Abstract
Study Design
Network Meta-analysis.
Objective
To evaluate the clinical efficacy and safety of minimally invasive (MIS-), full endoscopic (FE-), and biportal endoscopic (BE-) transforaminal lumbar interbody fusion (TLIF).
Methods
PubMed, Embase, Cochrane Library, and ClinicalTrials.gov registry were searched up to August 2025. Randomized controlled trials and nonrandomized studies of intervention comparing MIS-TLIF, FE-TLIF, and BE-TLIF for lumbar degenerative diseases were included. Network meta-analysis was conducted using the frequentist framework, with meta-regressions assessing effect modifiers.
Results
49 studies (4188 patients) were included. FE-TLIF and BE-TLIF significantly reduced back pain vs MIS-TLIF from 1 month postoperatively through final follow-up. However, leg pain showed no significant differences among the three approaches. FE-TLIF and BE-TLIF demonstrated superior short- and mid-term functional outcomes, with FE-TLIF achieving the shortest hospital stay. Both endoscopic techniques reduced blood loss compared to MIS-TLIF. Long-term functional improvement, fusion rates, and complication rates were comparable, though FE-TLIF and BE-TLIF required longer operative times. Meta-regression suggested greater back pain relief in older patients following FE-TLIF.
Conclusion
BE-TLIF and FE-TLIF are safe techniques offering advantages over MIS-TLIF, including greater back pain reduction, improved short-term function, shorter hospital stays, and reduced blood loss. However, both require longer operative times. Meta-regression analysis suggested a promising signal that FE-TLIF may provide greater back pain relief in older patients. MIS-TLIF remains viable for those with higher anesthesia risk or significant cardiopulmonary comorbidities. Further high-quality randomized controlled trials are needed to validate these findings and refine patient selection criteria.
Keywords
Introduction
Degenerative lumbar diseases, such as lumbar spondylosis and lumbar spondylolisthesis, are prevalent spinal disorders in adults. 1 Patients experiencing chronic low back pain and radiculopathy that are refractory to conservative treatment often require surgical intervention, such as lumbar decompression and interbody fusion. 2 Traditional fusion techniques, including posterior lumbar interbody fusion (PLIF) and transforaminal lumbar interbody fusion (TLIF), have been widely utilized. However, the open approach is associated with increased perioperative morbidity. It often causes damage to posterior structures such as muscles and ligaments, leading to greater tissue disruption and prolonged hospitalization. The introduction of minimally invasive transforaminal lumbar interbody fusion (MIS-TLIF) aimed to mitigate these issues, with studies demonstrating advantages including reduced blood loss, less tissue trauma, and shorter hospital stays.3-5
To achieve direct visualization for endplate preparation while minimizing vascular and neurological injury, full endoscopic transforaminal lumbar interbody fusion (FE-TLIF), also referred to as uniportal endoscopic TLIF, was developed.6,7 FE-TLIF utilizes a single coaxial working channel introduced through Kambin’s triangle. Both the endoscope and surgical instruments are inserted through the same portal, resulting in the smallest incision and minimal soft tissue disruption. However, this approach is constrained by a fixed, straight trajectory.8,9 In contrast, biportal endoscopic transforaminal lumbar interbody fusion (BE-TLIF) uses two separate portals—one for endoscopic viewing and the other for instrumentation. This configuration allows triangulation, bimanual manipulation, a broader working angle, and easier access for contralateral decompression. By separating the viewing and working channels, BE-TLIF offers different visualization characteristics and greater surgical flexibility and may facilitate endplate preparation, potentially reducing the risk of cage subsidence.10,11
Previous systematic reviews and meta-analyses have compared FE-TLIF and BE-TLIF with MIS-TLIF. These studies highlight that each approach has distinct advantages and limitations across various clinical outcomes. Overall, FE-TLIF and BE-TLIF appear to provide superior short-term pain relief, improved early functional recovery, and reduced blood loss and hospital stay compared to MIS-TLIF, while maintaining comparable long-term clinical outcomes, fusion rates, and complication rates. Nonetheless, both FE-TLIF and BE-TLIF are associated with longer operative times.7,12-20
Despite the growing body of research, definitive comparative analyses between these advanced surgical techniques remains limited. A prior meta-analysis comparing FE-TLIF and BE-TLIF has provided preliminary insights. 21 However, its conclusions were based on a small number of studies and lacked indirect comparisons involving MIS-TLIF, limiting its relevance to broader clinical practice. Network meta-analysis offers a robust methodological approach to overcome these limitations by integrating both direct and indirect evidence. Moreover, by incorporating both randomized and nonrandomized studies, it allows for a more comprehensive synthesis of available evidence and enhances the generalizability of the findings across diverse clinical contexts. In this study, we conducted a network meta-analysis to compare the clinical outcomes of FE-TLIF, BE-TLIF, and MIS-TLIF. Our analysis aims to provide a solid evidence-based framework to inform clinical decision-making and advance the refinement of minimally invasive lumbar spine surgical strategies.
