and sensory, lower value is worse.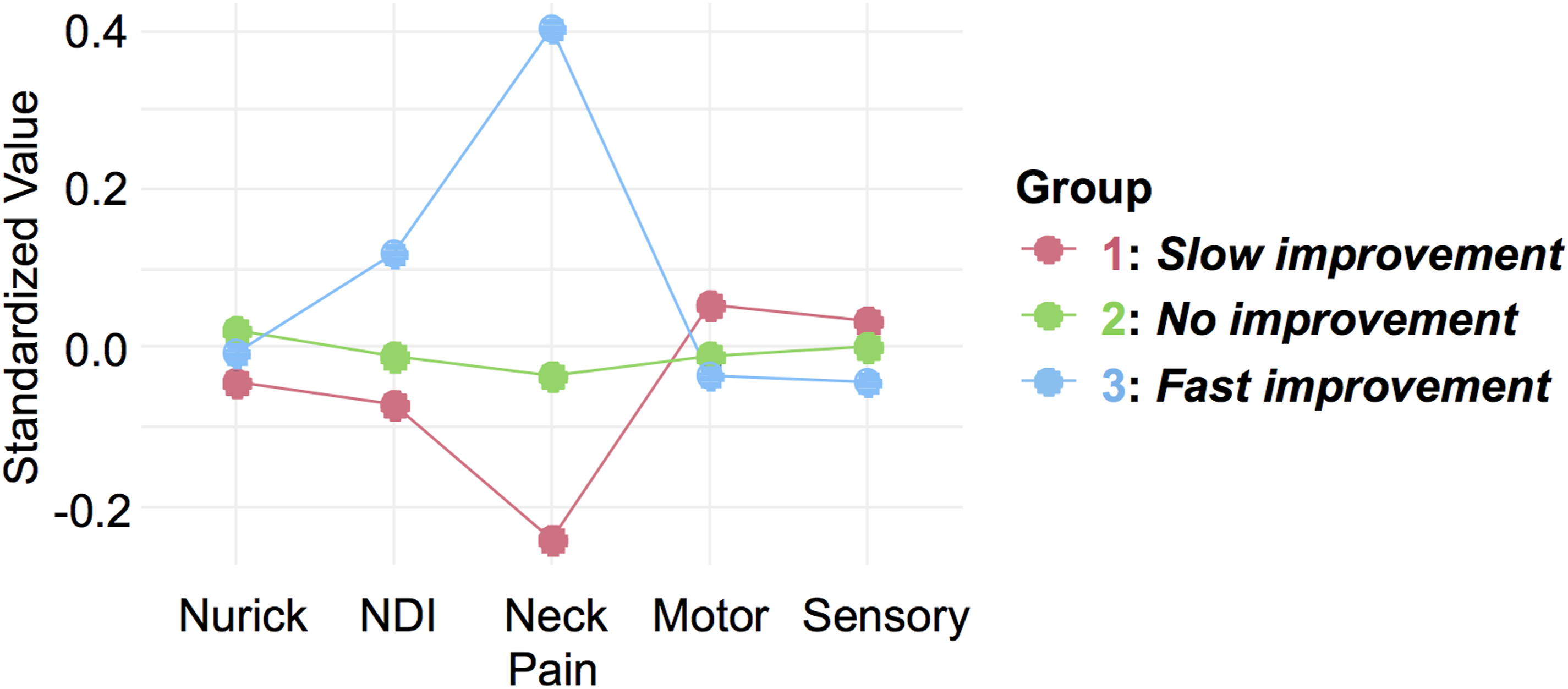
Slow Pain Improvement
Group 1, making up 25% of the study population, consisted of individuals who had minimal pain. They also displayed the highest scores for motor (mJOA motor subscore = 7.86 ± 2.17) and sensory (mJOA sensory subscore = 1.85 ± 0.62), alongside with having the least gait impairment (Nurick = 3.26 ± 0.99). Taken together, we considered this group as displaying
No Pain Improvement
Group 2 represented over half (55%) of our patient sample and was characterized by younger patients (mean age = 56.26 ± 11.35) who were less frail (mFI-5 = 0 = 27%). These patients were mostly ranked in the middle across the various symptom assessment scales relative to Groups 1 and 3, which suggested
Fast Pain Improvement
Comprising of 20% of the sample, Group 3 had older patients (mean age = 58.56 ± 11.29) and can be distinguished from other groups based on their profound neck pain. Additionally, they had the highest disability score (NDI = 42.53 ± 18.22), as well as the lowest scores for motor (mJOA motor score = 7.86 ± 2.17) and sensory (mJOA sensory subscore = 1.80 ± 0.56). Thus, patients in this group experienced
Identifying Factors Associated with Neck Pain Trajectory Groups
Neck Pain Trajectory Group Membership Predictors Based on Multinomial Logistic Regression Analysis of Covariates.
