Abstract
Study Design:
A multicentered retrospective case series.
Objective:
To determine the incidence and circumstances surrounding the development of a symptomatic postoperative epidural hematoma in the cervical spine.
Methods:
Patients who underwent cervical spine surgery between January 1, 2005, and December 31, 2011, at 23 institutions were reviewed, and all patients who developed an epidural hematoma were identified.
Results:
A total of 16 582 cervical spine surgeries were identified, and 15 patients developed a postoperative epidural hematoma, for a total incidence of 0.090%. Substantial variation between institutions was noted, with 11 sites reporting no epidural hematomas, and 1 site reporting an incidence of 0.76%. All patients initially presented with a neurologic deficit. Nine patients had complete resolution of the neurologic deficit after hematoma evacuation; however 2 of the 3 patients (66%) who had a delay in the diagnosis of the epidural hematoma had residual neurologic deficits compared to only 4 of the 12 patients (33%) who had no delay in the diagnosis or treatment (P = .53). Additionally, the patients who experienced a postoperative epidural hematoma did not experience any significant improvement in health-related quality-of-life metrics as a result of the index procedure at final follow-up evaluation.
Conclusion:
This is the largest series to date to analyze the incidence of an epidural hematoma following cervical spine surgery, and this study suggest that an epidural hematoma occurs in approximately 1 out of 1000 cervical spine surgeries. Prompt diagnosis and treatment may improve the chance of making a complete neurologic recovery, but patients who develop this complication do not show improvements in the health-related quality-of-life measurements.
Keywords
Introduction
Cervical spine surgery for multiple pathologies including persistent radiculopathy and spondylotic myelopathy has been associated with significant improvements in health-related quality-of-life (HRQOL) outcomes. 1 -4 However, there are significant risks associated with undergoing cervical spine surgery. Some complications, such as mild dysphagia after an anterior cervical discectomy and fusion, are common, but rarely result in any long-term sequelae. 5 -7 Conversely, rare complications such as a symptomatic epidural hematoma, esophageal injury, or vertebral artery injury can lead to significant morbidity and even death. 8 -15
Asymptomatic postoperative epidural hematomas after spine surgery are extremely common 16 -20 ; in a study that looked at patients who had any type of spine surgery, Mirzai et al reporting that magnetic resonance imaging can detect an epidural hematoma in up to 89% of patients postoperatively. 20 Fortunately, these hematomas are only symptomatic in 0.10% to 0.24% of all spine cases. 21 -25 When this complication is recognized quickly and the hematoma is evacuated, many patients can make a full neurologic recovery, but a delay in diagnosis and treatment can lead to an irreversible neurological injury. 8 -10,26,27
For surgeons to be able to accurately inform their patients about the risks and benefits of cervical spine surgery, it is important to establish the actual incidence of rare but potentially devastating complications. To date, almost all of the large studies published on this complication are single-institution studies that report the incidence of postoperative symptomatic epidural hematomas in all types of spine surgery. 21 -24,28 The published studies that are dedicated to postoperative epidural hematomas in the cervical spine are relatively small cases series. 8 -10 The purpose of the current study is to determine the incidence of symptomatic postoperative epidural hematomas following cervical spine surgery across multiple centers, and assess the impact of this complication on clinical outcomes.
Methods
The study is a large retrospective multicenter case series study involving 21 high-volume surgical centers from the AOSpine North America Clinical Research Network. Centers were included if they are members of the AOSpine North America Clinical Trial Research Network. Every center that is a member of the network was invited to participate. Those centers that had interest in participating were prescreened concerning their ability to provide data per study protocol. Altogether, 21 centers passed the screening and were included. Medical records for 17 625 patients who received cervical spine surgery, anterior or posterior (levels from C2 to C7), between January 1, 2005, and December 31, 2011, inclusive, were reviewed to identify occurrence of 21 predefined treatment complications. The complications included reintubation requiring evacuation, esophageal perforation, epidural hematoma, C5 palsy, recurrent laryngeal nerve palsy, superior laryngeal nerve palsy, hypoglossal or glossopharyngeal nerve palsy, dural tear, brachial plexopathy, blindness, graft extrusion, misplaced screws requiring reoperation, anterior cervical infection, carotid artery injury or cerebrovascular accident, vertebral artery injuries, Horner’s syndrome, thoracic duct injury, tetraplegia, intraoperative death, revision of arthroplasty and, pseudomeningocele. Trained research staff at each site abstracted the data from medical records, surgical charts, radiology imaging, narratives, and other source documents for the patients who experienced one or more of the complications from the list. Data were transcribed into study-specific paper case report forms. Copies of case report forms were transferred to the AOSpine North America Clinical Research Network Methodological Core for processing, cleaning, and data entry.
