Abstract
Study Design
Original research, cross-sectional study.
Objectives
Evaluate preferences of patients, caregivers, and patient-caregiver dyads for non-fusion vs fusion surgery.
Methods
A survey-based discrete choice experiment included patients 12-15 years of age with idiopathic scoliosis and their caregivers. Preference weights were estimated for 2 types of attributes that differed between fusion and non-fusion surgery: treatment outcomes (ie, appearance, confidence in planned correction, spinal motion, device failure, reoperation, and recovery period) and procedure-type requirements associated with each surgery type. Correlations between preference weights for patients, caregivers, and dyads were calculated to assess preference concordance between cohorts.
Results
A total of 344 respondents (124 patients, 92 caregivers, and 128 patient/caregiver dyads) completed the survey. Appearance was the most important treatment outcome for the average respondent in all cohorts. For patients, the least important treatment outcome was recovery time, whereas reoperation rate was the least important treatment outcome for caregivers and dyads. After controlling for treatment outcomes, patients and patient-caregiver dyads preferred the procedure-type requirements associated with fusion surgery, while caregivers preferred the procedure-type requirements associated with non-fusion surgery. Preferences of respondents who completed the survey prior to surgery were similar to those who completed the survey after surgery. Patient and caregiver preferences were more similar to the preferences of dyads than to each other.
Conclusions
Patients and patient-caregiver dyads preferred the procedure-type requirements associated with fusion surgery, while caregivers preferred the procedure-type requirements associated with non-fusion surgery. Appearance was the most important treatment outcome for patients, caregivers, and patient-caregiver dyads.
Keywords
Introduction
Idiopathic scoliosis is defined as a lateral curvature of the spine ≥10° associated with vertebral rotation that lacks an identifiable cause and is the most common type of pediatric spinal deformity. 1 Idiopathic scoliosis is most frequently diagnosed between 10 to 18 years and is referred to as adolescent idiopathic scoliosis (AIS). Idiopathic scoliosis of 10° or more occurs in about 2.5% of most adolescent populations, however, in only about 0.25% does a curve progress to where treatment is warranted.2,3 Nonoperative brace treatment is considered for skeletally immature patients with curves greater than 20-25°, while surgical treatment is considered for curves greater than 40-45° that are progressive despite brace treatment in skeletally immature patients, and for curves approaching or greater than 50° in skeletally mature patients.4,5 The surgical treatment for AIS with the longest track record is instrumented posterior spinal fusion which provides immediate, excellent, and durable correction of spinal curvatures with low rates of complications including revision surgery.6,7 However, fusion surgery results in permanent loss of motion and growth across the instrumented spinal levels and may lead to degeneration of adjacent spinal segments and reduced health-related quality of life in adulthood.8-10 Non-fusion spinal devices including anterior vertebral body tethers (AVBT) and posterior dynamic distraction (PDD) have received marketing authorization by the United States Food and Drug Administration (FDA) for treatment of pediatric scoliosis.11-13 These devices aim to correct spinal curvatures while preserving spinal growth, spinal mobility and delaying or preventing the need for spinal fusion.14,15 While AVBT and PDD have been shown to delay or avoid spinal fusion in appropriately selected patients, complications and reoperations are higher compared to fusion, and spinal deformity correction has been reported as less consistent and predictable compared to fusion.16-20 However, limited published data which compares long term outcomes of non-fusion vs fusion surgery for treatment of AIS is currently available to guide decision-making by patients and families.
