Abstract
Study Design
Exploratory prospective observational case-control study.
Objectives
Aim of this study was to compare clinical and radiologic outcome, as well as peri-operative complications, of anterior lumbar interbody fusion (ALIF) and full-endoscopic/percutaneous trans-Kambin transforaminal lumbar interbody fusion (pTLIF) with a large-footprint interbody cage.
Methods
Patients that underwent elective ALIF and pTLIF with a large-footprint interbody cage were prospectively evaluated. Clinical follow-up was measured pre- and post-operatively with Visual Analogic Scale and Oswestry Disability Index scores. Radiologic outcome was assessed with a computed tomography scan and standing films at 12 months.
Results
44 patients underwent ALIF and 43 pTLIF surgery (total 87 cases). Clinical pre- and post-operative scores were comparable between both groups with 33.4 months mean follow-up. Median surgical time was significantly lower for pTLIF (28 minutes) compared to ALIF (72 minutes). Radiologic outcome was favorable for ALIF with a significantly higher increase in segmental lordosis compared to pTLIF. Fusion rates did not significantly differ between both groups. ALIF showed significantly less cage subsidence than pTLIF. Complications included 9 (21%) cases with transitory post-operative radiculitis for ALIF and 12 (28%) for pTLIF; post-operative partial muscle weakness 3(6%) cases for ALIF and 4 (9%) for pTLIF. Two (4%) cases required revision surgery for pTLIF.
Conclusions
ALIF and trans-Kambin pTLIF obtained comparable clinical outcome and fusion rates, while segmental lordosis restoration was favorable for ALIF. pTLIF required less surgery time and presented less intra-operative complications, while ALIF reported lower rates of post-operative subsidence, revision surgery and complications during follow-up.
Keywords
Introduction
The advantages of large-footprint interbody cages for lumbar spine interbody fusion have been widely described and comprise a high interbody fusion rate due to a large window for bone graft, a lower risk of subsidence due to an increased endplate/cage contact surface area and a lower risk of cage migration.1-4
Anterior lumbar interbody fusion (ALIF) is an established treatment in lumbar spine surgery as it allows placement of a large-footprint interbody cage into a lumbar disk.3,4 However, wide-spread adoption of ALIF is restricted by a demanding abdominal surgical approach with risks of serious complications, i.e. severe venous bleeding, injury of intra-abdominal structures, retrograde ejaculation, etc.3,5 Moreover, ALIF is most convenient for L5/S1, as the anterior access of upper lumbar levels requires medial retraction of the vascular package (abdominal aorta and vena cava) with risk of catastrophic vascular injury. 6 In many countries, an access surgeon is required for ALIF procedures, limiting the adoption by orthopedic/neurologic surgeons.
Due to the mentioned limitations, open and minimally-invasive (MIS) transforaminal lumbar interbody fusion (TLIF) remain within the most widely used interbody lumbar fusion techniques. 5 However, the interbody cage size for a TLIF is usually limited to smaller footprint interbody cages due to the anatomic constraints of a posterior/ posterolateral approach. Banana-shaped TLIF cages can present a larger footprint than regular TLIF cages but are usually placed with open TLIF.7,8 Multiple comparative studies between ALIF and open/MIS TLIF have reported comparable clinical outcomes, while reports on radiologic outcome and other peri-operative variables seem to differ5,9-14 or are contradictory.9,10
In the last decade, full-percutaneous/endoscopic TLIF (pTLIF) has found increasing acceptance.15-21 pTLIF uses a postero-lateral extraforaminal approach through Kambin’s safety triangle to bypass the exiting nerve root and reach the disk (trans-Kambin approach). The advantages of pTLIF comprise minimal soft tissue disruption using sequential dilators (without dissection),15-21 a facet-sparing approach with limited bone resection,17,19 minimal intra- and post-operative bleeding,15-17 minimal risk of infection,15-19 early and opioid-free post-operative ambulation and hospital discharge,19-21 as well as direct decompression of the neuroforamen by foraminoplasty.15-18 Recently, technical advancements in the trans-Kambin approach have reported the feasibility to place a lumbar interbody cage with a larger footprint than regular-sized TLIF cages,
22
see Figure 1. However, the trans-Kambin approach also presents intrinsic limitations, like post-operative radiculitis and doubts on proper disk and endplate preparation.15-21 To the best of our knowledge, there are no comparative studies between ALIF and pTLIF with a large-footprint interbody cage. Visual comparison of a standard titanium ALIF interbody cage with medium-footprint, size L27xW36xH12 mm (Length × Width × Height) and 10° lordosis with (A) superior image: a large-footprint titanium expandable TLIF cage in collapsed status, size L39 × W12 × H8 mm and 3° lordosis; inferior image: the same TLIF cage after expansion with an increased anterior height of 13 mm and lordosis of 13°. (B) Axial view of the superposed interbody cages. The ALIF cage lies below and the TLIF cage is positioned diagonally in the same way it is positioned in the lumbar disk with pTLIF procedure. (C) Lateral view of the ALIF cage and the expanded TLIF cage with 13° lordosis. (D) Axial view of the TLIF cage (left image) and the ALIF cage (right image). (E) Frontal view of the collapsed TLIF cage (upper left image) and expanded TLIF cage (lower left image) with the ALIF cage (right images).
Aim of this study is to assess and compare the clinical and radiologic outcome, surgical efficiency, as well as peri-operative variables and complications for ALIF and pTLIF with a large-footprint interbody cage. Our main hypothesis is that patients treated with a large-footprint lumbar interbody cage will show favorable clinical and radiologic outcome independently from the surgical technique employed for its placement. Our secondary hypothesis is that peri-operative variables and complications will vary depending on the surgical technique employed.
Methods
Ethics Statement
This study was conducted according to principles of the Declaration of Helsinki and was approved by the ethics committee of our institution’s Institutional Review Board (approval number: 2023107-COT-CMT). All patients provided verbal and written consent for participation in the study.
Study Design
This was an Exploratory, Prospective Non-randomized Case-Control Study.
Study Population
Patients presenting with low back pain and/or associated uni- and/or bilateral leg pain were evaluated between 2018 and 2023 at one facility. Pre-operative assessment for all patients followed a standard protocol which included physical examination, pre-operative magnetic resonance imaging (MRI) and/or Computer Tomography (CT) scan of the lumbar spine.
Indications for spine surgery comprised degenerative disk disease of the lumbar spine, spondylolisthesis up to grade 2 and foraminal stenosis. A corresponding degenerative alteration had to be confirmed by imaging of the lumbar spine.
Inclusion Criteria
Patients had to be (I) at least 18 years old, (II) have obtained indication for ALIF or pTLIF surgery and (III) posterior screw fixation, and (IV) surgery had to exclusively target lumbar levels L4/L5 and/or L5/S1.
ALIF and pTLIF were respectively indicated after an individual and thorough assessment of each patient’s clinical symptoms and anatomy: i.e. patients with a very high BMI and/or previous abdominal surgery and/or history of vascular pathology (i.e. iliac vein thrombosis), etc. were preferably considered for pTLIF; while patients with a very high iliac crest and/or spondylolisthesis with a considerable anterior prominence, etc. were preferably considered for ALIF. The final decision on the surgical technique was always taken in consensual agreement with the patient after a thorough explanation of risk and benefits of all surgical and non-surgical options.
Exclusion Criteria
Patients with spondylolisthesis grade >2, neoplasm, trauma, infection, systemic diseases and not meeting the inclusion criteria.
ALIF Surgical Technique
The patient was placed in supine position on a radiolucent table and underwent general anesthesia. No access surgeon was used for ALIF in this study. A blunt left-sided retroperitoneal approach was performed according to standard technique.
3
A large-footprint titanium interbody cage (SCARLET AL-T, Spineart SA, Switzerland) packed with Demineralized Bone Matrix (DBM), was placed into the disk under fluoroscopic control. The cage was secured with self-locking screws. For L4/L5, the anterior retroperitoneal approach was left-sided and the ascending ilio-lumbar vein was identified, dissected and ligated. Interbody cage insertion was performed as previously described. Finally, the patient was flipped to prone position for percutaneous posterior spinal fixation, see Figure 2. Post-operative X-ray films with anterior/posterior (left image) and lateral (right image) views in standing position of (A) a single level ALIF case with a 20° hyperlordotic cage at L5/S1 and augmented percutaneous transpedicular screws (B) a two-level ALIF case with interbody cages with 15° lordosis at L5/S1 and L4/L5 and augmented posterior transpedicular screws.
