Abstract
Study Design
Retrospective cohort study.
Objectives
To determine the ideal cage placement position in transforaminal lumbar interbody fusion (TLIF) surgery to prevent vertebral endplate cyst (VEC) formation and improve bone union rates.
Methods
This study retrospectively analyzed 69 patients who underwent TLIF, and 88 intervertebral levels were observed. Patients were categorized into peripheral, central, and mixed types based on the cage placement. The incidence of VEC formation and endplate injuries (EIs) were assessed using computed tomography 1 year postoperatively. Factors associated with VEC formation and non-union, including patient demographics, clinical data, cage placement, Hounsfield unit values, and intraoperative EIs, were analyzed.
Results
VECs occurred in 55.7% of surgical levels and were significantly associated with non-union (P < .001). Center-type cage placement was a strong predictor of VEC formation (P = .028, OR = 16.3, 95% confidence interval [CI]: 1.36-195.0), along with EIs (P = .003, OR = 5.86, 95% CI: 1.80-19.0). In contrast, pre-existing VECs appeared to have a protective effect (P = .046, OR = 0.272, 95% CI: 0.076-0.979).
Conclusions
Cage placement in the central region significantly increases the risk of VEC formation and non-union. Periphery-type placement may be preferable to reduce endplate damage and improve fusion outcomes. Preventing EIs and optimizing cage positioning are critical to minimize VEC formation. Further prospective studies are needed to refine surgical strategies and improve long-term outcomes following TLIF.
Keywords
Introduction
Vertebral endplate cysts (VECs) are fluid-filled sacs that can develop adjacent to the vertebral endplates following spinal surgery, such as in posterior or transforaminal lumbar interbody fusion (PLIF/TLIF). These cysts are often associated with endplate injuries (EIs), micromotion, or mechanical stress at the bone-implant interface.1,2
The formation of VECs can be a complication after spinal fusion surgery, potentially leading to pain, reduced fusion rates, and the need for revision surgery.1-4 Therefore, strategies to prevent the formation of VECs are critical. Wu et al. 5 reported that EIs are particularly likely to occur at the ipsilateral epiphyseal ring and central endplate, with thresholds of 326.21 Hounsfield units (HUs) and 296.42 HUs, respectively, which were identified as risk factors. To avoid such EIs, they emphasized the importance of preoperative assessment of endplate HU values, use of optimal distraction techniques, careful cage placement, and consideration of additional posterior instrumentation.5,6 Based on studies of patients with degenerative disc disease, the association between osteoporosis and the occurrence of EIs suggests that treating osteoporosis could be considered a method to prevent EIs. 7
Based on these considerations, we hypothesized that placing the cage in the peripheral region of the intervertebral disc space or avoiding placement in the center of the disc space, may result in fewer EIs and reduced VEC formation. To date, no study has focused on cage placement location in relation to VEC formation. We, therefore, aimed to investigate the relationship between cage placement location and VEC formation and to determine the optimal cage placement position to prevent VEC formation and improve bone union rates. We then aimed to develop strategies to prevent the occurrence of VECs that may lead to non-union in PLIF/TLIF surgeries, thereby potentially increasing the number of cases where non-union can be avoided in PLIF/TLIF procedures.
Methods
Patient Population
We retrospectively analyzed 83 consecutive patients who underwent one- or two-level TLIF for lumbar degenerative diseases between January 2018 and December 2022. Patients who underwent concomitant laminectomy at other levels were also included. Fourteen patients were excluded because of prior spinal fusion, hemodialysis due to renal failure, severe scoliosis (Cobb angle >30°), or infection (Figure 1). All patients experienced lower back pain, with or without radicular pain that was unresponsive to conservative therapy for more than 3 months. Pain was considered attributable to the following conditions: low-grade lumbar spondylolisthesis (I or II), lumbar disc herniation, or lumbar spinal stenosis. All clinical and radiological interventions followed standard protocols and written informed consent was obtained from all participants. Flowchart for patient selection.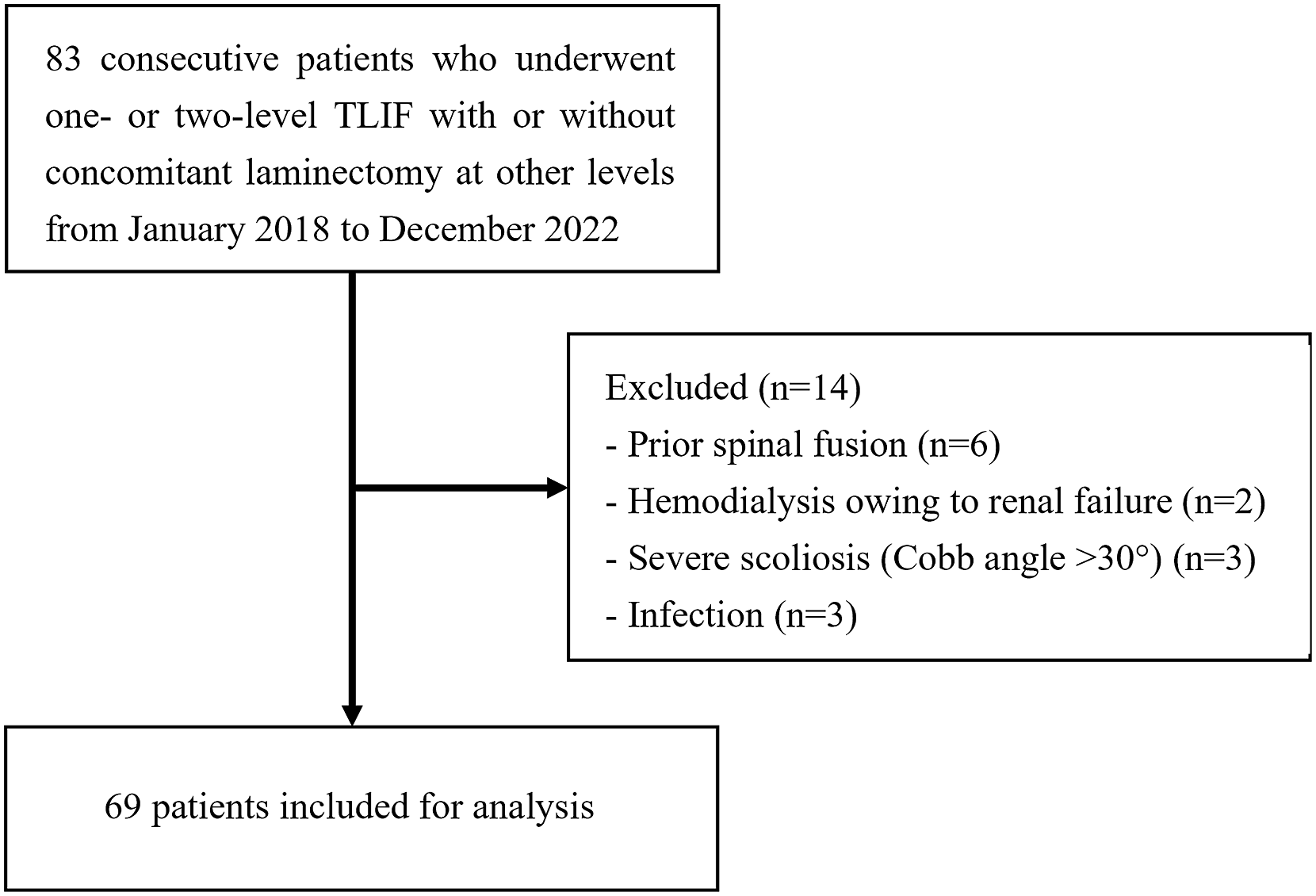
Surgical Procedure
Indications for TLIF included (1) spondylolisthesis with >3 mm of slippage and/or >5° posterior opening on dynamic lateral radiographs; and (2) stenosis requiring total facetectomy for decompression. All patients underwent TLIF with bilateral side grade 2 osteotomy. Following discectomy and endplate preparation on both sides, an autologous bone graft harvested from the posterior spinal elements was packed into the center of the disc space before inserting two identical cages bilaterally. A Tritanium PL cage (Stryker, Mahwah, NJ) packed with an autograft was implanted bilaterally in the disc space. We used two cages of 7-mm, 38 of 8-mm, 104 of 9-mm, and 32 of 10-mm height; the number of cages by angle were four of the 0° cage, 154 of the 6° cage, and 18 of the 12° cage. All cages were 9 mm wide and 23 mm long. Finally, bilateral compression was applied to the disc space using pedicle screw (PS) instrumentation.