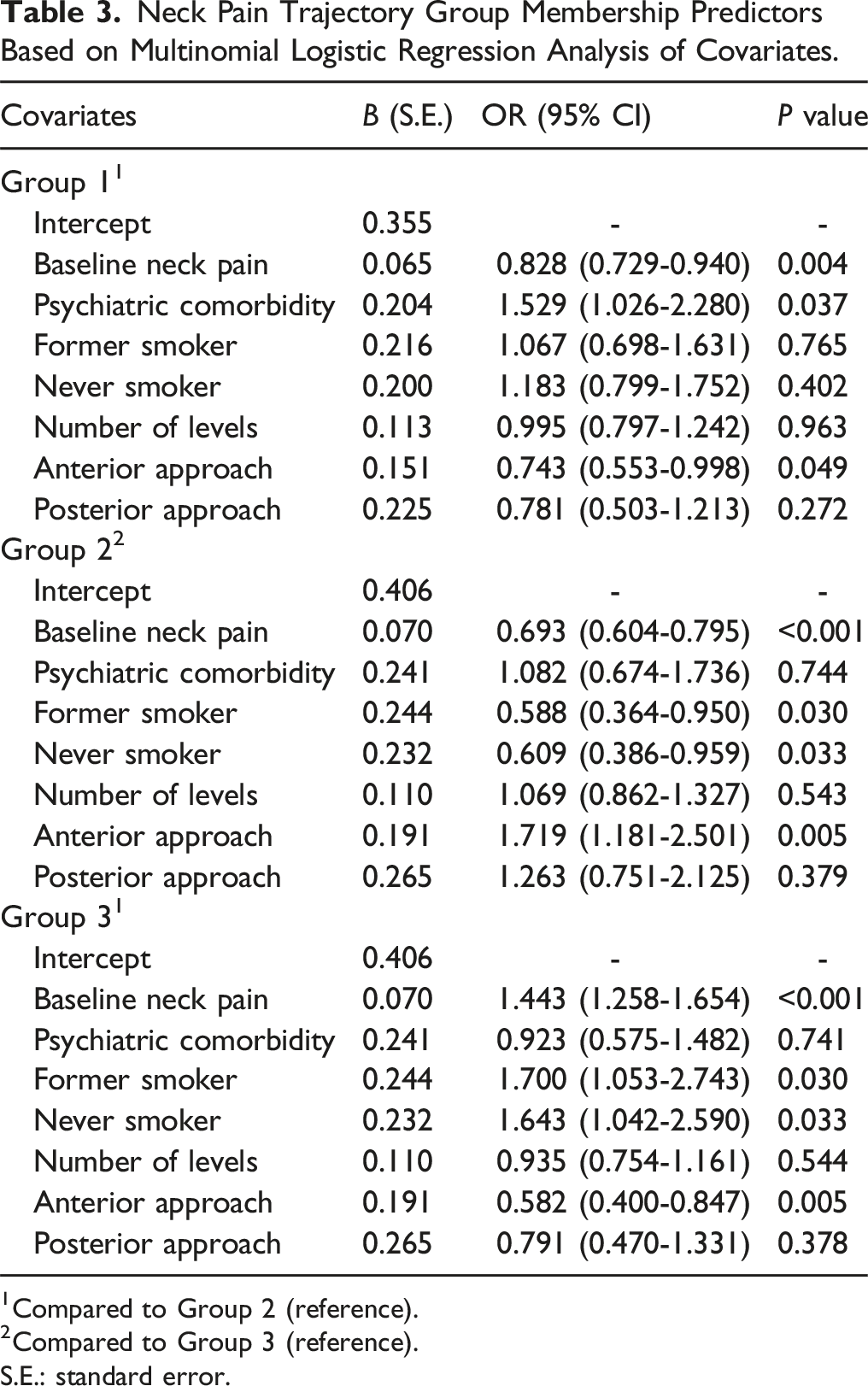
1Compared to Group 2 (reference).
2Compared to Group 3 (reference). S.E.: standard error.
Discussion
The present study identifies subpopulations of patients with DCM based on distinct neck pain trajectories following surgery. By applying an unsupervised group-based modeling algorithm, we were able to delineate patient groups that followed similar neck pain progression patterns over time. Our results show that patients fall into three different neck pain trajectory groups that range from possibly no pain improvement (i.e., failed to reach estimated MCID for pain reduction by one year) to fast pain improvement (i.e., reached estimated MCID by 6 months). Trajectory modeling based on neck pain has the potential to supplement the constraints of the conventional mJOA system by characterizing patient phenotypes and outlining the trajectory of clinical/functional outcome after surgery.