Trans-Kambin pTLIF Surgical Technique
The patient was placed in prone position on a radiolucent Wilson frame. All pTLIF patients underwent general anesthesia with intra-operative neuromonitoring. Somatosensory evoked potentials (SEP) and motor evoked potentials (MEP) were employed to monitor involved peripheral nerves. A 2 cm skin incision was made lateral to the midline on the side of the patient’s dominant radiculopathy. A standard trans-Kambin approach13-17 to the disk was performed with a specialized trans-Kambin fusion dilator (Maxfusion system, Hoogland Spine Products GmbH, Munich, Germany) under fluoroscopic imaging, see Figure 3A and B. A retractor sheet was placed in the caudal aspect of the foramen protecting the exiting nerve root, see Figure 3C and D A foraminoplasty was progressively performed either with manual reamers (Maxmorespine, Hoogland Spine Products GmbH, Munich, Germany), see Figure 3E and F, and/or with a high-speed burr under endoscopic view. Disk preparation was performed using regular-sized punches, curettes and sharp spoons under fluoroscopic imaging and haptic feedback. A DBM-packed large-footprint titanium interbody cage (Vertaconnect, Signus Medizintechnik GmbH, Alzenau, Germany) was placed into the disk under fluoroscopic imaging, see Figures 1 and 3G and H. (A) Intra-operative view of a specialized dilator that is being introduced with guidance of a smaller-diameter dilator that has already been placed inside the target disk. (B) Intra-operative view of the specialized dilator after being placed inside the target disk. The dilator is now separated into two stages: a blunt dilator stage guided by the smaller dilator will be removed while a protective retractor sheet will remain inside the disk. (C) Intra-operative view of the retractor sheet in place. Note the open design of the sheet that allows full access to the disk (without the diameter constraints of a tube and/or endoscopic sleeve). (D) corresponding intra-operative fluoroscopic lateral view of the retractor sheet anchored inside the disk and in the caudal aspect of the neuroforamen protecting the exiting nerve root. (E) Intra-operative view of a manual reamer with a blunt tip. The reamer is placed on the retractor sheet that serves as a guide into Kambin’s triangle. The reamer only cuts when turned clockwise and is used to progressively ream the SAP until reaching the disk. The retractor sheet protects the exiting nerve root during the