Abstract
Study Design
Retrospective Cohort Study.
Objective
Evidence on chemoprophylactic anticoagulation efficacy in preventing venous thromboembolism (VTE) after spine surgery for metastasis is scarce. We hypothesized that early heparin administration (0-2 days post-surgery) significantly reduces VTE risk post-arthrodesis. We also examined VTE incidence and risk over 2 months and severe postoperative bleeding (SPOB).
Methods
We conducted a retrospective cohort study using the TriNetX network, identifying adults who had a spinal arthrodesis for spinal metastasis in the past 20 years. Patients were grouped by anticoagulation use: a heparin cohort and a non-anticoagulated control. After propensity matching for key VTE risk factors, we examined VTE risk ratios (RR) with 95% confidence intervals (CI) up to 2 weeks post-surgery, and incidence and RR of VTE and SPOB over 2 months.
Results
There were 847 patients per cohort after matching. There was no statistically significant difference in incidence of VTE in the heparin group compared to the no anticoagulation cohort through 2-weeks’ (4.0% vs 2.7%; RR: 1.48; 95% CI: 0.88, 2.49; P = 0.1383) and 2 months’ follow-up (9.4% vs 7.9%; RR: 1.2; 95% CI: 0.88, 1.63; P = 0.2619). Furthermore, there was no statistically significant difference in incidence of SPOB in the heparin group compared to the no anticoagulation group at 2-weeks’ (1.5% vs 1.7%; RR: 0.93; 95% CI: 0.44, 1.96; P = 0.8462) and 2-months’ follow-up (2.8% vs 2.5%; RR: 1.1; 95% CI: 0.64, 2.04; P = 0.6504).
Conclusion
Early heparin administration did not significantly affect VTE or SPOB rates compared to controls. Future research should explore whether other anticoagulants lower VTE risk post-spinal metastasis fusion.
Introduction
There is no consensus regarding the use of chemoprophylactic anticoagulation after spine surgery to prevent venous thromboembolism (VTE) among individuals with spinal metastasis.1-5 In general, chemoprophylactic anticoagulation conveys a potential benefit of preventing VTE and its associated morbidity and mortality; however, there is also a concern regarding the potential increased risk of severe postoperative bleeding (SPOB), such as epidural hematoma.2-4 The effectiveness of chemoprophylactic anticoagulation has been examined more extensively for spine surgery among individuals without cancer6-8 as compared to patients undergoing surgery for spinal metastasis, a population with an even greater baseline risk of VTE. 9 Accordingly, there remains a gap in the literature regarding chemoprophylactic anticoagulation in the context of surgical management of spinal metastasis, thereby limiting surgeons’ ability to make an evidence-informed decision regarding the use of this therapy.6,10
While the incidence of VTE after spine surgery in the absence of metastasis is relatively low (3%), 7 the incidence among those with spine metastasis is substantially greater, ranging from 8% to 11%.9,11,12 Of the current 3 publications which examine VTE and/or SPOB occurring after spine surgery for metastasis, only a single study has examined the effectiveness of chemoprophylactic anticoagulation in mitigating VTE.9,11,12 In this small cohort study (n = 65), adults receiving delayed anticoagulation had a greater probability of VTE compared to those receiving early anticoagulation (≤3 days postoperative), with an odds ratio of 6.43 (P = 0.049). 11 While this study suggested that anticoagulation may be beneficial in these patients, the study was not adequately powered to examine SPOB or extensively control for confounding variables affecting thrombogenesis, and as a single study, warrants corroboration.
The currently limited evidence regarding the relative effectiveness and safety of chemoprophylactic anticoagulation for spine surgery for metastasis has hindered spine surgeons’ ability to predict and mitigate the risk of VTE and SPOB. Given the limited existing data on this topic, we sought to compare the incidence and risk of VTE and SPOB among individuals undergoing spine surgery for metastasis treated with chemoprophylactic anticoagulation (ie, heparin) vs controls without anticoagulation after spine surgery.
Materials and Methods
Study Design
This retrospective cohort study examined the risk of VTE and SPOB after spine arthrodesis surgery for spinal metastasis with heparin vs no anticoagulation and adhered to a protocol registered on Open Science Framework (https://osf.io/mnuv2/). The University Hospitals Institutional Review Board ([IRB] Cleveland, OH, US) considers study designs which use de-identified data from TriNetX in accordance with standard operating procedures to meet criteria for Not Human Subjects research, thereby making the present study exempt from IRB review and waiving the need for consent. Our data query was performed on April 10, 2024.