Descriptive statistics were provided for baseline patient characteristics. Paired t test was used to analyze changes in clinical outcomes at follow-up compared to preoperative status.
Results
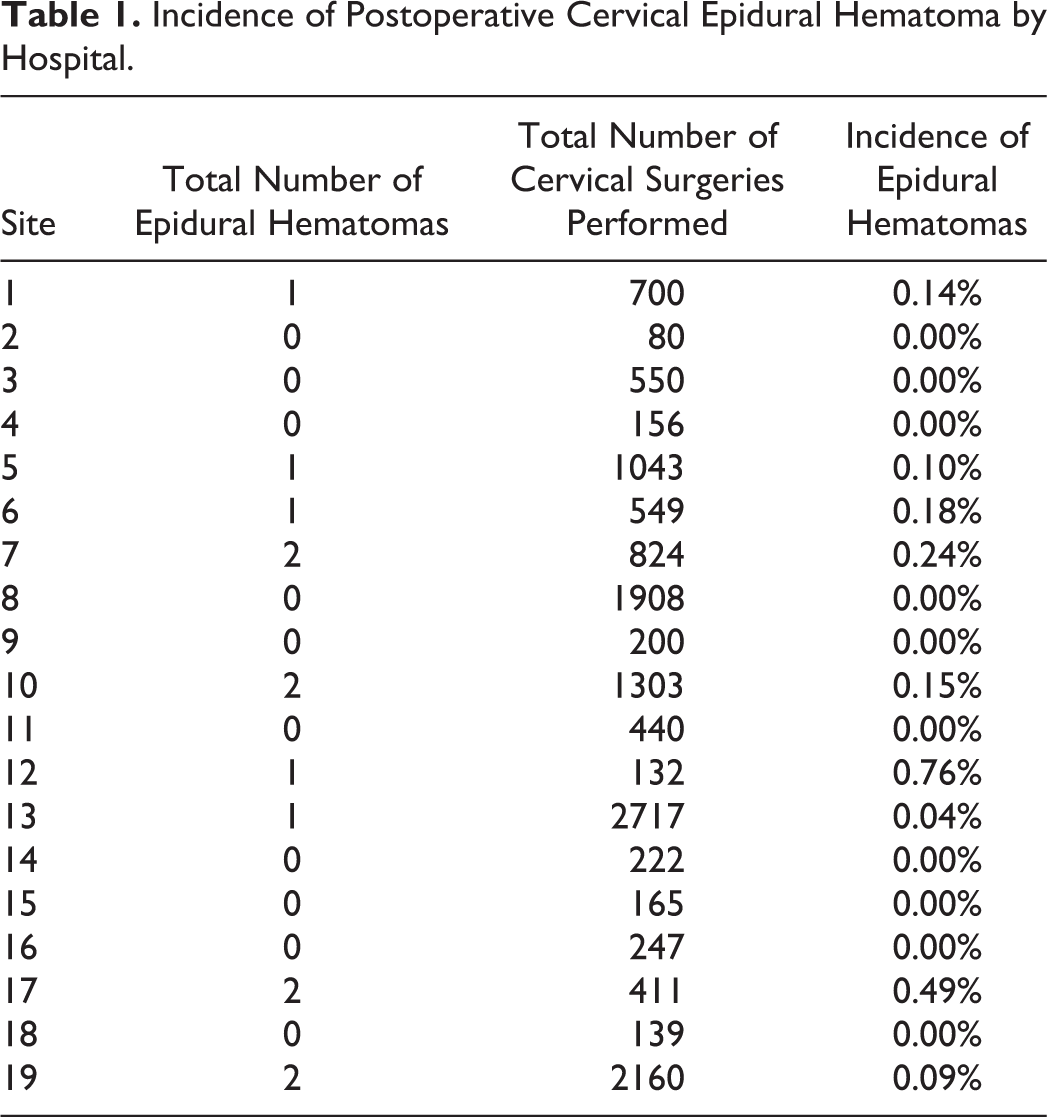
Of the 21 involved sites, only 19 reported the incidence of postoperative epidural hematomas. A total of 16 582 cervical spine surgeries occurred between January 1, 2005, and December 31, 2011, at 19 different institutions, and 15 patients developed a postoperative epidural hematoma, for a total incidence of 0.090%. A total of 8887 anterior procedures and 7695 posterior procedures were evaluated. While rate of epidural hematoma was less in anterior procedures (5.63 per 10 000 anterior cases) than