Abstract
Objective
This study was performed to compare fusion rates and clinical outcomes of posterior decompression by posterolateral fusion (PLF) versus transforaminal lumbar interbody fusion (TLIF) for treatment of lumbar spinal stenosis.
Methods
This retrospective cohort study involved 157 patients with lumbar spinal stenosis treated with
Results
The fusion rate was similar between the two groups. Among patients with two- and three-level fusion, improvements in the clinical outcome scores were significantly greater in Group B than Group A.
Conclusion
Combining TLIF with PLF provides better clinical outcomes than PLF alone when multilevel fusion is indicated. TLIF augmentation does not improve the fusion rates in either single- or multi-level surgery.
Keywords
Introduction
Degenerative lumbar spinal stenosis is the most common indication for spinal surgery in patients of advanced age. 1 It is characterized by narrowing of the lumbar spinal canal, lateral recesses, or neural foramina. The resultant pressure on the neural structures causes an array of symptoms including pain, fatigue, and hypoesthesia in the lower extremity and lower back. The chronic nature of the disease has profound effects on social and business life, reducing the patient’s quality of life and eventually restricting his or her activities of daily living. Extensive surgical decompression, which is often required to alleviate symptoms, may lead to instability of the vertebral column. In such cases, instrumented fusion is indicated. 2
Instrumented posterolateral fusion (PLF) is the most commonly used surgical technique to prevent instability following extensive posterior decompression. 3 Spinal fusion is defined as arthrodesis of two adjacent vertebral bodies. It can be achieved by placing a bone graft alongside the rods on decorticated posterior elements as in PLF or by placing graft-loaded cages inside a curetted disc space. Various types of intervertebral cages have been developed to maintain the stability of the fused sections until solid arthrodesis occurs.4,5 Inserting these cages prevents the disc space from collapsing and creates a suitable biomechanical environment for successful fusion.
Achieving solid fusion is vital for a successful outcome. A well fused segment should show successful integration with the surrounding bone tissue through remodeling, function as well as the original tissue, and have sufficient mechanical strength. 6 Failure to achieve any of these characteristics is considered non-fusion. Studies have shown that poorer radiological fusion grades result in worse pain scores in patient outcome questionnaires (visual analog scale [VAS], Oswestry Disability Index [ODI], and 36-Item Short-Form Health Survey [SF-36]).7,8 The risk of non-fusion is higher in surgeries where multilevel arthrodesis is attempted.
In transforaminal lumbar interbody fusion (TLIF), an intervertebral cage is placed between the vertebral bodies through the foraminal space. Approaching the disc space from a more lateral location eliminates the need for rigorous traction of the dura, as in posterior lumbar interbody fusion. The TLIF procedure maintains the decompression of the nerve elements after disc removal, theoretically solves post-decompression instability in patients undergoing laminectomy, and expands the foraminal space.9,10 Augmenting PLF with TLIF reportedly increases fusion and lowers the risk of repeat surgeries in patients with spondylolisthesis. 11
Our clinical experience involving patients with both degenerative disc and lumbar spinal stenosis has shown that patients requiring disc space decompression and interbody fusion in addition to posterior decompression have good clinical outcomes and fusion rates despite the fact that they have a potentially more complicated etiology. In 2015, we began using interbody fusion as an adjunct to instrumented PLF in patients with spinal stenosis without evident disc pathology. In this study, we compared the fusion rates and clinical outcomes between posterior decompression with PLF alone and PLF augmented with TLIF for treatment of lumbar spinal stenosis.
Materials and methods
This retrospective cohort study involved patients with degenerative lumbar spinal stenosis who underwent posterior decompression and instrumented fusion from 2010 to 2018 at the Uludag University Faculty of Medicine Orthopaedics and Traumatology Clinic. The study was approved by the Uludag University Faculty of Medicine Clinical Research Ethics Committee (2011-KAEK-26 Decision reference number: 2019-10/13). Written consent was obtained from all patients enrolled in the study.
Patients with pathological curvature of the lumbar region in the coronal and sagittal planes, patients with a lumbar global alignment and proportion score of >2, patients with dysfunction, and patients who required cemented pedicle screws (T score of >−2.5) were excluded from the study.