Setting and Data Source
Data for this study was be obtained from TriNetX, a United States research network based on electronic health records of academic medical centers and their affiliated outpatient offices, which includes over 125 million patients across 89 health care organizations. The database may be queried using standardized nomenclature such as International Classification of Disease codes, 10th edition (ICD-10) and Current Procedural Terminology codes (CPT), among others (eg, RxNorm for medications). ICD-10 codes are interconverted automatically by the software to ninth edition as needed. TriNetX is compliant with the Health Insurance Portability and Accountability Act (HIPAA), the US federal law which protects the privacy and security of health care data. TriNetX is certified to the International Organization for Standardization 27,001:2013 standard and maintains an Information Security Management System to ensure the protection of the health care data it has access to and to meet the requirements of the HIPAA Security Rule. Any data displayed on the TriNetX Platform in aggregate form, or any patient-level data provided in a data set generated by the TriNetX Platform, only contains de-identified data as per the de-identification standard defined in Section §164.514(a) of the HIPAA Privacy Rule. The process of de-identifying data is attested to through a formal determination by a qualified expert as defined in Section §164.514(b) (1) of the HIPAA Privacy Rule. The TriNetX network contains data provided by participating health care organizations, each of which represents and warrants that it has all necessary rights, consents, approvals, and authority to provide the data to TriNetX under a Business Associate Agreement , so long as their name remains anonymous as a data source and their data are utilized for research purposes. The data shared through the TriNetX Platform are attenuated to ensure that they do not include sufficient information to facilitate the determination of which health care organization contributed which specific information about a patient.
This study also employed the use of TriNetX’s built-in natural language processing function software (Averbis, Freiburg im Breisgau, DE), which has been previously validated against manual chart review.13-15 This function screens clinical chart text and radiology notes for the presence or absence of terms used in our selection criteria and outcomes and understands context and negation of such terms.
Participants
Adults (≥18 years old) who underwent first-time spine surgery for spinal metastasis from 3 months to 20 years prior to the database query, did not have VTE within the prior 6 months, and did not have an inferior vena cava filter were divided into 2 cohorts based on anticoagulation administration within 2 days before or after of spine surgery (Supplemental Table 1). Patients in the heparin group needed to be given heparin on the same day or up to 2 days after spinal surgery due to the inherent short half-life of heparin. 16 Patients in the control group could not have received any of a broad range of anticoagulation medications up to 2 days before or after spinal surgery. Both cohorts were required to have a diagnosis of metastatic disease co-occurring on the same date of spinal arthrodesis surgery. As a measure to improve data completeness, we required all patients to have had platelet and glucose measurements within 6 months preceding the surgery date.
Variables
To minimize bias, we used propensity score matching to control for confounding variables present within 20 years up to inclusion and associated with an increased risk of VTE. Matched variables included coagulation defects, coagulation-modifying medications prior to inclusion/surgery, demographics (ie, age at index, female/male, ethnicity), heart disease, nicotine dependence status, overweight or obese status, diabetes mellitus, end stage renal disease, remote history of VTE (>6 months), use of chemotherapy, insertion of central venous catheter, relevant laboratory findings (ie, platelets and glucose), and markers of disease complexity and immobility (eg, bed confinement status and dependence on a wheelchair).
Outcome
Our primary composite outcome included several diagnosis codes indicative of VTE following spine surgery for spinal metastasis, including those affecting the upper and/or lower extremities, renal vein, unspecified veins, and pulmonary embolism (Supplemental Table 2). VTE diagnoses in this study relied on ICD-10 codes and validated NLP algorithms to identify relevant terms in clinical and imaging reports. However, this approach may not detect subclinical VTE events, representing a limitation of the dataset. Our outcome assessment window commenced the day following surgery and was assessed at 2 weeks and 60 days to assess the impact of heparin anticoagulation. A secondary outcome regarding safety was examined regarding SPOB, a feared complication after surgery. This study is subject to certain limitations inherent to the data available in the TriNetX network. Specifically, granular information regarding the dose, duration, and precise timing of heparin administration beyond the defined inclusion window (up to 2 days before or after spinal arthrodesis) was not available. Variability in these treatment factors, which may influence VTE risk or SPOB, could not be accounted for in this analysis. Similarly, neurological function—an important clinical parameter that may affect both the indication for anticoagulation and the risk of thrombotic or bleeding complications—was not accessible due to the de-identified nature of the dataset.
Statistical Analysis
Initial statistical analysis was completed using the built-in statistical suite within the TriNetX network software. For comparisons of patient baseline demographics, this study use an independent-samples t test or Pearson chi-squared test. We used standardized mean difference (SMD) to evaluate meaningful between cohort differences with a threshold of SMD >0.1. Logistic regression was used to calculate the propensity scores for patients in each of the 2 cohorts. This was followed with a propensity score density graph to visually compare the scores before and after propensity matching. We calculated risk ratios (RR) with 95% confidence intervals (CIs), evaluating significance at P < 0.05, and incidence of our outcome with 95% CIs. In addition, we used R (version 4.2.2, Vienna, AT) and ggplot2 to calculate incidence with 95% CIs and plot cumulative incidence as a sensitivity analysis.
Study Size
A required total sample size of 638 patients per cohort was calculated after reviewing data from the prior publications regarding risk of VTE after spinal surgery for spinal metastasis.9,11,12,17 Using GPower (Kiel University, DE) with the z-testing function, we powered the study to test for a difference in incidence between cohorts of 4.0% (hypothesized as 5.0% in the heparin group and 9.0% in the control group), using an allocation ratio of 1, two-tailed α-error of 0.05, and power of 0.80.
Results
Patients
Baseline Characteristics Before and After Matching.
