Abstract
Study Design
Exploratory prospective observational cohort.
Objectives
Aim of this study was to evaluate clinical and radiologic outcome, and surgical efficiency of a novel universal delivery system that allows full-percutaneous extraforaminal, trans-Kambin lumbar interbody fusion with a large-footprint lumbar interbody cage using only fluoroscopic imaging and open-surgery instrumentation.
Methods
We prospectively evaluated patients that underwent elective trans-Kambin TLIF surgery with a large-footprint interbody cage using a novel universal delivery system. Clinical follow-up was evaluated pre-and post-operatively with Visual Analogic Scale (VAS) and Oswestry Disability Index (ODI) scores, while radiologic follow-up was performed with a computed tomography scan and standing films post-operatively at hospital discharge and 1 year follow-up.
Results
A total of 47 patients were evaluated. Clinically, post-operative VAS and ODI scores significantly (P < 0.001) improved compared to pre-operative scores and 29.4 months mean follow-up. Radiologic evaluation yielded an intervertebral fusion rate of 90% of the operated levels and a significant increase in segmental lordosis by 3.7°. Median surgical time for interbody cage insertion per level was 28 minutes. Complications included 14 (29%) cases with transitory post-operative radiculitis, 4 (8%) cases with partial muscle weakness and 2 (4%) cases that required revision surgery. Post-operative ambulation started at a median 5 hours and median hospital length of stay was 28 hours.
Conclusions
A new universal delivery system allows overcoming most limitations of current full-endoscopic trans-Kambin fusion as it allows a time- efficient full-percutaneous insertion of a large-footprint interbody cage under fluoroscopy imaging only, with standard open-surgery instruments and optional endoscopic visualization.
Keywords
Introduction
Full-endoscopic spine surgery has seen widespread adoption in the last decade due to excellent anatomic visualization, decreased intra-operative blood loss and post-operative pain, leading to early patient mobilization and hospital discharge.1-8 Early patient mobilization significantly reduces the rate of complications and morbidity along with the average hospital length of stay.9,10 Recent studies have shown the superior cost-effectiveness and non-inferior clinical outcome of spinal endoscopic procedures.11,12
The first reported and still most widely adopted approach for full-endoscopic/percutaneous transforaminal lumbar interbody fusion (TLIF) surgery 13 is a uniportal approach to Kambin’s triangle that allows safely bypassing the exiting nerve root and insertion of an interbody cage into the lumbar disk (trans-Kambin approach).1-8,13 Full-endoscopic, trans-Kambin TLIF has seen an increasing adoption in the last decade.1,2 Advantages of trans-Kambin TLIF comprise an approach without soft tissue dissection, minimal blood loss and risk of infection, and a rapid, opioid-free post-operative recovery 8 with early hospital discharge (out-patient surgery)1-8,13. However, its wide-spread acceptance is still hindered by 3 important limitations: first, endoscopic visualization is mandatory for most trans-Kambin fusion systems, which implies investment in expensive endoscopic equipment and instrumentation, as well as overcoming the steep learning curve of endoscopic spine surgery. 14 Second, most full-endoscopic trans-Kambin systems use a tubular sleeve for endoscopic disk access and endplate preparation. The tubular sleeve’s and/or the endoscope’s working channel diameter is usually small (less than 12 mm), representing a main constraint to the size of an interbody cage that can be placed through that tube. Therefore, expensive, small-footprint interbody cages with an intricate expansion mechanism for interbody fusion are employed in most full-endoscopic fusion systems. Third, also the size of the disk preparation instruments is usually small as they must fit through a small endoscopic working channel. Small disk preparation instruments are expensive and frail, and it takes more surgical time with small instruments to complete a disk preparation.
The main aim of this study is to evaluate the clinical and radiologic outcome, and surgical efficiency of a novel universal delivery device that was developed to allow an efficient, full-percutaneous insertion of a large-footprint lumbar interbody cage with regular open-surgery instrumentation under fluoroscopic guidance only (endoscopic visualization is optional). Our hypothesis is that this novel device allows overcoming the mentioned limitations of current full-endoscopic/percutaneous fusion procedures.
Methods
Ethics Statement
This study was conducted according to principles of the Declaration of Helsinki and was approved by the ethics committee of our institution’s Institutional Review Board (approval number: 2023107-COT-CMT). All patients provided verbal and written consent for participation in the study.
Study Design
This is an exploratory prospective study with consecutive cases that underwent elective percutaneous TLIF surgery at 1 facility.
Study Population
Patients presenting with low back pain and/or associated uni- and/or bilateral leg pain were evaluated in our facility. Pre-operative assessment followed a standard protocol which included physical examination, pre-operative magnetic resonance imaging (MRI) and/or Computer Tomography (CT) scan of the lumbar spine. Indications for surgery comprised degenerative disk disease of the lumbar spine, spondylolisthesis up to grade 2 and foraminal stenosis. A corresponding degenerative alteration had to be confirmed by imaging of the lumbar spine.
Inclusion Criteria
(I) Patients aged 18 years and older with no previous lumbar spine surgery, (II) with an indication for percutanoues TLIF, (III) with percutaneous posterior fixation with transpedicular screws and (IV) limited to a one-, two- or three-level fusion surgery of the lumbar spine.
Exclusion Criteria
Patients with spondylolisthesis grade >2, neoplasm, trauma, infection, systemic diseases and not complying with the inclusion criteria.
Surgical Technique
Position, anesthesia and equipment setting
The patient was placed in prone position on a radiolucent Wilson frame. All patients underwent general anesthesia with intra-operative neuromonitoring. Somatosensory evoked potentials (SEP) and motor evoked potentials (MEP) were employed to monitor involved peripheral nerves. If required, direct probe stimulation was performed. Nerve root distance was considered acceptable at signal intensities ≥10 mA. The fluoroscopic C-arm was placed opposite to the main surgeon’s side.