All patients available for final assessment were divided into two groups: the PLF group (Group A), in which posterior decompression with instrumented PLF alone was performed, and the cage-augmented group (Group B), in which interbody fusion using TLIF cages was performed in addition to PLF. Six subgroups were created based on the number of fused levels (A1–A3 and B1–B3). The patients’ clinical status before and after surgery was noted. Plain radiographs were used to assess fusion. Long-cassette standing radiographs were used to evaluate the preoperative lumbar lordosis angle. Patients in each subgroup with the same number of fusion levels were compared with each other with respect to demographic factors, operation length, and clinical and radiological outcomes. The effects of the lumbar lordosis angle, age, and surgical technique on the fusion rate were evaluated both cumulatively and for each number of fused segments.
Surgical technique
All patients underwent operations through the posterior surgical approach using the Wagoner technique. 12 Polyaxial pedicle screws were placed into the predetermined spinal levels under fluoroscopic control. The hypertrophic ligamentum flavum was excised and laminectomy was performed for decompression. 13 TLIF cages with autografts were placed inside the disc space following discectomy. Rods of the appropriate size were fixed onto the screws, and grafts were placed on the vertebral segments alongside the rods. When interbody fusion was not used, discectomy was omitted and only PLF was performed. The duration of the operation was noted.
Radiological evaluation
The patients’ preoperative lumbar lordosis angle was measured on long-cassette standing radiographs. The Brantigan–Steffee classification was used to confirm the presence of radiological fusion. 14 This classification describes the status of fusion by grading it from 1 to 5. Grades 1 to 3 indicate non-fusion, and grades 4 and 5 indicate radiographic fusion. For an arthrodesis region to be considered grade 4 or 5, the bone in the fusion area must be denser than that on early postoperative imaging, no space must be present between the cage and the vertebral body, and bone trabeculae must be seen in the fusion area. If one of these three criteria is not met, the patient is considered to have non-fusion. In the Brantigan–Steffee classification, grade 1, 2, and 3 fusions were considered non-fusion and grades 4 and 5 were considered fusion. We used standard radiographic criteria and a numeric scale to interpret the success of radiographic fusion. 15