Since its initial description, the term DCM had referred to a heterogenous population of patients. 24 Addressing this heterogeneity remains a key challenge, and it is difficult to predict patient outcome following surgical intervention. Setting the expectations of patients and their families via outcome predictions is an important goal in informed consent to reduce the expectation-actuality discrepancy (EAD). Accordingly, if preoperative counselling establishes expectations that are more in line with the actual surgical outcome, patients will overall perceive a better outcome, leading to improved mental health, patient satisfaction, and overall recovery. 8 For DCM, it is especially important to manage EAD by setting appropriate expectations prior to surgery when patients are experiencing both neck pain and neurological deficits, since they might believe surgery can alleviate all of their symptoms. Unfortunately, despite neck pain being the number one reason why patients seek surgery, it has only recently begun to garner attention as a research focus within the DCM literature9,25,26; many older clinical studies had opted to forego measurements of neck pain as this metric was traditionally not recognized as relevant in DCM. Ultimately, the disconnect between research priorities vs patient needs has resulted in a poor understanding of how neck pain is affected with surgery for different patients. Our work reveals potential trajectories to expect in terms of pain improvement based on the patients’ corresponding clinical phenotype.
Recently, our group published a study that delineated a novel approach of classifying the phenotypes of patients with DCM by supplementing standard motor and sensory assessments (via the relevant mJOA subscores) with important measures for functional disability (via the Neck Disability Index; NDI), gait impairment (via Nurick), and neck pain (via the NDI pain subscore). 9 The rationale was that combining these 5 measures results in more realistic grouping that better resembles the complex range of symptoms that patients typically present with in the clinical context. Notably, these 5 symptoms correspond with current systematic reviews that identify the most prevalent DCM presentations. 27 Based on this classification scheme, we found in our current study that each pain trajectory group trended towards a specific phenotype. Consistent with our previous publication, 9 neck pain was by far the pivotal classifier, with each trajectory group having a different baseline level of pain (i.e., low, medium, and high for Groups 1, 2, and 3, respectively) that was statistically significant when assessed with either univariate or multivariate approaches. Further characterization when considering the other symptom measures showed a pattern where the low, medium, and high pain groups trended towards having minimal, moderate, and severe impairment, respectively. Nevertheless, given a lack of statistical significance, the clinical relevance of classifying our pain trajectory groups with these other symptom measures remains to be determined.
A previous study suggested that greater preoperative pain is associated with a higher likelihood of persistent postoperative pain for a range of surgical procedures such as limb amputation, thoracotomy, and breast surgery. 28 However, this does not appear to be the case here since our results showed that patients with DCM who have the greatest preoperative neck pain (i.e., Group 3) in fact experienced the fastest improvement in pain postoperatively. This was potentially due to the nature of cervical spine surgery potentially affecting pain differently compared to other types of surgeries, as a recent study had suggested that cervical cord decompression for DCM can influence pain perception by affecting central sensitization. 26 Indeed, such a mechanism of action would be especially profound for patients where pain is the predominant complaint. Evidently, this subgroup (20% of sample) appeared to benefit most from surgery for pain reduction. On the contrary, the group consisting of over half of the sample population (i.e., Group 2) had medium baseline pain that remained unchanged after one year of follow-up. This suggested that surgery may not be effective in alleviating neck pain for everyone, notably in patients where pain is not the primary complaint. Furthermore, these patients had the worst quality of life, functional, and neurological outcomes at the one-year timepoint. This finding was consistent with a recent cohort study demonstrating that persistent pain after spine surgery can lead to poor long-term patient-reported outcomes. 29 Overall, our findings suggest that surgical intervention for DCM can alleviate neck pain in certain cases but not all, thereby supporting the notion that cervical spine surgery is intrinsically not designed to cure pain. 30
The application of a data-driven approach with high granularity data could provide predictions about disease progression with better accuracy in the field of spine surgery. In our present study, 1047 patients with uniform DCM diagnoses were aggregated via a merger of three datasets, representing one of the largest prospective compiled datasets for DCM. From this total population, we identified 968 patients with data for neck pain, which accounted for 92% of the total combined sample. When averaging these patients, we see an overall trend of neck pain reduction postoperatively over time. To enhance our analysis, we employed a form of finite mixture modeling that incorporates the GBMT method to reveal meaningful subgroups with distinct pain trajectories. A key advantage of group-based trajectory modeling over traditional regression-based approaches is its ability to identify distinct, data-driven subpopulations without requiring a priori assumptions about trajectory shapes. Unlike conventional methods such as linear or logistic regression, which assess pain as a continuous or categorical outcome based on predefined covariates, GBMT accommodates non-linear patterns and heterogeneity within the study population. 15 This allows for a more nuanced understanding of recovery trajectories, particularly in conditions like DCM, where pain progression is highly variable. While direct performance comparisons with traditional models were not conducted in this study, future research could explore whether GBMT offers superior predictive accuracy or clinical utility by integrating validation analyses against established statistical techniques.