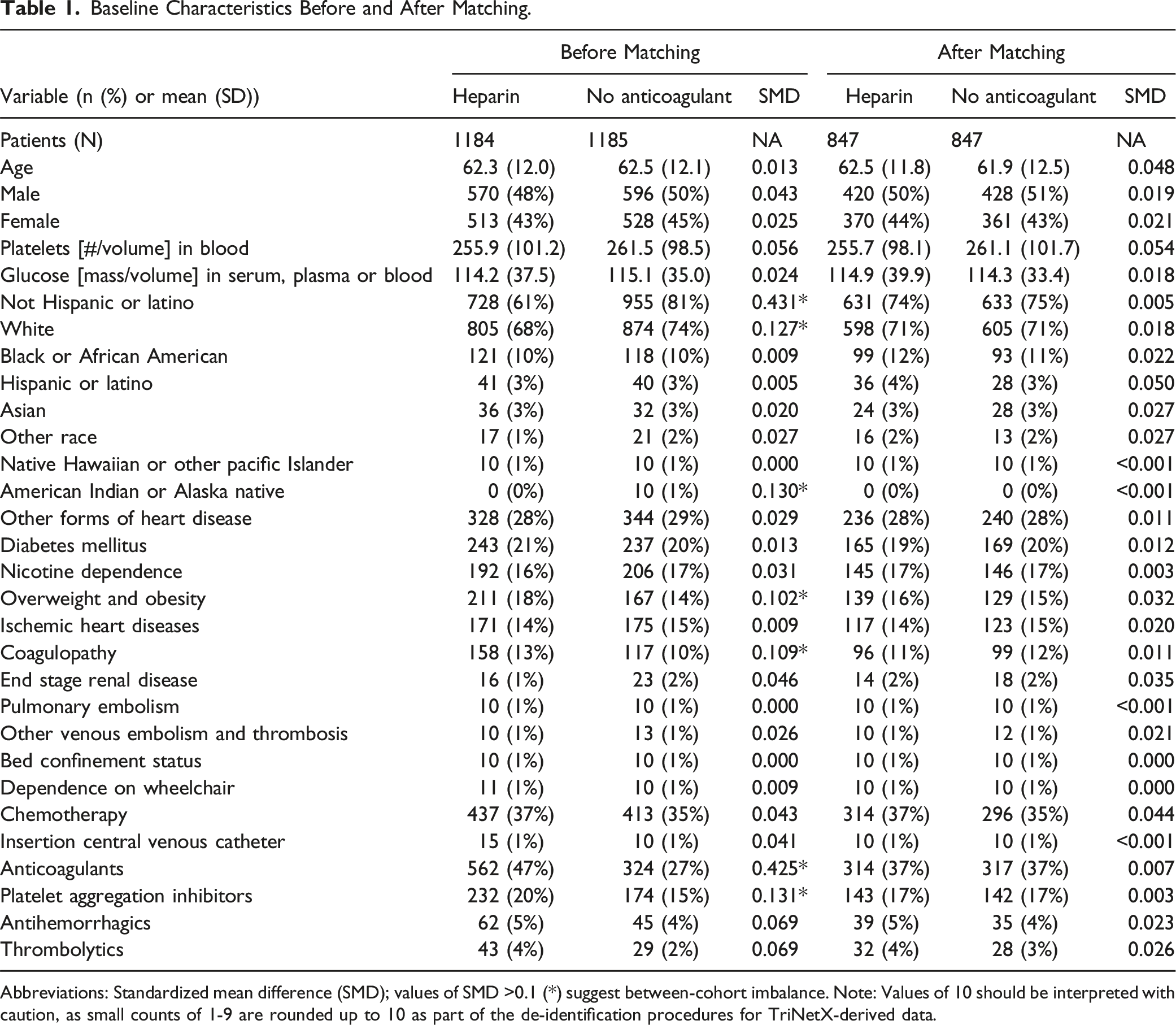
Abbreviations: Standardized mean difference (SMD); values of SMD >0.1 (*) suggest between-cohort imbalance. Note: Values of 10 should be interpreted with caution, as small counts of 1-9 are rounded up to 10 as part of the de-identification procedures for TriNetX-derived data.
