Abstract
Study Design
Retrospective cohort study.
Objectives
This study seeks to highlight differences in 1) postoperative complications, 2) recovery course, and 3) associated costs between patients with and without postoperative dysphagia who underwent single-level CDA.
Methods
The National Inpatient Sample (NIS) was queried to identify patients who underwent single-level CDA between 2016 and 2020. Patients were divided into dysphagia and control groups based on the presence or absence of postoperative dysphagia. Propensity score-matching (1:5) was performed, accounting for age, sex, race, and comorbidities. Primary outcomes included length of stay (LOS), hospital costs, and discharge disposition. Chi-square and t-tests were used for statistical comparisons. Significance was set at the P < .05 level.
Results
The final analysis included 640 patients in the dysphagia group and 3,200 controls after matching. Patients in the dysphagia group experienced a significantly higher rate of perioperative complications (7.8% vs 2.8%; P = .006). The mean LOS was longer for the dysphagia group (2.9 ± .3 days vs 1.5 ± .1 days; P < .001), and hospitalization costs were significantly higher ($27,100 vs $21,700; P < .001). Additionally, dysphagia patients were more likely to have a non-routine discharge (15.6% vs 9.2%; P = .030).
Conclusions
Postoperative dysphagia following CDA significantly escalates healthcare resource utilization, leading to prolonged hospital stays, increased costs, and a greater risk of non-routine discharge. This underscores the need for targeted interventions to reduce the incidence of dysphagia and improve recovery outcomes, ultimately enhancing patient care and reducing the financial burden on healthcare systems.
Keywords
Introduction
Dysphagia remains one of the most common complications following procedures of the cervical spine. Although transient in most cases, postoperative dysphagia has been associated with a worsened health-related quality of life for patients, underscoring the need for its prevention and treatment in the clinical decision-making process.1,2 A study conducted by Lee et al. found that dysphagia was common throughout the two-year recovery period following cervical spine surgeries, with rates declining from over 50% at one month to around 13% at two years. 3 The authors also noted that gender, revision surgeries, and multilevel surgeries were the main drivers of long-term dysphagia during the postoperative period. These findings emphasize the enduring impact of dysphagia on recovery from cervical spine surgery and highlight the importance of recognizing patient-specific risk factors to provide effective spinal care.
As the field of spine surgery transitions towards the incorporation of more motion-preserving technologies that limit the onset of adjacent segment disease, procedures like cervical disc arthroplasty (CDA) have gained significant favor among both physicians and patients.4,5 While anterior cervical discectomy and fusion (ACDF), the long-established standard of care, has faced a plateau in utilization, CDA has experienced a 654.24% increase in use over the past decade, reflecting a shift in the management of cervical spine disorders. 6 Compared to ACDF, CDA has been associated with improved functional outcome scores, success rates, less symptomatic adjacent segment disease, and fewer secondary surgeries. 7 In addition, CDA has been associated with decreased rates of dysphagia. A meta-analysis conducted by Zhong et al. highlights this trend, demonstrating significantly lower rates of dysphagia in patients undergoing CDA compared to those receiving ACDF. 8 Despite this improvement, the literature recognizes dysphagia as a common occurrence across all cervical spine procedures and calls for studies to determine the postoperative effects of dysphagia following CDA, given the complication’s substantial challenges to recovery following spine surgery. 8 A study conducted by Altman et al. utilized the National Hospital Discharge Survey to evaluate the co-occurrence of dysphagia with common comorbid conditions. 9 The authors found that the presence of dysphagia during the postoperative period for patients diagnosed with an intervertebral disc disorder significantly increased mortality rate. Early detection and treatment of dysphagia was advised to decrease length of stay in these patients and improve the prognosis of their recovery. Given these findings, further research is warranted to comprehensively characterize the postoperative course for patients who develop dysphagia following CDA, especially in light of the growing adoption of motion-preserving technologies in cervical spine surgery.