Treatment of pediatric spinal deformity was recognized by the FDA as a preference-sensitive priority area for medical devices due to the patient benefit-risk tradeoffs and impact of uncertainty related to treatment decisions regarding non-fusion spinal surgery vs spinal fusion. 21 In 2019 a multidisciplinary team comprised of health professionals including spine surgeons, researchers, a patient advocacy organization, and FDA staff developed a survey to identify the benefits, risks, and key attributes of fusion surgery and non-fusion surgery. This survey was designed as a discrete choice experiment (DCE) and collected quantitative patient preference information for a set of treatment related attributes associated with fusion surgery and non-fusion surgery (AVBT). Results of this initial study showed that a single cohort comprised of patients and their caregivers valued appearance and preservation of spinal motion and, to a lesser extent, reoperation rates when considering non-fusion vs fusion surgery. 22 To our knowledge, this was the first study to quantitatively evaluate patient and parent preferences for fusion surgery compared to non-fusion surgery for treatment of adolescent idiopathic scoliosis using a discrete choice experimental design. This follow-up study was conducted to address the research question of whether differences in preferences exist for non-fusion vs fusion surgery from the perspectives of patients, caregivers, and patient-caregiver dyads. The aim of this study is to better understand how the preferences of pediatric patients, caregivers, and patient-caregiver dyads interact and influence decision-making, and to utilize these data to help inform future clinical and regulatory decision-making.
Materials and Methods
The preferences of pediatric patients, caregivers, and patient-caregiver dyads associated with fusion and non-fusion surgery were evaluated using an online DCE. DCEs elicit stated-preference information using a survey-based design that requires respondents to choose between profiles for a medical intervention that are defined in terms of attributes and various manifestations of the attributes (attribute levels) associated with a specific medical intervention. DCEs are based on the hedonic principle that choices between alternatives correspond with the relative importance of their features or attributes, and that systematic variations in choices can be leveraged as an indication of the relative importance (or preference) associated with attribute changes. The collection of these data is based on measurement theories in cognitive psychology. 23 The analysis of the choice data generated through DCEs is based on random-utility theory, which links choices and utility. 24
Survey Design
Survey Treatment Outcome Attributes and Attribute Levels for Fusion and Non-Fusion Scenarios Used to Generate Discrete-Choice Experiment Questions Presented to Respondents for Treatment Outcomes for Adolescent Idiopathic Scoliosis.

Note: The discrete-choice experiment questions were labeled. This means every question presented a fusion and non-fusion option. While this label was not experimentally assigned to the alternatives, they represent an attribute meant to capture all aspects of each of the 2 types of surgeries presented to respondents (ie, fusion surgery, and non-fusion surgery (AVBT)) not represented by the outcomes in the table. We identify this attribute as procedure-type requirements.
As outcomes associated with fusion surgery for adolescent idiopathic scoliosis are well-established with a high level of certainty, a fixed profile was identified for the fusion attribute levels to reflect outcomes associated with posterior spinal fusion and posterior spinal instrumentation using contemporary surgical techniques: 90% confidence in planned correction of the spinal curve, excellent improvement in trunk (back) appearance (level 3), no motion after surgery within the treated spinal area, 1% risk of spinal device failure, 3% reoperation rate, and a 6-week recovery period. The non-fusion surgery attribute levels were varied and were defined to encompass the range of outcomes considered clinically plausible based on published literature and expert consensus among the 8 spinal surgeon members of the multidisciplinary team using a nominal group technique. The spinal surgeon investigators were orthopedic surgeons and neurosurgeons from a mix of academic and private practice settings widely distributed across the US and had extensive experience in both fusion and non-fusion surgery for pediatric scoliosis.
A draft survey was created and contained 3 parts. Part 1 collected basic demographic information (patient age, patient sex, caregiver educational level) and prior experiences of patients and caregivers with scoliosis. Part 2 provided a tutorial on the benefits and risks of surgery for idiopathic scoliosis and an overview of fusion surgery vs non-fusion surgery. Part 3 asked the respondents to choose between non-fusion and fusion surgery for various experimentally designed case profiles. The draft survey instrument was pretested in 8 interviews with patients and their caregivers that met the study inclusion criteria. The results from pretesting were used to refine the survey and ensure respondents comprehended the choice tasks and the information contained in the survey. Pretesting was also used to ensure that respondents were trading off between the outcomes.