reaming. (F) corresponding fluoroscopic lateral view of the manual reamer after reaching the lateral aspect of the disk. The sheet is placed in the caudal aspect of the foramen to protect the exiting nerve root. (G) Intra-operative view of a DBM-packed large-footprint interbody cage placed on the sheet right before insertion into the disk (H) fluoroscopic lateral view (right image) of the cage guided by the sheet into Kambin’s triangle and fluoroscopic A/P view (left image) of the cage in place in the center of the disk.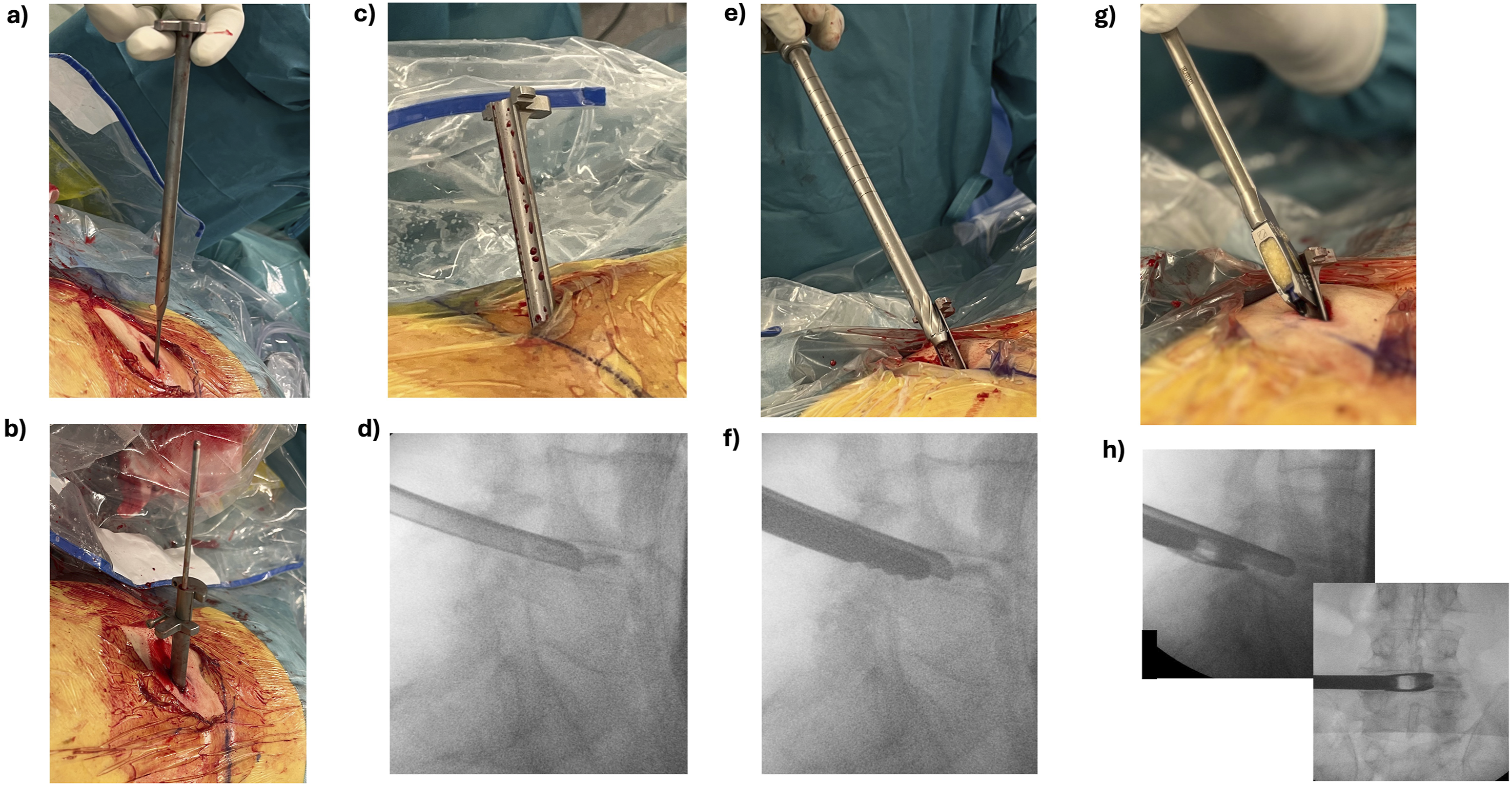
Posterior Fixation
Interbody cage placement was complemented with a percutaneous posterior spinal fixation, see Figures 2 and 4, consisting of transpedicular screws and rods, (ROMEO 2 MIS, Spineart SA, Switzerland). For cases with severe osteoporosis, screw augmentation was performed with polymethyl-methaacrylate (PMMA). Lordosis and sagittal alignment were considered during the fusion procedure and the patient’s position was adjusted correspondingly. In cases with neurogenic claudication and symptomatic severe central canal stenosis, an endoscopic unilateral laminectomy for bilateral decompression was additionally performed at the symptomatic level. Finally, fascia, subcutaneous tissue and skin were sutured. Post-operative X-ray films with anterior/posterior (left image) and lateral (right image) views in standing position of (A) a single level pTLIF case with an expanded interbody cage with 13° lordosis at L4/L5 and posterior augmented percutaneous screws. (B) A two-level pTLIF case with a interbody cages at L4/L5 and L5/S1 and posterior percutaneous screws.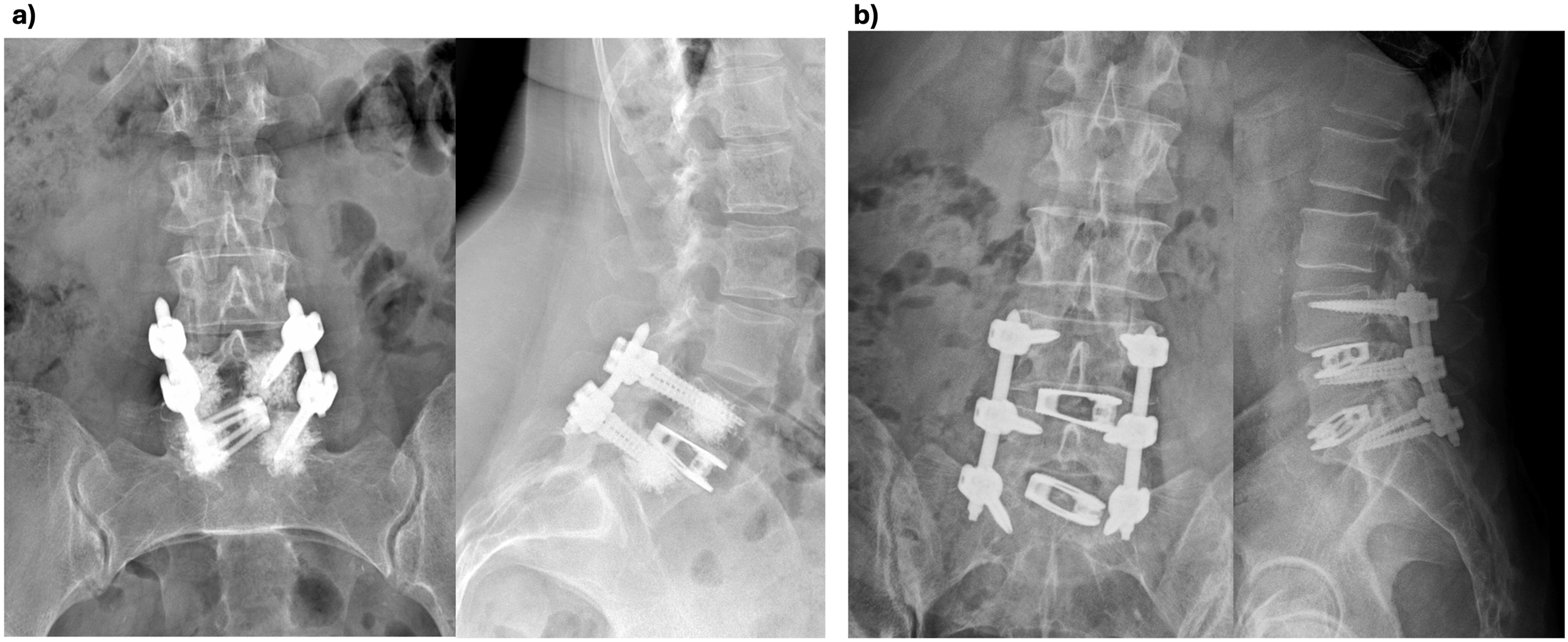
Clinical and Radiologic Outcome Measurement
Pre-operative and post-operative Visual Analogic Scale (VAS) for lumbar back and leg pain were quantitatively assessed (range 0 to 10). Oswestry Disability Index (ODI) scores (range 0 to 50) 23 were pre-operatively assessed. Post-operative follow-up controls were established at hospital discharge, 1, 3 and 6 months, 1 year and thereafter successively every 12 months.
A computer tomography scan was taken in the immediate postoperative period (within 24 hours after surgery) and at 1 year post-operatively, conventional films at 6 weeks and 1 year after surgery.
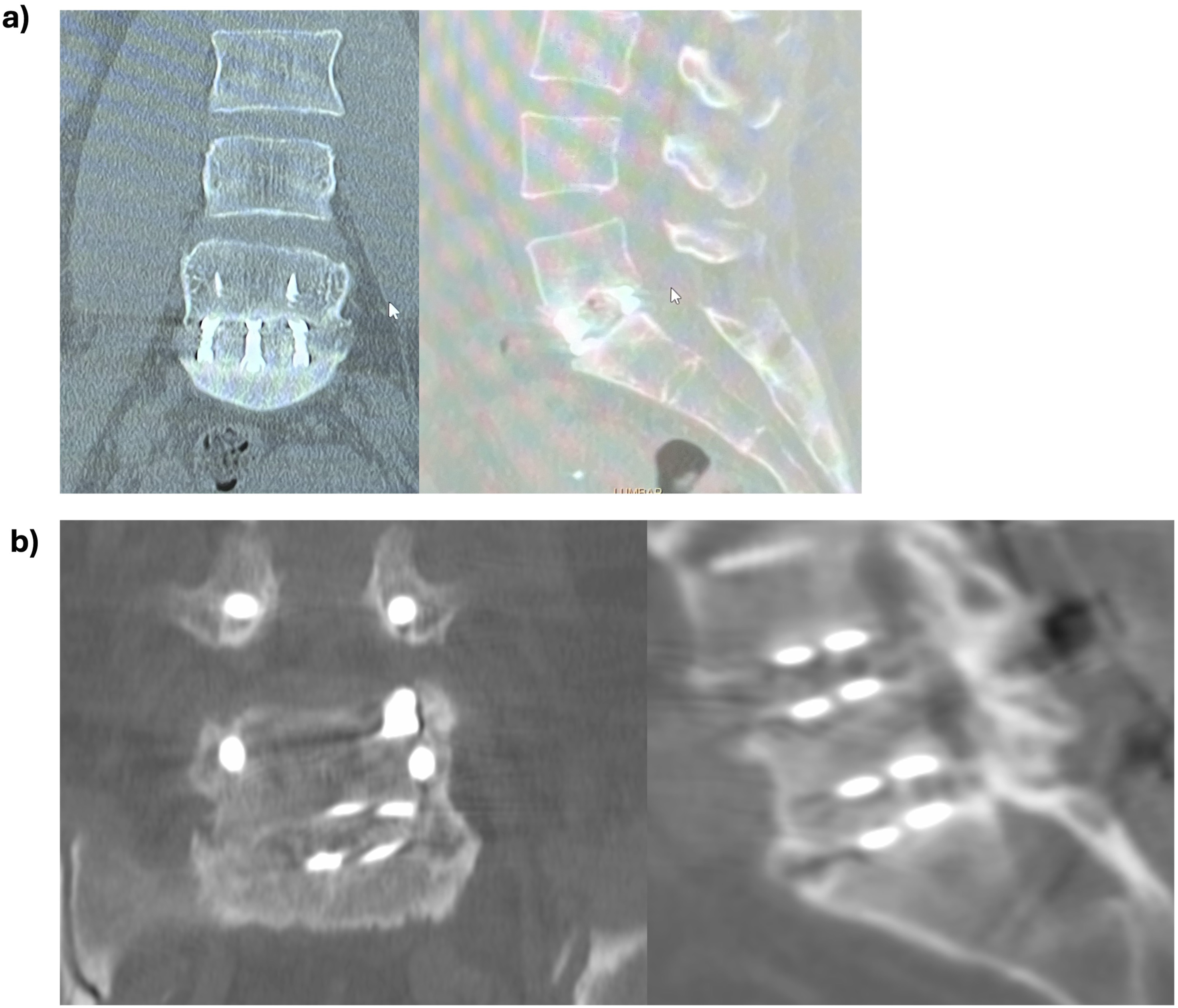