Titanium and polyetheretherketone cages were used. Titanium cages do not allow for confirmation of bone fusion in the cage because of radiological artifacts. In the present study, when a radiolucency appeared around the cage, we concluded that a gap was present. The presence of a radiolucency-free zone surrounded by a sclerotic region was evaluated on a lateral radiograph and was considered to indicate non-fusion. A bone bridge was defined as radiological fusion of the bone with no other surface or gap, without distortion of the bone density, and with trabecular continuity from the end plate of one vertebra to the end plate of another vertebra (Figure 1(a), (b)).
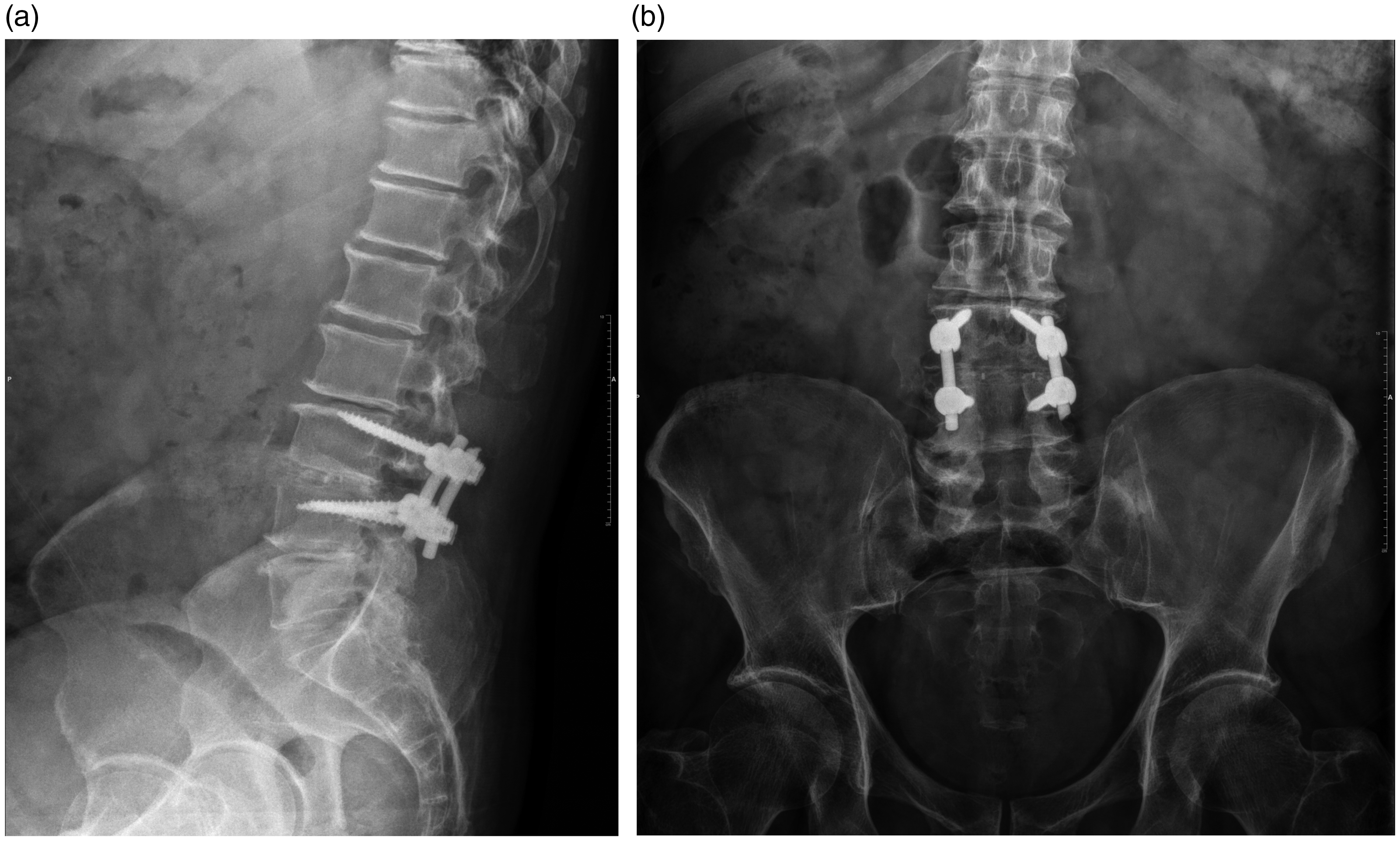
Postoperative radiographs at 1 year after single-level laminectomy with posterolateral fusion augmented with transforaminal lumbar interbody fusion cage. (a) Lateral view. (b) Anteroposterior view.
Although computed tomography was not used as a criterion to show fusion in our study, we considered computed tomography to confirm fusion in patients who exhibited fusion on radiographs (Figure 2(a), (b)).

Postoperative computed tomography scan at 1 year after single-level laminectomy and posterolateral fusion augmented with transforaminal lumbar interbody fusion cage. (a) Anteroposterior view. (b) Lateral view.
Clinical outcome
The ODI, VAS, and SF-36 scores were used to evaluate the clinical outcome.16,17 The percentage change between the preoperative and postoperative scores in each subgroup corresponding to the same number of fusion levels were compared.
Statistical analysis
The Shapiro–Wilk test was used to determine whether the data showed a normal distribution. The Mann–Whitney U test was used to compare non-normally distributed data between the two groups. Fisher’s exact test and the chi-square test were used to analyze categorical data. Logistic regression analysis was performed to evaluate the effect of several factors on fusion rates. The significance level was set at α = 0.05.
Results
Among 175 patients with degenerative lumbar spinal stenosis who underwent posterior decompression and instrumented fusion, 157 patients (115 females and 42 males) were available for the final assessment. The patients’ mean age was 61.6 years (range, 35–81 years). The minimum follow-up duration was 1 year. Seventy-three patients underwent PLF alone and were included in Group A, whereas 84 patients required TLIF augmentation (Group B). The numbers of patients who underwent each procedure and the operative times in the subgroups according to the numbers of fused segments are outlined in Table 1.