The final DCE survey was a labeled design in which a fixed profile for fusion was compared to various experimentally designed case profiles in which the attribute levels for non-fusion varied (Figure 1). The design of the non-fusion surgery options relied on a D-efficient design with 420 questions divided into 60 blocks of 7 questions using Lighthouse Studio 9.1 (Sawtooth Software, Inc, Provo UT). Surveys were presented to the patient and caregiver together, and survey respondents identified who completed the survey (patient, caregiver, or both together). Example question from the survey-based discrete-choice experiment presented to respondents.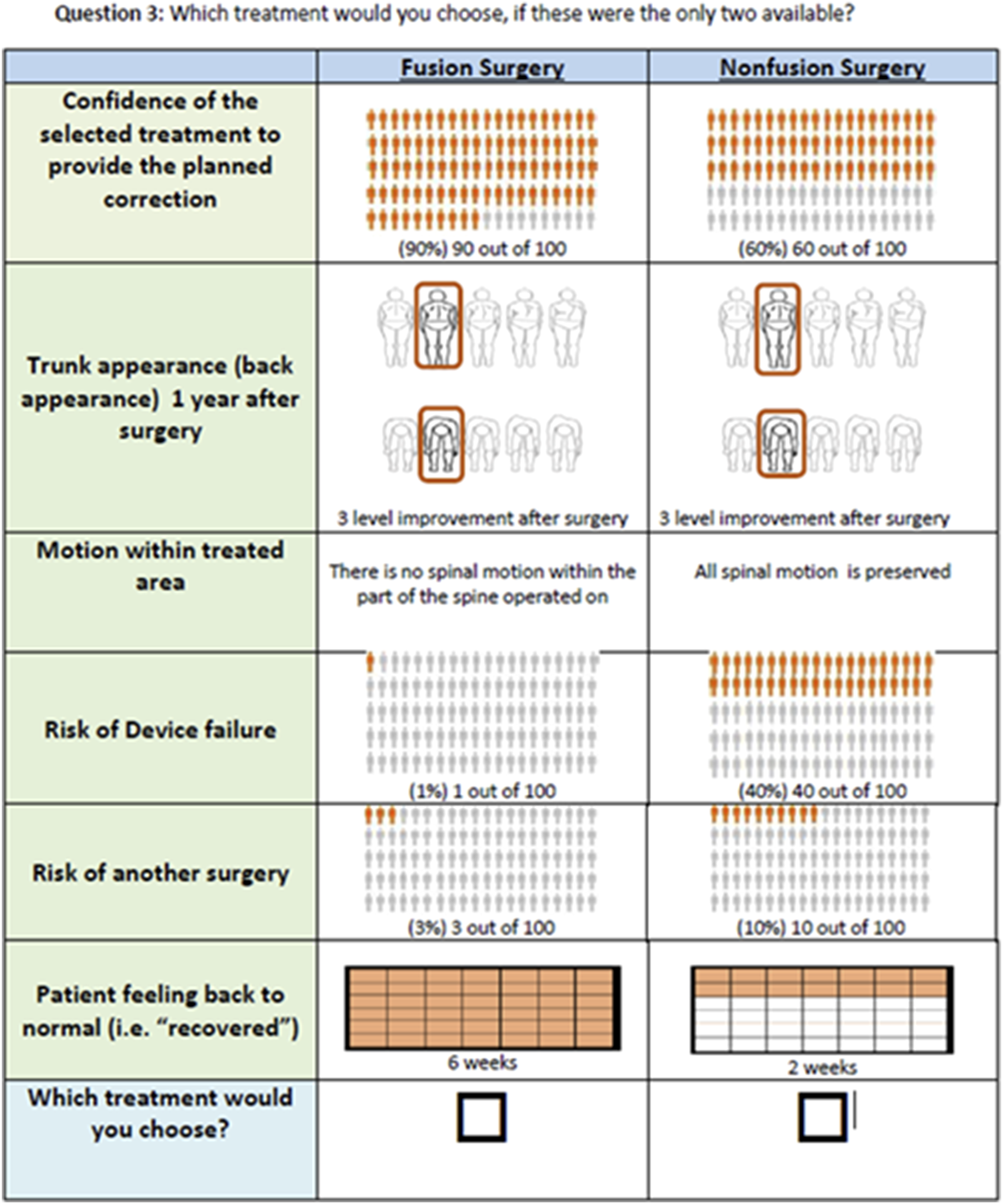
Survey Participants
The DCE was offered to all eligible patients and caregivers evaluated at 7 pediatric scoliosis spine centers distributed across the United States with expertise in both fusion and non-fusion surgery over a 1-year time period. Inclusion criteria were: (1) 10-21-year-old patients who were considering AIS surgery, (2) 10-21-year-old patients who had undergone either fusion surgery or non-fusion surgery for AIS in the past 3 years, and (3) caregivers of the AIS patients. Study participants were recruited in the clinic and the DCE was completed via an online platform either in the clinic space (for those without internet connectivity or online access) or at home via a website link without assistance from research or clinical personnel. Respondents were stratified into 3 cohorts based on who completed the survey: (1) DCE completed by the patient, (2) DCE completed by the caregiver, and (3) DCE completed jointly by the patient and caregiver (dyads). All research activities were Institutional Review Board (IRB) approved at the central study coordination site and at each participating site.
Data Analysis
Three random-parameters logit (RPL) models were used to analyze choices collected from each of 3 respondent groups. Each model regressed choices against differences in the profiles presented in each question. Preference weights were represented as log-odds indicating the rate at which the relative probability of choice changed when a profile included specific attribute levels, all else equal. 27 Cohort-specific preference weights were estimated.