Despite extensive research on dysphagia rates following CDA, a comprehensive characterization of the postoperative course in patients with dysphagia remains limited. Therefore, this study seeks to address this gap by utilizing propensity score matching to highlight differences in 1) postoperative complications, 2) recovery course, and 3) associated costs between patients with and without postoperative dysphagia who underwent CDA. We hypothesize that patients who develop postoperative dysphagia after CDA will experience significantly higher rates of perioperative complications, longer hospital stays, and increased healthcare costs compared to those without dysphagia.
Methods
Data Collection
The National Inpatient Sample (NIS) database was accessed for analysis and merged with the respective cost-to-charge ratio database from January 2016 through December 2020. The NIS database, sponsored by the Healthcare Cost and Utilization Project (HCUP) under the Agency for Healthcare Research and Quality, is the largest publicly available all-payer inpatient healthcare database designed to produce U.S. regional and national estimates of inpatient utilization, access, cost, quality, and outcomes. 10 The database samples 20% of all inpatient encounters from U.S. acute-care hospitals representing over 95% of the U.S. population. 10 In all analyses, weighted estimates were derived using HCUP’s discharge weights. Institutional Review Board (IRB) approval was not required, and informed consent was deemed exempt due to the use of a publicly available, de-identified dataset.
Patient Population
Patients who underwent CDA were selected using the International Classification of Diseases, 10th Revision (ICD-10) procedural codes 0RR307Z, 0RR30JZ, and 0RR30KZ. Patients who underwent multi-level CDA were identified and subsequently excluded based on the presence of multiple ICD-10 procedural codes for CDA during the admission period. Patients under the age of 18 were excluded. Cohorts were assigned based on the presence of dysphagia vs the absence of dysphagia (control) as a perioperative complication. Dysphagia was identified in the NIS database using the ICD-10-CM diagnosis code R13.1. Additionally, non-elective patients and those with missing data for sex, race, income quartile, insurance payer, hospital bed size, teaching hospital/location status, total costs, and length of stay (LOS) were excluded. Discharge disposition was classified as routine (patients who went home), non-routine (patients sent to a short-term hospital, skilled nursing facility, intermediate care facility, or home with health care services), and other (patients leaving against medical advice, died in hospital, or unknown destination). Patients with “other” discharge disposition were excluded.
Variable Selection
Independent variables collected included age, sex, race, hospital bed size, teaching hospital/location status, region, charges, and LOS. Charges were converted to costs using the cost-to-charge ratios provided by the cost-to charge ratio database. Since data from multiple years were used, all cost data were adjusted for inflation to reflect 2020 US dollar values using specific weights. 11 Patient comorbidities were identified using the Elixhauser comorbidity software refined for ICD-10-CM provided through HCUP, identifying 38 different pre-existing conditions based on secondary diagnoses. 12
Primary Outcome Variables
The primary outcomes analyzed included postoperative complications such as acute post-hemorrhagic anemia, wound disruption, surgical site infection, mechanical complication, hematoma, nervous system complication, acute deep vein thrombosis, myocardial infarction, cerebrovascular accident, venous thromboembolism, pneumonia, acute kidney injury, sepsis, and anesthesia-related complications. Dysphagia was excluded from this list of perioperative complications, as it was the defining characteristic of the experimental group in this analysis, serving as the basis for comparison. Additional outcomes included LOS, total hospital cost of admission, and discharge disposition.
Statistical Analysis
For this analysis, 1:5 propensity score-matching was performed between cohorts based on age, sex, race, and the Elixhauser Comorbidity Index (ECI). Statistical analyses were conducted using R statistical software (version 4.4.0; R Project for Statistical Computing, Vienna, Austria). Chi-square and Student’s t-tests were employed to compare differences in categorical and continuous outcomes between age groups, respectively, with statistical significance set at the P < .05 level.
Results
Demographics
Baseline Demographic Characteristics of Patients Prior to Propensity Score-Matching.