Methods
Search Strategy and Selection Criteria
This systematic review and network meta-analysis was planned, conducted, and reported in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines. 22 The protocol of this study was registered in PROSPERO (CRD42024625884).
Three electronic databases (PubMed, Embase, Cochrane Library, and ClinicalTrials.gov registry) were searched up to August 2025. Randomized controlled trials (RCTs) and nonrandomized studies of intervention (NRSIs) that compared the effects among FE-TLIF, BE-TLIF, and MIS-TLIF for lumbar degenerative diseases were included. The inclusion criteria and search strategy were summarized in Appendix 1 and Appendix 2, respectively. There were no limitations to language, country, publication date, or publication status.
Two reviewers (HYC and CWL) independently extracted important data and resolved disagreements regarding trial inclusion and data extraction by consulting with the third reviewer (CLL). The following information was collected from each study: bibliography, study design, grouping, sample size, disease level, cage material, patient sex, patient age, follow-up duration, and outcomes.
Risk of Bias Assessment
Using the revised Cochrane risk-of-bias tool for randomized trials (RoB 2) and Risk Of Bias In Non-randomised Studies - of Interventions (ROBINS-I), two reviewers (HYC and CWL) independently assessed the risk of bias of the included studies. Any disagreements were resolved by consultation with the third reviewer (CLL).
Outcome Measures
The primary outcomes were pain (back pain and leg pain, measured by the visual analogue scale, the numerical rating scale, or any other published scales), function (measured by Oswestry Disability Index, the Japanese Orthopaedic Association scoring system, or any other published scales), hospital stay duration, and intraoperative blood loss. The secondary outcomes were operation time, fusion rate, and complications.
Data Synthesis and Analysis
The data used in the analysis of continuous outcomes in this network meta-analysis comprised scores at the endpoint. A random-effects network meta-analysis was performed within a frequentist framework using the netmeta package of R (version 4.0.4). Effect sizes for pain and function were reported as standardized mean differences (SMDs), enabling the pooling of results across studies using different measurement tools by converting them to a common metric. Mean differences (MDs) were used for other continuous outcomes, and risk ratios (RRs) for dichotomous outcomes. The effect estimates were reported with 95% CIs. To evaluate the relative efficacy of the treatments, the network forest plots were constructed, and the treatments were ranked using the P-scores. To validate the results, the network meta-analysis was also performed under the Bayesian framework.
The transitivity assumption was evaluated by comparing the distribution of potential effect modifiers (publication year, sample size, female ratio, age, disease level, and cage material) across studies grouped by comparison. We assumed a common heterogeneity parameter across the various treatment comparisons and presented the between-study variance τ 2 for each outcome. We assessed global inconsistency using the design-by-treatment model and the Cochrane’s Q test, with P < 0.1 indicating significant global inconsistency. We also applied back-calculation methods to separately assess indirect and direct evidence, allowing for the evaluation of local inconsistency. We further explored residual heterogeneity and inconsistency by several meta-regressions (based on covariates of study design, publication year, sample size, female ratio, age, disease level, and cage material). In the meta-regressions assessing study design and publication year, we assumed common coefficients across all treatments comparisons. For other effect modifiers, we assumed treatment-specific coefficients. The meta-regressions were performed under the Bayesian framework. The posterior distributions were obtained using the Markov-chain Monte Carlo process with 5000 burn-ins and 500 000 iterations of 4 chains. They were then thinned after every 10th simulation to reduce autocorrelation. The convergence of the model was assessed by evaluating the trace plots and Gelman-Rubin diagnostics with a cut-off value of 1.05.
To assess publication bias, comparison-adjusted funnel plots were generated, and Egger’s regression asymmetry tests were conducted.
Evaluation of Certainty of Evidence
The certainty of evidence was assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach, following the method outlined by Salanti et al. 23 GRADE classifies evidence quality into 4 levels: very low, low, moderate, and high. These levels reflect the degree of confidence that the estimated effects approximate the true effects. 24 Evaluation covered 6 domains: risk of bias, reporting bias, indirectness, imprecision, heterogeneity, and incoherence.25,26
Results
Study Selection
Figure 1 illustrates the process of identification, screening, and selection of relevant studies. The initial search of the databases yielded 4906 publications. After the titles and abstracts were screened to eliminate publications that did not align with the inclusion criteria, 418 publications were selected for full-text review. Subsequently, 369 publications were excluded (Appendix 3), resulting in the identification of 49 publications, comprising three RCTs and 46 NRSIs (Appendix 4). Flow diagram of study inclusion
Characteristics of the Included Studies
A total of 49 studies with 4188 patients were included (Appendix 4 and Appendix 6). Although our search strategy placed no restrictions on publication date, all eligible articles retrieved were published between 2019 and 2025. Of the 46 studies that provided age information, the study-specific mean age ranged from 39.6 to 70.5 years (median 60.1 years), with most studies clustering between 55 and 65 years. The enrolled patients had an approximately equal distribution of males and females. Two studies reported follow-up limited to the period of hospitalisation without specifying a numeric duration; in the remaining 47 studies, follow-up ranged from 0.5 to 43 months (mean 16.1 months, median 14 months). Most studies monitored participants for at least three months. Only one study had a follow-up shorter than a month, and 13 studies extended beyond 18 months.