Wound incision and placement
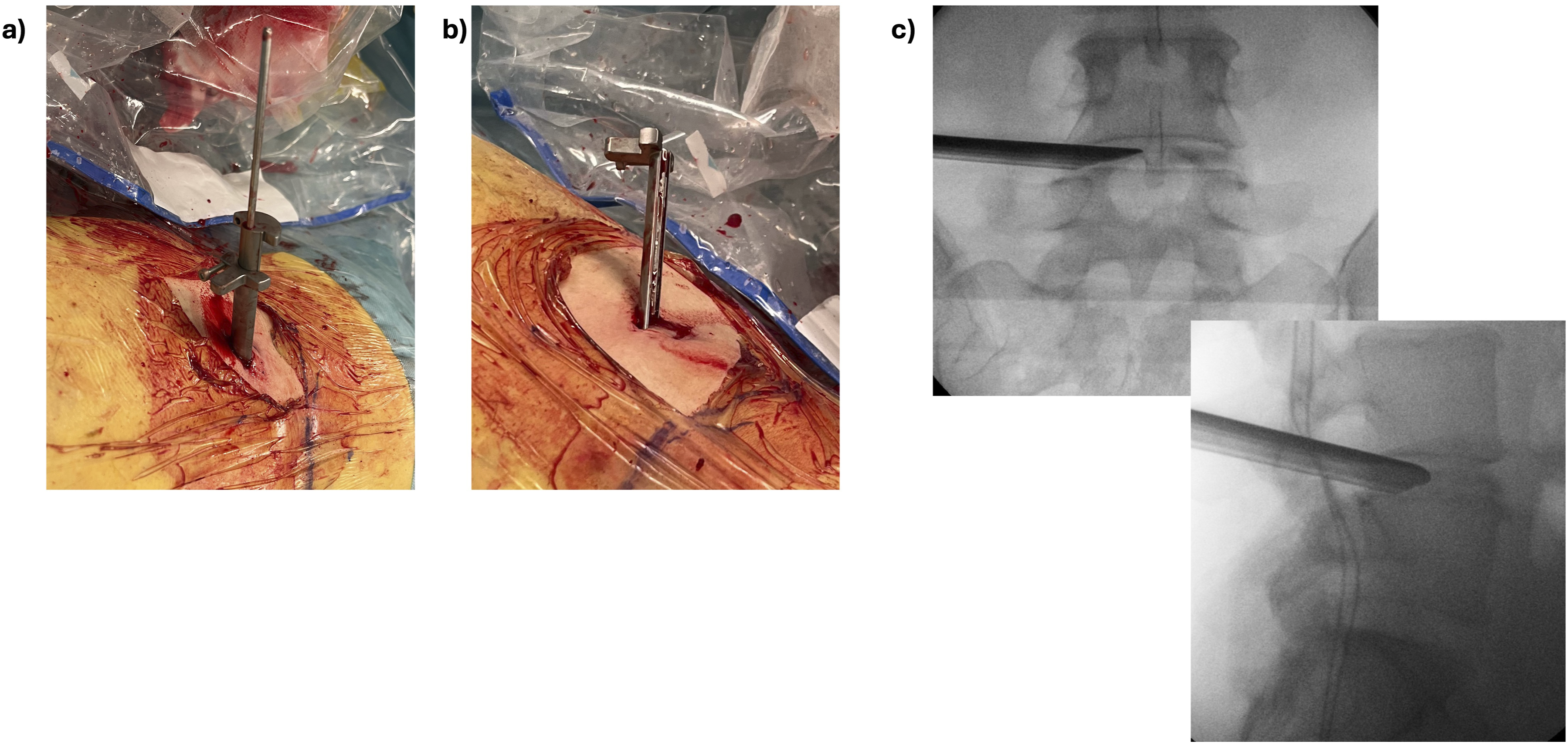
The corresponding pathological level was located, and the trajectory was estimated intra-operatively by fluoroscopic imaging. A 1.5 to 2 cm skin incision was made lateral to the midline on the side of the patient’s dominant radiculopathy. An 18G needle was advanced into the disk with an approximate 55 to 65° angle under fluoroscopic imaging using a standard trans-Kambin approach,5,7,15 see Figure 1A. A discography was performed with a 9:1 diluted solution of Ioperamide contrast and Indigo-Carmine colorant. A guiding wire was placed into the disk and the needle was retrieved. Soft tissues were progressively dilated with guided dilators. Then, a specially designed dilator with an incorporated universal delivery sheet (Maxfusion system, Hoogland Spine Products GmbH, Munich, Germany) was placed into the disk under fluoroscopic imaging only, see Figure 1B and C. The dilator was then retrieved, see Figure 2A, leaving the universal delivery sheet anchored inside the disk, Figure 2B and C. The delivery sheet is always placed in the caudal aspect of the foramen and facing cranially to protect the exiting nerve root
16
, see Figure 2C, and remains in place until successful completion of the interbody cage placement. Intra-operative imaging of (A) a 2.0 mm small dilator has been inserted using a guiding wire into the disk via Kambin’s triangle (B) fluoroscopic A/P and lateral views of the small dilator inside the disk (C) placement of the universal delivery dilator that includes the protection sheet using the small dilator as accessory guide (D) fluoroscopic lateral view of the dilator at the lateral border of the disk (E) an annulotomy has been performed by placing the dilator inside the disk (F) fluoroscopic lateral view showing the dilator inside the disk. (A) decoupling of the dilator and the protective sheet. (B) view of the sheet after completely retrieving the dilator. The sheet is placed in the caudal part of the foramen and facing to the cranial part of the foramen in order to protect the exiting nerve root (C) fluoroscopic A/P and lateral views of the sheet anchored inside the disk in the caudal part of the foramen.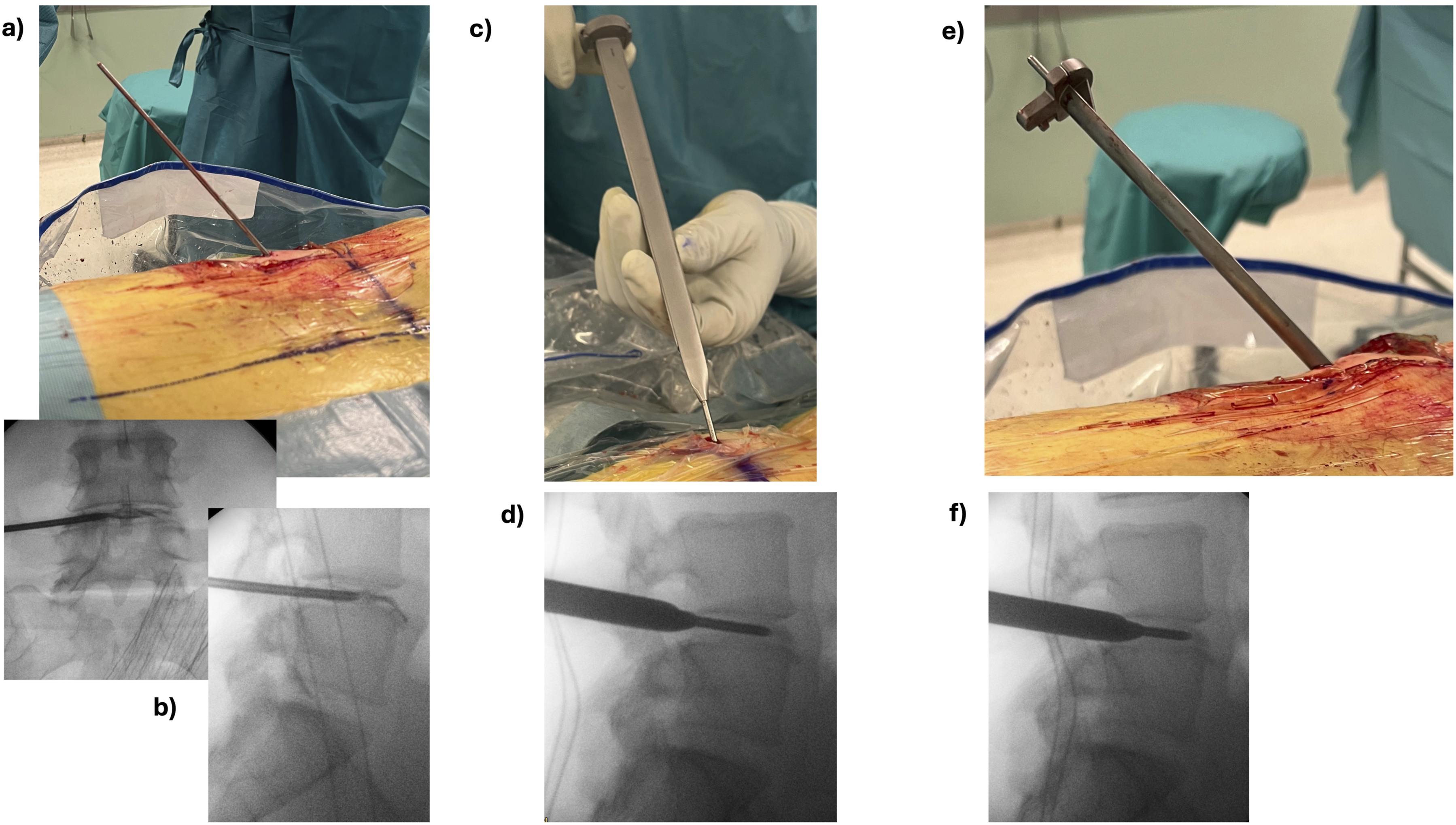