posterior procedures (13.00 per 10 000 posterior cases), this did not meet statistical significance (P = .188). Substantial variation between institutions was noted, with 11 sites reporting no epidural hematomas, and 1 site reporting an incidence of 0.76% (Table 1). The overall demographic and operative details are reported in Table 2. The average time to presentation of symptoms was 4.67 ± 7.90 days after surgery, and the average length of stay for patients with the complication was 9.36 ± 9.35 days.
Incidence of Postoperative Cervical Epidural Hematoma by Hospital.
Patient Demographics and Operative Detailsa.
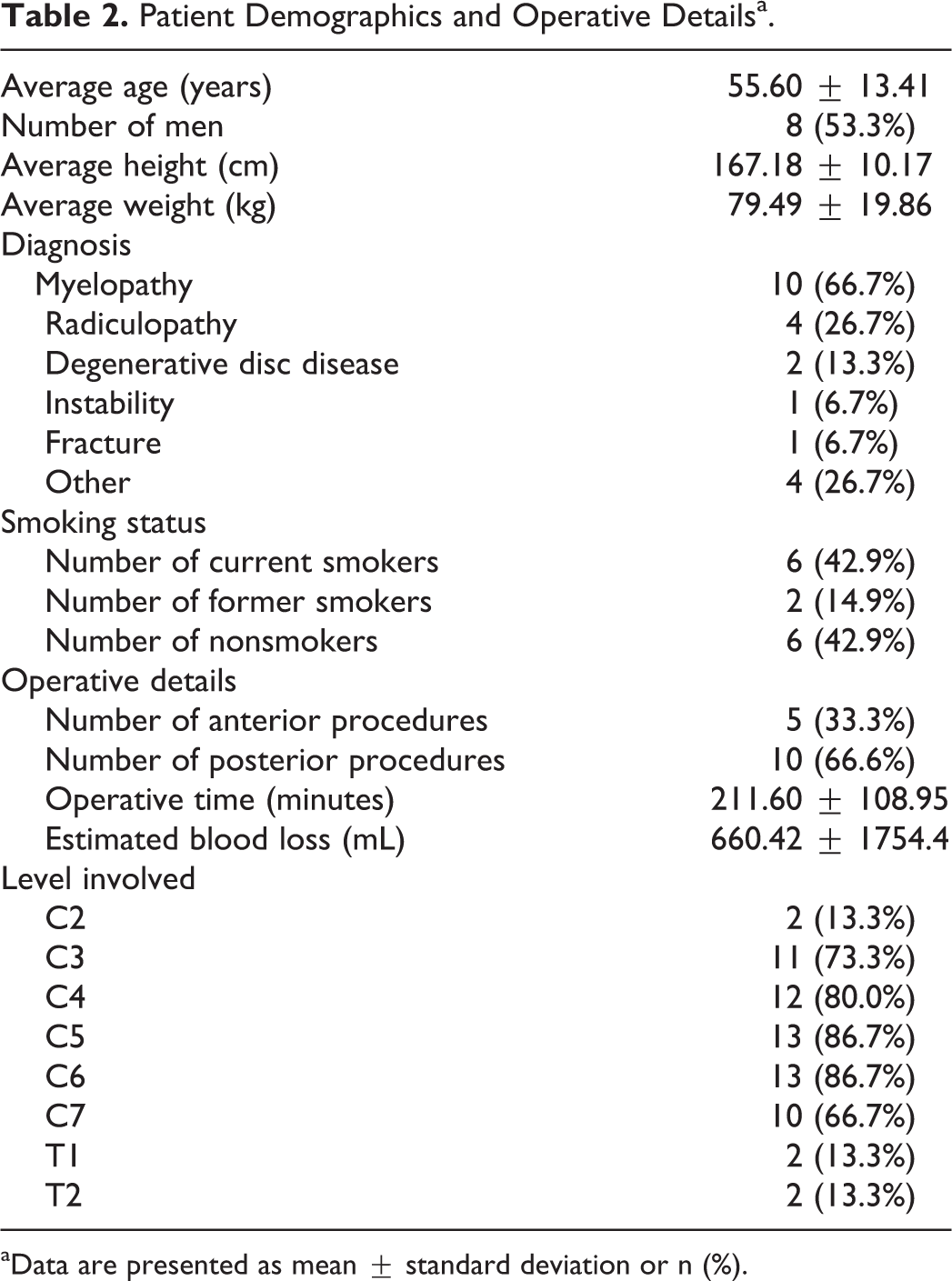
aData are presented as mean ± standard deviation or n (%).