Demographic Data
Age at the time of surgery, sex, body mass index (BMI), smoking, 5-item modified frailty index (mFI-5), 8 bone mineral density assessed by the T-score measured by dual-energy X-ray absorptiometry (DEXA) at the proximal femur (T-score), the number and level of PLIF segments, the Japanese Orthopedic Association scoring system (JOA) performed before surgery and at 1 year postoperatively, and osteoporosis treatment were obtained from medical charts and operative notes. The mFI-5 consisted of five variables: (1) history of severe chronic obstructive pulmonary disease, (2) congestive heart failure within 30 d before surgery, (3) functional health status prior to surgery (independent vs partially or totally dependent), (4) hypertension requiring medication, and (5) diabetes mellitus with oral agents or insulin. The combined mFI score was calculated for each patient by adding the number of frailty variables present (one point per variable). The mFI-5 was categorized as 0 (robust), 1 (pre-frail), or 2 (frail). Osteoporosis was diagnosed in cases that met one of the following criteria: (i) history of vertebral or proximal femoral fracture; (ii) history of other fragility fractures and a bone density below 80% of YAM; or (iii) in the absence of a history of fragility fracture, a bone density below 70% of YAM or −2.5 SD. 9 The JOA scoring improvement rate (%) was calculated using the following formula: JOA scoring improvement rate (%) = (JOA score 1 year after surgery - JOA score before surgery) x 100/(29 - JOA score before surgery).
Radiographic Assessment via Computed Tomography (CT) Scans
The CT images were acquired using a Discovery CT750 HD scanner (GE Health care, Tokyo, Japan). Images were processed at our institution using built-in three-dimensional imaging software (Synapse Vincent; FUJIFILM Medical Systems USA, Lexington, MA). Radiological images were examined by two examiners blinded to the clinical outcome. Continuous variables were expressed as the mean of each examiner’s measurements. The presence or absence of a finding was considered as determined by both examiners. This study analyzed CT scans obtained preoperatively, immediately postoperatively, and at the 1-year follow-up for analysis.
VECs
VECs were defined as a visible low-density circular or oval area located at the cephalad and/or caudal vertebral endplate on coronal and/or sagittal computed tomographic images, as reported by Fujibayashi et al. 1 The size of the low-density areas was not considered relevant. CT scans were obtained preoperatively and at the 1-year follow-up for analysis. Patients without preoperative VECs were classified as VECs(+) at 1 year if new VECs developed postoperatively, whereas those with preoperative VECs were classified as VECs(+) if there was evidence of VECs enlargement or the appearance of new VECs at 1 year.
HUs
HUs were measured on both the upper and lower end plates of the treated level on preoperative CT images. The preoperative axial CT image layer used for HU measurement was taken from the subchondral bone of the vertebral endplate at the vertebra directly above and below the disc space scheduled for PLIF, selecting the layer that visualized the largest area of the subchondral bone in the endplate region being measured. The region of interest was manually selected to conform to the endplate contour, excluding the peripheral cortical bone. Intervertebral segments were recorded as where the median HU values of the cephalic and caudal endplates in the measured intervertebral segments were greater than 189.1; the median endplate HU value for the entire study population was defined as HU >190.
Cage Placement
The positions of the cages on both sides were examined using coronal CT images obtained immediately after surgery. Vertebral width was divided into three parts in the central part of the vertebral body. If the cages on both sides were placed in the outer third, they were classified as the periphery type, whereas those placed in the central area on both sides were classified as the center type. If 80% or more of the cage width was contained within the outer third, it was considered the periphery type. Cases in which one side was classified as the periphery type and the opposite side as the center type were defined as mixed type.
EIs and Cage Subsidence
EIs were defined as iatrogenic damage to the cortical endplate detected on immediate postoperative sagittal or coronal CT scans that were absent preoperatively. Cage subsidence was defined as the cage entering the vertebral endplate by ≥ 1 mm. Cage subsidence immediately after surgery was identified as a severe form of EI. CT scans were obtained immediately after the surgery for analysis.
Loosening of PS
PS loosening was defined as a translucent zone measuring >1 mm around the PS. Loosening was assessed using axial, sagittal, and coronal CT images 1 year after surgery.
Union/non-union
Union was defined as the absence of loosening in the four PSs or the achievement of continuity of the grafted bone on CT images 1 year after surgery. Cases that did not meet this criterion were defined as non-union. However, this non-union classification was based on the evaluation at 1 year postoperatively within this study and did not represent the final assessment of non-union as a surgical outcome.
Intra-/inter-observer Reliability
Two spine surgeons independently evaluated EIs and VECs before surgery and at the 1-year follow-up, and non-union on two separate occasions, with a 2-week interval between assessments. Both intra- and inter-observer Cohen’s kappa agreement rates were calculated. In cases where discrepancies were observed between the two examiners, both re-evaluated the mismatched data to reach a consensus. The final dataset used in this study consisted of the concordant data agreed upon by both examiners, including both the initially matching data and the re-evaluated consensus data.