Foraminoplasty
A foraminoplasty was progressively performed with manual reamers (Maxmorespine, Hoogland Spine Products GmbH, Munich, Germany) under protection of the delivery sheet, see Figure 3. The reamers were employed to progressively ream the superior articulating process (SAP). The foraminoplasty usually starts with a 6 mm diameter reamer. The reamer is advanced on the delivery sheet under fluoroscopic imaging until reaching the disk. Then, the reamer is retrieved and a new reamer with progressively increasing diameter in 1 mm steps is applied, until reaching up to 12 mm of diameter, see Figure 3. (A) manual reamer with a blunt tip. The reamer is placed on the sheet that serves as a guide into Kambin’s triangle. The reamer only cuts when turned clockwise and is used to progressively ream the SAP until reaching the disk. (B) fluoroscopic lateral view of the reamer that has reached the lateral aspect of the disk while the sheet is protecting the exiting nerve root.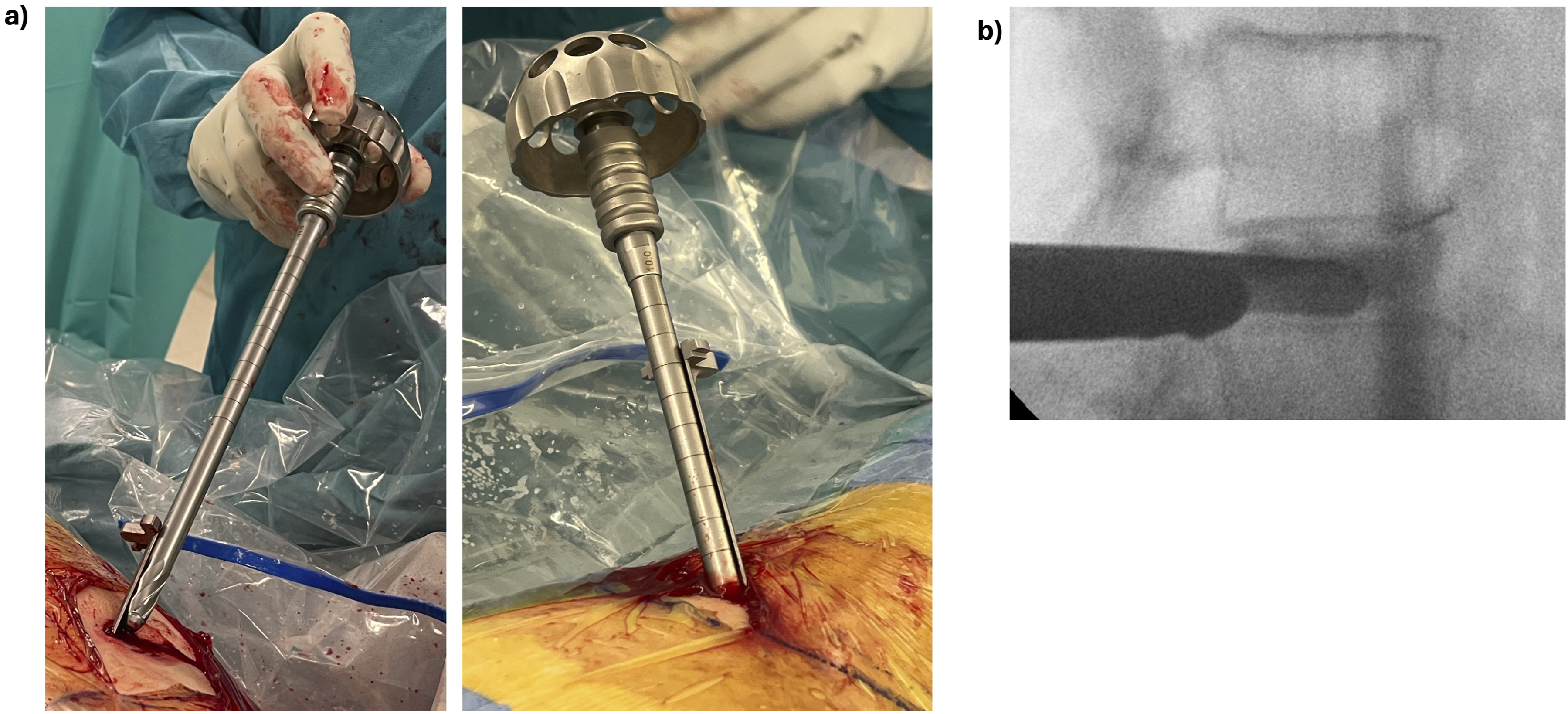
In selected cases with a challenging anatomy, ie, severe foraminal stenosis, foraminal osteophytes, extremely collapsed disks with disk height <5 mm and/or approaches to L5/S1 on males with a very high iliac crest, an additional foraminoplasty can be performed using a high-speed burr (Nouvag GmbH, Switzerland) under endoscopic visualization (Maxmorespine, Hoogland Spine Products GmbH, Munich, Germany). Foraminoplasty under endoscopic visualization consisted in completely removing the SAP until reaching the articular joint (tip of the SAP) cranially and the yellow ligament medially. The yellow ligament was usually preserved as a protecting layer for the traversing nerve root.