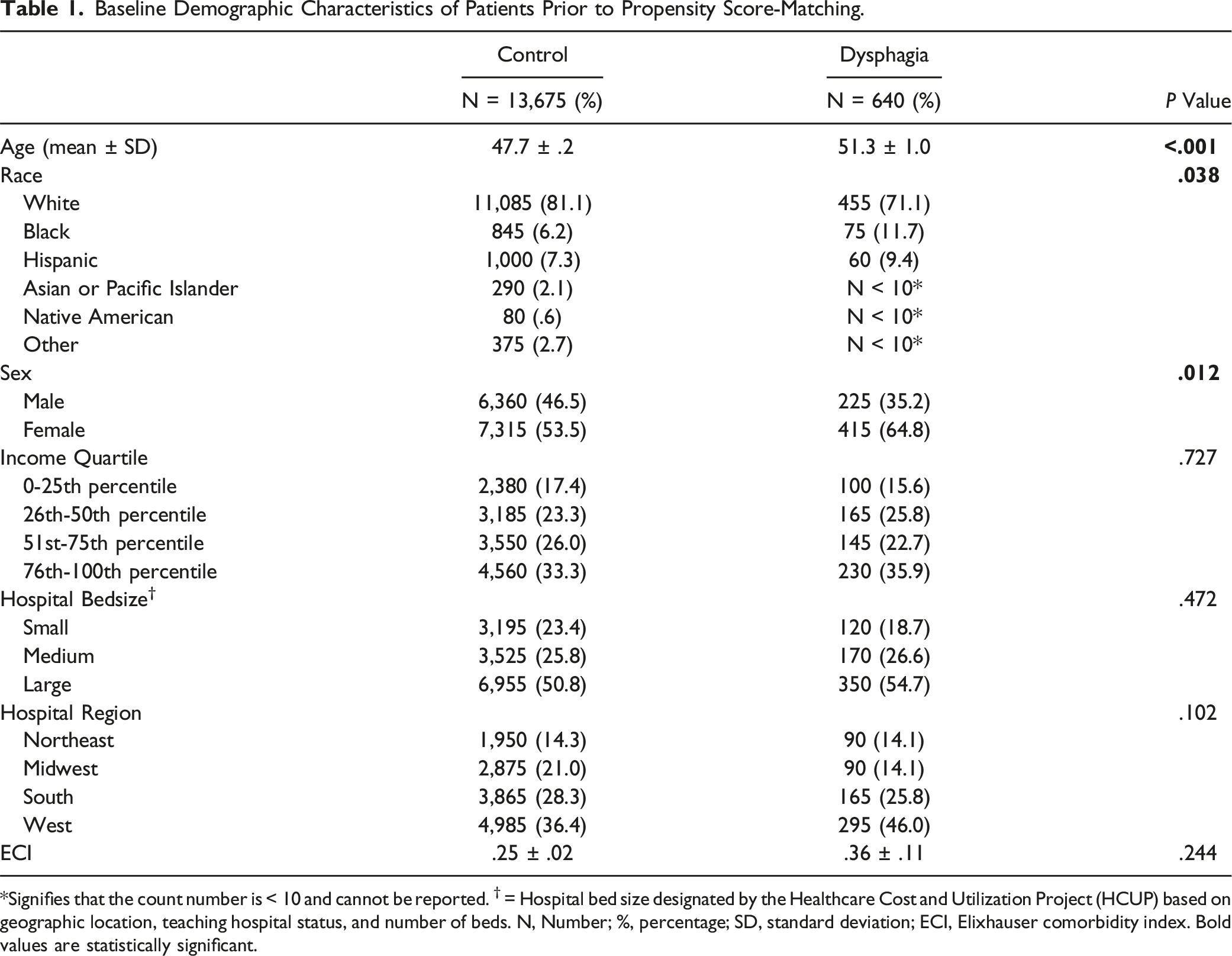
*Signifies that the count number is < 10 and cannot be reported. † = Hospital bed size designated by the Healthcare Cost and Utilization Project (HCUP) based on geographic location, teaching hospital status, and number of beds. N, Number; %, percentage; SD, standard deviation; ECI, Elixhauser comorbidity index. Bold values are statistically significant.