Pre- and postoperative radiologic parameters consisting of lumbar lordotic angle (LLA), segmental lordotic angle (SLA) of the operated level(s) and pelvic incidence (PI), as well as the difference between PI and LLA (PI–LL) were measured in pre- and post-operative standing X-rays, see Figure 5. Intervertebral bone fusion was radiologically assessed at 1-year post-op with a CT-scan and a standard film. Intervertebral bone fusion was confirmed with presence of trabecular bone in the CT scan imaging, see Figure 6. Pre-operative (left image) and post-operative (right image) standing whole spine X-ray films with radiologic measurements and their corresponding values in the image at the lower right corner, respectively of (A) and (B) two-level ALIF case with cages at L4/L5 and L5/S1 (C) and (D) a two-level pTLIF case with cages at L4/L5 and L5/S1; Note the correction of the radiologic values after surgery (B) and (D) in comparison to the pre-operative (A) and (C) values for each case, respectively. Post-operative CT scan images with coronal (left image) and lateral (right images) cuts at 1 year post-operative follow-up showing completed interbody fusion with trabecular bone inside the interbody cage for (A) a single-level ALIF cage at L5/S1 (B) a two-level pTLIF case with cages at L4/L5 and L5/S1. Note the trabecular bone inside the interbody cage of L5/S1.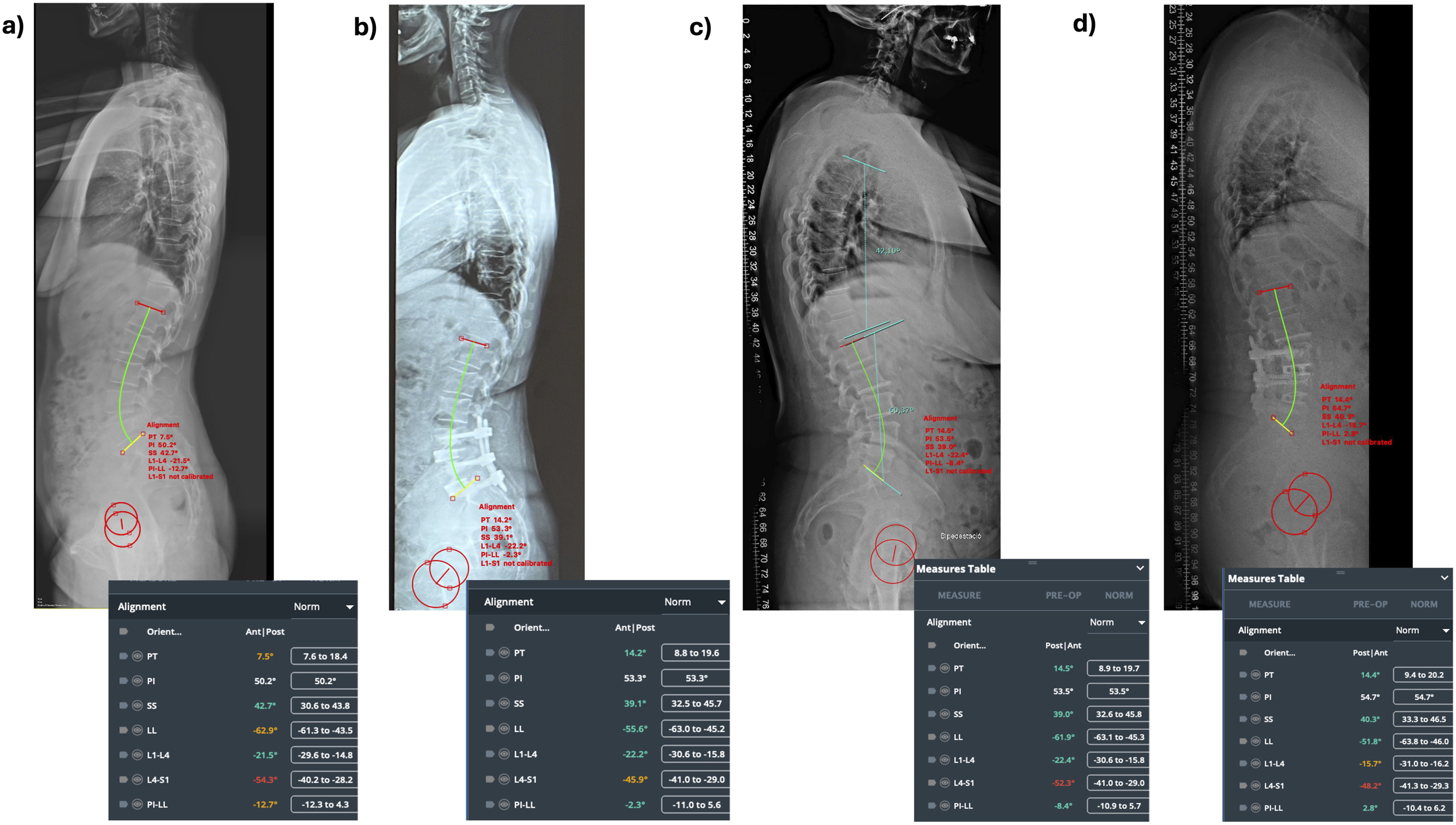
Surgical time for interbody cage insertion was measured as time elapsed between first skin incision and skin suture (skin-to-skin) for ALIF and pTLIF, respectively, while surgical time for posterior screw fixation and for patient re-positioning for the posterior screw fixation was not included.
Statistical Analysis
Kolmogorov-Smirnov test was employed to evaluate normal distribution of the data series. Paired and unpaired student’s T tests were employed to statistically compare intra- and inter-cohort variables, respectively. Statistical significance was defined at P < 0.05. A power analysis calculation was performed for two independent groups (G* Power R3.1.9.7) to assess the robustness of the study design.
Results
Demographics and Surgery Data
An overall of 87 patients were prospectively included in this study: 44 (51%) patients underwent ALIF and 43 (49%) pTLIF. A power analysis with a significance level (α) of 0.05, a target power (1 - β) of 0.80 and a standardized effect size (Cohen’s d) of 0.54 yielded sample sizes of 44 and 43 patients, respectively.
Demographics and Surgical Characteristics of the Operated Patients.
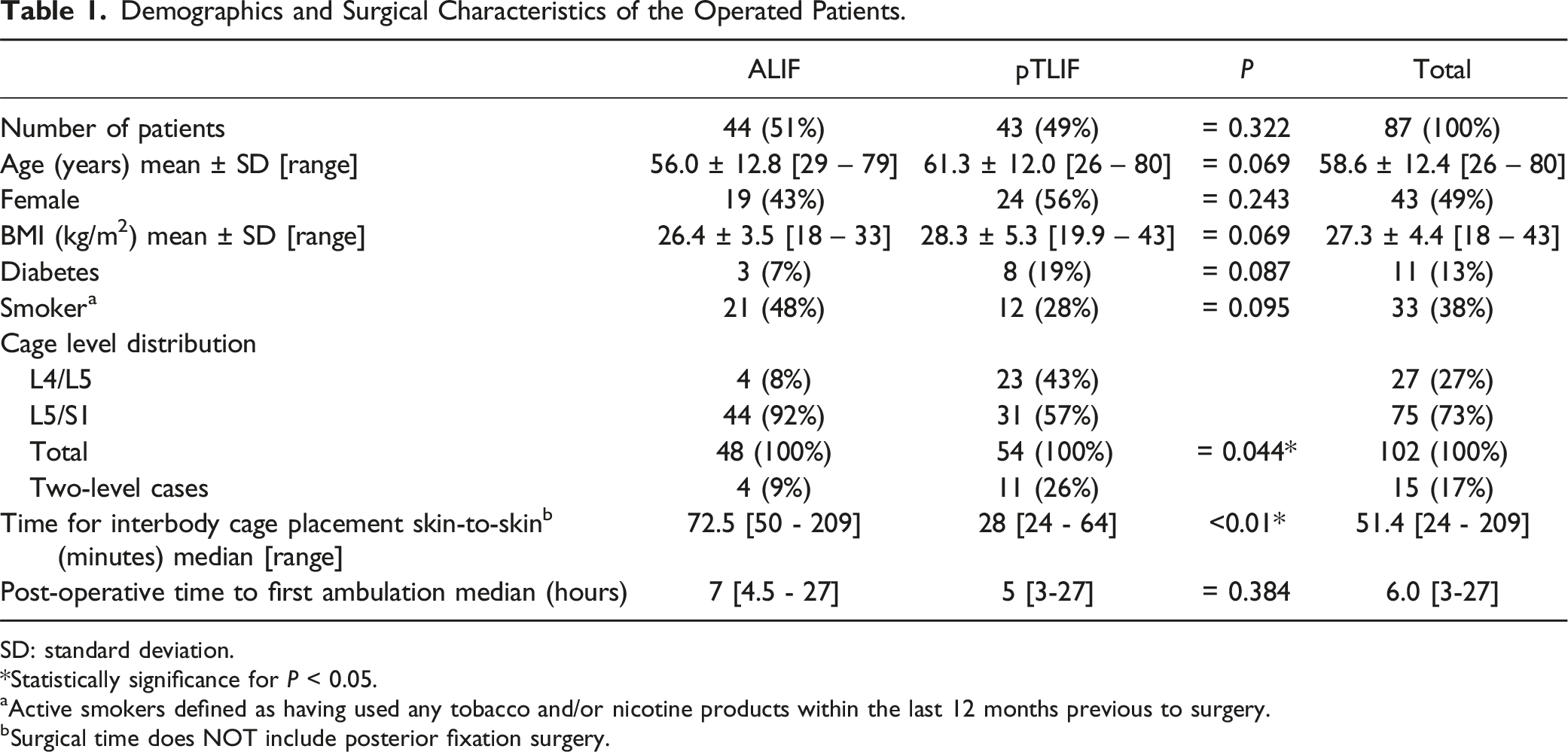
SD: standard deviation.