Statistical Analysis
Data are presented as mean ± standard deviation for continuous variables and as numbers and percentages for categorical variables. Statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), a graphical user interface for R (R Foundation for Statistical Computing, Vienna, Austria).
10
The t test was used for continuous variables, and Fisher’s exact test was applied to categorical variables. A P-value <0.05 was considered statistically significant. For categorical variables, the odds ratio and 95% confidence interval (CI) were calculated, while for continuous variables, Cohen’s d and the 95% CI were determined. Univariate analysis was conducted to identify potential predictors of VECs development at 1 year postoperatively. Variables with P < 0.05 in the univariate analysis were included in the subsequent multivariate logistic regression analysis. Cage subsidence was considered to be included in EIs and omitted from the multivariate analysis factors. The multivariate analysis was performed to determine independent risk factors for VEC development. Receiver operating characteristic (ROC) curve analysis was conducted to evaluate the discriminatory ability of the final multivariate model (Figure 2). The area under the curve (AUC) was calculated to assess the predictive performance. An AUC value closer to 1.0 indicates a higher discriminative ability of the model. ROC curve for the three significant risk factors (EIs, center-type cage placement, and no pre-existing VECs).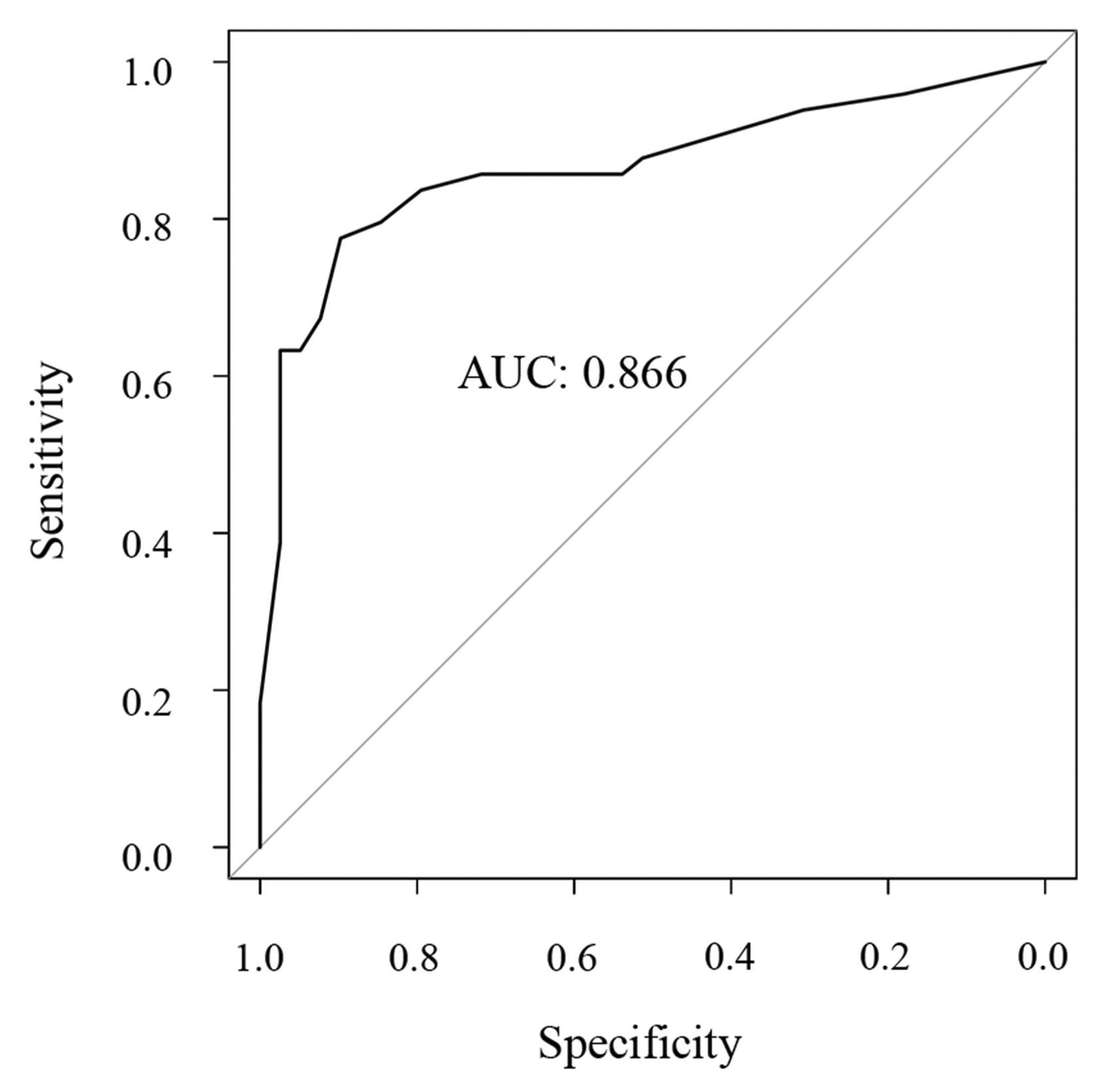
Results
Demographic and Clinical Data of the Patients.