Disk and endplate preparation
After completion of the foraminoplasty, nucleotomy was performed using regular-sized punches under fluoroscopic imaging and haptic feedback, see Figure 4A. In collapsed disks, paddle distractors were employed to expand the disk space. After a satisfactory disk preparation, endplates were prepared with curettes under fluoroscopic imaging, see Figure 4B. Endoscopic visualization remains optional for the whole disk and endplate preparation procedure, but it could be employed to visually check the final endplate preparation. (A) fluoroscopic A/P and lateral views of a forceps inside the disk performing nucleotomy. The forceps is being guided into the disk by the sheet that is protecting the exiting nerve root (B) fluoroscopic A/P and lateral images showing straight and angled curettes, respectively for endplate preparation (C) fluoroscopic A/P image showing a graft injector inside the disk (D) photo of removed disk material after disk and endplate preparation using the trans-Kambin approach.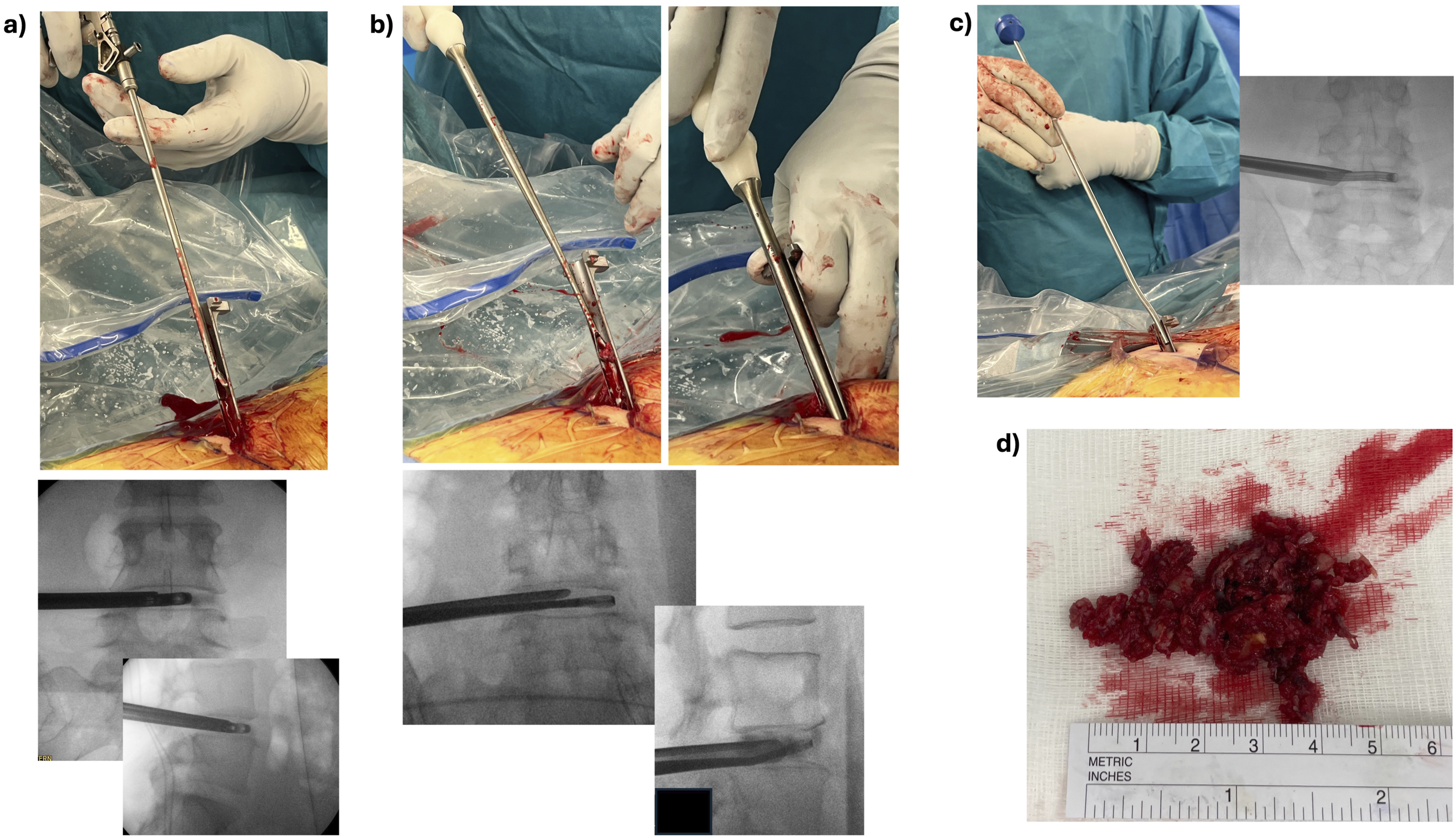
Bone grafting, cage insertion and percutaneous screw insertion
Bone graft was injected into the disk using a graft injector under fluoroscopic imaging, see Figure 4C. In this study, Demineralized-Bone Matrix (DBM) was systematically employed as bone graft material for all cases. A DBM-packed large-footprint (size 39 × 13 × 15 mm, 13° lordosis) titanium interbody cage (Vertaconnect, Signus Medizintechnik GmbH, Alzenau, Germany) was placed into the disk under fluoroscopic imaging, see Figure 5. In the case of a small lumbar disk as measured in pre-operative imaging, a smaller cage (32 × 9 × 9 mm) (Avenue-P Exp Ti, Zimvie Inc. USA; Juliet TO, Spineart SA, Switzerland) was employed. (A) a DBM-packed large-footprint interbody cage is placed on the sheet prior to insertion into the disk (B) fluoroscopic