Matched Demographic Characteristics of Patients After Propensity Score-Matching.
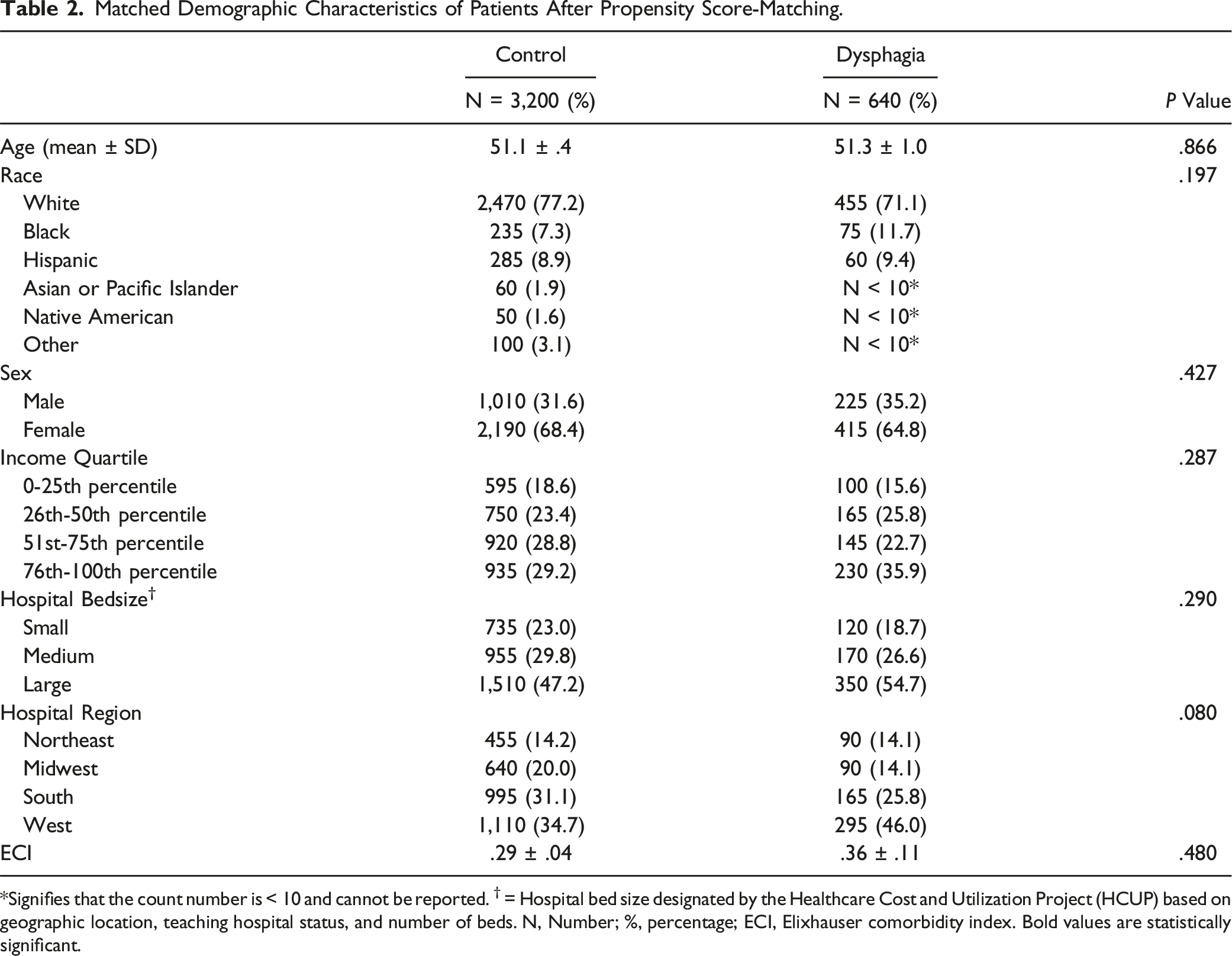
*Signifies that the count number is < 10 and cannot be reported. † = Hospital bed size designated by the Healthcare Cost and Utilization Project (HCUP) based on geographic location, teaching hospital status, and number of beds. N, Number; %, percentage; ECI, Elixhauser comorbidity index. Bold values are statistically significant.