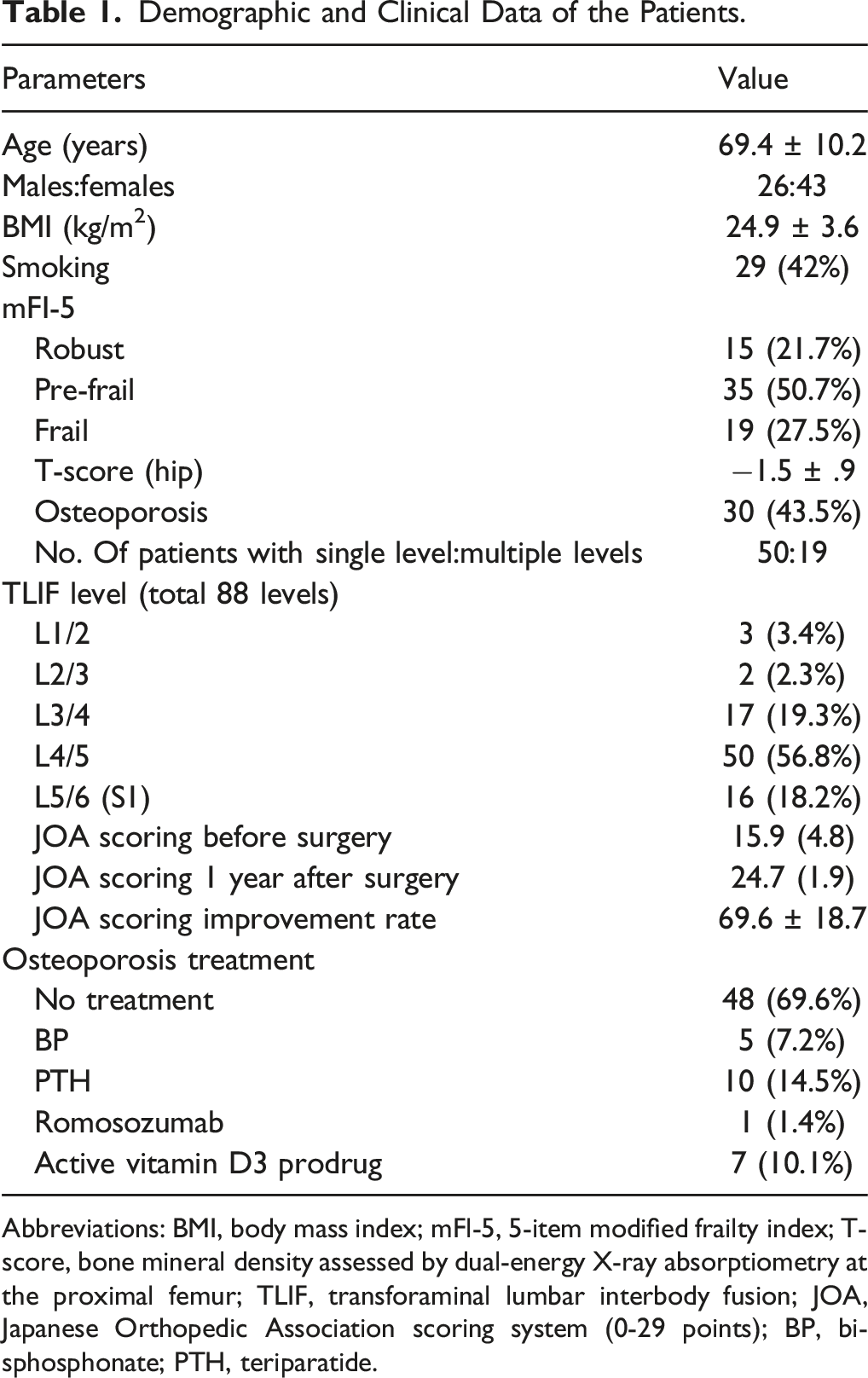
Abbreviations: BMI, body mass index; mFl-5, 5-item modified frailty index; T-score, bone mineral density assessed by dual-energy X-ray absorptiometry at the proximal femur; TLIF, transforaminal lumbar interbody fusion; JOA, Japanese Orthopedic Association scoring system (0-29 points); BP, bisphosphonate; PTH, teriparatide.
Relationship Between VEC and Non-union.
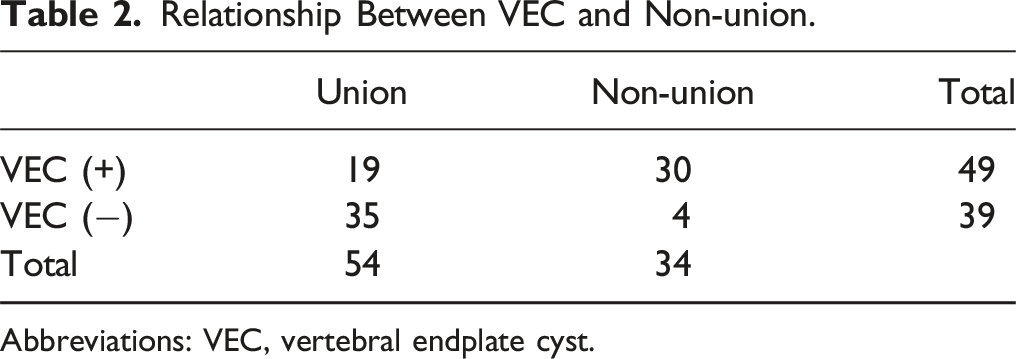
Abbreviations: VEC, vertebral endplate cyst.
Factors Associated With EIs: Osteoporosis, Cage Placement, and Cage Characteristics.
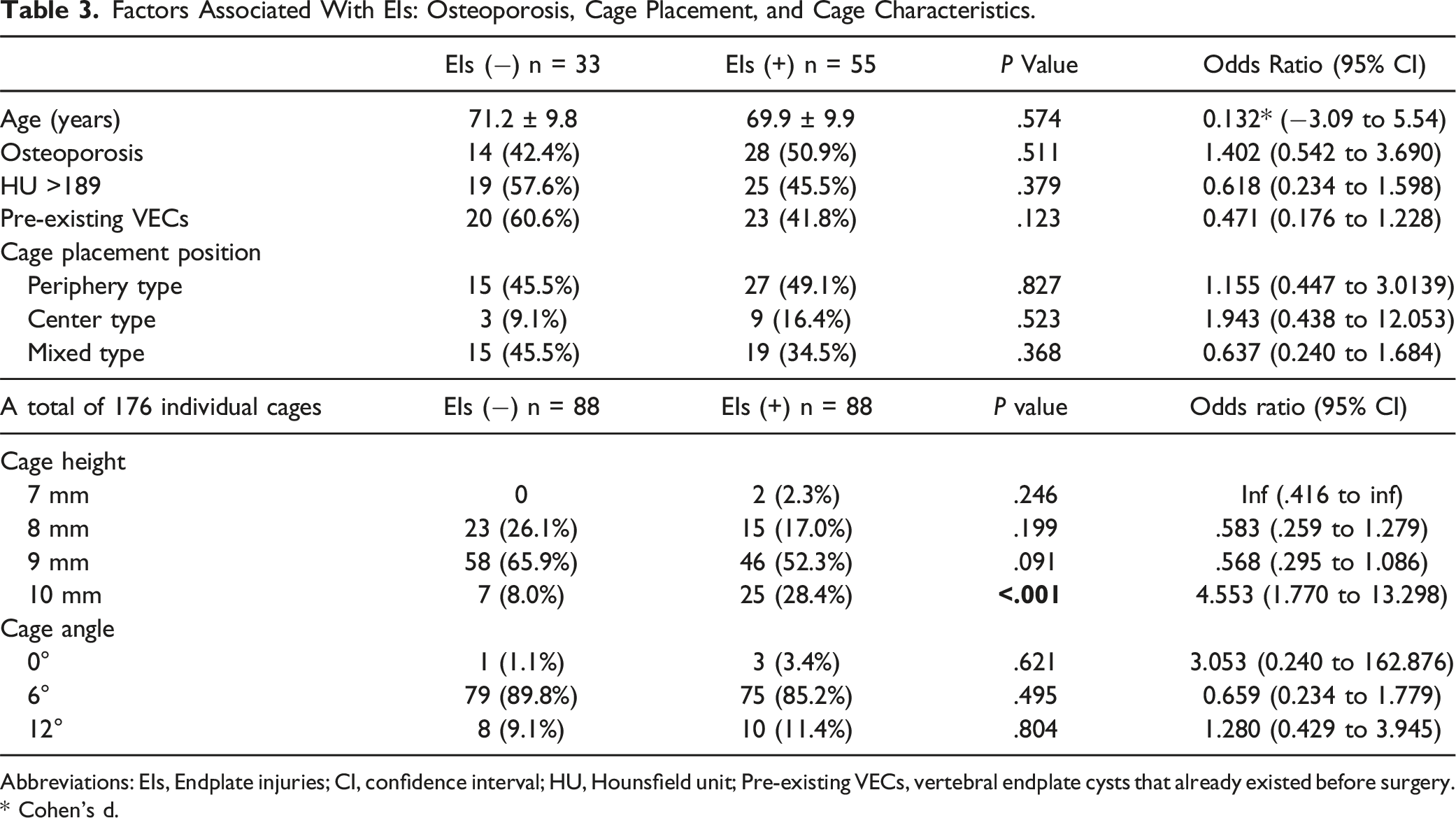
Abbreviations: EIs, Endplate injuries; CI, confidence interval; HU, Hounsfield unit; Pre-existing VECs, vertebral endplate cysts that already existed before surgery. * Cohen’s d.