A/P and lateral views of the cage on the lateral aspect of the disk guided by the sheet (C) final view of the inserted cage. Note the sleeve protecting the exiting nerve root during the whole cage placement procedure (D) fluoroscopic A/P and lateral views of the cage placed inside the center of the disk (symmetric view).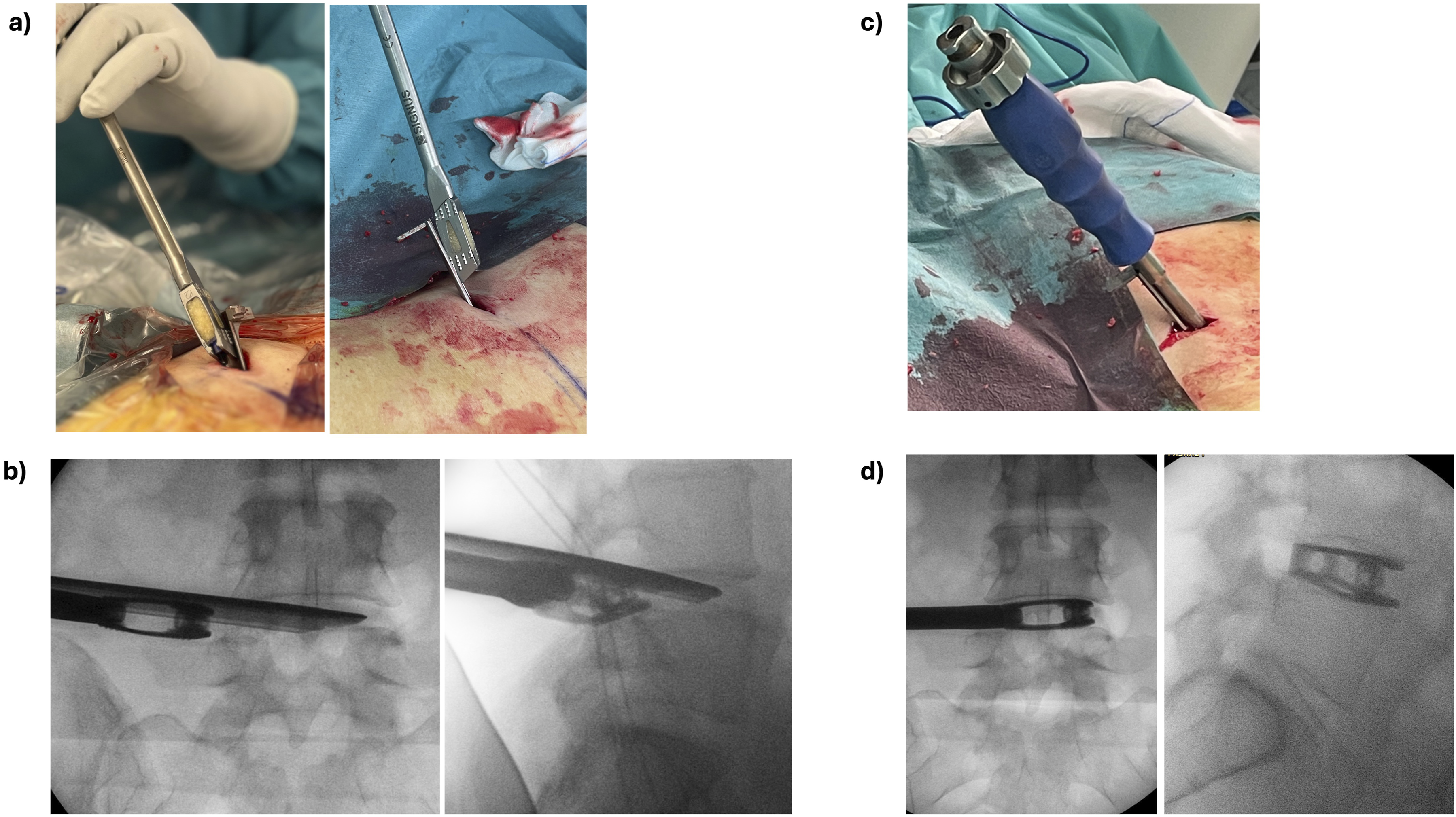
The cage placement is complemented with a percutaneous posterior spinal fixation, see Figure 6 A and C, consisting of transpedicular screws and rods, (ROMEO 2 MIS, Spineart SA, Switzerland). In cases with severe osteoporosis, screw augmentation was performed with polymethyl-methaacrylate (PMMA). Lordosis and sagittal alignment were considered during the fusion procedure and the patient’s position was adjusted correspondingly. In cases with neurogenic claudication and symptomatic severe central canal stenosis, an endoscopic unilateral laminectomy for bilateral decompression was additionally performed at the symptomatic level. (A) Standing post-operative X-rays films showing A/P and lateral views of a case with a one-level interbody cage and posterior fixation with transpedicular screws at L4/L5 (B) photo of the patient’s back showing immediately post-operative wounds. Note the 2.5 cm skin incision on the lateral side employed for the trans-Kambin cage placement (C) Standing post-operative X-rays films showing A/P and lateral views of a case with two-level interbody cages and posterior fixation with transpedicular screws at L4/L5 and L5/S1 (D) photo of the patient’s back showing immediately post-operative wounds. Also in this case only one 2.5 cm skin incision on the lateral side (marked in a red circle) was employed to place both cages using the trans-Kambin approach.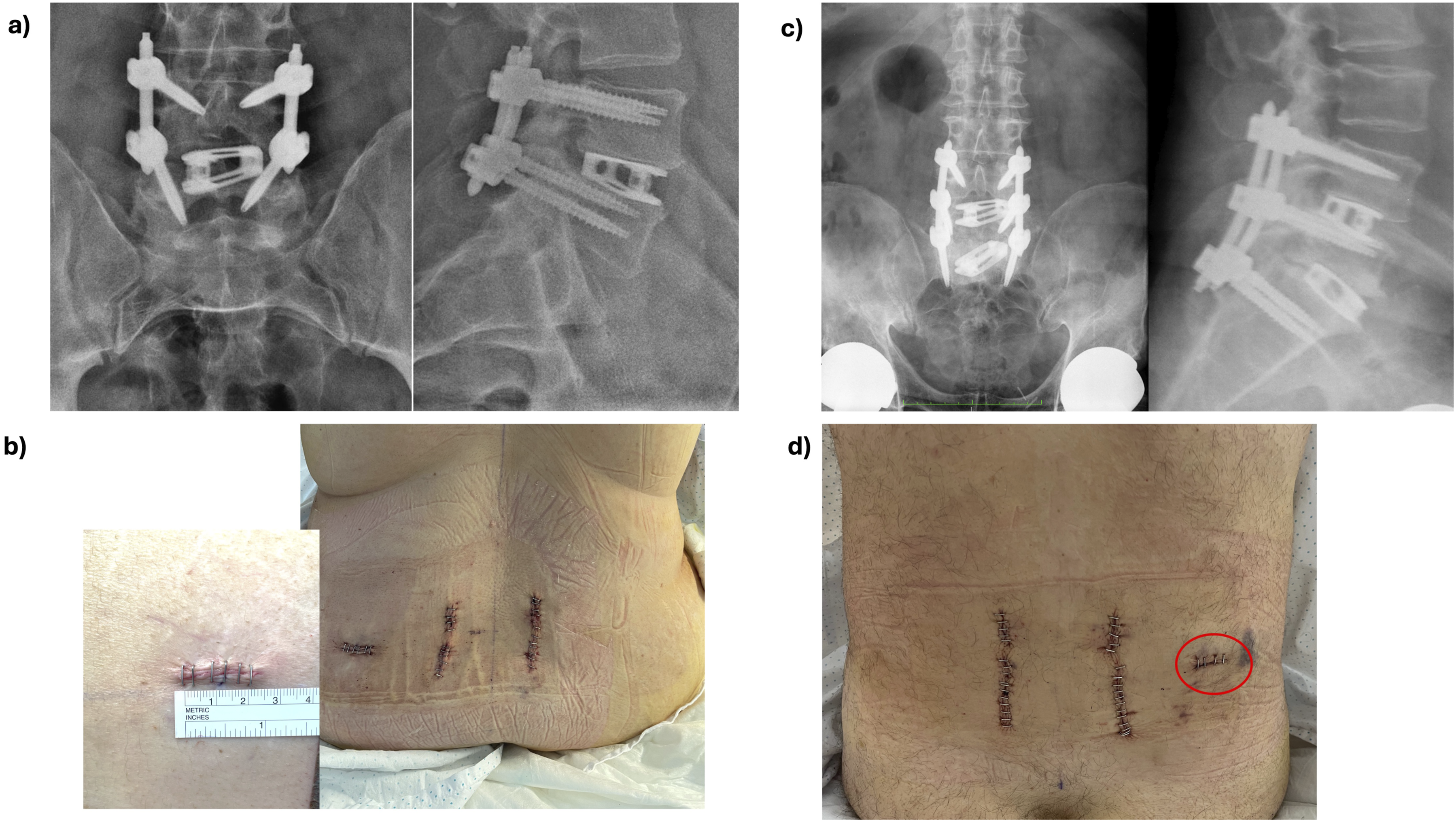
Finally, fascia, subcutaneous tissue and skin were sutured, see Figure 6 B and D.