Descriptive Data
There was an adequate mean number of data points per patient in each cohort: 7084 in the heparin cohort and 6859 in the no anticoagulation cohort. After propensity matching, there were similar proportions of unknown variables (SMD <0.1) per cohort including unknown sex (7% per cohort; SMD = 0.005), unknown ethnicity (21% in the heparin cohort vs 22% in the no anticoagulation cohort; SMD = 0.017), and unknown race (13% per cohort; SMD = 0.004). Adequate covariate balance was further highlighted by a propensity score density plot (Figure 1). Propensity score density plot. Density distributions of cohorts’ propensity scores before (A) and after (B) matching are shown. Orange shading represents the heparin cohort, while blue represents the no anticoagulant cohort. After matching, propensity score distributions overlap closely, indicating adequate balance of key covariates.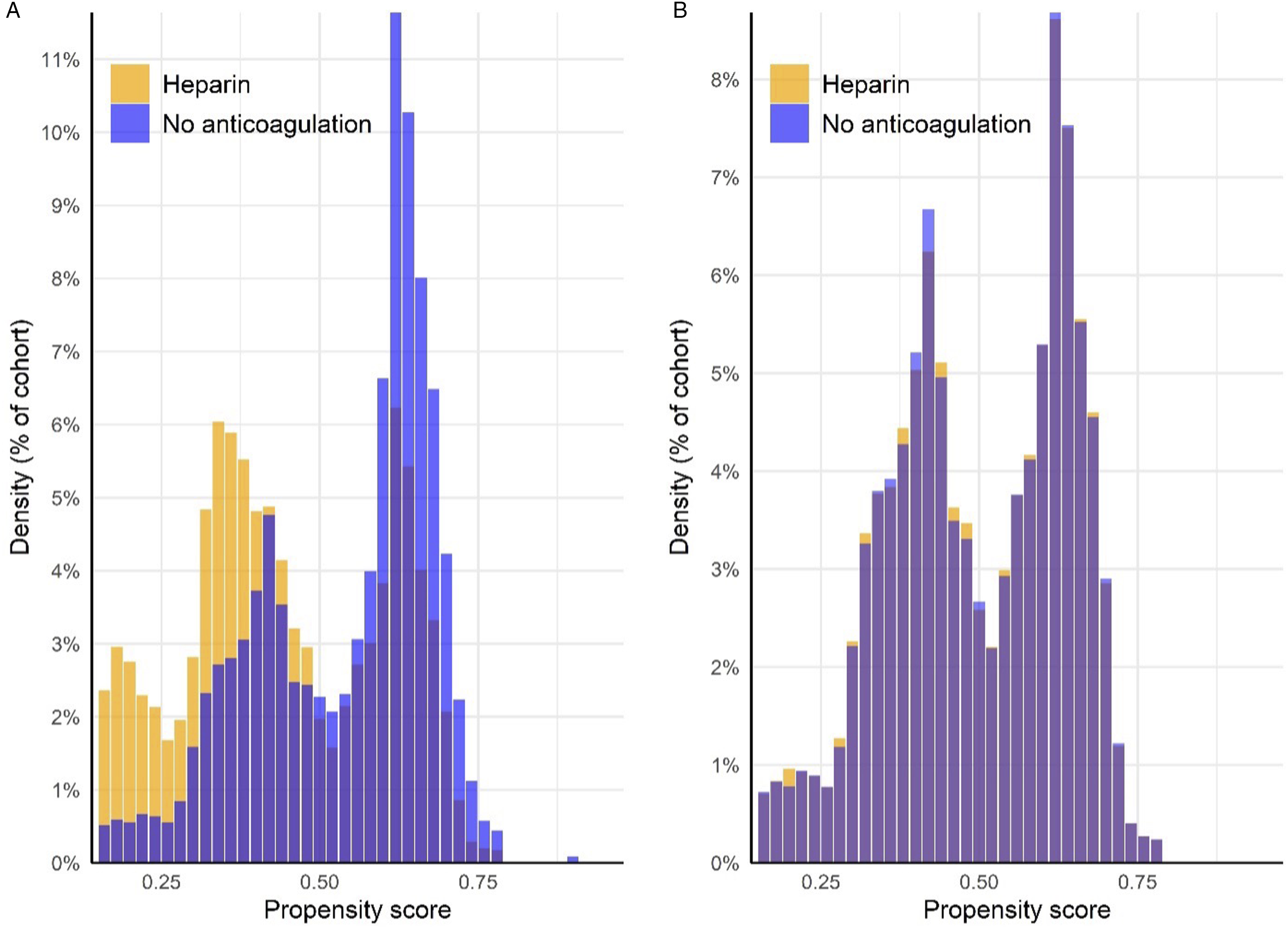