All patients initially presented with a new neurologic deficit from the hematoma, but 9 (60%) patients had complete resolution of the neurologic deficit after hematoma evacuation. Table 3 reports the specific surgery, the time of symptom onset, the new symptoms, if there was a delay in diagnosis, and if there were residual neurologic symptoms for each of the 15 cases. Importantly, 2 of the 3 patients (66%) who had a delay in the diagnosis of the epidural hematoma had residual neurologic deficits compared to only 4 of the 12 patients (33%) who had no delay in the diagnosis or treatment (P = .53). All 6 of the patients with persistent neurologic deficits had motor weakness, with 2 of the 6 having an ASIA (American Spinal Injury Association) C spinal cord injury. Importantly, among the patients who were diagnosed with a symptomatic postoperative epidural hematoma, there was no significant improvement in HRQOL metrics between the preoperative evaluation and the final follow-up evaluation (Table 4).
The Time of Symptom Onset, if There Was a Delay in Diagnosis, and if There Were Residual Neurologic Symptoms for Each of the 15 Cases.
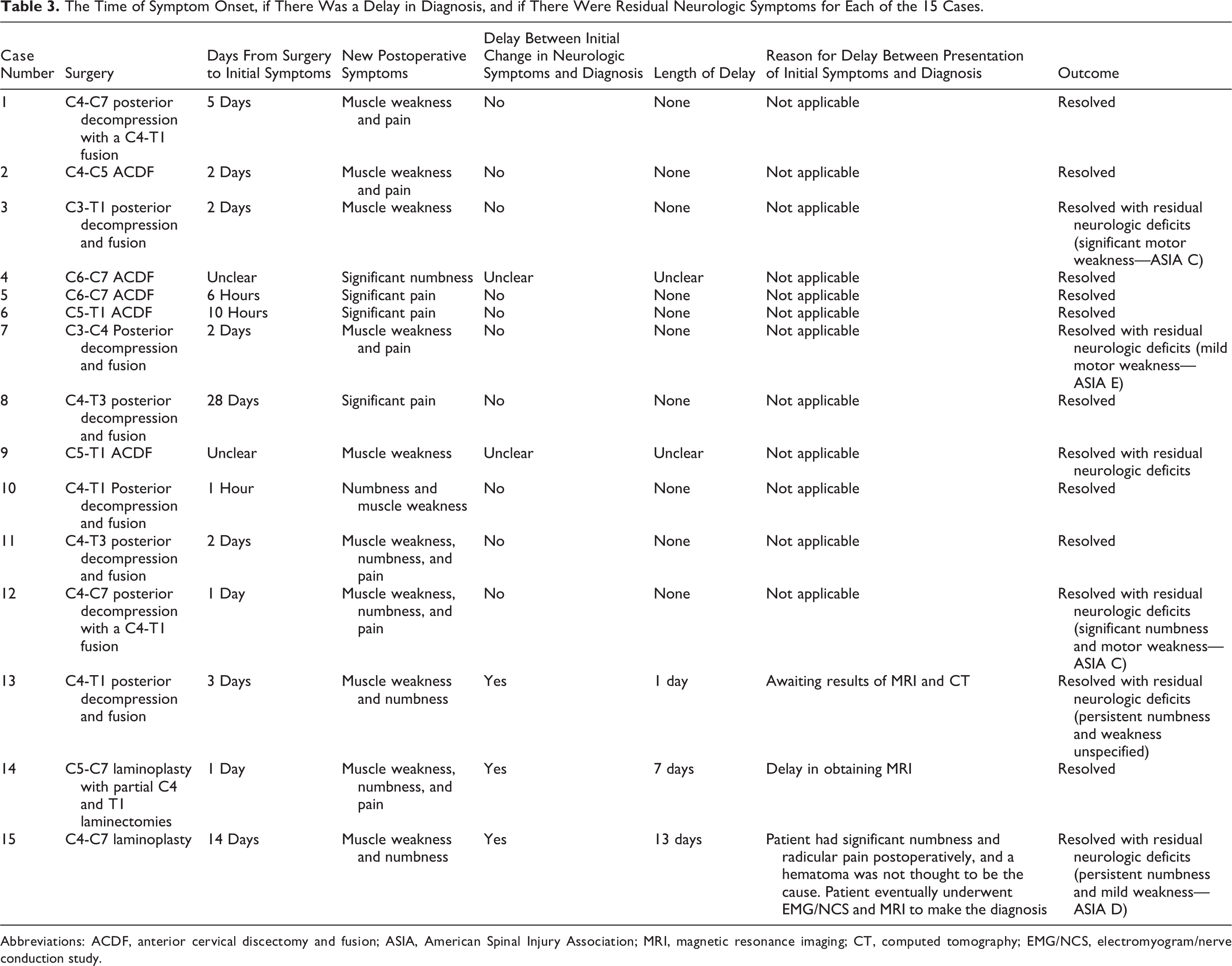
Abbreviations: ACDF, anterior cervical discectomy and fusion; ASIA, American Spinal Injury Association; MRI, magnetic resonance imaging; CT, computed tomography; EMG/NCS, electromyogram/nerve conduction study.
The Health-Related Quality-of-Life Metrics for Patients Who Developed a Postoperative Epidural Hematoma.