*Statistically significance for P < 0.05.
aActive smokers defined as having used any tobacco and/or nicotine products within the last 12 months previous to surgery.
bSurgical time does NOT include posterior fixation surgery.
Clinical Outcome
Clinical Outcome for 87 Patients After ALIF and pTLIF Surgery. (P Values Reflect Statistical Differences Between the Groups (Inter-Cohort).
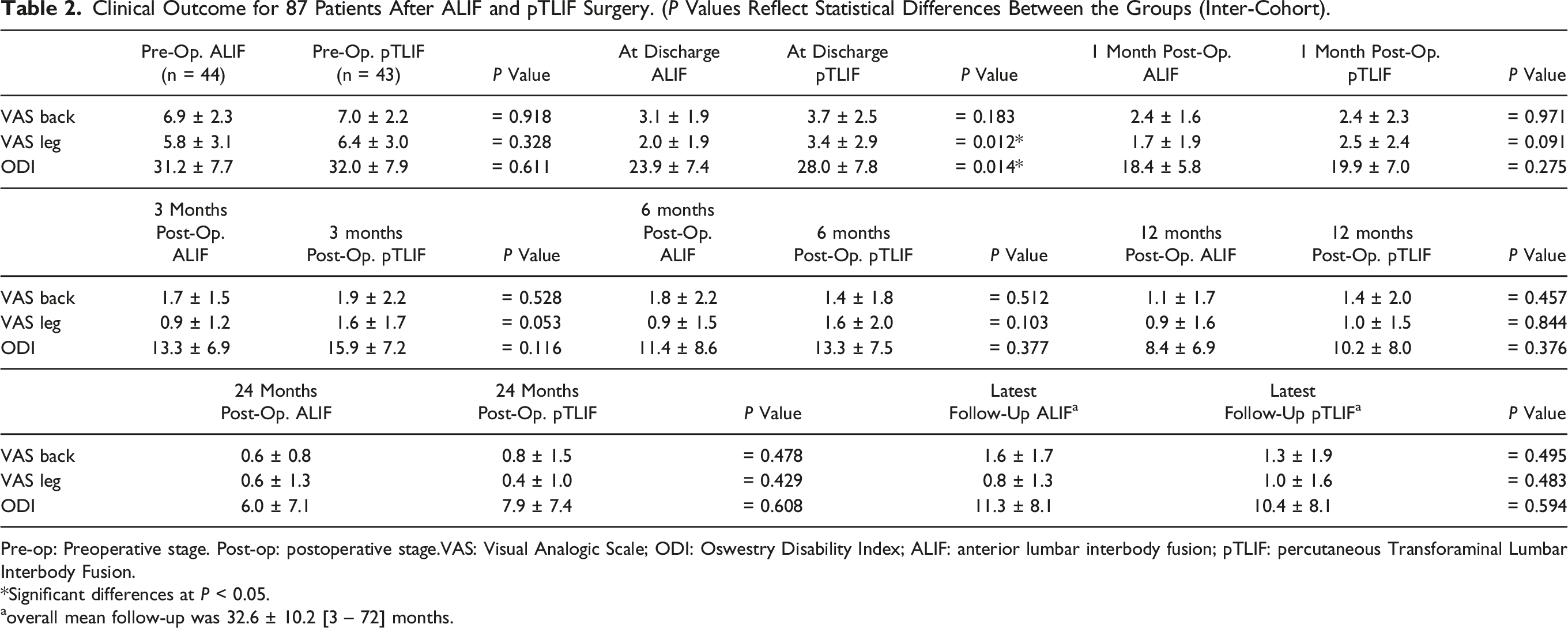
Pre-op: Preoperative stage. Post-op: postoperative stage.VAS: Visual Analogic Scale; ODI: Oswestry Disability Index; ALIF: anterior lumbar interbody fusion; pTLIF: percutaneous Transforaminal Lumbar Interbody Fusion.
*Significant differences at P < 0.05.
aoverall mean follow-up was 32.6 ± 10.2 [3 – 72] months.
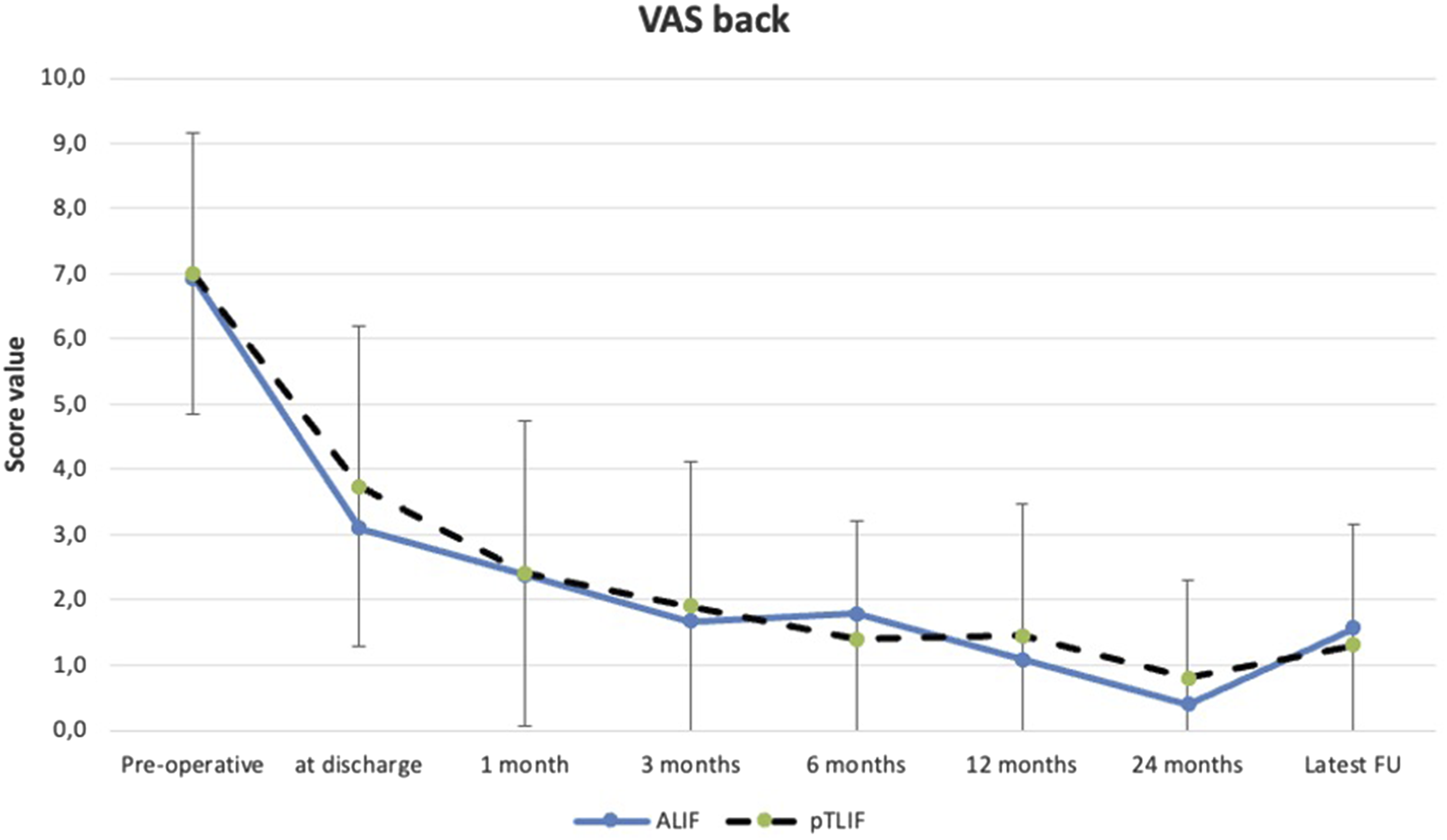
Error bar plot displaying mean ± standard deviation of pre-operative and post-operative VAS back scores of the ALIF and pTLIF group at different stages of the post-operative follow-up.