Attribute relative-importance weights for each cohort were calculated from the preference weights for each outcome in the DCE. Preference weights are relative in nature and are not directly interpretable. However, greater differences between preference weights indicate greater changes in the probability of choice. Relative importance weights indicate the maximum impact that changes in the study attributes had on the likelihood of choosing a treatment option in the DCE. The sum of the overall importance of attributes was normalized to add up to 1 within groups. 28
The impact of previous patient experience with fusion or non-fusion surgery was evaluated by including whether this experience affected the relative importance of procedure-type requirements. This was accomplished by generating an interaction term between a dummy-coded variable indicating whether the patient had already completed surgery for scoliosis or not, and the ASC capturing the type of surgery respondents could choose (ie, fusion or non-fusion). New cohort-specific preference models were estimated including the interaction term between the ASC and the patient timing for survey completion (ie, before or after surgery). Significance of this interaction would indicate that completion of the survey after surgery was associated with systematically different preferences for the procedure-type requirements. To assess the magnitude of the interaction effect, we used the estimates with the interaction term to generate expected choice probabilities (ECPs) between fusion and non-fusion surgery by cohort and surgery experience. These ECPs represent the probability of choosing 1 surgery over another by the average respondent in the cohort and experience group, assuming all outcomes are expected to be the same. This had the effect of isolating the impact of procedure-type requirement on choice. Lastly, pairwise correlations of preference weights across cohorts were calculated using a simple Pearson’s correlation coefficient.
Results
Demographics
Survey Respondent Demographics for Adolescent Idiopathic Scoliosis Discrete Choice Experiment Survey.
Relative Preference for Procedure Type Across Respondent Cohorts
Procedure-Type Requirements: Predicted Choice Probabilities by Cohort for Fusion and Non-Fusion Surgery.

a95% confidence intervals are in parentheses.