Primary Outcome at Two Weeks
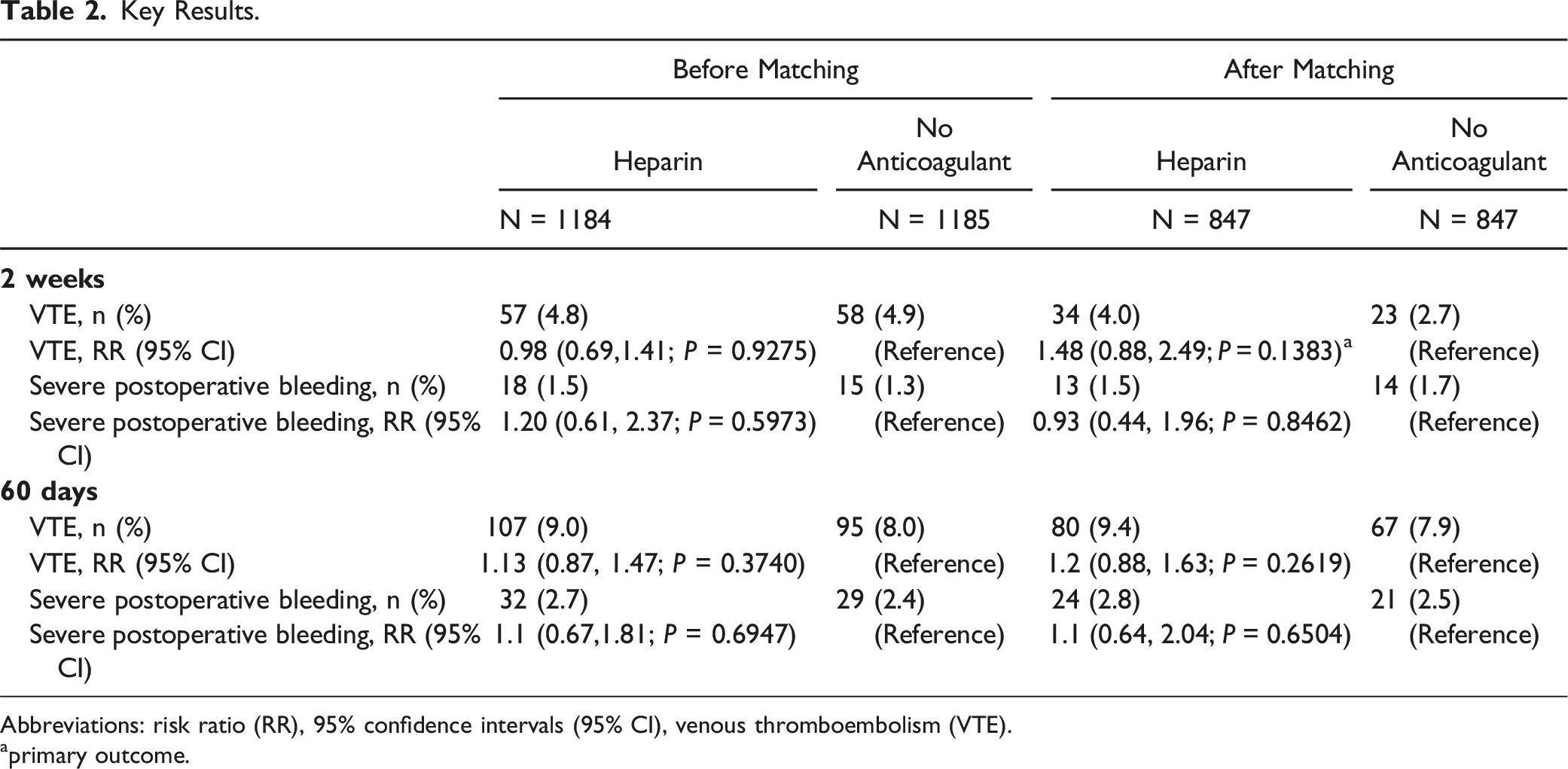
There was no statistically significant difference in the incidence of VTE in the heparin group compared to the no anticoagulation group after spine fusion for spinal metastasis through 2-weeks (4.0% vs 2.7%; RR: 1.48; 95% CI: 0.88, 2.49; P = 0.1383) (Figure 2; Table 2). Furthermore, there was no statistically significant difference in the rate SPOB in the heparin group as compared to the no anticoagulation group at 2-weeks’ follow-up (1.5% vs 1.7%; RR: 0.93; 95% CI: 0.44, 1.96; P = 0.8462) (Figure 3; Table 2). Cumulative incidence of the occurrence of venous thromboembolism per cohort, after matching. Incidence curves in the heparin cohort (orange) and no anticoagulant cohort (blue) are illustrated over the 60-day follow-up period. Shaded bands indicate 95% confidence intervals. Key Results. Abbreviations: risk ratio (RR), 95% confidence intervals (95% CI), venous thromboembolism (VTE). aprimary outcome. Cumulative incidence of the occurrence of severe postoperative bleeding per cohort, after matching. Incidence curves in the heparin cohort (orange) and no anticoagulant cohort (blue) are illustrated over the 60-day follow-up period. Shaded bands indicate 95% confidence intervals.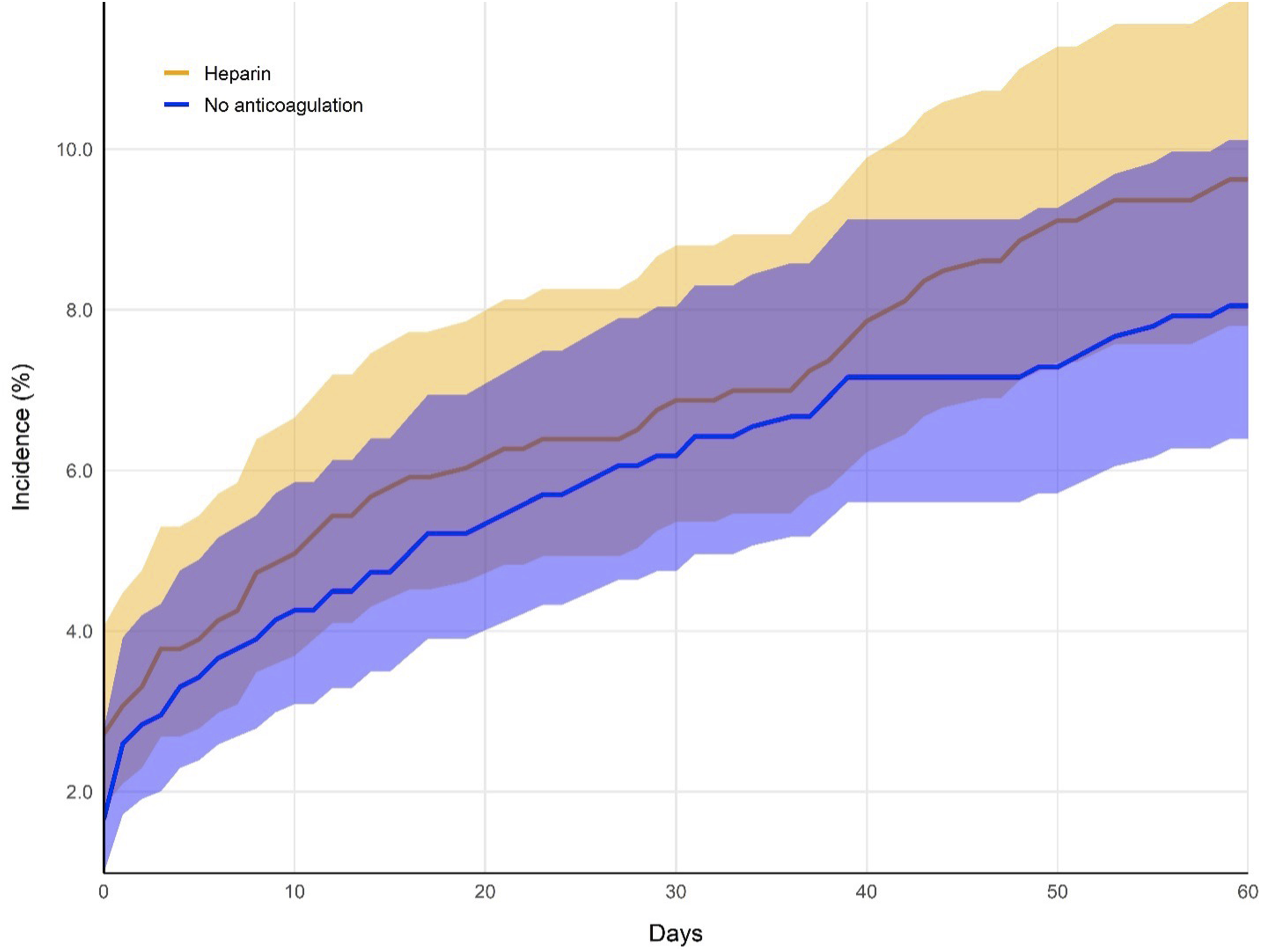
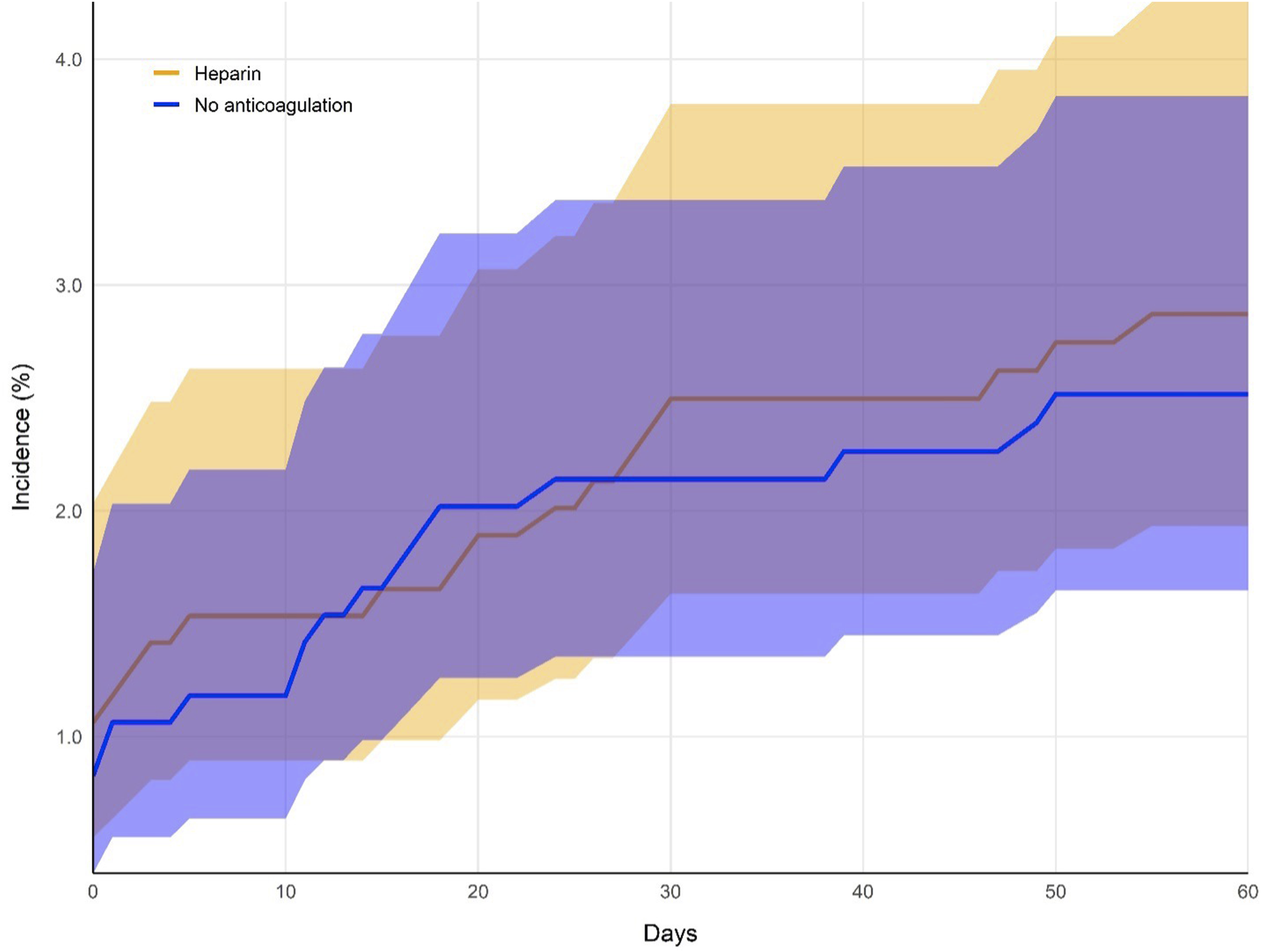
Secondary Outcomes at Two Months
There was no statistically significant difference in the proportion of VTE in the heparin group as compared to the no anticoagulation group after spinal fusion through 2 month’s follow-up (9.4% vs 7.9%; RR: 1.2; 95% CI: 0.88, 1.63; P = 0.2619) (Figure 2) (Table 2). Furthermore, there was no statistically significant difference in the proportion of SPOB in the heparin group compared to the no anticoagulation group through 2-months’ follow-up (2.8% vs 2.5%; RR: 1.1; 95% CI: 0.64, 2.04; P = 0.6504) (Figure 3; Table 2).