Univariate Analysis for Predictors of Development of a Vertebral Endplate Cyst at 1 year After Surgery.
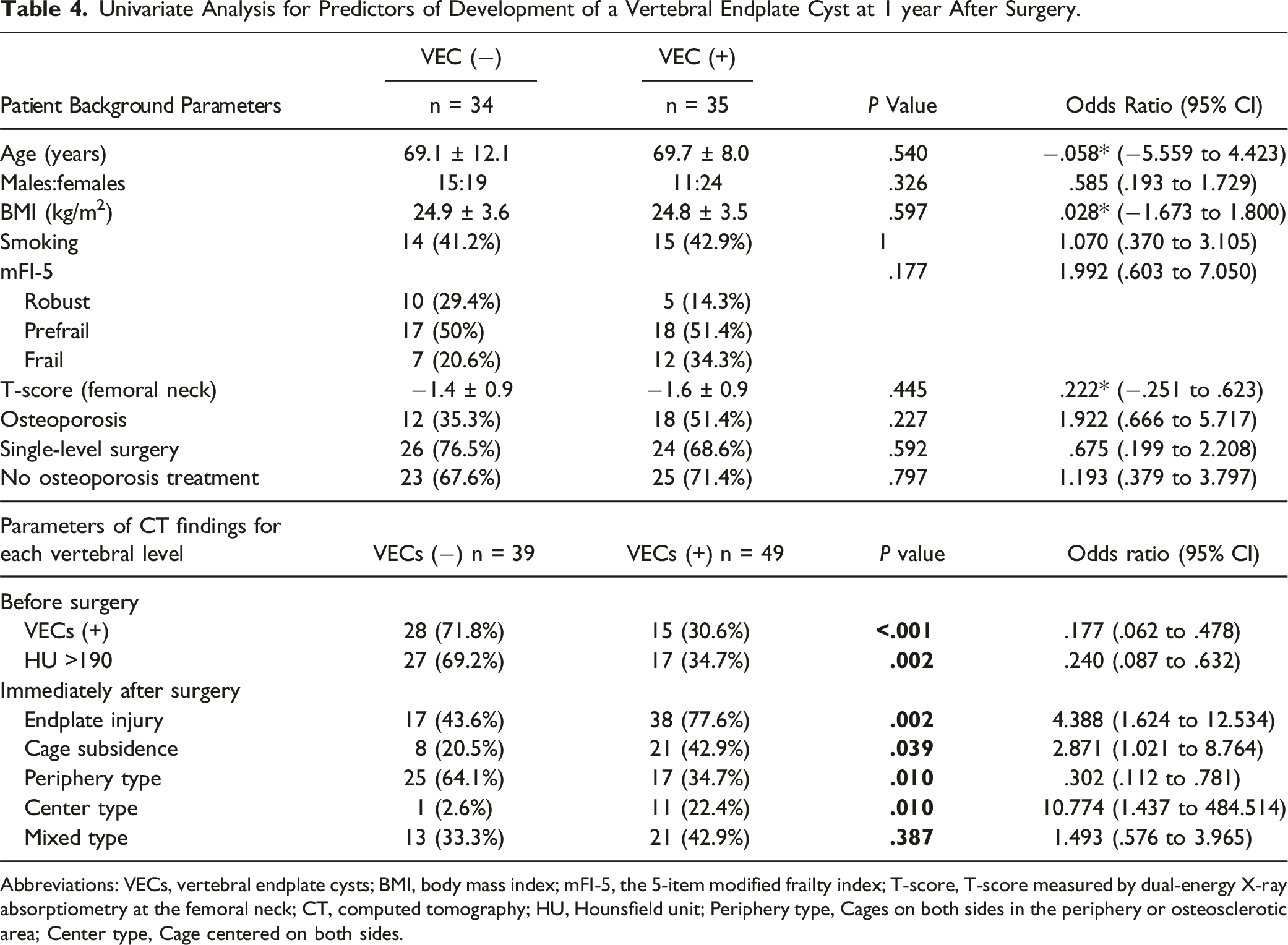
Abbreviations: VECs, vertebral endplate cysts; BMI, body mass index; mFI-5, the 5-item modified frailty index; T-score, T-score measured by dual-energy X-ray absorptiometry at the femoral neck; CT, computed tomography; HU, Hounsfield unit; Periphery type, Cages on both sides in the periphery or osteosclerotic area; Center type, Cage centered on both sides.
According to the analysis of the 88 intervertebral levels that underwent surgery, there were significantly fewer patients with preoperative VECs in the VECs(−) group than that in the VECs(+) group (P < .001). The occurrence of VECs was significantly lower in intervertebral segments where the average preoperative HU values of the cephalic and caudal endplates were greater than 190, the median endplate HU value for the entire study population (P = .002). There were significantly fewer EIs in the VECs(−) group than in the VECs(+) group (P = .002). Fewer cases of cage subsidence were observed in the VECs(−) group than in the VECs(+) group (P = .040). Periphery-type cage positioning was more common in the VECs(−) group than in the VECs(+) group (P = .010). Center-type cage positioning was more common in the VECs(+) group than in the VECs(−) group (P = .010).
Multivariate Analysis for Development of VECs at 1 year After Surgery.