Treatment Outcome Relative Importance Weights for Different Respondent Cohorts
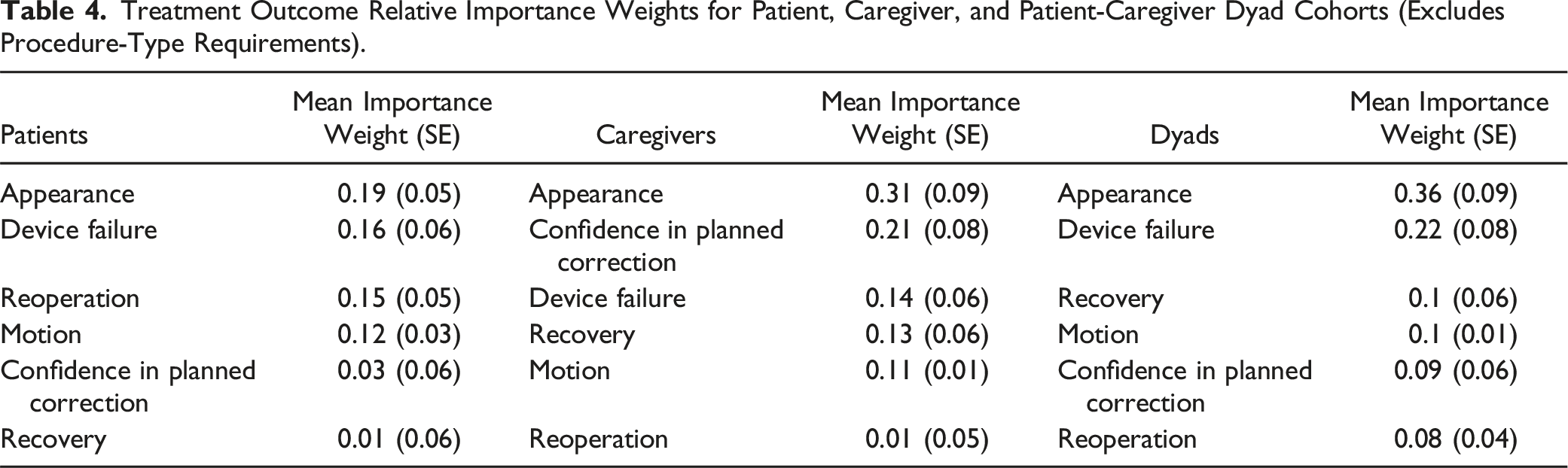
Treatment Outcome Relative Importance Weights for Patient, Caregiver, and Patient-Caregiver Dyad Cohorts (Excludes Procedure-Type Requirements).
Correlations of Preference Weights for Procedure-Type Requirements Across Cohorts
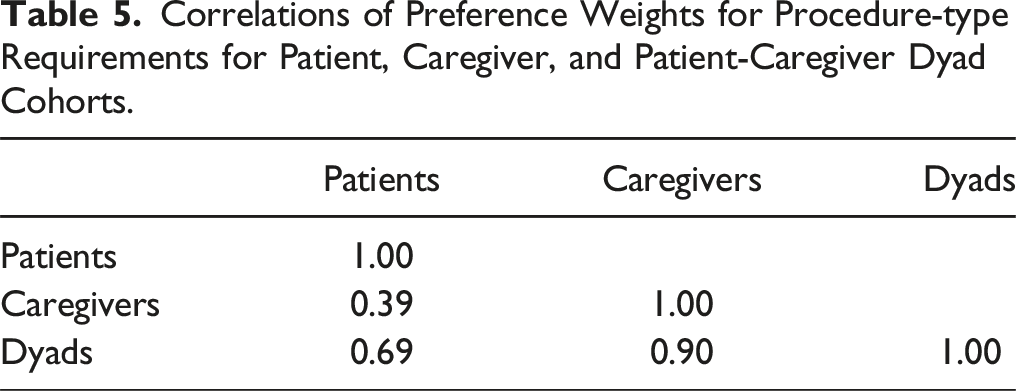
Correlations of Preference Weights for Procedure-type Requirements for Patient, Caregiver, and Patient-Caregiver Dyad Cohorts.
Impact of Completing the Survey Before or After Surgery
Predicted Choice Probability by Cohort and Previous Experience With Scoliosis Surgery Based on Procedure-Type Requirements.