Using GBMT, the optimal number of trajectory groups was objectively evaluated using posterior probabilities and goodness-of-fit criteria. This led to the identification of three distinct subgroups of patients with unique traits who experience pain differently: patients in Group 1 have low baseline pain and experience pain reduction at one year (“slow pain improvement”); Group 2 patients have medium baseline pain that stays constant (“no pain improvement”); and those in Group 3 who have high baseline pain benefit from pain reduction by 6 months (“fast pain improvement”).
Our multinomial regression findings reveal potential predictors for group membership in addition to baseline neck pain status. Group 1 “slow pain improvement” was primarily defined by the presence of psychiatric comorbidities. Mental health deterioration is an extremely prevalent issue for patients with spinal cord disabilities. A recent longitudinal cohort study found that these patients had a 79% increased risk of developing any psychiatric comorbidity compared to control adults. 31 Previous work from our team noted that approximately a third of patients with DCM had psychiatric comorbidities, which the authors expressed as being much higher than expected. 32 Furthermore, a previous longitudinal prospective study assessing patients after spine surgery reported that their mental health was substantially impacted in a negative manner that did not improve over 6 months. 33 This timeframe was consistent with our present study’s finding that Group 1 displayed an increase in neck pain at 6 months postoperatively. Despite not reaching the estimated MCID for pain increase, patients in this group had the worst quality of life and functional outcomes at 6 months. Taken together, our results supported the idea that psychiatric comorbidities could potentially contribute to delayed pain improvement following spine surgery, with broader implications on other outcome measures. 34
Anterior surgery had a significant association with membership in Group 2 “no pain improvement”, which had reduced segment involvement and a more focal pathology. This result is consistent with current best practices that favor applying anterior operations to pathology of one to three levels. 35 In contrast to the evidence suggesting higher functional status and quality of life in patients with axial neck pain following anterior operations, 36 when considering patients with DCM specifically, Group 2 exhibited the least improvements in terms of neurological outcomes, functional status, and quality of life at the one-year timepoint. However, rather than the anterior approach contributing to the poorer outcomes of Group 2, it appears more likely that the lack of improvement in neck pain had greater impact. 29 Notably, this finding should not be interpreted to suggest that anterior surgery is associated with poorer pain outcomes in general; rather, anterior approaches are typically associated with less postoperative pain compared to posterior approaches, likely due to differences in surgical disruption of musculature and postoperative immobilization. The observed association between anterior surgery and membership in the “no pain improvement” trajectory is likely coincidental, reflecting underlying patient or disease characteristics of DCM rather than the surgical approach itself.
Lastly, Group 3 “fast pain improvement” was associated with being non-smokers. It has been previously shown that cigarette smoking decreased the odds of improvement after spine surgery, and a smoking history was associated with a lower recovery in Nurick score that was independent of the length of symptoms. 37 Moreover, it was further demonstrated that smoking, especially if long-term (>25 pack years), can directly reduce the spinal cord’s inherent healing capacity. 38 Thus, this is consistent with our finding that non-smokers benefit from having the quickest improvement in pain and best overall outcomes starting at 6 months.
Our work employed a robust dataset that came from worldwide multicenter studies carried out according to strict protocols to ensure consistent diagnosis and outcome evaluation. Nonetheless, this study had several limitations, which include possible data quality constraints, missing information, and importantly, restrictions imposed by utilizing only the parts that were common to all three datasets. This latter point restricted us to using the NDI pain subscore to assess neck pain, which raised two key challenges:
1) Given the limited number of scores on the pain intensity scale of NDI (i.e., zero to 5), there were potentially both ceiling and floor effects when used alone to assess pain status. 39 When compared to the more robust 10-point Numeric Rating Scale (data that were only collected in one of the three studies), some patients may experience changes in pain that the NDI scale could not detect due to lack of sensitivity. 2) Since the neck pain trajectory groups were identified using the NDI pain subscore, our analyses inherently focused on a component of functional disability. However, this deviated from the main goal of the original studies, which were designed to assess neurological outcome as the primary focus. Thus, our current findings should be applied cautiously when considering the broader clinical context. To help bridge this discrepancy, we show in Supplemental Table 8 that improvement in NDI parallels the improvement in both quality of life and neurological outcomes.