Effect of surgical technique and number of fused segments on operation length.

TLIF, transforaminal lumbar interbody fusion; PLF, posterolateral fusion.
There was no significant difference in the fusion rate between the two groups regardless of the number of fusion levels. Both surgical procedures yielded similar radiological outcomes with single-, two-, or three-level fusion. The fusion rate in each subgroup is outlined in Table 2.
Comparison of fusion rates between PLF and TLIF with same number of fused segments.

TLIF, transforaminal lumbar interbody fusion; PLF, posterolateral fusion.
The lumbar lordosis angle, age, and surgical technique did not affect the fusion rate regardless of the number of fused segments (Table 3). Combining PLF with TLIF did not cause a significant increase in the operation time for all levels of fusion (Table 1).
Effect of age, lumbar lordosis angle, fusion type, and fusion levels on fusion rates.

Improvement in the ODI and VAS score of patients with single-level fusion was significantly greater in the PLF group (Group A1) than in the cage-augmented group (Group B1) (p < 0.05). No significant difference was observed between two groups in the percentage change in SF-36 scores except for the “restriction due to physical problems” domain, in which patients with a TLIF cage performed better (p < 0.05) (Table 4).
Patient-reported outcomes of two surgical techniques for single-level fusion.

Data are presented as mean (range).
TLIF, transforaminal lumbar interbody fusion; PLF, posterolateral fusion; ODI, Oswestry Disability Index; VAS, visual analog scale; SF-36, 36-Item Short-Form Health Survey.
Improvement in the ODI and VAS scores of patients with both two- and three-level fusion was significantly greater in the cage-augmented subgroups (Group B2 and B3) than in the PLF subgroups (Groups A2 and A3) (p < 0.05). There was no significant difference in the percentage change in SF-36 scores between the subgroups (Tables 5 and 6).
Patient-reported outcomes of two surgical techniques for two-level fusion.

Data are presented as mean (range).
TLIF, transforaminal lumbar interbody fusion; PLF, posterolateral fusion; ODI, Oswestry Disability Index; VAS, visual analog scale; SF-36, 36-Item Short-Form Health Survey.
Patient-reported outcomes of two surgical techniques for three-level fusion.

Data are presented as mean (range).
TLIF, transforaminal lumbar interbody fusion; PLF, posterolateral fusion; ODI, Oswestry Disability Index; VAS, visual analog scale; SF-36, 36-Item Short-Form Health Survey.
Discussion
Degenerative lumbar spinal stenosis is the most common indication for spinal surgery among patients 65 years of age. Decompressing the stenotic segments often requires sacrificing important osseous and connective tissues that are essential for biomechanical stability. The resultant instability must be surgically addressed, and several methods including TLIF and PLF have been described. Without proper fusion, spinal implants alone do not provide adequate biomechanical stability, causing persistent pain and loss of disc space height.
Despite the high number of surgeries performed for spinal stenosis, few studies have compared the fusion rates of these procedures. Many of these studies compared one fusion technique with another, 18 and the studies that focused on simultaneous use of PLF and interbody fusion generally involved patients with spondylolisthesis with single-level interbody fusion. 19
Successful intervertebral fusion after TLIF is defined as formation of a bone bridge across the disc space and an increase in bone density without radiolucency between the cage and the vertebral body. 20 McAfee 21 emphasized that the presence of a bone bridge is the most important parameter in measuring fusion success. Similarly, in our study, all patients who underwent TLIF and achieved successful fusion had a bone bridge evident in all surgical segments. Kim et al. 22 reported that 35% of patients with fusion following posterior lumbar interbody fusion had achieved formation of a bone bridge around the cage at 12 months. In about 82% of these patients, the bone fusion had extended to the posterior cortex of the vertebra 4 years after the procedure. Our study focused on TLIF, and literature describing the radiological outcome of this procedure is scarce. Kim et al. 22 showed that bone remodeling is a continuous process and that the shape of the bone bridge constantly changes throughout time. Our study involved a follow-up period of 8 years but did not have standard follow-up times for each patient. Therefore, for patients with a short follow-up period, whether fusion will develop in the future remains unclear.