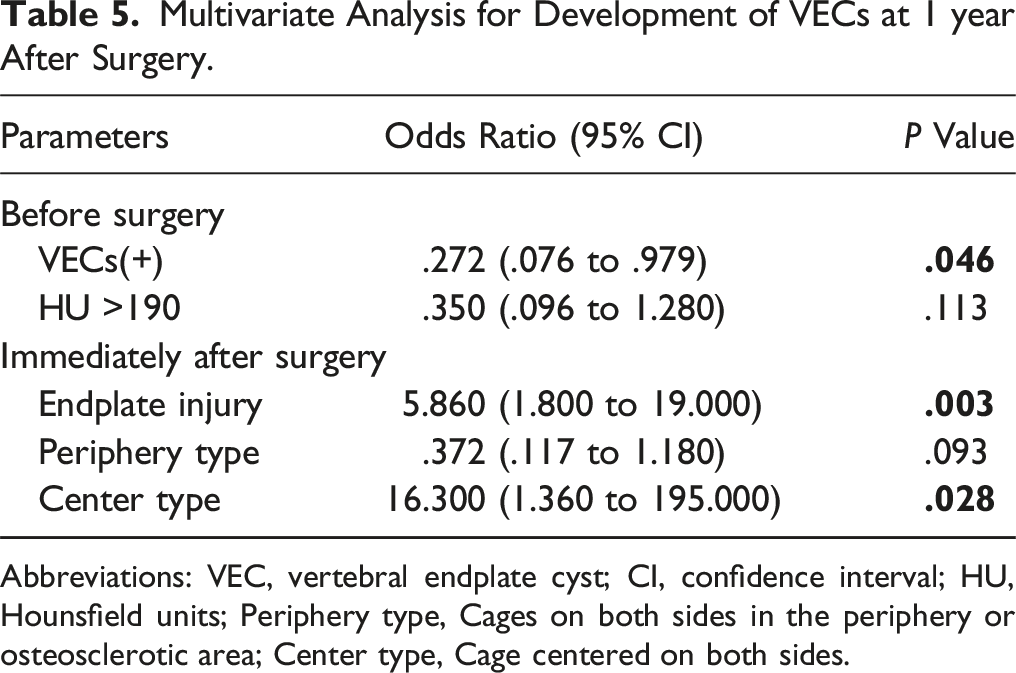
Abbreviations: VEC, vertebral endplate cyst; CI, confidence interval; HU, Hounsfield units; Periphery type, Cages on both sides in the periphery or osteosclerotic area; Center type, Cage centered on both sides.
Figure 3 shows a case of the periphery type, where the union was determined 1 year postoperatively, with the JOA score improving favorably from 20 points preoperatively to 27 points at 1 year postoperatively. The patient was a 77-year-old male with no history of osteoporosis treatment, a T-score of 0.4, and an mFI-5 score of 0, indicating robustness. During the TLIF procedure, cages were placed bilaterally, and their position was classified as periphery type (Figure 3A). No endplate injury was observed, and CT scans 1 year postoperatively showed no formation of VECs around the cages (Figure 3B) or loosening of the PS (Figure 3C), with continuity of trabeculae in the grafted bone between the vertebral bodies (Figure 3B). Periphery-type cage placement without EIs. (A) Coronal CT image immediately postoperatively; the cages on both sides were placed in the peripheral region when dividing the width of the vertebral body into three equal parts, without EIs. (B), (C) Coronal CT images 1 year postoperatively; no VEC formation or cage subsidence was observed around the cages on both sides. There was no loosening of the pedicle screws (within the black circle in (C)), and the continuity of trabeculae in the grafted bone between the vertebral bodies was noted (arrow in (B)).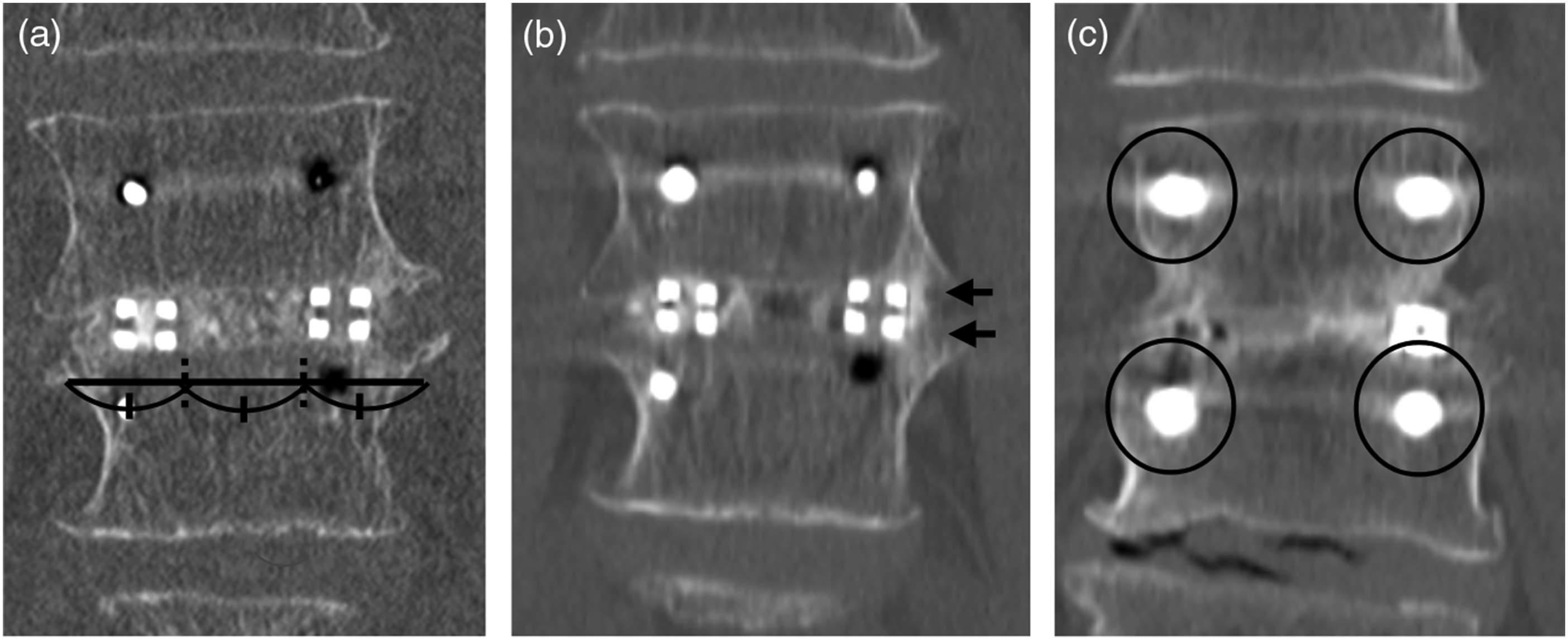
Figure 4 illustrates a case of the center type in which non-union was determined 1 year postoperatively. The patient was a 78-year-old man with no history of osteoporosis treatment, a T-score of −2.0, and an mFI-5 score of 2 points, indicating frailty. During the TLIF procedure, the cages were placed bilaterally, and their positions were classified as the center type (Figure 4A). Endplate injury was observed on the cranial side of the right cage; however, the injury was approximately 1 mm in length (arrow in Figure 4B). One year postoperatively, CT showed the formation of VECs around the cages (Figure 4C), loosening of pedicle screws, and no continuity of trabeculae in the grafted bone between the vertebral bodies, leading to a diagnosis of non-union. The preoperative JOA score was 20 points, and was 22 points at 1 year postoperatively, indicating insufficient symptom improvement. Center-type cage placement with EIs. (A) Coronal CT image immediately after surgery; the cages on both sides were not located in the peripheral region when dividing the width of the vertebral body into three equal parts, thus classified as the center-type cage placement. (B) Coronal CT image immediately after surgery; EIs were observed at the cranial side of the right cage (arrow). (C) Coronal CT image 1 year postoperatively; multiple VECs were observed around both cages (triangles). Loosening of pedicle screws was observed (within the black circle in (C)). The continuity of trabeculae in the grafted bone between the vertebral bodies was not seen.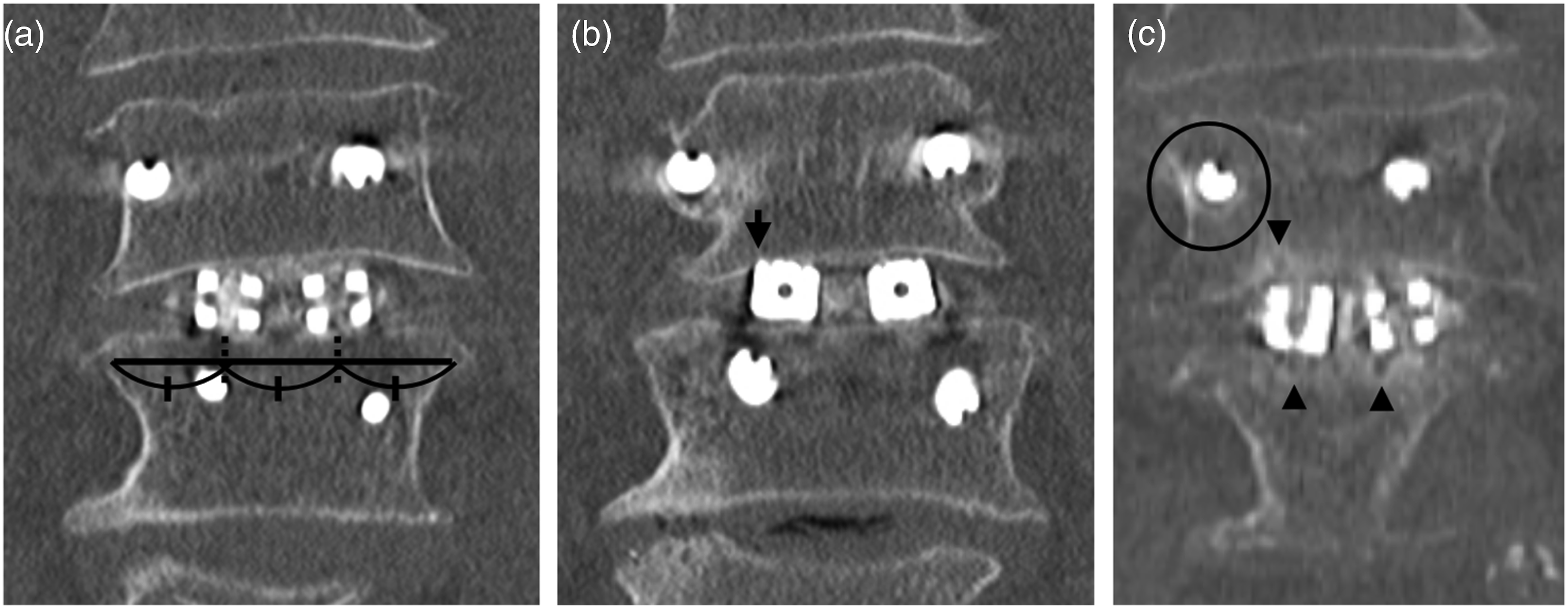
Discussion
This study investigated the relationship between cage placement location and VECs formation and bone union following TLIF surgery. Our findings demonstrated that EIs and center-type cage placement were significant risk factors for the development of VECs 1 year after surgery. Conversely, pre-existing VECs appeared to have a protective effect against new VEC formation. These findings emphasize the importance of proper cage positioning and the minimization of endplate injury to reduce postoperative complications and improve fusion outcomes.