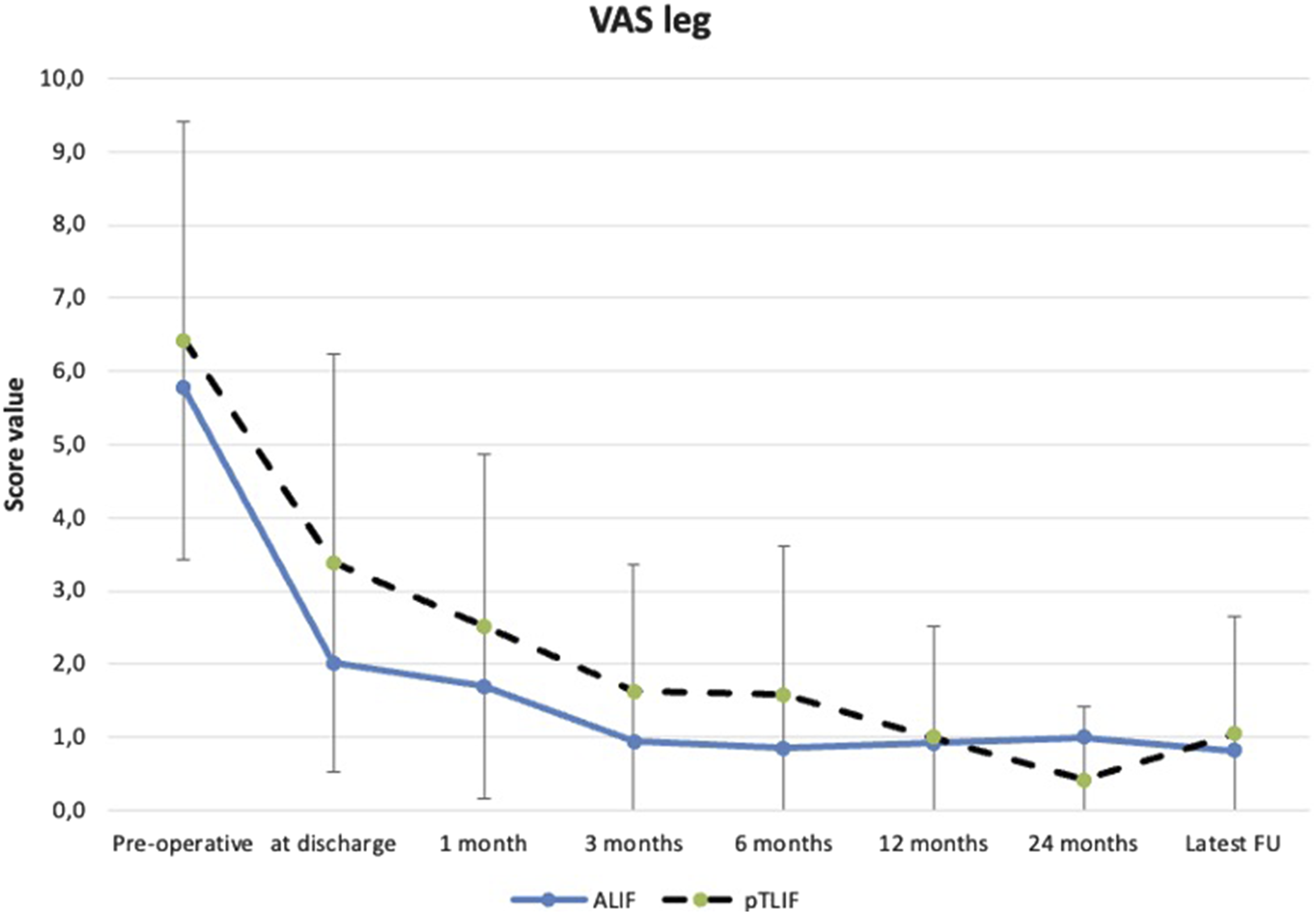
Error bar plot displaying mean ± standard deviation of pre-operative and post-operative VAS leg scores of the ALIF and pTLIF group at different stages of the post-operative follow-up.
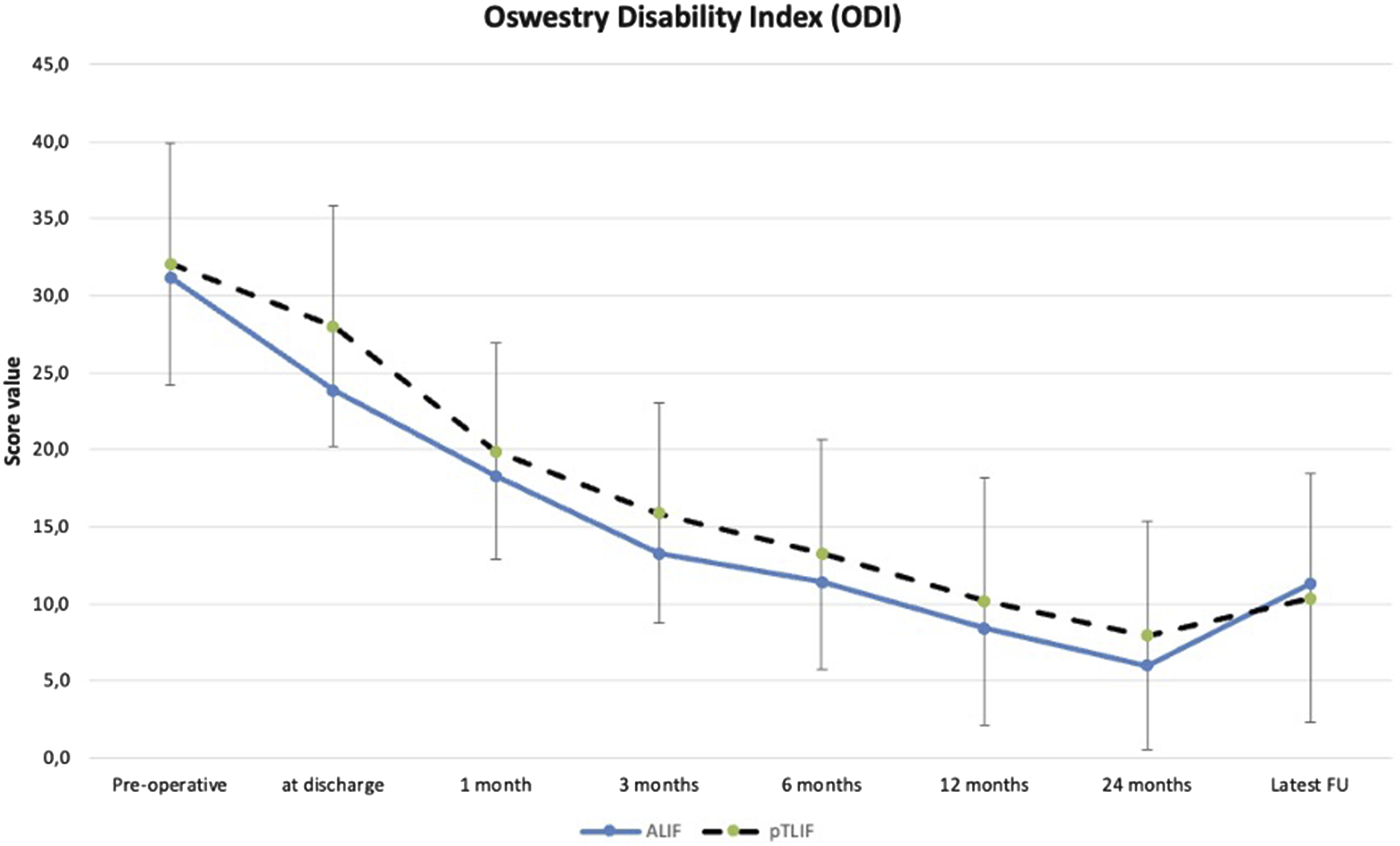
Error bar plot displaying mean ± standard deviation of pre-operative and post-operative ODI scores of the ALIF and pTLIF group at different stages of the post-operative follow-up.