We found no significant difference between the fusion rates of the two groups regardless of the fusion levels. Both surgical procedures yielded similar radiological outcomes for single-, two-, or three-level fusion. This is contrary to the results of Macki et al., 11 who reported increased pseudarthrosis in patients with isolated PLF. A direct comparison with their study may not be conclusive because they included only patients with spondylolisthesis and single-level fusion. Such patients often require reduction of the slipped segment, and the fused segment is subjected to greater loads. Similar to our study, Høy et al. 23 reported no difference in fusion rates in patients with a broader spectrum of degenerative lumbar diseases who underwent operations on multiple segments.
Our results indicate that augmenting PLF with single-level TLIF has no additional benefits over PLF alone. In fact, these patients had worse clinical outcomes than patients who underwent PLF alone. This is similar to the report of Macki et al., 11 who reported worse clinical outcomes when PLF was combined with TLIF for patients with single-level fusion, although the difference did not reach statistical significance.
This tendency was reversed with increased numbers of segments, for which combined multilevel fusion yielded better clinical outcomes than PLF alone. This is contrary to most of the studies in the literature, which reported similar outcomes with the two techniques.11,18,19,23–25 A direct comparison with most of these studies may not be conclusive because they included only patients with spondylolisthesis, performed single-level interbody fusion even when multiple levels were decompressed, or limited the PLF to one side. The studies by Høy et al. 23 and Divi et al. 25 are methodologically closest to ours; however, they included patients with degenerative disc disease, listhesis, spinal stenosis, or failed back surgery, whereas we included only patients with spinal stenosis with no prior surgery. Furthermore, although they decompressed multiple segments, they did not perform an analysis based on the number of fused segments, whereas we stratified the outcome based on the number of fused segments. The more uniform patient selection criteria and separate analysis by stratification of fused segments during the statistical analysis might have helped us to detect the difference in the patient-reported outcome measures between the two surgical techniques in our study.
Adding TLIF cages to PLF yielded better ODI and VAS scores. This may have been due to the instability tending to increase in parallel with the extent of decompression. Even when PLF occurs, it may not be enough to prevent micromotion in the facet joints and disc spaces in an extensively decompressed spine. In such cases, the benefits of additional stabilization using interbody fusion may outweigh the downsides of prolonged surgery and a more invasive approach.
The main strength of our study is the relatively uniform group of patients despite a large sample size. We included only patients with degenerative spinal stenosis, and there was no baseline age difference between the study groups. As patients’ age and preoperative morbidities increase, surgeons tend to prefer PLF over TLIF because of its technical ease, resulting in younger patients in the TLIF group. 25
Limitations of our study include its retrospective design, substantial loss to follow-up, use of different cages, and possible effects of confounding variables such as osteoporosis, infection, smoking, hormonal imbalance, corticosteroid or nonsteroidal anti-inflammatory drug use, and chemotherapy. Another limitation is that we did not have a postoperative computed tomography scan of each patient, which could have yielded different results than the Brantigan–Steffee classification. Additionally, using a historical cohort (PLF alone) resulted in longer follow-ups, which might have caused bias in clinical outcomes.
Conclusion
Augmenting PLF with TLIF improves patient-reported outcome measures in multilevel decompression despite no difference in fusion rates. This finding is partly contradictory to previous reports in the literature, which yielded similar results with different fusion techniques. We believe that this is due to the heterogeneity of the study designs.
Supplemental Material
sj-pdf-1-imr-10.1177_0300060520910025 - Supplemental material for Augmenting posterolateral fusion with transforaminal lumbar interbody fusion cage improves clinical outcome, but not fusion rate, of posterior decompression
Supplemental material, sj-pdf-1-imr-10.1177_0300060520910025 for Augmenting posterolateral fusion with transforaminal lumbar interbody fusion cage improves clinical outcome, but not fusion rate, of posterior decompression by Teoman Atici, Selcan Yerebakan, Cenk Ermutlu and Ali Özyalçın in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