Perioperative Complications
Comparison of Complications and Hospital Course Between Cohorts After Propensity Score-Matching.
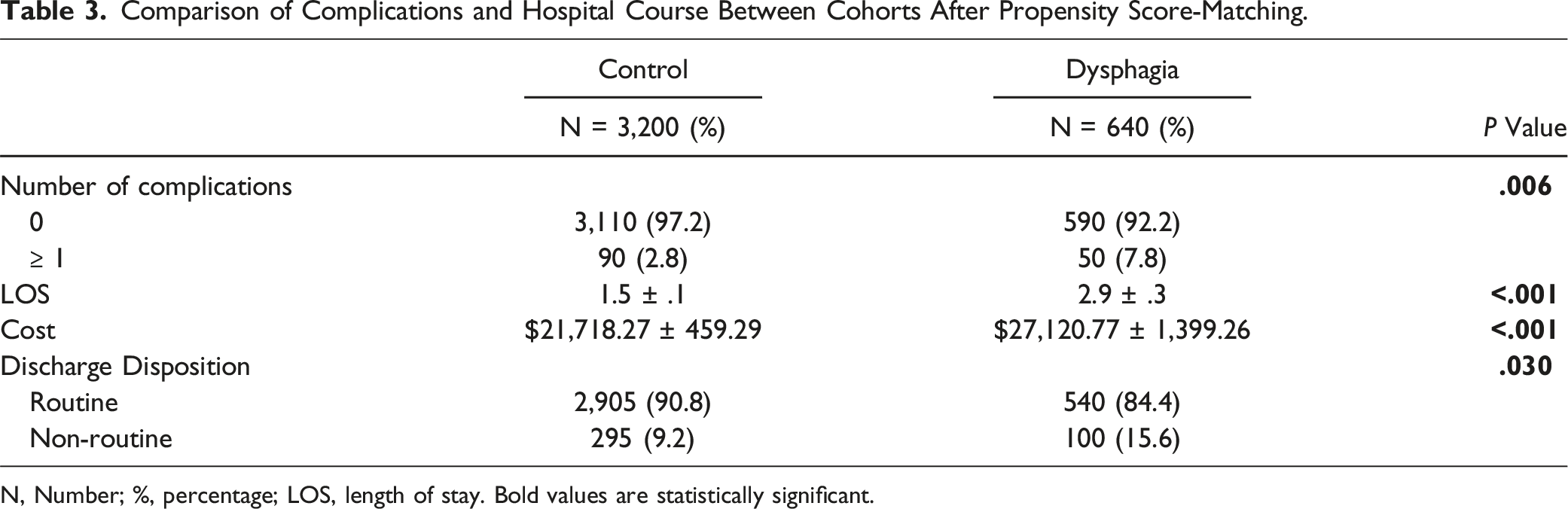
N, Number; %, percentage; LOS, length of stay. Bold values are statistically significant.
Hospital Course
The LOS was significantly longer for patients in the dysphagia group, averaging 2.9 ± .3 days compared to 1.5 ± .1 days for the control group (P < .001). Similarly, the cost of hospitalization was substantially higher in the dysphagia group, with an average cost of $27,100 compared to $21,700 for the control group (P < .001). Additionally, discharge disposition differed significantly between the two groups, with dysphagia patients more likely to be discharged non-routinely (15.6% vs 9.2%, P = .030; Table 3).