Clinical and Radiologic Outcome Measurement
Pre-operative and post-operative Visual Analogic Scale (VAS) for lumbar back and leg pain were quantitatively assessed (range zero to 10). Oswestry Disability Index (ODI) scores (range zero to 50) 17 were pre-operatively assessed. Post-operative follow-up controls were established at hospital discharge, 1 month, 3 months, 6 months, 1 year and thereafter successively every 12 months. Time of first post-operative patient ambulation (hours), and hospital length of stay was registered.
A computer tomography scan was taken in the immediate postoperative period (within 24 hours after surgery) and at 1 year post-operatively. Conventional standing X-ray films were performed at 6 weeks and 1 year after surgery. Bony interbody fusion rates were radiologically evaluated at 1-year post-op with a CT-scan and a standard film. Bone fusion was confirmed with presence of trabecular bone in the CT scan imaging, see Figure 7. (A, B) Post-operative CT scan images at 1 year post-op showing successful intervertebral fusion with trabecular bone inside a large-footprint interbody cage in the coronal (left image) and lateral (right image) CT scan cuts for 2 cases, respectively.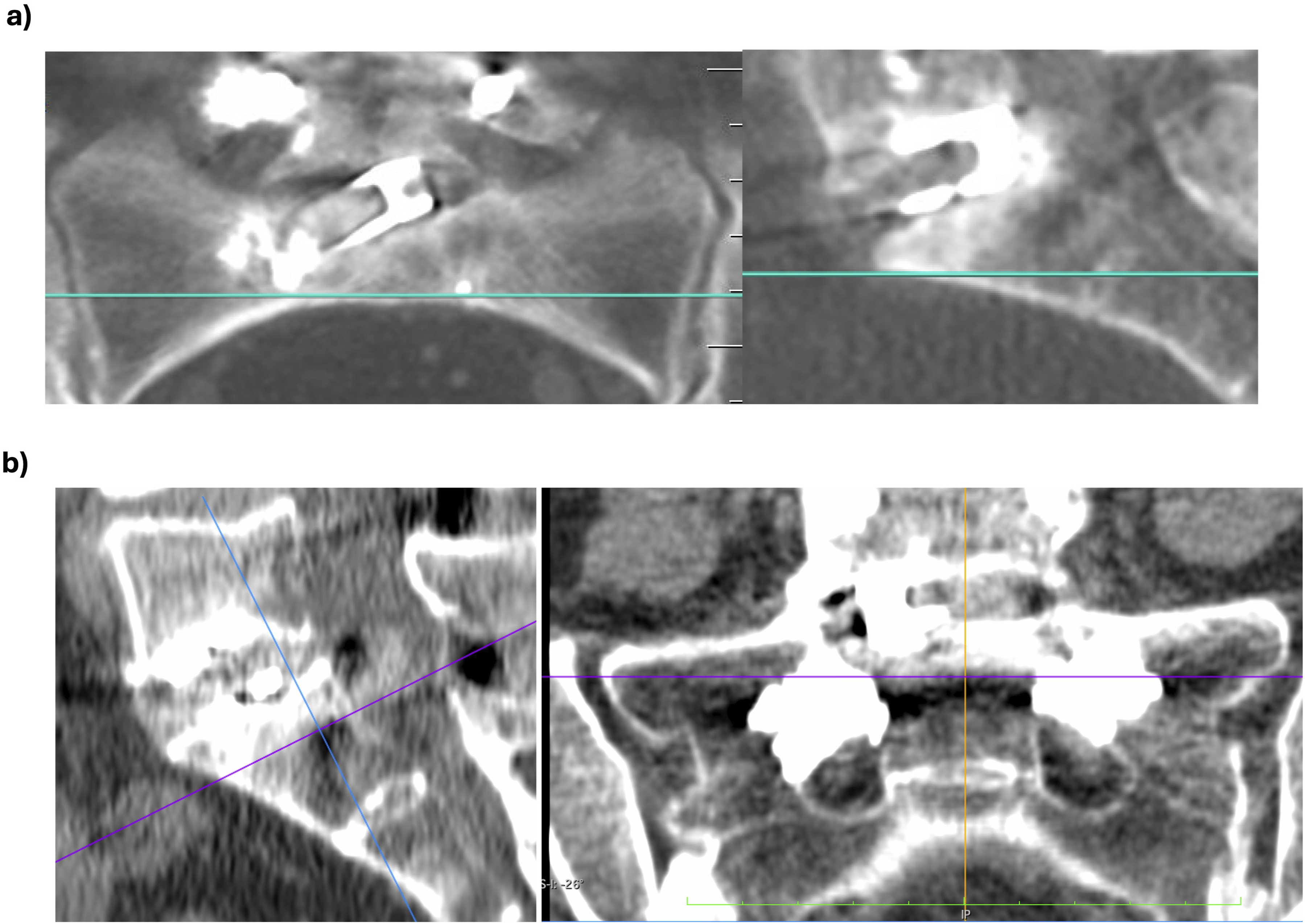
Statistical Analysis
Kolmogorov-Smirnov test was employed to evaluate normal distribution of the data series. Paired student’s t test was employed to statistically compare intra-cohort variables. Statistical significance was defined at P < 0.01.
Results
Demographics and Surgery Data
Demographics and Surgical Characteristics of 47 Patients.

SD: standard deviation.
Clinical Outcome
Pre- and Post-operative Clinical Outcome for 47 Patients After Percutaneous TLIF Surgery.