Discussion
To our knowledge, the present study is the largest to examine the association between early heparin chemoprophylaxis for spine fusion for spinal metastasis and VTE and SPOB. As VTE remains a relevant complication after spine surgery, especially among those with spinal metastasis,17,18 additional research was needed to guide clinical management.9,11,12 This study found that early heparin administration after spine fusion for spinal metastasis was not associated with a significant difference in VTE or SPOB through 2 months after spinal fusion.
The findings of this study cautiously suggest that the use of early heparin utilization as chemoprophylaxis has no clear superiority over no early anticoagulation for the prevention of VTE after spine fusion for spinal metastasis. However, our findings also suggest that there is no increase in risk of SPOB with early heparin administration. In the context of a lack of a clear risk/benefit profile regarding early anticoagulation, the complexity and variability of individual clinical circumstances must be acknowledged. Accordingly, clinicians should tailor care recommendations regarding prophylactic anticoagulation to patients on a case-by-case basis, considering any relevant risk factors for VTE and SPOB, including prior use of anticoagulants, and pre-, intra-, and post-operative factors.
The present findings further support the lack of a general clear consensus regarding VTE chemoprophylaxis for spine surgery, for example, as summarized by the 2009 clinical guidelines put forth by the North American Spine Society (NASS).2-4,6 Importantly, those guidelines indicated that there was insufficient high-quality data to comment on the overall rate of VTE in patients undergoing spine surgery for spinal malignancy with or without chemoprophylaxis. 6 Our findings reinforce the lack of a straightforward role of early VTE chemoprophylaxis among patients undergoing spine fusion for metastasis.
Our findings agree with a previous case control study by Groot et al. (2019) which found no significant association between chemoprophylaxis and VTE among patients undergoing spine surgery for metastasis after controlling for demographics and comorbidities (n = 637; odds ratio = 1.34; 95% CI: 0.62, 2.90; P = 0.459). 9 In this previous study, chemoprophylaxis agents included either heparin, aspirin, or enoxaparin, reflecting the real-world variation in chemoprophylaxis for surgical management of spinal metastasis. 9 To examine whether variation in anticoagulant profiles could have explained the null finding of this prior study, we concentrated on heparin only, yet arrived at the same conclusion.
The present findings align with those of a recent study by Jiang et al. (2023), which suggested that heparin administration did not increase the incidence of SPOB among individuals undergoing spinal surgery for metastasis. 12 In their study (n = 100), patients who had preoperative deep vein thrombosis (DVT) and subsequently received anticoagulation (enoxaparin) (n = 8) were compared with patients without preoperative DVT who did not receive enoxaparin. 12 None of the patients in each group developed major bleeding, although minor bleeding events occurred in the group who did not receive anticoagulation.
Our findings contradict those of a small retrospective study (n = 65) by De la Garza Ramos et al (2019). After controlling for smoking and surgical characteristics, that study found that patients who had delayed anticoagulation (n = 14) had a significantly higher risk of VTE compared to patients who had early anticoagulation (n = 22) 30 days after spine surgery (odds ratio: 6.43; 95% CI: 1.01, 41.2; P = 0.049). 11 Several relevant differences between the prior study and ours may explain the discrepancy in findings. For example, the previous study (1) was smaller in sample size, raising the possibility of a spurious finding, (2) examined a range of anticoagulants [heparin, dalteparin, and enoxaparin] which may have varying effectiveness, and (3) did not control for covariates that may influence the risk of VTE (eg, platelets, glucose, previous pulmonary embolism, coagulopathy, heart disease, and chemotherapy). Consideration of VTE risk factors is relevant to minimizing confounding by indication, as surgeons often administer prophylactic anticoagulation when anticipating a higher risk of post-operative VTE.