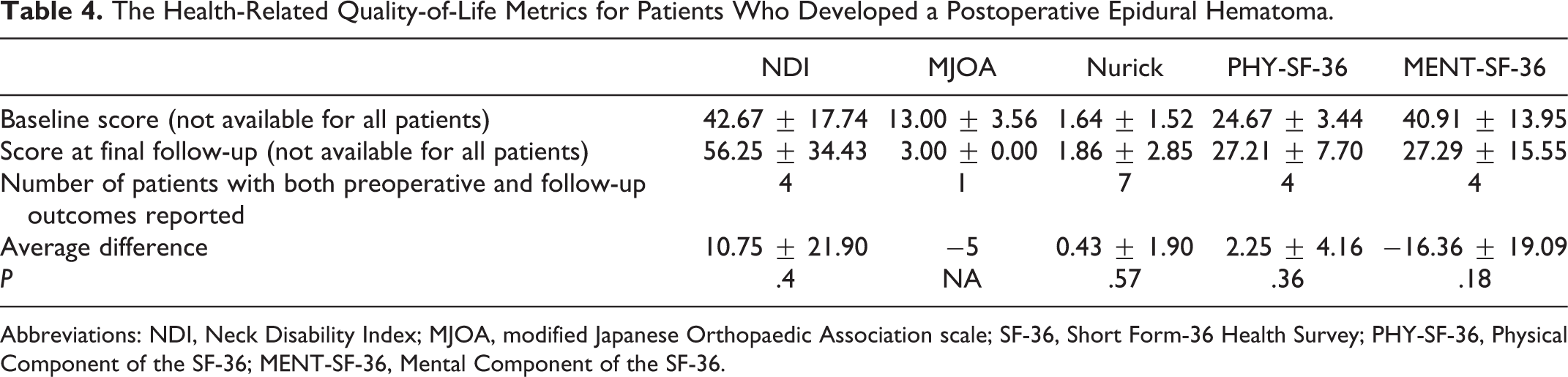
Abbreviations: NDI, Neck Disability Index; MJOA, modified Japanese Orthopaedic Association scale; SF-36, Short Form-36 Health Survey; PHY-SF-36, Physical Component of the SF-36; MENT-SF-36, Mental Component of the SF-36.
Discussion
This study finds that symptomatic postoperative epidural hematomas in the cervical spine are a rare event, occurring in approximately 1 in 1000 cervical spine cases. The results of this study are consistent with other large studies that identify the rate of symptomatic postoperative epidural hematomas throughout the entire spine. In a review of 14 932 spine cases that were performed at a single institution over 18 years, Awad et al identified 32 symptomatic postoperative epidural hematomas for an incidence of 0.20%. While the methodology of the current study did not allow for the identification of risk factors for this complication, Awad et al reported 3 preoperative risk factors, including the use of nonsteroidal anti-inflammatory medication, Rh-positive blood, and patients greater than 60 years of age. Additionally, surgeries involving 6 or more levels, greater than 1 L of blood loss, and a hemoglobin of less than 10 g/dL were identified as intraoperative risk factors; last, if the international normalized ratio rose above 2.0 in the first 48 hours, patients were also at an increased risk of a symptomatic epidural hematoma. 21 In a similar, large, single-institution retrospective study of 12 000 spine surgeries, Kou et al 22 reported that the incidence of a symptomatic postoperative epidural hematoma was 0.10%, and they identified preoperative coagulopathy as well as multilevel surgery as risk factors. Both the incidence rate reported in the current study and the fact that 11/15 cases were multilevel fusions are consistent with the results of Kou et al.
There are 3 large single-institution studies that reported the incidence of a symptomatic epidural hematoma in patients undergoing cervical surgery. 8,28,29 Aono et al identified 1376 patients who underwent cervical spine surgery, and they reported only a single epidural hematoma in 466 anterior cervical spine procedures (0.21%), and 4 symptomatic epidural hematomas in 910 patients who underwent a cervical laminoplasty (0.44%). 28 These results are similar to those reported by Amiri et al, who reported an overall incidence of 0.22% in 4568 spine cases, and a rate of 0.15% in 1962 patients who underwent cervical surgery. 29 Last, Goldstein et al reported on 529 patients undergoing posterior cervical surgery over 10 years at a single intuition, and they reported that 1.5% of patients developed a symptomatic postoperative epidural hematoma. 8 This incidence by Goldstein et al is substantially higher than that identified in the current study or previous reports, and Goldstein et al postulate that this may be due to the fact that almost 50% of the patients in the study underwent a posterior cervical decompression and fusion; however, this is contradicted by their results, as a stepwise regression analysis identified an increased Charlson Comorbidities Index as well as the use of nonsteroidal anti-inflammatory medication in the postoperative period as independent risk factors for a hematoma, but not the surgical treatment (laminoplasty, laminectomy, or the use of instrumentation). 8 Because the study by Goldstein et al is a relatively small, single-institution study, it is possible that the substantial increase in the risk of an epidural hematoma may be due to institutional protocols or population-specific factors. Specifically, the rate of asymptomatic epidural hematomas has been reported to be almost 90%, 20 so if the surgeons at the institution were more inclined to order advanced postoperative imaging, they may have attributed a neurologic deficit, such as a C5 palsy to the hematoma.