Surgical Technique
All 98 surgical cohorts performed direct neural decompression. The most frequent technique was a combined facetectomy and laminectomy, whereas isolated facetectomy or isolated foraminotomy were less common. MIS-TLIF procedures typically employ a posterior approach using the Wiltse plane or a tubular retractor system, with placement of polyetheretherketone (PEEK) cages. FE-TLIF utilized a full-endoscopic transforaminal approach through Kambin’s triangle, performing foraminotomy and discectomy, followed by insertion of small expandable titanium or PEEK cages. BE-TLIF employed dual portals with continuous irrigation, facilitating wider decompression commonly via facetectomy and laminectomy, with subsequent cage insertion transforaminally using PEEK or expandable titanium devices.
Risk of Bias within Individual Studies
Appendix 5 summarizes the risk of bias of the included studies. All 3 RCTs had a high overall risk of bias, mainly due to bias in the randomization process and bias in outcome measurement. Among the 46 NRSIs, most had a moderate risk of bias due to confounding. The risk of bias was generally low in the selection of participants, classification of interventions, deviations from intended interventions, and missing data. Most studies had a serious risk of bias in outcome measurement and a critical risk of bias in the selection of reported results.
Primary Outcomes
44 studies with 3495 patients presented results for back pain (Figure 2 and Appendix 7). In the analysis within 1 month postoperatively, FE-TLIF demonstrated statistically significant improvements in back pain reduction compared to MIS-TLIF (SMD −0.63, 95% CI −0.90 to −0.37, P-score 0.75), as did BE-TLIF (SMD −0.63, 95% CI −0.91 to −0.35, P-score 0.75). As the follow-up duration extended, the pain reduction effect gradually diminished. However, both techniques maintained a statistically significant advantage over MIS-TLIF at the final follow-up (FE-TLIF: SMD −0.15, 95% CI −0.23 to −0.06, P-score 0.68; BE-TLIF: SMD −0.19, 95% CI −0.29 to −0.09, P-score 0.81). Notably, no significant differences were observed between FE-TLIF and BE-TLIF across all follow-up periods. Forest plots of the pain and function. Treatments are ranked according to their P-scores. Effect sizes are presented as SMDs with 95% CIs. Abbreviations: BE, biportal endoscopic transforaminal lumbar interbody fusion; CI, confidence interval; FE, full endoscopic transforaminal lumbar interbody fusion; MIS, minimally invasive transforaminal lumbar interbody fusion; SMD, standardized mean difference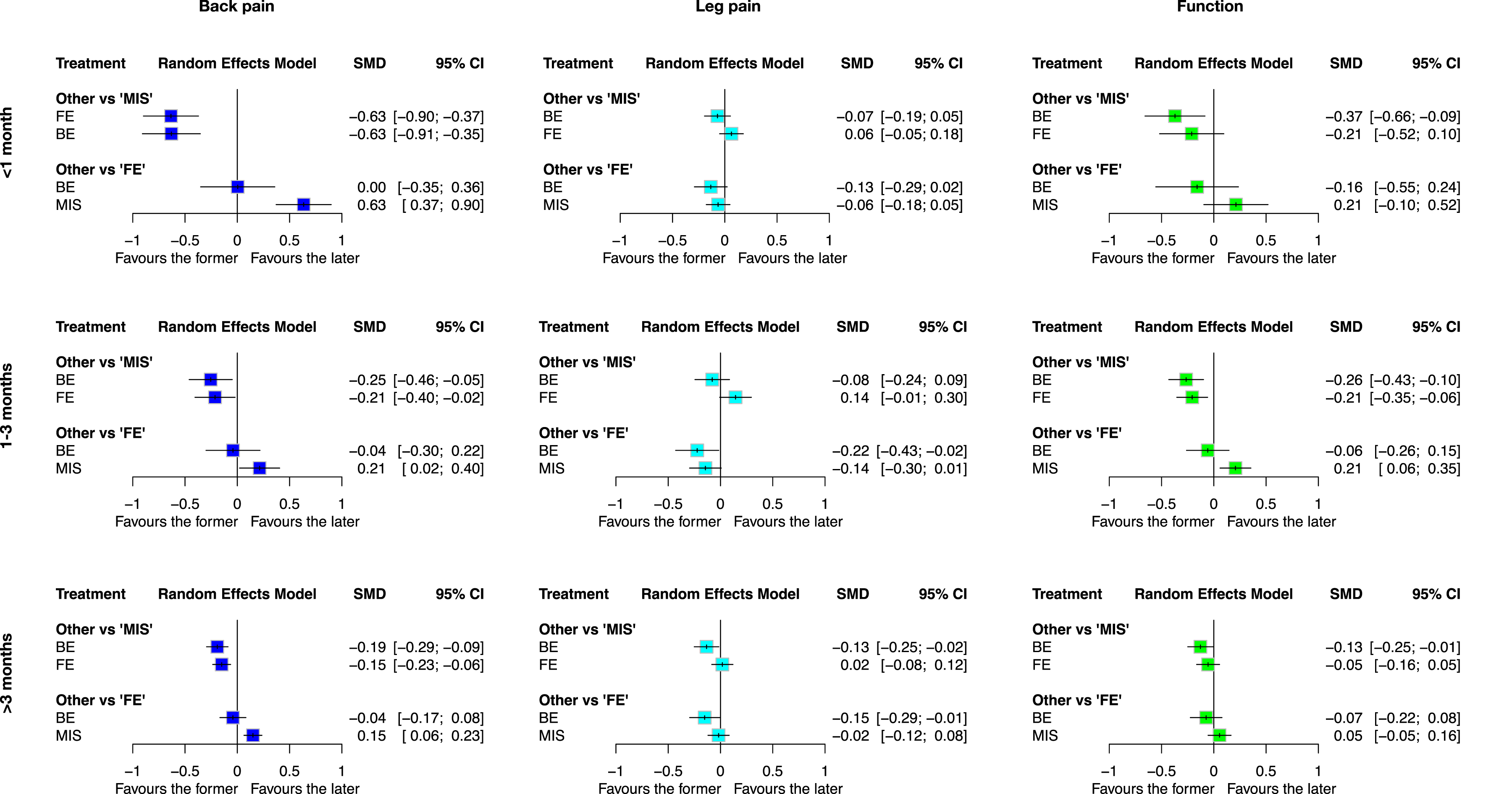
45 studies with 3697 patients presented results for leg pain (Figure 2 and Appendix 7). Although BE-TLIF showed a trend to be superior to MIS-TLIF at the final follow-up, there were generally no significant differences among the three approaches in reducing leg pain.
A total of 45 studies involving 3703 patients reported functional outcomes (Figure 2 and Appendix 7). In the short term, FE-TLIF (SMD −0.21, 95% CI −0.52 to 0.10, P-score 0.56) and BE-TLIF (SMD −0.37, 95% CI −0.66 to −0.09, P-score 0.89) demonstrated the largest effect sizes compared to MIS-TLIF, although the differences were not statistically significant. The effect size diminished gradually over time. While FE-TLIF (SMD −0.21, 95% CI −0.35 to −0.06, P-score 0.64) and BE-TLIF (SMD −0.26, 95% CI −0.43 to −0.10, P-score 0.86) demonstrated superiority over MIS-TLIF in the mid-term functional outcomes, no significant differences were observed among the three techniques at the final follow-up.
36 studies involving 2958 patients reported data on the duration of hospital stay (Figure 3 and Appendix 7). Compared to MIS-TLIF, FE-TLIF significantly reduced hospital stay (MD −1.86 day, 95% CI −2.43 to −1.30 day, P-score 0.99), as did BE-TLIF (MD −1.11 day, 95% CI −1.69 to −0.52 day, P-score 0.51). Among the three techniques, FE-TLIF ranked highest in minimizing the length of hospitalization. Forest plots of hospital stay. Treatments are ranked according to their P-scores. Effect sizes are