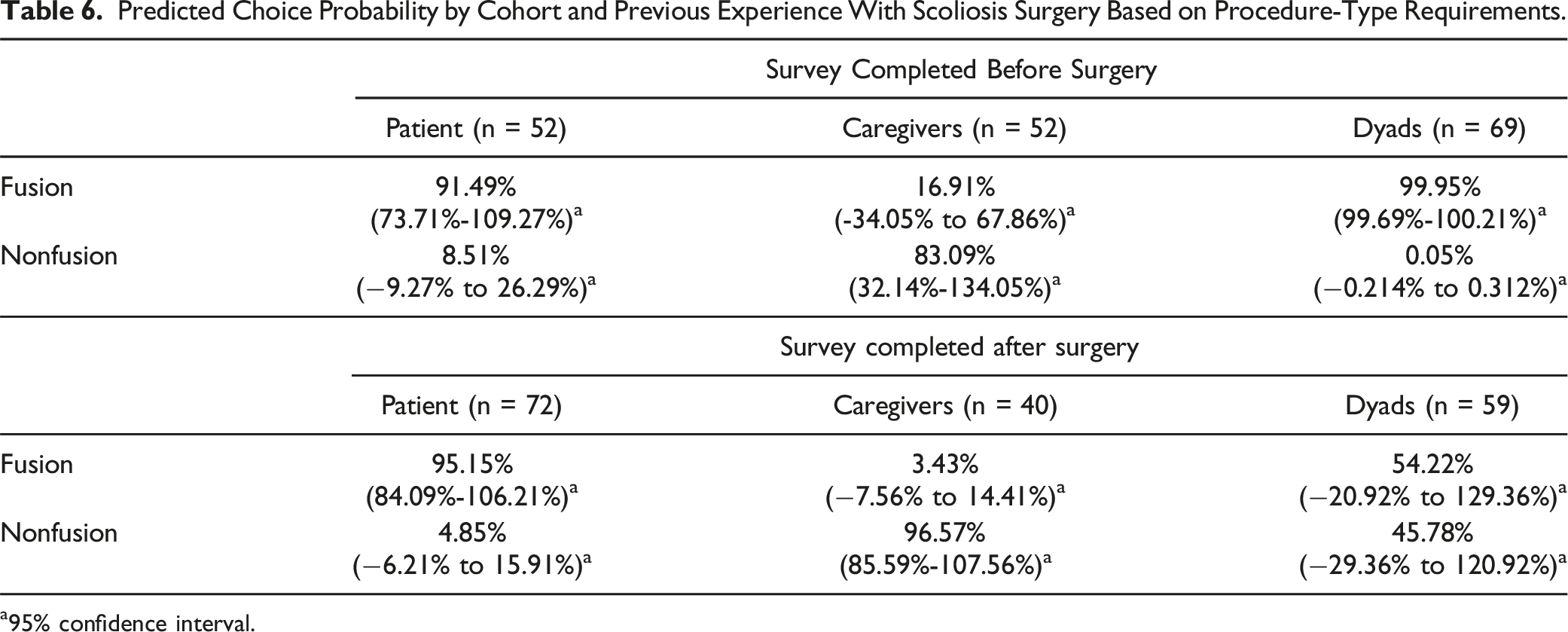
a95% confidence interval.
Discussion
In our study, notable differences were observed among the 3 study cohorts regarding relative preference for fusion vs non-fusion surgery and the attributes that drove these choices. On average, patients and patient-caregiver dyads preferred the procedure-type requirements associated with fusion surgery, while caregivers preferred non-fusion surgery. The average respondent in each cohort identified appearance as the most important treatment outcome attribute that impacted the likelihood of choosing a fusion or non-fusion surgery. For patients and patient-caregiver dyads, the second most important treatment outcome attribute was device failure rate, whereas for caregivers the second most important treatment outcome attribute was confidence in planned correction. For patients, relative preference weights were least influenced by changes in recovery time, whereas relative preference weights of caregivers and patient-caregiver dyads were least influenced by changes in reoperation rates. Correlations of preference weights across the 3 cohorts for procedure-type requirements demonstrated that patient preferences were more highly correlated with the preference of dyads than with the preferences of caregivers. Caregiver preferences are also more highly correlated with the preferences of dyads than with the preferences of patients. These correlations between attribute preference weights of both caregivers and patients with the preference weights of dyads support the result that caregivers are more likely driving the discussions about surgery type when the decisions are considered in dyads. Predicted choices by surgery type for patients and caregivers appeared stable when completing the survey before or after surgery. For dyads, results show that respondents who completed the survey prior to surgery strongly preferred fusion when considering only the procedure-type requirements, while the ECPs for respondents who completed the survey after surgery were more equally apportioned between fusion and non-fusion.