Significant intra-group differences were found at all stages of follow-up between pre- and post-operative scores for VAS back (P < 0.001), VAS leg (P < 0.001) and ODI (P < 0.001) for the ALIF group as well as the pTLIF group, respectively.
The inter-group comparison between the clinical scores for the ALIF and pTLIF groups can be found in Table 2. The VAS leg and ODI scores were significantly better for the ALIF group at hospital discharge. No significant differences were found for all scores between both groups at any other time during follow-up, see Table 2. Hospital length of stay for the ALIF group was median 52 [range 25 – 98] hours and for the pTLIF group median 27 [range 19 - 65] hours.
Radiologic Outcome
Radiologic Outcome of the Operated Patients. Values are Represented as Mean ± Standard Deviation [Range].
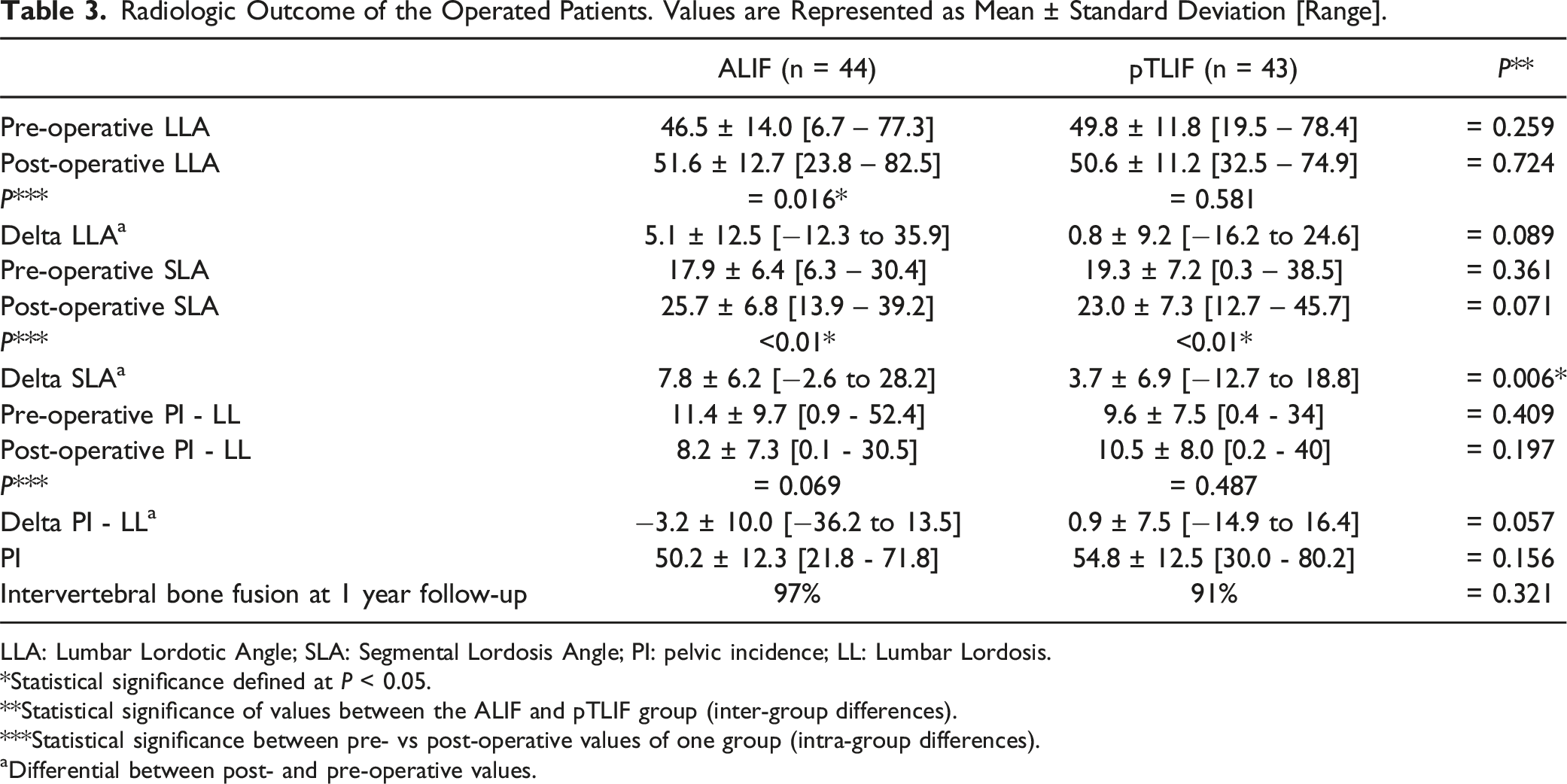
LLA: Lumbar Lordotic Angle; SLA: Segmental Lordosis Angle; PI: pelvic incidence; LL: Lumbar Lordosis.
*Statistical significance defined at P < 0.05.
**Statistical significance of values between the ALIF and pTLIF group (inter-group differences).
***Statistical significance between pre- vs post-operative values of one group (intra-group differences).
aDifferential between post- and pre-operative values.
Peri-Operative Complications of ALIF and pTLIF.
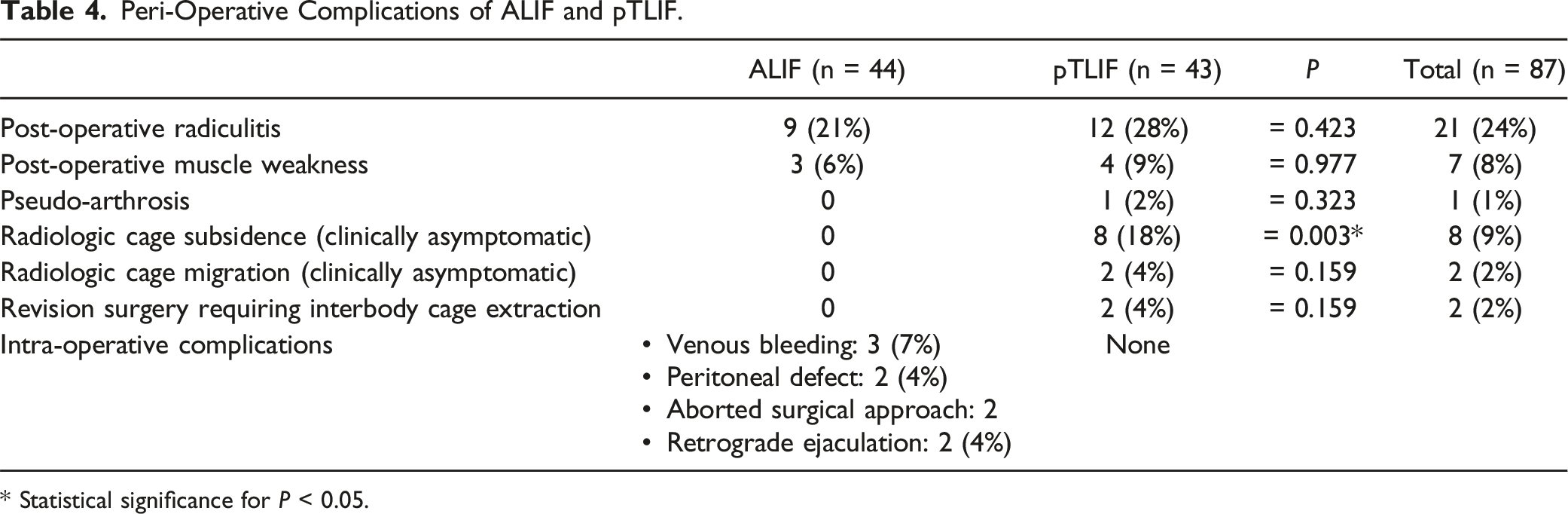
* Statistical significance for P < 0.05.