presented as MDs with 95% CIs. Abbreviations: BE, biportal endoscopic transforaminal lumbar interbody fusion; CI, confidence interval; FE, full endoscopic transforaminal lumbar interbody fusion; MD, mean difference; MIS, minimally invasive transforaminal lumbar interbody fusion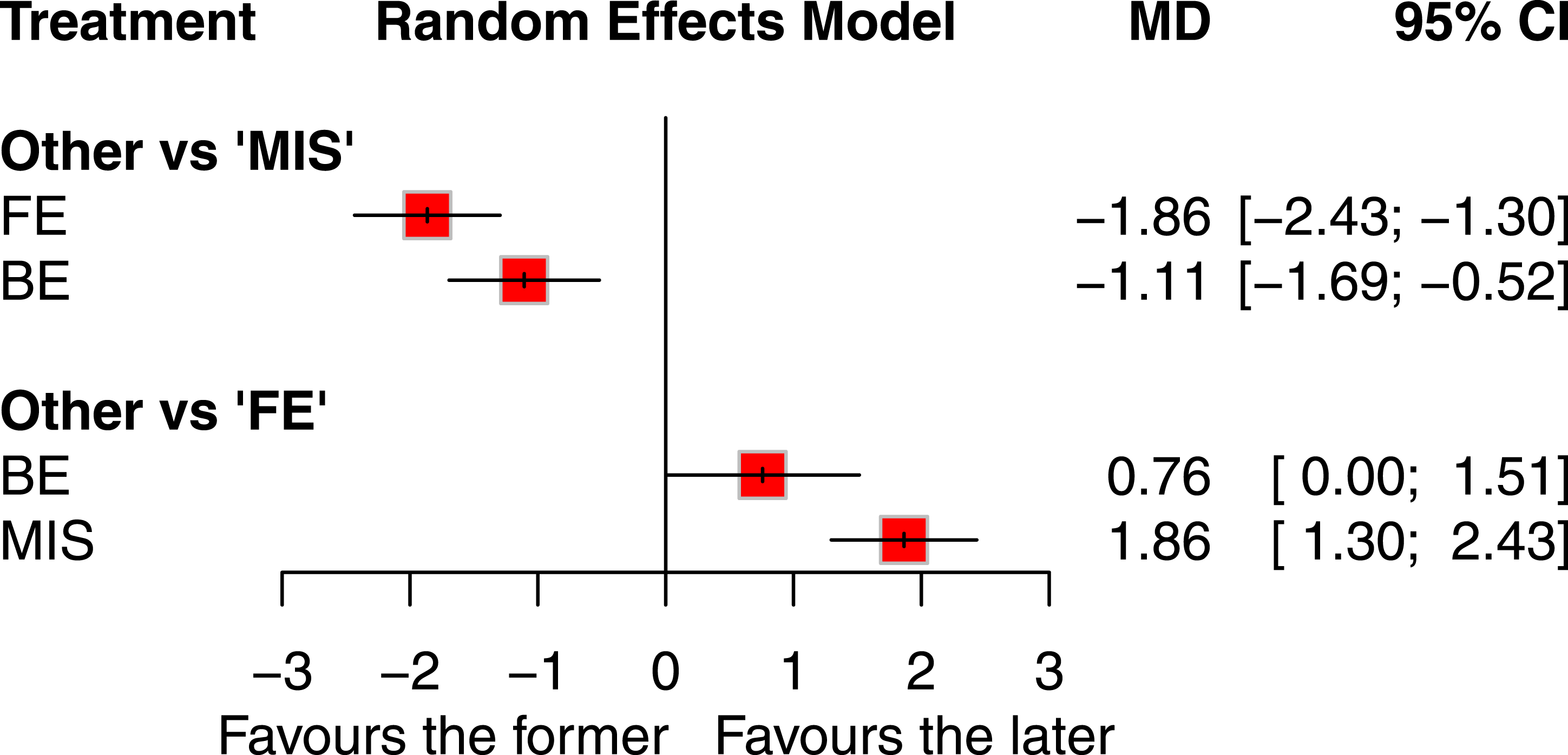
34 studies involving 2866 patients reported intraoperative blood loss outcomes (Figure 4 and Appendix 7). Both FE-TLIF (MD −72.60 mL, 95% CI −96.89 to −48.32 mL, P-score 0.84) and BE-TLIF (MD −63.64 mL, 95% CI −94.56 to −32.72 mL, P-score 0.66) were associated with significantly reduced blood loss compared to MIS-TLIF. Among the three techniques, FE-TLIF ranked as the most effective in minimizing intraoperative blood loss. Forest plots of intraoperative blood loss. Treatments are ranked according to their P-scores. Effect sizes are presented as MDs with 95% CIs. Abbreviations: BE, biportal endoscopic transforaminal lumbar interbody fusion; CI, confidence interval; FE, full endoscopic transforaminal lumbar interbody fusion; MD, mean difference; MIS, minimally invasive transforaminal lumbar interbody fusion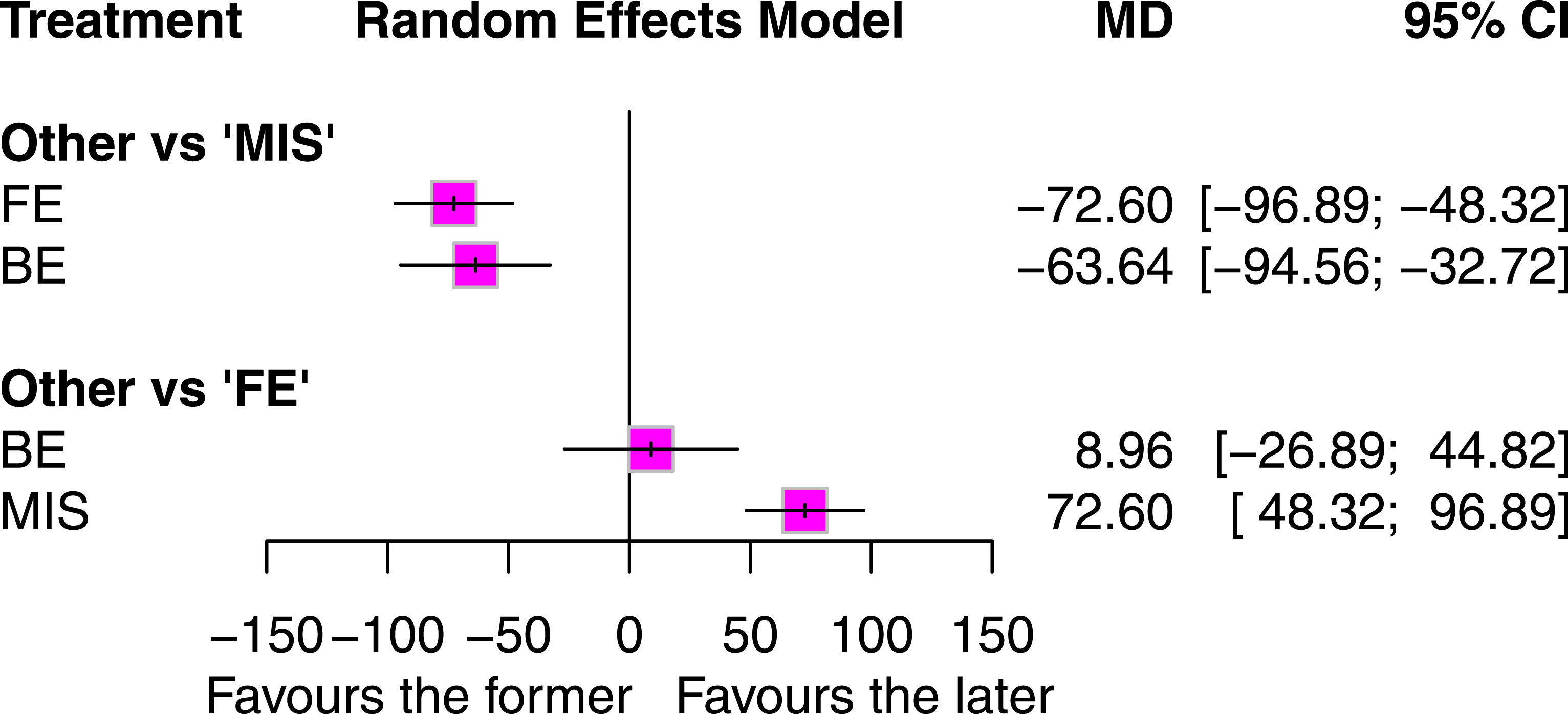
To verify the findings, we also conducted analyses within the Bayesian framework (Appendix 7.4), which yielded results consistent with those obtained under the frequentist framework.
High heterogeneity was prominently observed in the networks examining short-term back pain (τ2 = 0.35) and short-term functional outcomes (τ2 = 0.33), indicating substantial variability in these early-stage assessments. Transitioning to mid-term evaluations, moderate levels of heterogeneity were detected in networks analyzing back pain (τ2 = 0.13), leg pain (τ2 = 0.06), and functional outcomes (τ2 = 0.06). In contrast, long-term networks demonstrated low heterogeneity (back pain τ2 = 0.00, leg pain τ2 = 0.02, and function τ2 = 0.03) and non-significant global inconsistency (back pain P = 0.80, leg pain P = 0.61, and function P = 0.96), suggesting more stable and consistent patterns