As specified in the initial trial guidelines, all study participants received a maximum of 12 months of follow-up after surgery. This consequently limited the number of time points for GBMT modeling, which is more effective over a longer period of time. 14 In addition, to account for missing data in some cases, we imputed data using the multiple imputation method as outlined in the CSM-P trial’s statistical analysis plan.
There were several other limitations to the current work. Firstly, sagittal alignment and cervical lordosis are known to influence postoperative pain and functional recovery. 40 Nonetheless, these variables were not consistently available across the three clinical trials that our current dataset was compiled from and thus could not be directly analyzed. Future studies integrating radiographic parameters, including pre- and postoperative sagittal alignment, could help clarify the role of spinal alignment in shaping pain trajectories. Additionally, examining the interaction between surgical approach, alignment correction, and postoperative pain recovery may further refine predictive models and improve patient-specific surgical planning.
Moreover, our study did not assess biomechanical factors such as subsidence, implant positioning, or bone quality, which are known to influence postoperative pain outcomes. 41 While cage placement within the disc space can affect stress distribution and the risk of subsidence, retrospective imaging analysis was not feasible in our dataset. Future studies incorporating detailed postoperative imaging and bone quality assessments could help clarify the impact of these factors on pain trajectories and further refine predictive models for surgical outcomes.
In addition, early postoperative pain assessments (i.e., within the first few weeks after surgery) were not available in our dataset, limiting our ability to determine whether patients in the “no improvement” group potentially experienced either an initial worsening of pain or a lack of early postoperative relief. As a result, our findings primarily captured mid- to long-term pain trajectories rather than immediate postoperative pain trends. Future studies incorporating early pain measurements could provide additional insight into the timing and progression of pain recovery, further refining trajectory classifications and their clinical implications.
Lastly, a key challenge in applying trajectory-based pain modeling to clinical decision-making is the variability in DCM etiology and surgical approaches. While our analysis did not incorporate radiological findings, the identified pain trajectories offer valuable insights for patient counseling. Specifically, preoperative pain severity and baseline functional status emerged as critical factors distinguishing recovery patterns. Neurosurgeons can use these data to set realistic expectations for postoperative pain relief, particularly for patients in the “no pain improvement” trajectory, who may require additional interventions such as multidisciplinary pain management. Moreover, integrating trajectory-based risk stratification with radiological and surgical variables in future studies could refine personalized counseling strategies, ultimately enhancing shared decision-making and postoperative care planning.
Conclusion
Three novel neck pain trajectories following surgery for DCM were identified with the use of a patient-centered group-based trajectory analysis, highlighting the heterogeneity of the DCM patient population. Our findings imply the possible presence of subgroups of patients who experience fast, slow, or no improvement in neck pain postoperatively. Furthermore, this work provides empirical data suggesting that when neck pain is the patients’ predominant complaint, they can derive tremendous benefit from surgery when it comes to alleviating pain. However, when pain is not the main issue, it can remain as a residual complaint that either gradually improves or may even stay constant. Neck pain in DCM remains an underrecognized issue; our study suggests that presence of residual pain can potentially impact other measures postoperatively such as quality of life, functional, and neurological outcomes.
Supplemental Material
Supplemental Material - Role of Neck Pain in Defining Clinical Trajectories of Outcomes in Patients With Degenerative Cervical Myelopathy: Results of a Novel Machine Learning Algorithm
Supplemental Material for Role of Neck Pain in Defining Clinical Trajectories of Outcomes in Patients With Degenerative Cervical Myelopathy: Results of a Novel Machine Learning Algorithm by Raymond Wong, Mohammed Ali Alvi, Ayesha Iqbal Quddusi and Michael G. Fehlings in Global Spine Journal
Footnotes
Acknowledgments
We would like to recognize Dr. Jefferson Wilson and Dr. Christopher Witiw for their invaluable assistance.
Author contributions
RW: conceptualization, methodology, access to and verification of the underlying data, formal analysis, validation, visualization, writing-original draft, writing-review and editing. MAA: data curation, validation, methodology, writing-review and editing. AQ: validation, methodology, writing-review and editing. MGF: conceptualization, methodology, data curation, access to and verification of the underlying data, investigation, project supervision, administration, funding acquisition, validation, writing-original draft, writing-review and editing. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
Deidentified data available from corresponding author Dr. Michael Fehlings upon reasonable request –
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