The results of this study indicate that non-union was significantly more frequent at surgical intervertebral levels where VECs were present at 1 year postoperatively. This aligns with previous findings suggesting that VEC formation is associated with micromotion at the bone-implant interface, which can lead to impaired fusion and mechanical instability.1,2,11 Although the positive predictive value (64.8%) was not particularly high, the high sensitivity (88.2%) and negative predictive value (89.7%) observed in this study further support the notion that the absence of VECs correlates with a lower likelihood of non-union. Given that non-union is associated with poorer JOA scores (as shown in Supplemental Table 1) and although the position of the cage placement does not affect the JOA score (Supplemental Table 2), these findings suggest that the prevention of VEC formation is crucial to promote bone union and improve clinical outcomes.
One of the risk factors for the development of VECs was endplate injury. The incidence of endplate damage in TLIF has been reported as 72.06% in previous studies, 12 and the incidence of EIs in this study was 62.5% (55/88 cases), which was considered to be roughly the same. In this study, even minor injuries detectable on CT were classified as EIs, whereas another study, which defined endplate damage as a depression of 2 mm or more, reported an incidence of 8.6%. 13 This suggests that the definition of endplate damage significantly influences the reported incidence rates. The importance of avoiding endplate damage during cage placement is further highlighted by our findings. The occurrence of EIs was not associated with osteoporosis, HU values, existing VECs or cage placement. A 10-mm cage height, not cage angle, was significantly associated with an increased risk of EIs, likely due to excessive stress exerted on the endplate. This is in line with previous reports suggesting that larger cage sizes can lead to greater mechanical stress and increased risk of iatrogenic endplate fractures.5,12,14
Among the factors analyzed, center-type cage placement was found to be an independent risk factor for VEC formation. In contrast, periphery-type placement was associated with a lower incidence of VECs, suggesting that placing the cage in a more peripheral position may be advantageous for preventing VECs and improving bone union. These results are consistent with biomechanical studies indicating that the central region of the endplate is structurally weaker, making it more susceptible to damage and subsidence.15-17 Furthermore, the HU values in the periphery suggest that peripheral regions have greater bone strength, which may contribute to better load distribution and reduced micromotion. 5 No significant difference in the incidence of EIs was HU values (Table 3). However, in this study, HU values of the endplate were not measured separately for the central and peripheral regions. Instead, the reported HU values represent the median across the entire endplate, combining both the central and peripheral regions. As a result, the relationship between HU values at the specific cage placement location and the occurrence of EIs may not have been accurately assessed.