Future studies on this topic may consider replicating our design while examining different anticoagulants, such as rivaroxaban or apixaban, which may convey different risk profiles for VTE and/or SPOB.3,7 In addition, due to the lack of studies on the topic, variations of study design methods and populations are encouraged, which may produce different risk estimates compared to ours. As evidence accrues, a meta-analysis on the topic could be considered to synthesize risk estimates while considering other factors such as study quality and precision. Additionally, in many spine centers, LMWH is a more commonly used prophylactic agent compared to unfractionated heparin, which may limit the generalizability of our findings. However, our focus on heparin provided an opportunity to evaluate its specific impact. Future studies should compare heparin to other agents like LMWH to determine the optimal chemoprophylactic strategy for this population. This comparison is particularly relevant given the differing pharmacokinetic profiles and associated risk-benefit balances of these agents. Finally, updated clinical practice guidelines are needed to guide surgeons’ use of early anticoagulation for patients with metastasis undergoing spine surgery. Such a process could include a broader scope of evidence, including expert opinion (eg, Delphi process), to derive evidence-based recommendations for clinicians.
Strengths and Limitations
Strengths of this study include a large sample size and robust propensity matching strategy. Focusing on a single anticoagulant removed potential bias related to differential efficacy of between anticoagulant medications. Another strength was that the incidence of VTE and SPOB in or study was similar to the expected range of incidences reported previously, thereby serving as a marker of validity of our findings.9,11,12
However, there are multiple limitations that warrant caution with our study findings. The retrospective nature of this study precludes us from determining a causal inference between heparin and presence or absence of VTE and/or SPOB. There may be unmeasured confounding related to pre-operative or intra-operative factors, such as the patient’s surgical complexity, number of spinal levels operated on, or duration of surgery.5,9 Despite our attempt to standardize the chemoprophylactic agent, the dose or timing of heparin likely varied. In addition, the present methods using matching and a fixed index date do not allow us to adjust risk estimates for post-operative VTE mitigation strategies, such as the use of intermittent pneumatic compression devices, compression stockings, early walking/mobilization, leg elevation, and hydration levels. Importantly, the lack of data on neurological status, which could influence VTE risk, represents a limitation of the database. This information was unavailable due to the de-identified and aggregate nature of the dataset, preventing adjustment for this potentially relevant factor. Furthermore, our study has a limited granularity regarding the type of cancer or primary source of metastasis, which was often unknown and/or inconsistently documented in the preoperative period according to our pilot queries. It is possible that some patients developed clinical warning signs of VTE immediately following surgery, thus prompting administration of anticoagulation (ie, confounding by indication). The use of routine vs symptom-driven doppler ultrasounds for DVT diagnosis could not be determined, as this detail is not consistently recorded in the database and may influence the reported incidence of DVT. While our propensity matching strategy aimed to minimize between-cohort variation with respect to factors that could lead to confounding by indication, it remains impossible to eliminate this source of bias entirely. While confounding by indication can be overcome by randomized trials, none exist on this topic at this time, and multicenter participation may be needed to attain sufficient sample size to conduct such a trial. A final limitation is the inability to stratify by histotype of metastasis due to inconsistent data. Future studies should include this detail to better understand its impact on VTE and bleeding risk. While newer agents such as LMWHs and DOACs are gaining favor, unfractionated heparin remains widely used, as demonstrated in prior studies and our own clinical practice. This may limit the generalizability of our findings to settings where newer agents are more commonly utilized.
Conclusion
In this large, propensity-matched retrospective cohort study, early heparin administration after spine fusion for spinal metastasis was not associated with a significant difference in VTE or SPOB incidence compared to matched controls without early anticoagulation through 2 months after spinal fusion. Given the current limited and conflicting findings available, future studies should examine if the risk of VTE and SPOB varies according to the type of anticoagulant used. In addition, evidence synthesis on the topic may be warranted. Considering the lack of clear superiority of early heparin administration for VTE prophylaxis in surgical management of spinal metastasis in the present study, yet limited and conflicting available evidence, surgeons should only recommend this therapy on a case-by-case basis.
Supplemental Material
Supplemental Material - Association Between Early Heparin Prophylaxis and Risk of Venous Thromboembolism Through Two Months After Spine Fusion for Spinal Metastasis: A Retrospective Cohort Study of United States Academic Health Centers
Supplemental Material for Association Between Early Heparin Prophylaxis and Risk of Venous Thromboembolism Through Two Months After Spine Fusion for Spinal Metastasis: A Retrospective Cohort Study of United States Academic Health Centers by Anthony N. Baumann, Robert J. Trager, Omkar Anaspure, John T. Strony, Aditya Muralidharan, Tyler Sanda, and Jacob C. Hoffmann in Global Spine Journal
Footnotes
Acknowledgements
This study is made possible via the support from the Clinical Research Center of University Hospitals Cleveland Medical Center. The contents of this paper are solely the responsibility of the authors and do not encompass the views or opinions of the Clinical Research Center of University Hospitals Cleveland Medical Center.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is supported by the Clinical and Translational Science Collaborative of Northern Ohio which is funded by the National Institutes of Health, National Center for Advancing Translational Sciences, Clinical and Translational Science Award grant, UM1TR004528. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