Discussion
Dysphagia remains a significant postoperative complication following procedures of the cervical spine, adversely impacting the recovery process. 13 Early detection and careful preoperative screening have been advised throughout the literature to ensure that patients receiving cervical spine care have an optimal prognosis.14,15 Although increasingly utilized motion-sparing procedures like CDA reduce postoperative dysphagia rates, the postoperative effects of dysphagia in this patient population have been poorly characterized.8,16 Therefore, this study aimed to address this gap by using propensity score matching to compare the postoperative course of patients who developed dysphagia after CDA with those who did not. Patients who underwent CDA and developed postoperative dysphagia were significantly associated with having one or more additional postoperative complications, longer hospital stays, higher rates of non-routine discharge, and increased healthcare costs compared to those without dysphagia.
Patients who developed dysphagia following CDA were significantly more likely to experience one or more additional postoperative complications compared to those without dysphagia. Dysphagia has been well-documented in the literature as a factor that increases the likelihood of related complications, including aspiration, aspiration-induced pneumonia, malnutrition, dehydration, and even increased mortality.17,18 Anterior cervical spine approaches can often cause mechanical damage to surrounding soft tissue, potentially injuring the recurrent laryngeal nerves and contributing to postoperative prevertebral thickness, both of which can lead to dysphagia and its related complications. 18 These procedures also require significant intraoperative retraction of soft tissue to achieve proper visualization for completing the discectomy and placing an adequately sized artificial disc or spacer. 2 Consequently, the nature of the anterior approach predisposes patients to an increased incidence of dysphagia. 19 Although standardized methods to reduce dysphagia onset have not been established, research suggests that mean surgical retraction time is a significant factor contributing to higher rates of dysphagia. 2 In addition, numerous studies have proposed parameters surrounding intervertebral distraction for predicting dysphagia. A retrospective study conducted by Yi et al. analyzed a group of 289 patients who underwent anterior cervical spine surgery at a single institution. 20 The authors identified an optimal distraction cutoff of 6.10 mm for predicting dysphagia one week after surgery. The authors suggested that intervertebral distraction at or above this threshold was independently associated with early dysphagia. Ultimately, spine surgeons may reduce the incidence of dysphagia in patients undergoing CDA by minimizing retraction time intraoperatively, releasing retractors when possible, and employing a careful surgical approach to avoid mechanical damage to soft tissue and neural elements, while closely monitoring postoperative distraction parameters.
Dysphagia was associated with significantly longer hospital stays and increased rates of non-routine discharge in patients undergoing CDA. Throughout the literature, dysphagia has been recognized as a notable risk factor for increased hospitalization lengths.13,21-23 As patients with dysphagia face challenges with swallowing, managing proper nutrition becomes a critical concern. 24 Treatment protocols often include postural adjustments, sensory input enhancement, swallowing maneuvers, and diet modification. 25 In cases where dysphagia persists following cervical spine surgery, hospital discharge to rehabilitation centers may become necessary, as reflected in this study’s findings. 26 This is further supported by literature that identifies length of stay as a major predictor of non-home discharge across the field of spine surgery.27,28 A multidisciplinary approach, involving surgeons, speech therapists, dietitians, and specialized nursing care, is essential for managing dysphagia in these patients. Early involvement of speech-language pathologists can facilitate the development of individualized swallowing therapies, reducing the need for prolonged hospital stays and non-routine discharges. 29 Studies also emphasize that early detection and prevention of dysphagia are key factors in avoiding extended hospitalizations.9,30 Among the preventative measures discussed, perioperative local retropharyngeal steroid administration to reduce soft-tissue swelling is frequently recommended.21,31,32 Ultimately, in order to decrease the length of stay and reduce the rate of non-routine discharge in patients undergoing CDA, early recognition of dysphagia is essential, and all preventative and treatment strategies should be considered to optimize patient outcomes.