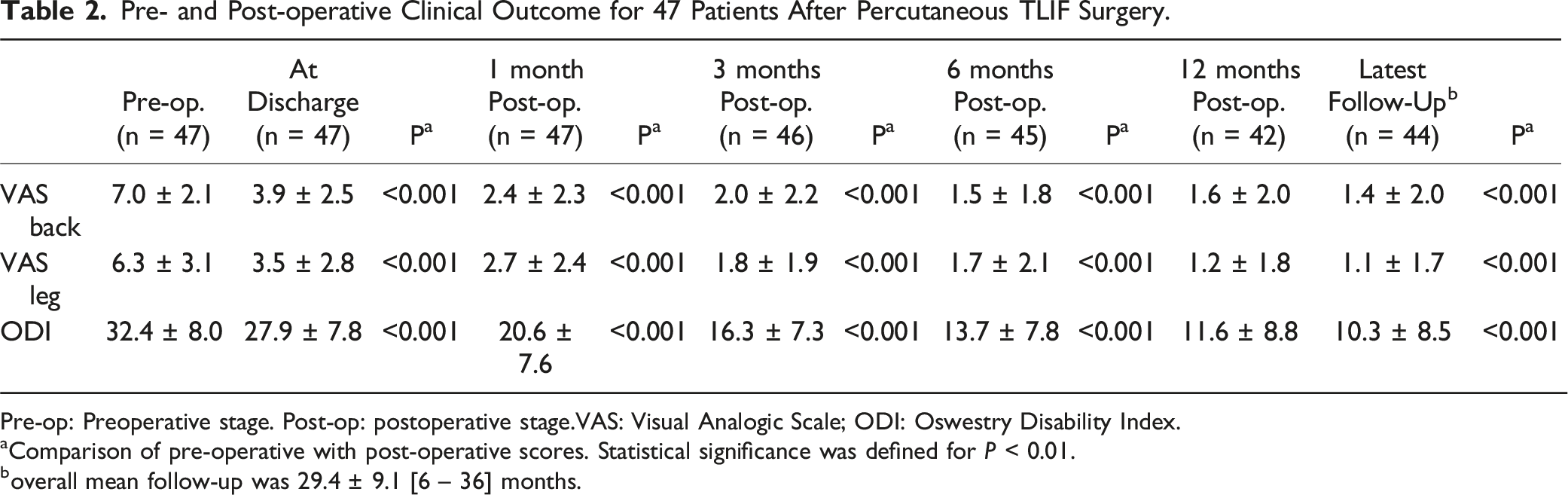
Pre-op: Preoperative stage. Post-op: postoperative stage.VAS: Visual Analogic Scale; ODI: Oswestry Disability Index.
aComparison of pre-operative with post-operative scores. Statistical significance was defined for P < 0.01.
boverall mean follow-up was 29.4 ± 9.1 [6 – 36] months.
Total surgery duration and surgical time for interbody cage placement are shown in (Table 1). Post-operative time to first ambulation and median total hospital length of stay are detailed in (Table 1).
Radiologic Outcome
Radiologic Outcome of the 47 Operated Patients.

SD: standard deviation; LLA: Lumbar Lordotic Angle; SLA: Segmental Lordosis Angle; PI: pelvic incidence; LL: Lumbar Lordosis.
astatistical significance defined at P < 0.01.
bstatistical significance of values between the ALIF and pTLIF group (inter-group differences).
cstatistical significance between pre- vs post-operative values of 1 group (intra-group differences).
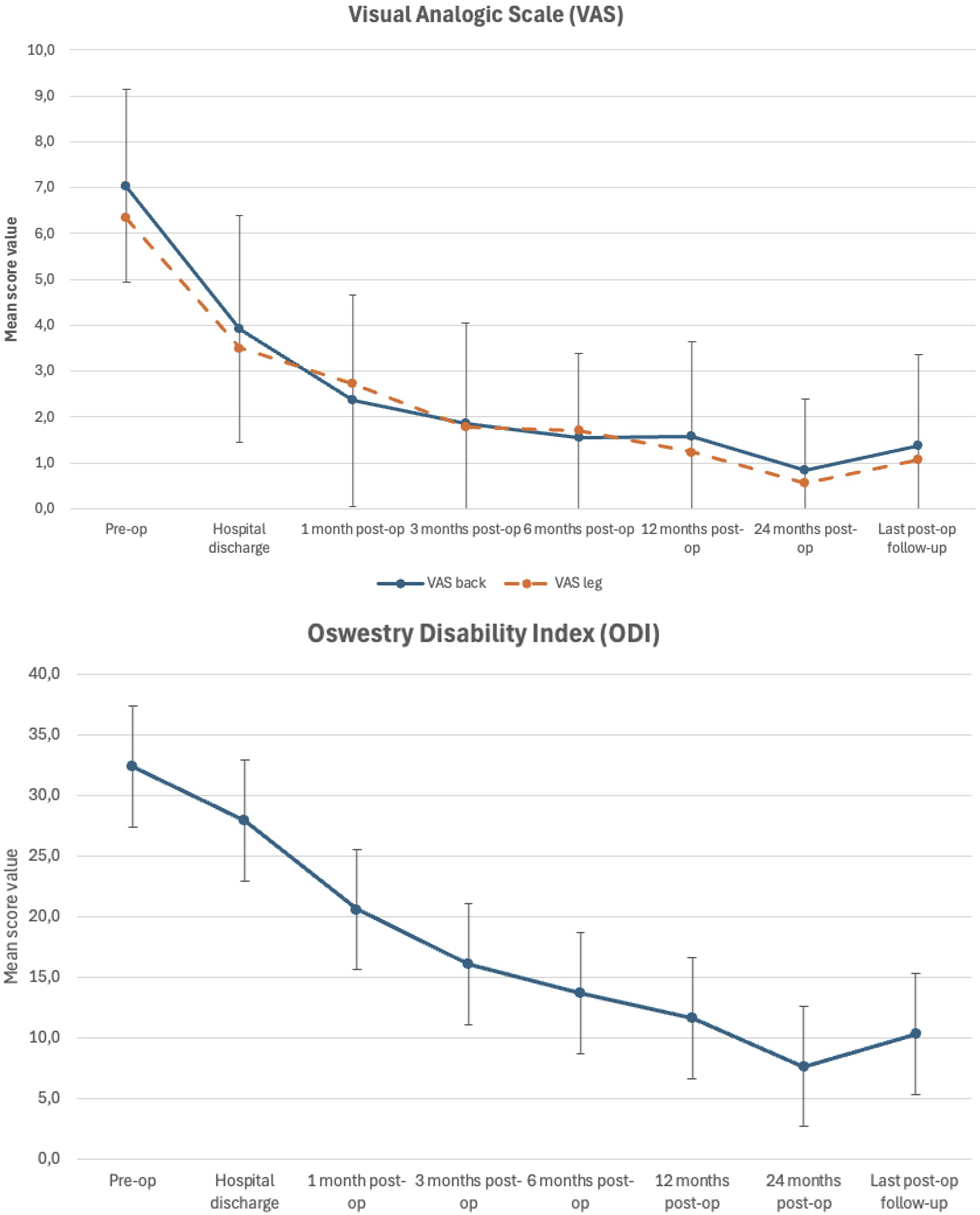
Error bar plot displaying mean ± standard deviation of pre-operative and post-operative VAS back and leg scores (upper image) as well as ODI scores (lower image) in relationship to post-operative follow-up.
Complications
Post-operative transitory radiculitis was reported by 14 patients (29%) at L4/L5 (1 case), L5/S1 (8 cases) and by two-level cases (5 cases). Bilateral post-operative radiculitis was observed for 5 (36%) of these cases. No relevant changes in somatosensory and motor potentials were observed during intra-operative neuromonitoring for cases reporting post-operative radiculitis. All cases with post-operative radiculitis were self-limited and resolved after a median of 6 weeks with oral pregabaline treatment and 2 weeks of daily intramuscular injections of dexamethasone 4 mgr.
Post-operative muscle weakness was reported by 4 patients (8%) in form of neuropraxia. All patients (at least partially) recovered muscle function after post-operative corticoid treatment and intensive physiotherapy. In this case series, no dural injuries were reported during cage and screw placement.