Complications
Intra- and post-operative complications are detailed in Table 4. There was no significant difference in post-operative radiculitis between both groups. All cases with post-operative radiculitis were self-limited and resolved after a median of 6 weeks with oral pregabaline treatment and 2 weeks of intramuscular injections of dexamethasone 4 mgr.
Post-operative muscle weakness was observed in form of neurapraxia. All patients recovered muscle function (at least partially) after post-operative corticoid treatment and intensive physiotherapy.
In the ALIF group the surgical approach was aborted in 2 cases due to the impossibility to access the disk because of unfavorable vascular anatomy. One of these cases was converted to pTLIF, the other case obtained posterior fixation without interbody cage. None of the aborted surgery cases was considered in the total case count for the ALIF group, i.e. all 44 patients reported in the ALIF group successfully obtained an interbody cage. The ALIF group also showed some approach-specific intra-operative complications: 3 cases (7%) with venous bleeding that were successfully solved with hemostatic gel, two cases (4%) with retrograde ejaculation in male patients that spontaneously self-resolved during follow-up and two cases with a small peritoneal defects(<2 cm) that were solved with suture.
In the pTLIF group, 2 cases (4%) required revision surgery with interbody cage removal due to a pseudo-arthrosis and an intra-operatively mal-positioned cage that protruded into the neuroforamen, respectively. In the ALIF group, no revision surgery with interbody cage removal was necessary.
Discussion
The aim of this study was to critically assess the advantages and limitations of pTLIF by comparing it to ALIF as an established lumbar fusion technique. For this purpose, we evaluated cases treated either with ALIF or pTLIF using interbody cages with a larger footprint than most regular-sized TLIF cages. To our best knowledge, there is no consensus on a threshold that allows categorizing “large” and “small” footprint cages with absolute values for size, surface and/or proportion of cage-to-endplate contact area.24-28 The TLIF interbody cage employed here has a surface of 468 mm2 which represents a 1.5 to two-fold increase compared to most regular-sized MIS TLIF and endoscopic TLIF cages. Hence, despite the obvious differences in size and surface between an ALIF cage and the TLIF cage employed here, see Figure 1, we considered that both cages have a “large” footprint when compared to most regular-sized TLIF cages used in clinical routine.
The learning curves for the trans-Kambin approach 29 and for the anterior retroperitoneal approach3-5 are both challenging. In our opinion, the retroperitoneal approach (without an access surgeon) is the more challenging one: i.e. we were forced to abort an anterior approach in two cases due to a vascular situation that impeded access to the disk. Complications like venous bleeding, retrograde ejaculation and retroperitoneal defects are specific to the anterior retroperitoneal approach and usually not an issue for the trans-Kambin approach. Hence, surgical time for cage placement for ALIF (approximately 1 hour per level) doubled the time required for pTLIF (approximately 30 minutes per level). As all cases in this study obtained posterior fixation, the additional time required to flip the patient from supine to prone needs also to be considered in the overall surgical time of the ALIF group. However, for selected indications, stand-alone ALIF could be considered as a valid alternative that could reduce the overall surgical time for an ALIF procedure.
Clinical outcome was similar for both groups and is in line with reported results elsewhere.5,10-12 The ALIF group presented significantly better VAS leg and ODI scores at hospital discharge. This could be related to the significantly longer hospital stay for ALIF. In our facility, ALIF patients usually stay for at least one day in-patient for observation and are usually discharged within 48 hours after surgery. Consequently, hospital length of stay was, as expected, longer for ALIF than for pTLIF and should not be considered clinically relevant here.
It is remarkable that there were no significant differences in the rate of post-operative radiculitis and muscle weakness between the ALIF and pTLIF group. The rate of 28% post-operative radiculitis for our pTLIF group lies within the reported incidence of 5% to 35% for trans-Kambin fusion.15-22 Interestingly, we also report 21% of cases with post-operative radiculitis for the ALIF group, confirming the rate of 18% to 34% of post-operative radiculitis that has been reported for ALIF in other studies.30,31 The most probable cause for radiculitis after ALIF is indirect distraction of the neurological structures with a hyperlordotic, large-footprint interbody cage.30,31 Cage over-sizing is also the most probable cause for the 3 cases with post-operative muscle weakness reported here for this group.
The reported rate of radiculitis for the pTLIF group is probably also related to the large-footprint cage employed here. In contrast to ALIF, direct compression of neurologic structures with the large-footprint interbody cage inside Kambin’s triangle is probably an additional cause for radiculitis during the trans-Kambin approach.15-22 Hence, a careful and extensive foraminoplasty is key to ensure the safety of neurologic structures during the trans-Kambin approach. Still, all cases with post-operative radiculitis were self-limited and completely resolved after a median of 6 weeks for both groups.
Radiologic post-operative values were favorable for ALIF in comparison to pTLIF, which is in line with other reported results.13,14 This is probably related to the release of the anterior longitudinal ligament and the feasibility to place a hyperlordotic interbody ALIF cage with >20° of lordosis. Furthermore, ALIF did not present post-operative cage migration, pseudo-arthrosis nor required revision surgery with interbody cage removal during follow-up. This is probably related to the additional stability provided by the self-locking screws of ALIF directly attaching the interbody cage to both vertebral bodies.4,5 Moreover, a regular ALIF cage still presents an at least two-fold increment in surface than even a large TLIF cage, see Figure 1, providing increased stability and endplate contact surface. 1
In contrast, pTLIF was able to increase post-operative SLA compared to pre-operative values but the increment was significantly smaller compared to the ALIF group and had no impact on the pTLIF’s post-operative global LLA and PI-LL ratio. In our opinion, the reasons for this are that a regular TLIF cage usually offers less than 10° of lordosis and the cage is positioned diagonally on the disk without bilateral facetectomy. All this limits the changes in sagittal alignment that can be achieved with a pTLIF. 15
Limitations
This study has some weaknesses: the results presented here are reported by a single surgeon at a single center. As our research represents one of the first attempts to compare ALIF and pTLIF using large-footprint interbody cages, we approached this work as an exploratory prospective observational case-control study designed to generate preliminary data. The distribution of operated levels differed between both groups, while underlying sagittal alignment types (i.e Roussouly classification) were not explicitly radiologically assessed. While this limitation reflects real-world surgical decision-making (where the choice of ALIF vs pTLIF was partly guided by patient-specific anatomy and comorbidities), this could remain a potential confounder. In future studies, controlling for or stratifying by Roussouly type would probably allow a more nuanced comparison of sagittal plane correction and functional outcome. Furthermore, even though the moderate effect size in the power analysis suggests that the sample size employed was sufficiently robust to detect meaningful differences for the examined primary outcome measures, a larger, multi-center randomized controlled trial would offer a stronger level of evidence, especially for the subgroup analyses.
Conclusion
ALIF and pTLIF obtained comparable clinical outcome and bone fusion rates, while segmental lordosis restoration was favorable for the ALIF group. pTLIF presented less intra-operative complications and shorter surgical time, while ALIF reported lower rates of subsidence, revision surgery and post-operative complications at 2.5 years of mean follow-up. Careful surgical indication is advised by taking the advantages and limitations of each surgical technique into consideration.
Footnotes
Author Contributions
CM and RM contributed to the study conception and design. Material preparation and data collection were performed by CM, FN and GD. Data analysis was performed by CM and GD. The first draft of the manuscript was written by CM, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CM: Consulting and royalty fees Hoogland Spine GmbH and Signus Gmbh. Consulting and royalty fees by Unintech Gmbh and consulting fees by Spineart SA outside the submitted work. FN, GD and RM: no conflict of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclosure
CM: Consulting and royalty fees Hoogland Spine GmbH and Signus Gmbh. Consulting and royalty fees by Unintech Gmbh and consulting fees by Spineart SA outside the submitted work. RM: royalty fees: DePuy-Synthes, Joimax GmbH outside of the submitted work.