Patients with postoperative dysphagia incurred significantly higher healthcare costs following CDA compared to those without dysphagia. Throughout the literature, increased costs have been documented for patients who develop dysphagia postoperatively after undergoing cervical spine surgery. 33 By prolonging hospital stays and increasing the rate of non-routine discharges, dysphagia directly contributes to higher healthcare expenses. A study conducted by Starmer et al. analyzed 1,649,871 patients who underwent anterior cervical discectomy of fewer than four vertebrae using the NIS database. 34 The authors highlighted that dysphagia was a significant predictor of healthcare costs, as well as increased length of hospitalization. These findings align with the results of this study, showcasing the substantial impact dysphagia has on both hospital resource utilization and patient outcomes across various cervical spine procedures. The financial burden of dysphagia in CDA patients is significant, as extended hospital stays, specialized care, and the need for non-routine discharges to rehabilitation centers or skilled nursing facilities all contribute to higher costs. Furthermore, the increased incidence of dysphagia places staffing demands on hospital systems, as they are required to add additional specialized care and resources, such as speech therapists, nutritional support, and extended nursing services, to ensure adequate postoperative care. Given these significant economic and resource implications, early detection and prevention of dysphagia are essential for improving patient outcomes and alleviating the strain on healthcare systems.
Although this study has several strengths, it is important to acknowledge certain limitations. The retrospective design limits our ability to establish a causal relationship between dysphagia and postoperative outcomes following CDA. Additionally, while the NIS database provides a broad dataset, it is susceptible to coding inaccuracies and lacks detailed clinical information, such as the severity of dysphagia or specific treatment interventions. Notably, only the diagnosis of dysphagia, rather than its degree, could be captured, limiting the study’s ability to differentiate between mild and severe cases. Furthermore, the database does not specify whether dysphagia was primarily pharyngeal or esophageal in origin, limiting insights into the underlying mechanisms contributing to prolonged hospitalization. Operative factors such as the laterality of the surgical approach (right vs left) and the specific cervical level (eg, C3–4 vs C6–7) are also not available in the NIS, limiting our ability to assess their potential impact on dysphagia risk. The exclusion of multi-level CDA cases may also restrict the generalizability of the results to more complex surgical scenarios. Despite the use of propensity score matching to minimize confounding, unmeasured variables, such as preoperative swallowing function or other comorbidities, may still influence the findings. Lastly, the study focuses on short-term outcomes and does not account for long-term complications or functional recovery, both of which are critical for understanding the full impact of dysphagia on patients undergoing CDA. Future research should aim to address these limitations by incorporating prospective data with long-term follow-up and a more detailed range of patient and procedural factors. Additionally, preventive and management strategies, such as the use of intraoperative steroids, minimizing retraction time, and smaller cervical plate designs, could be considered for further study to reduce the risk of postoperative dysphagia.
Conclusion
While CDA offers a motion-preserving alternative to fusion procedures and has been associated with lower overall rates of dysphagia compared to ACDF, it is clear that postoperative dysphagia remains a significant complication for a subset of patients. In order to optimize outcomes for these individuals, early recognition and intervention are crucial. Preventative strategies, such as perioperative local retropharyngeal steroid administration to reduce soft-tissue swelling, have shown promise in minimizing the incidence and severity of dysphagia. Additionally, a multidisciplinary approach, involving speech therapists, dietitians, and specialized nursing care, should be standardized to provide comprehensive care for patients who develop this complication. By implementing these evidence-based strategies, healthcare providers can improve recovery outcomes, reduce hospital stays, and lessen the economic burden associated with dysphagia following CDA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclosure
Mitchell K. Ng is a paid consultant at Pacira BioSciences Inc., Sage Products Inc., Alafair Biosciences Inc., Next Science LLC, Bonutti Technologies Inc., Johnson & Johnson Ethicon Inc., Hippocrates Opportunities Fund LLC, and Ferghana Partners Inc. Christopher K. Kepler has the following disclosures: Clinical Spine Surgery (editorial or governing board), Curetiva (IP royalties), and Regeneration Technologies, Inc. (IP royalties). All other authors have no other relevant financial disclosures.