Revision surgery with interbody cage removal was required for 2 patients (4%) during follow-up. The first revised case presented a symptomatic pseudo-arthrosis at L3/L4 about 6 months after surgery. The posterior fixation was removed and the interbody cage at L3/L4 was removed using an anterior-to-psoas lateral approach. Then, a lateral cage filled with Bone-Morphogenic-Protein (BMP-2) was placed at L3/L4 with its corresponding posterior fixation. Post-operative clinical and radiologic outcome was favorable during follow-up.
The second patient presented an intra-operatively mal-positioned cage that protruded into the neuroforamen. At 1 year follow-up the patient reported that she was undergoing revision surgery elsewhere and follow-up was lost.
Discussion
In this study, we have shown the feasibility to overcome what we have identified as the 3 main limitations in current endoscopic/percutaneous TLIF: a full-percutaneous insertion of a lumbar interbody cage using only intra-operative fluoroscopic imaging (without the need of endoscopic visualization); the placement of a large-footprint interbody cage without the limitation of a tube’s and working channel’s diameter; and the possibility to use standard, open-surgery instrumentation for a time- and cost-efficient disk and endplate preparation.
Key to overcome these difficulties was the development of a specialized dilator that incorporates a universal delivery sheet, as seen in Figure 2. This sheet allows protecting the exiting nerve root in Kambin’s triangle 16 during foraminoplasty, disk preparation and cage insertion, while its non-tubular shape allows a diameter-independent insertion of instruments and a large-footprint lumbar interbody cage. The fact that endoscopic visualization is not mandatory to perform a full-percutaneous TLIF, should make this technique accessible to surgeons without previous endoscopic experience. The surgical steps of this procedure are reproducible, while the handling of the fluoroscope feels familiar to percutaneous transpedicular screw insertion. Hence, in our opinion, the learning curve for this full-percutaneous TLIF should be shorter than for a standard endoscopic TLIF.
Furthermore, this full-percutaneous TLIF surgery allows a time-efficient interbody cage placement in less than 30 minutes per level and is cost-efficient, as standard interbody cages and regular instruments for disk and endplate preparation can be used (no need for expensive specialized interbody cages and endoscopic instruments).
We have also shown the feasibility of placing a large-footprint interbody cage using the trans-Kambin approach. This is remarkable, given the anatomic constraints of Kambin’s triangle which have led current endoscopic TLIF systems to use small, expandable interbody cages specially designed to fit through an endoscopic tube and/or working channel. These specialized cages usually have a complex, centrally placed expansion mechanism that is expensive and impedes bone growth through the cage. In contrast, large-footprint interbody cages usually have a big window for bone graft and a high bone/cage surface contact area, hence decreasing the risk for subsidence and increasing the rate of intervertebral bone fusion. 18 Hence, in this study, we were able to translate the advantages of a large-footprint interbody cage to trans-Kambin fusion. In fact, the large-footprint TLIF cage employed here has a footprint of 39x12mm (surface 468mm2) which represents a 1.5 to two-fold increase in surface compared to most regular-sized MIS TLIF and endoscopic TLIF cages, and our reported interbody fusion rate at 12 months follow-up is 90%.
The rate of post-operative, transitory radiculitis is still an issue in trans-Kambin fusion. Our rate of post-operative transitory radiculitis is within the range of 15% to 35% reported for the trans-Kambin technique by other studies that inserted a considerably smaller interbody cage.1-7 In contrast, anterior lumbar interbody fusion (ALIF) that allows placing a large-footprint interbody cage, has reported post-operative transitory radiculitis of 18% to 34%.19,20 The main cause for radiculitis for ALIF is usually indirect distraction of the neurological structures with a (hyperlordotic) large-footprint interbody cage. In our trans-Kambin fusion procedure, the rate of transitory radiculitis is probably also related, in part, to the large-footprint cage employed here, as bilateral post-operative radiculitis after pTLIF was observed for every third case with radiculitis. This is probably also due to indirect distraction of both neuroforamina caused by the large-footprint cage (and/or its expansion). Yet, additionally to indirect distraction, also direct compression of neurologic structures is probably an issue during the trans-Kambin approach given the anatomic constraints of Kambin’s triangle.1-7 The purpose of the delivery sheet is to protect the exiting nerve root, which is the most exposed neurologic structure during the trans-Kambin approach. 16 Additionally, an extensive foraminoplasty is essential for a safe placement of the interbody cage and instrumental manipulation inside Kambin’s triangle. However, the limited foraminoplasty that can be achieved with manual reamers with progressively increasing diameter in a full-percutaneous approach could be a limitation of our technique especially if a large-footprint interbody cage is employed. Further studies are necessary to determine if an even more extensive foraminoplasty under endoscopic visualization with a high-speed burr leads to a decreased rate of post-operative transitory radiculitis without considerably increasing the surgery time.
No predictive changes were recorded during intra-operative neuro-monitoring that allow a reliable prediction of post-operative radiculitis. We believe that this is probably due to the transitory radiculitis being an inflammatory process of the sensitive part of the dorsal root ganglion that occurs in the post-operative period (usually post-operatively within the first 72 hours) and self-resolves once the inflammation process winds down. Some studies have tried to establish a safety threshold with direct stimulation inside Kambin’s triangle during the cage placement21,22 but results are yet non-conclusive. Nonetheless, it should be remarked that all cases affected by post-operative radiculitis were self-limited and completely resolved after a few weeks.
Limitations
This study has some limitations. The system presented here was evaluated by a single surgeon at a single center. As our research represents one of the first attempts to describe a full-percutaneous TLIF technique using a large-footprint interbody cage, we approached this as an exploratory prospective observational study to obtain preliminary results. Even though the radiologic and clinical outcome is favorable after 2.5 years post-operative follow-up, a randomized, controlled, multi-center study with a larger sample size and control arms with comparable surgical techniques would offer a stronger level of evidence to confirm our preliminary results.
Conclusion
We have presented a promising, novel system for a full-percutaneous placement of a large-footprint interbody cage using the trans-Kambin approach. It allows a time-efficient cage insertion in less than 30 minutes, a shorter learning curve as endoscopic visualization is not mandatory, and it is cost-effective as there is no need for expensive, specialized interbody cages and endoscopic instrumentation. Clinical and radiologic outcome at 2.5 years post-operative follow-up is favorable with a fusion rate of 90% and a significant post-operative increase in segmental lordosis. However, further improvements need to be made to reduce the rate of post-operative, transitory radiculitis, especially when employing large-footprint interbody cages.
Footnotes
Acknowledgments
We would like to thank Geoffrey Delbos for his support.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by CM. The first draft of the manuscript was written by CM, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CM: Consulting and royalty fees Hoogland Spine GmbH and Signus GmbH. Consulting and royalty fees Unintech GmbH, as well as consulting fees Spineart SA outside of the submitted work. RM: royalty fees: DePuy-Synthes, Joimax GmbH outside of the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