in pain and functional outcome measurements over extended time frames (Appendix 7.3). Substantial heterogeneity with nonsignificant global inconsistency was found in the networks evaluating hospital stay (τ2 = 1.37) and intraoperative blood loss (τ2 = 3242) (Appendix 7.3). In the meta-regressions analyses (Appendix 7.4), RCTs tended to report greater short-term reductions in back pain for FE-TLIF and BE-TLIF compared with MIS-TLIF than did NRSIs. Older patients experienced greater back pain reduction following FE-TLIF at the final follow-up. No significant effect modifiers were identified for leg pain, function, hospital stay, or intraoperative blood loss.
Secondary Outcomes
Appendix 8 summarizes the results of the secondary outcomes. 42 studies with 3381 patients demonstrated that FE-TLIF (MD 25.71 min, 95% CI 13.97 to 37.45 min, P-score 0.17) and BE-TLIF (MD 22.38 min, 95% CI 9.73 to 35.04 min, P-score 0.33) significantly increased the operation time compared to MIS-TLIF. The outcomes of the fusion rate from 38 studies involving 3392 patients indicated that FE-TLIF and BE-TLIF had an equivalent effect to MIS-TLIF. Furthermore, data from 45 studies with 3693 patients revealed no significant differences in complication rates among FE-TLIF, BE-TLIF, and MIS-TLIF. Due to low incidence rates and inconsistent reporting of specific complication types across studies, further analyses by complication category were not performed to avoid insufficient statistical power and potential bias.
Publication Bias
Comparison-adjusted funnel plots were constructed, and Egger’s regression asymmetry tests were performed (Appendix 9). No significant publication bias was observed for the outcomes in this study.
Certainty of Evidence
Given the predominance of NRSIs, the certainty of evidence was initiated at a low level, in contrast to RCTs, which commence at a high level. Taking into account the risk of bias and the heterogeneity, the certainty of evidence for the outcomes in this network meta-analysis was judged to be low to very low.