Prior literature regarding preferences related to surgery for adolescent idiopathic scoliosis has focused on key attributes identified for posterior fusion surgery from the perspectives of patients, parents, and health care providers 29 and evaluation of parents’ and patients’ preferences and concerns regarding anterior, posterior, and combined fusion surgery. 30 Our study is the first to quantitatively explore preferences for treatment of adolescent idiopathic scoliosis with fusion vs non-fusion surgery from the perspectives of patients, caregivers, and patient-caregiver dyads using the same survey instrument. The present study also offers a novel approach to assess the decision dynamics between patients and caregivers and sheds light on the decision makers who are influencing treatment decisions in this population. Medical decision-making regarding AIS surgery is complex due to proxy decision-making by caregivers and the developmental maturation and increasing decision-making capabilities of adolescent patients over time which allows for their inclusion in treatment decisions. 31 Surgery for AIS is most frequently undertaken to address concerns regarding body image due to trunk asymmetry and to prevent future adverse consequences related to curve progression including back pain, increased deformity, and pulmonary symptoms in patients with large thoracic curves. However, long term natural history studies have shown that the majority of patients with untreated moderate AIS function well in society as adults with respect to employment, marriage including childbearing, and maintenance of activity levels into older adulthood. 32 Both fusion and non-fusion treatment of AIS are not without risks which include postoperative pain, infection, device failure, reoperation, and neurologic injury. Balancing the acute and long-term benefits and risks of surgical intervention pose challenges for adolescents and their caregivers when treatment preferences and perspectives on the roles each has in decision making differ, 33 particularly in light of concerns about respecting adolescents’ autonomy. 34 Priorities of each stakeholder might differ, with adolescents potentially more concerned about short-term outcomes, whereas caregivers might focus on long-term impacts of treatment decisions. 35 There exists a need for additional future research regarding intergenerational decision making related to treatments for AIS 36 and the authors are currently developing a shared decision-making tool as a pragmatic way to facilitate communication among patients, caregivers, and clinicians considering surgical treatment.
This study has several notable strengths and limitations. Regarding strengths, this study was designed by a multidisciplinary team comprised of a patient advocacy group, scoliosis experts, patients with scoliosis and their families, and FDA staff, and undertaken in accordance with recommended best practices for patient-preference studies. The study included both preoperative and postoperative patients with AIS who were considering surgical options as well as patients who had undergone fusion or non-fusion surgery in order to evaluate the diverse range of patients encountered in clinical practice. Limitations of this study included variability in administration of the DCE as it was completed by either the patient, caregiver or patient-caregiver dyad, and separate surveys were not completed by each, and versions of the survey were not randomized across respondents. This cross-sectional study design was selected as it was considered representative of real-world decision-making and minimized respondent burden and disruption of clinical workflow. Also, the patients and caregivers who entered this study were selected from scoliosis centers where surgeons had extensive experience performing both fusion and non-fusion surgery which may not be representative of all hospitals where scoliosis surgery is performed. Lastly, decisions regarding surgery were subject to influence by a range of sources including discussions with clinicians involved in the study and their care team members, non-study health care providers, other patients and family members, the internet and social media. It is not possible to determine how informed respondents were about the treatment options when they completed the survey, but in general, patients and their caregivers had access to many resources including scoliosis patient education web sites and web-based patient support groups. We attempted to minimize disparities or gaps in respondents’ understanding of their options by providing pre-survey education.