With 16582 cervical spine cases, this study is the largest study in the literature looking at individual patients who developed a symptomatic postoperative epidural hematoma in the cervical spine, and because patients from 19 different institutions were reviewed, individual surgeon and institution-based risks have been significantly mitigated. While this methodology has many benefits, including that a series of 15 patients with a postoperative cervical epidural hematoma is the largest reported in any publication to date, there are still significant limitations in the study that must be acknowledged. While a series of 15 patients is the largest published, it was still too small to definitively establish if there is a significant difference in the neurologic outcome of patients who had a delay in the diagnosis. Additionally, a substantial increase in the rate of epidural hematomas occurred in posterior surgeries; however, with only 15 patients in total with this complication, it did not reach statistical significance (anterior procedures—5.63 per 10 000 cases; posterior procedures—13.00 per 10 000 cases; P = .188). Furthermore, each institution identified all patients with a postoperative symptomatic epidural hematoma, but the individual patient data for all of the unaffected patients were not available. Because of this, it was not possible to identify specific risk factors for a hematoma, such as the use of anticoagulation, or the presence of specific comorbidities; additionally, it is not possible to determine the institutional risk factors. The incidence rate ranged from 0.00% to 0.76% at different institutions; however, without reviewing the entire cohort from all the hospitals, it is not possible to determine which factors affected the hospital’s rate of epidural hematomas. Conversely, this methodology allowed for a large sample size, and because individual patient data were used rather than ICD-9 codes, it is not subject to flaws of large, administrative database studies. 30,31
Another limitation to this study is the heterogeneity in the data that were reported. Because the study spanned 7 years and 19 institutions, the available data varied significantly. Only 4 patients (26.7%) had pre- and postoperative SF-36 (Short Form-36 Survey) and NDI (Neck Disability Index) data, while MJOA (modified Japanese Orthopaedic Association scale) and Nurick grades were available for 1 (6.7%) and 7 (46.7%) patients, respectively. In spite of the limited numbers and variable outcomes measures, the current study is the only study in the literature to report any HRQOL outcome measures after this rare complication, and regardless of the outcome measure reported, patients with a postoperative epidural hematoma showed no significant change from preoperative scores. A final limitation is that the diagnosis of a symptomatic postoperative epidural hematoma was left up to the treating physician. Undoubtedly, some surgeons have a lower threshold for obtaining advanced imaging postoperatively, and therefore they are more likely to identify and treat an epidural hematoma than surgeons who rarely obtain advanced imaging.
Conclusion
The current study is the largest series to date to analyze the incidence of an epidural hematoma following cervical spine surgery. The results of this study suggest that an epidural hematoma is a very rare event, occurring in approximately 1 out of 1000 cervical spine surgeries, and it may be slightly more common in posterior surgeries. Prompt diagnosis and treatment resulted in a complete neurologic recovery in the majority of patients, but even without a delay in the diagnosis, 33% of patients still had persistent neurologic deficits. Additionally, the sequelae of this complication results in patients having no improvement in HRQOL outcomes from their preoperative state.