Discussion
This is the first network meta-analysis to compare the efficacy and safety of FE-TLIF, BE-TLIF, and MIS-TLIF. In this systematic review, we identified 3 RCTs and 46 NRSIs, comprising a total of 4188 patients. Both FE-TLIF and BE-TLIF were associated with significant back pain reduction across all follow-up periods and with improved functional outcomes in the short and mid term. These techniques also demonstrated shorter hospital stays and reduced intraoperative blood loss compared to MIS-TLIF. FE-TLIF demonstrated additional advantages over BE-TLIF, including greater back pain relief in older patients and shorter hospitalization. However, both endoscopic approaches were associated with longer operative times than MIS-TLIF.
Prior to our study, a few pairwise meta-analyses had evaluated the efficacy and safety of FE-TLIF and BE-TLIF compared to MIS-TLIF.14-18 Our findings largely aligned with previous studies, confirming the superiority of FE-TLIF and BE-TLIF in reducing back pain, short- to mid-term functional outcome, hospital stay, and intraoperative blood loss. However, all three techniques provide comparable effects on leg pain relief, a result not reported by previous meta-analyses. Because all TLIF variants address the same compressive pathology, radicular pain relief reaches a similar ceiling. In contrast, endoscopic approaches minimize paraspinal muscle disruption, which may result in greater relief of axial back pain.
Despite its relative inferiority in the aforementioned outcomes, MIS-TLIF was associated with a shorter operative time. This finding is consistent with those of previous studies.14-18 In cases with high anesthesia risk, significant cardiovascular comorbidities, or severe pulmonary conditions, a shorter operative time is crucial, and MIS-TLIF may be a more suitable option. Moreover, MIS-TLIF involves a shorter learning curve, relies on equipment that is widely available, and requires lower capital investment compared to endoscopic systems. These features enhance its accessibility, particularly in resource-limited settings.
Although previous meta-analyses have highlighted the benefits of FE-TLIF and BE-TLIF, direct comparisons between these two techniques remain limited in clinical research. To date, only one pairwise meta-analysis has directly compared FE-TLIF and BE-TLIF, 21 but its findings were constrained by a small number of studies and limited sample size. This study presents the most comprehensive network meta-analysis to date comparing MIS-TLIF, FE-TLIF, and BE-TLIF, integrating both direct and indirect evidence to provide a more precise and robust evaluation of their comparative effectiveness.
In our network meta-analysis, we found that FE-TLIF was associated with a significantly shorter hospital stay than BE-TLIF, whereas the previous pairwise meta-analysis reported no difference. Given the advantages of shorter hospital stays in reducing complications, lowering costs, and optimizing resource utilization, FE-TLIF may be a favorable surgical option.27-29 Additionally, although a prior meta-analysis suggested that BE-TLIF was associated with shorter operative time and higher fusion rates, our analysis found no significant differences between FE-TLIF and BE-TLIF in either fusion rate or operative time. Despite these discrepancies, our results aligned with previous findings on intraoperative blood loss, complication rates, and functional improvement. Overall, our analysis supports FE-TLIF as an effective alternative to BE-TLIF, particularly in enhancing postoperative recovery efficiency.
Although endoscopic techniques are less invasive, they require specialized endoscopic systems, high-definition imaging equipment, and steeper learning curves, which increase initial setup costs. The need for disposable endoscopic instruments and specialized implants may also elevate per-case expenditures compared to MIS-TLIF. Although reduced hospital stay and faster recovery can offset some costs, the overall cost-effectiveness of endoscopic TLIF largely depends on surgical volume and institutional resources. Centers with established endoscopic programs and experienced surgeons are more likely to achieve economic viability through shorter hospitalization and faster patient turnover. However, comprehensive cost-effectiveness analyses comparing endoscopic fusion procedures with other minimally invasive techniques remain limited. Future studies incorporating direct and indirect costs, quality-adjusted life years, and long-term economic outcomes are needed to definitively establish the cost-effectiveness of endoscopic fusion procedures in different healthcare settings.
While our pooled data did not reveal a higher incidence of dural tears in endoscopic TLIF compared to MIS-TLIF, this may reflect underreporting, as many included studies lacked detailed complication profiles. Furthermore, approach-related complications such as exiting or traversing nerve irritation, neural element manipulation, or epidural fluid retention have been described in the literature. These factors warrant consideration in clinical decision-making. The lack of standardized complication classification across studies may have masked endoscopic-specific risks. Future trials should adopt unified reporting criteria to enable more accurate risk profiling.
Heterogeneity analysis revealed that pain and functional outcome networks exhibited greater variability in the short-term measurements, but became more stable over longer follow-up periods. Additionally, meta-regression analysis revealed that older patients experienced greater back pain reduction at the final follow-up with FE-TLIF. Given this advantage, along with the shortest hospital stay associated with FE-TLIF, it shows potential benefit for older patients. This exploratory meta-regression finding, while subject to ecological bias inherent to aggregate-level data, provides a hypothesis-generating signal for age-related treatment effects. However, since the included studies did not stratify outcomes by predefined age thresholds or provide individual participant data, subgroup analyses by patient age in this network meta-analysis were not feasible. Future randomized controlled trials with prospective age-stratified analyses would help validate this finding and establish more precise age cutoffs for optimal surgical selection.