Conclusions
Patients and patient-caregiver dyads preferred the procedure-type requirements associated with fusion surgery, while caregivers preferred the procedure-type requirements associated with non-fusion surgery. The most important treatment outcome for patients, caregivers, and patient-caregiver dyads related to surgery for adolescent idiopathic scoliosis was trunk (back) appearance after surgery.
Footnotes
Acknowledgments
The Harms Study Group Investigators include Aaron Buckland, MD, Melbourne Orthopaedic Group & Royal Childrens Hospital; Ahmet Alanay, MD, Acibadem Maslak Hospital; Amer Samdani, MD, Shriners Hospitals for Children, Philadelphia; Amit Jain, MD, Johns Hopkins Hospital; Baron Lonner, MD, Mount Sinai Hospital; Benjamin Roye, MD, Columbia University; Bob Cho, MD, Shriners Hospitals for Children, Pasadena CA; Burt Yaszay, MD, Seattle Children’s Hospital; Caglar Yilgor, MD, Acibadem Maslak Hospital; Dan Hoernschmeyer, MD, University of Missouri Health Care; Daniel Hedequist, MD, Boston Children’s Hospital; Daniel Sucato, MD, Texas Scottish Rite Hospital; David Clements, MD, Cooper Bone & Joint Institute; Firoz Miyanji, MD, BC Children’s Hospital; Harry Shufflebarger, MD, Paley Orthopedic & Spine Institute; Jack Flynn, MD, Children’s Hospital of Philadelphia; Jean Marc Mac-Thiong, MD, CHU Sainte-Justine; Josh Murphy, MD, Children’s Healthcare of Atlanta; Joshua Pahys, MD, Shriners Hospitals for Children Philadelphia; Keith Bachmann, MD, University of Virginia; Kevin Neal, MD, Nemours Children’s Clinic Jacksonville; Laurel Blakemore, MD, Pediatric Specialists of Virginia/Children’s National; Lawrence Haber, MD, Ochsner Health Center for Children New Orleans; Lawrence Lenke, MD, Columbia University; Mark Abel, MD, University of Virginia; Mark Erickson, MD, Children’s Hospital Colorado; Michael Glotzbecker, MD, Rainbow Children’s Hospital; Michael Kelly, MD, Rady Children’s Hospital; Michael Vitale, MD, Columbia University; Michelle Marks, PT, MA, Setting Scoliosis Straight Foundation; Munish Gupta, MD, Washington University; Nicholas Fletcher, MD, Emory University; A. Noelle Larson, MD, Mayo Clinic; Patrick Cahill, MD, Children’s Hospital of Philadelphia; Paul Sponseller, MD, Johns Hopkins Hospital; Peter Gabos, MD, Nemours/Alfred I. duPont Hospital for Children; Peter Newton, MD, Rady Children’s Hospital; Peter Sturm, MD, Cincinnati Children’sHospital; Randal Betz, MD, Institute for Spine & Scoliosis; Stefan Parent, MD, CHU Sainte-Justine; Stephen George, MD, Nicklaus Children’s Hospital; Steven Hwang, MD, Shriners Hospitals for Children Philadelphia; Suken Shah, MD, Nemours/Alfred I. duPont Hospital for Children; Sumeet Garg, MD, Children’s Hospital Colorado; Tom Errico, MD, Nicklaus Children’s Hospital; and Vidyadhar Upasani, MD, Rady Children’s Hospital.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided through FDA BAA- HDDISWP#83. NOTE: In addition to the above funding, this study was funded in part by grants to the Setting Scoliosis Straight Foundation in support of Harms Study Group research from DePuy Synthes Spine, EOS imaging/Atec, Stryker Spine, Medtronic, NuVasive, ZimVie, and Biedermann Motech.
IRB approval
IRB approval was obtained for all aspects of this study (Western IRB study number 1281165, tracking number 20200565).
Ethical Statement
Data Availability Statement
Authors are not authorized for participation in research data sharing.