Footnotes
Authors’ Note
This study was ethically approved by the institutional ethics committees at all participating sites.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Gregory D. Schroeder reports grants from AOSpine North America during the conduct of the study, other from AOSpine, grants from Medtronic, outside the submitted work; Alan S. Hilibrand reports grants from AOSpine North America during the conduct of the study, other from Amedica, Vertiflex, Benvenue, Lifespine, Paradigm Spine, PSD, Spinal Ventures, outside the submitted work, and in addition, Dr. Hilibrand has a patent Aesculap, Amedica, Biomet, Stryker, Alphatec, with royalties paid; David E. Fish reports grants from AOSpine North America during the conduct of the study; Jeffrey C. Wang reports grants from AOSpine North America during the conduct of the study; Jeffrey L. Gum reports grants from AOSpine North America during the conduct of the study, personal fees from Medtronic, Alphatec, Stryker, LifeSpine, Acuity, Pacira, PAKmed, Gerson Lehrman Group, personal fees from OREF, AOSpine, personal fees from Acuity, other from Medtronic, personal fees from MiMedx, Pacira Pharmaceuticals, Alphatec, grants from Fischer Owen Fund, nonfinancial support from American Journal of Orthopaedics, nonfinancial support from American Journal of Orthopaedics, The Spine Journal, outside the submitted work; Zachary A. Smith reports grants from AOSpine North America during the conduct of the study; Wellington K. Hsu reports grants from AOSpine North America during the conduct of the study, personal fees from Medtronic, personal fees from Stryker, personal fees from Bacterin, personal fees from Graftys, personal fees from Ceramtec, personal fees from Relievant, personal fees from Bioventus, personal fees from Globus, personal fees from SpineSmith, outside the submitted work; Ziya L. Gokaslan reports grants from AOSpine North America during the conduct of the study, grants from AOSpine North America, personal fees from AO Foundation, grants from AOSpine, outside the submitted work; Robert E. Isaacs reports grants from AOSpine North America during the conduct of the study, grants and personal fees from NuVasive, Inc., personal fees from Association for Collaborative Spine research, outside the submitted work; Thomas E. Mroz reports other from AOSpine, grants from AOSpine North America during the conduct of the study; personal fees from Stryker, personal fees from Ceramtec, other from Pearl Diver, outside the submitted work; Ahmad Nassr reports grants from AOSpine North America during the conduct of the study; Michael G. Fehlings reports grants from AOSpine North America during the conduct of the study; Zorica Buser reports grants from AOSpine North America during the conduct of the study; Peter I. Cha reports grants from AOSpine North America during the conduct of the study; Erica L. Gee reports grants from AOSpine North America during the conduct of the study; Elizabeth L. Lord reports grants from AOSpine North America during the conduct of the study; Erik N. Mayer reports grants from AOSpine North America during the conduct of the study; Owen J. McBride reports grants from AOSpine North America during the conduct of the study; Emily C. Nguyen reports grants from AOSpine North America during the conduct of the study; P. Justin Tortolani reports grants from AOSpine North America during the conduct of the study, other from Globus Medical, grants from Spineology, other from Innovasis, outside the submitted work, and in addition, Dr. Tortolani has a patent Globus with royalties paid; D. Alex Stroh reports grants from AOSpine North America, during the conduct of the study; and K. Daniel Riew reports personal fees from AOSpine International, other from Global Spine Journal, other from Spine Journal, other from Neurosurgery, personal fees from Multiple Entities for defense, plantiff, grants from AOSpine, grants from Cerapedics, grants from Medtronic, personal fees from AOSpine, personal fees from NASS, personal fees from Biomet, personal fees from Medtronic, nonfinancial support from Broadwater, outside the submitted work; Paul M. Arnold reports grants from AOSpine North America during the conduct of the study; other from Z-Plasty, other from Medtronic Sofamore Danek, other from Stryker Spine, other from FzioMed, other from AOSpine North America, other from Life Spine, other from Integra Life, other from Spine Wave, other from MIEMS, other from Cerapedics, other from AOSpine North America, outside the submitted work; Rick C. Sasso reports grants from AOSpine North America during the conduct of the study; personal fees from medtronic, grants from medtronic, grants from K2, outside the submitted work; Mohamad Bydon reports grants from AOSpine North America during the conduct of the study; Dhananjay Chatterjee reports grants from AOSpine North America during the conduct of the study; Allison K. Roe reports grants from AOSpine North America during the conduct of the study; D. Alex Stroh reports grants from AOSpine North America during the conduct of the study; Marisa Y. Yanez reports grants from AOSpine North America during the conduct of the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by AOSpine North America Inc, a 501(c)3 nonprofit corporation.