Our findings suggest that careful selection of cage size and optimal placement away from the central region of the endplate may help mitigate the risk of VEC formation and promote successful fusion, even in cases where EIs have already occurred during surgery. Even if EIs occur, in the periphery, the bone formation force surpasses the bone resorption force, which is considered to be related to the occurrence of VECs. As a result, the occurrence of VECs is suppressed, potentially favoring bone fusion. The reason for this is that periphery-type cage placement is considered to be more advantageous for osteogenesis (Wollf’s law) than center-type cage placement, as the stresses from the PS overlap vertically, resulting in better mechanical stress. 18 Periphery-type cage placement may be less prone to VECs and more favorable for bone healing in the event of endplate damage, as it is more likely to achieve moderate bone resorption, osteogenesis, and remodeling.
The process leading to the formation of VECs around the cage placement remains unclear. The proposed hypothesis suggests that micromotion may occur between the cage and the vertebral endplate when the primary stability of the screws or cage is insufficient. These micromotions can cause localized mechanical stress, leading to microfractures in the endplate, which may result in the development of VECs. Similarly, if endplate damage (fracture) occurs during cage placement surgery, it can trigger an inflammatory response and subsequent bone resorption at the site of the endplate injury. When bone damage occurs, inflammation mediated by damage-associated molecular patterns and inflammatory cells develops in the surrounding tissue, 19 and some osteocytes undergo cell death, resulting in necrosis of the bone tissue.20,21 The lacunae of dead osteocytes attract osteoclasts, initiating bone resorption, 22 followed by bone formation as part of the repair process. 23 Once instability occurs in the intervertebral area after surgery, the ongoing bone damage may perpetuate inflammation and bone resorption, potentially leading to VEC formation and subsequent bone loss around the cage. Vertebral endplate damage induces spinal degeneration in animal models.24,25 Preoperative VECs were present at 43 levels throughout the study period. Among them, 15 levels showed new occurrence or enlargement of VECs postoperatively, whereas 28 levels did not exhibit new occurrence or enlargement of VECs. This finding indicates that pre-existing VECs(−) are more likely to develop into postoperative VECs(+). When comparing the HU values of the endplate, pre-existing VECs(+) had higher values (Supplemental Table 3), suggesting that after the formation of VECs, a reparative response might occur, leading to higher HU values indicative of bone sclerosis. Interestingly, pre-existing VECs(+) seemed to suppress the development of postoperative VECs(+). Future studies should explore whether this phenomenon represents an adaptive response to chronic mechanical stress.
Our findings underscore the importance of optimal surgical techniques to prevent VEC formation and improve fusion outcomes. Based on the results of this study, the following recommendations can be made. First, avoid center-type cage placement. Given the strong association between center placement and VEC formation, surgeons should consider positioning cages more peripherally, when feasible. Second, minimize endplate injury. Proper surgical technique, including avoiding excessive distraction forces and selecting appropriate cage sizes, may help prevent iatrogenic endplate fractures. If the cage placement result is center type and EIs also occur, it is considered necessary to restrict postoperative activity, ensure prolonged and strict use of orthotic devices, and administer bone-forming drug therapy. Third, monitor VECs as a potential predictor of non-union. Given the strong correlation with fusion failure, VECs may serve as a radiographic marker for early detection of at-risk patients.
This study had some limitations. First, the retrospective nature thereof may limit the ability to establish causality between specific risk factors, such as cage placement and VEC formation, and outcomes, such as non-union. A prospective randomized controlled trial would provide stronger evidence to confirm these associations. Second, this study was conducted at a single center, which may limit the generalizability of the findings to other settings or populations. Multicenter studies are needed to confirm these results across different clinical environments and patient groups. Third, the sample size may be relatively small, particularly when considering subgroups based on cage placement or VEC status. This limitation may have affected the statistical power to detect significant differences or associations. Fourth, the follow-up period in this study was limited to 1 year postoperatively. The definition of bone union in this study was based on the absence of PS loosening and continuity of the grafted bone. This definition was selected because this study focused on the formation of VECs, which may directly influence the outcome of PS loosening. Additionally, approximately 70% of the study participants had no treatment for osteoporosis; if bone resorption inhibitors or bone formation agents had been administered, they might have influenced the study results. Long-term follow-ups are necessary to clarify the long-term effects of VEC formation on bone union and other clinical outcomes. Fifth, although the study aimed to standardize the surgical procedure, there could still be variations in techniques that might affect the outcomes. For instance, the amount of force used during cage placement or the precision in avoiding endplate injury could vary among surgeons. Sixth, the identification of VECs and EIs was based on CT imaging, which may have limitations in detecting very small or early-stage lesions. Additionally, the study did not employ advanced imaging techniques, such as MRI, which could provide more detailed information about soft tissue changes. Seventh, there might be unmeasured confounding factors, such as variations in bone quality beyond what is captured by HU values or differences in patient activity levels postoperatively, which could influence the outcomes. Finally, HU measurements were obtained from the entire endplate, which may not accurately reflect regional variations in bone quality, especially in areas critical to cage placement. Future studies should consider more localized HU evaluations to improve the accuracy.
Conclusions
This study highlights the significant role of cage placement location and endplate integrity in the formation of VECs following TLIF surgery. Center-type cage placement and endplate injury were identified as independent risk factors for VEC formation, whereas pre-existing VECs appeared to have a protective effect. These findings emphasize the importance of careful cage placement and surgical technique to minimize endplate injury and improve fusion outcomes. Further prospective studies are warranted to validate these findings and refine surgical strategies to optimize patient outcomes.
Supplemental Material
Supplemental Material - Impact of Cage Placement on Vertebral Endplate Cyst Formation and Bone Union in Transforaminal Lumbar Interbody Fusion
Supplemental Material for Impact of Cage Placement on Vertebral Endplate Cyst Formation and Bone Union in Transforaminal Lumbar Interbody Fusion by Chikara Ushiku, Taku Ikegami, Shinji Saito, Hiroki Wakiya, Tomoaki Kanai, Naomu Sawada, and Mitsuru Saito in Global Spine Journal.
Footnotes
Author contributions
CU was responsible for data curation and the writing of the original draft of the manuscript. TI was responsible for the formal analysis, investigation, review, and editing of the manuscript. TK and NS were responsible for formal analysis. SS and HW were responsible for curating the data. MS reviewed and edited the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