By contrast, no significant associations were identified in the meta-regressions for other effect modifiers. While variations in surgical techniques such as cage type and decompression method could theoretically affect transitivity, meta-regression analyses showed no significant impact of these factors on primary outcomes, and key modifiers were similarly distributed across comparisons (Appendix 6), supporting the validity of our indirect comparisons. Nonetheless, heterogeneity was not fully unexplained by the meta-regressions. Residual heterogeneity may stem from unmeasured confounders such as surgeon experience, patient comorbidities, and intraoperative variations (eg, irrigation extent in BE-TLIF, visualization quality in FE-TLIF), which were inconsistently reported across studies. Standardized reporting of these factors in future research would help clarify residual sources of heterogeneity.
This study has several limitations. First, the majority of included studies were NRSIs, with a limited number of RCTs. Due to their non-randomized design, NRSIs are inherently prone to confounding bias. Although many of the included NRSIs employed methodological strategies to mitigate this bias, it cannot be entirely excluded. In addition, while we conducted meta-regressions to assess the potential influence of study design and study quality, and the results indicated no significant effect, all included RCTs were at high risk of bias. Given the predominance of NRSIs, the presence of heterogeneity, and these methodological limitations, the overall certainty of evidence was rated as very low to low across most outcomes according to GRADE criteria. Future research would benefit from more rigorously designed RCTs to improve the certainty of evidence for clinical decision-making. Second, substantial heterogeneity was observed in outcomes such as short-term back pain, short-term functional outcomes, hospital stay, and intraoperative blood loss. This heterogeneity could not be fully accounted for by the predefined effect modifiers included in our analysis. Future studies should explore additional factors contributing to the observed between-study variability. Third, the meta-regression analyses may have been underpowered and remain vulnerable to confounding and ecological biases. Therefore, the results should be interpreted with caution. Fourth, the study population was predominantly aged 55-65 years with single-level disease, which may limit the generalizability of our findings to younger or older patients and those undergoing multilevel surgical intervention.
Conclusion
FE-TLIF and BE-TLIF are safe and minimally invasive alternatives that may offer certain advantages over MIS-TLIF, including better back pain relief, improved short- and mid-term function, shorter hospital stays, and less intraoperative blood loss. In clinical practice, FE-TLIF may provide greater back pain relief in older patients, while MIS-TLIF remains a viable choice for those with higher anesthesia risk. However, both BE-TLIF and FE-TLIF require longer operative times and may be associated with approach-specific complications that must be considered. From a research perspective, further prospective, long-term, and comparative randomized studies are needed to validate these findings and optimize patient selection.
Supplemental Material
Supplemental Material - Comparative Efficacy and Safety of Minimally Invasive, Full-Endoscopic, and Biportal Transforaminal Lumbar Interbody Fusion for Lumbar Degenerative Diseases: A Network Meta-Analysis
Supplemental Material for Comparative Efficacy and Safety of Minimally Invasive, Full-Endoscopic, and Biportal Transforaminal Lumbar Interbody Fusion for Lumbar Degenerative Diseases: A Network Meta-Analysis by Hsiao-Yi Cheng, Chun-Wei Liang, Yuan-Fu Liu, Chao-Jui Chang, Wei-Lun Chang, Ting-An Cheng, Hao-Chun Chuang, Yu-Chia Hsu, Cheng-Li Lin in Global Spine Journal
Footnotes
Acknowledgement
We are grateful to the Skeleton Materials and Bio-compatibility Core Lab, Clinical Medicine Research Center, National Cheng Kung University Hospital for assistance with this study.
Author Contributions
Hsiao-Yi Cheng and Chun-Wei Liang contributed equally to this work. Conceptualization: Hsiao-Yi Cheng, Chun-Wei Liang, Yuan-Fu Liu, Chao-Jui Chang, Wei-Lun Chang, Ting-An Cheng, Hao-Chun Chuang, Yu-Chia Hsu, and Cheng-Li Lin; data curation: Chun-Wei Liang and Hsiao-Yi Cheng; formal analysis, validation, and visualization: Chun-Wei Laing and Hsiao-Yi Cheng; writing - original draft: Chun-Wei Liang and Hsiao-Yi Cheng; writing - review and editing: Yuan-Fu Liu, Chao-Jui Chang, Wei-Lun Chang, Ting-An Cheng, Hao Chun Chuang, Yu-Chia Hsu, Cheng-Li Lin.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Taiwan National Science and Technology Council (Award Number: NSTC 112-2314-B-006-075, NSTC 114-2314-B-006-050). This study was also supported by National Cheng Kung University Hospital, Tainan, Taiwan (Award Number: NCKUH-11402019, NCKUH-11406014).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
PROSPERO
CRD42024625884.
Supplemental Material
Supplemental material for this article is available online.
