Abstract
Study Design
Case-control study.
Objectives
Degenerative Cervical Myelopathy (DCM) is a progressive neurological condition caused by mechanical stress on the cervical spine. Surgical exposure in the preceding months to a DCM diagnosis is a common theme of Patient and Public Involvement (PPI) discussions. Such a relationship has biological plausibility (e.g. neck positioning, cord perfusion) but evidence to support this association is lacking.
Methods
We analysed UK Hospital Episode Statistics (HES) data for participants in the UK BioBank cohort. We defined cases as those episodes with a primary diagnosis of DCM and generated controls using non-DCM HES episodes. Cases and controls were propensity score-matched by age, sex and date of episode, and a directed acyclic graph was used to robustly control for confounders. We defined the exposure as any surgical procedure under general or regional anaesthetic occurring within the 6-24 months prior to the episode.
Results
We analysed 806 DCM and 2287432 non-DCM hospital episodes. On multivariable logistic regression analysis, the odds ratio (95% CI) for the effect of a binarised (0 vs ≥ 1) exposure on risk of developing DCM was 1.20 (1.02-1.41), and for categorised (0 vs 1 and 0 vs ≥ 2) exposure was 1.11 (0.882-1.39) & 1.33 (1.075-1.65).
Conclusions
This study supports the patient narrative of surgery as a risk factor for the development of DCM. The association displays temporality, dose-response relationship, and biological plausibility. Further work is needed to confirm this in other cohorts, explore mediating mechanisms, and identify those at greatest risk.
Introduction
Degenerative Cervical Myelopathy (DCM) is a slow-motion spinal cord injury estimated to affect 1 in 50 adults. 1 It arises when degenerative changes in the cervical spine, such as disc prolapse, osteophyte formation, ligament hypertrophy and/or calcification, compress and injure the spinal cord. 2 DCM is typically characterised by a loss of dexterity, imbalance, urinary dysfunction, and pain; the symptoms are commonly mistaken for aging.3,4 Surgery to alleviate spinal cord compression is the only treatment for DCM. This can stop further spinal cord injury, but due to the limited intrinsic capacity of the spinal cord to repair, recovery is dependent on the extent of existing injury. Ensuring timely access to treatment is therefore critical to prevent long-lasting disability. 5 Unfortunately, today it is estimated less than 10% of DCM is diagnosed, 1 with those that are waiting on average 1 to 3 years. 6 Surgery therefore rarely restores patients fully, and DCM is associated with high levels of disability 7 and societal cost (£0.7bn in the UK). 8
The aetiology of DCM is poorly understood. Whilst spinal cord compression is a hallmark of diagnosis it is observed that compression is 10 times more likely to be an incidental finding (so called asymptomatic spinal cord compression), 9 and correlates poorly with disease severity. 10 This has led to a new framework that defines DCM as a function of mechanical stress (the mechanical loading of the spinal cord), vulnerability (an individual’s capacity to retard or tolerate spinal cord loading) and time (length of time under mechanical loading), in the presence of spinal cord compression. 11 The framework proposes that to manifest DCM, mechanical loading must overwhelm intrinsic repair processes and any functional reserve capacity. 11
Over the last 5 years, the RECODE-DCM (Research Priorities and Common Data Elements in DCM) initiative has formed a multidisciplinary community of people living and working with DCM 12 with the ultimate goal of enabling patient-centred research. 13 Patient and public involvement (PPI) work through this initiative has revealed a common patient experience of an unrelated surgical procedure in the build-up to their diagnosis of DCM.
Several candidate hypotheses may explain this observation. Coincidence: people with DCM are frequently misdiagnosed and perhaps treated for other conditions prior to a diagnosis.14,15 Confounding: it is conceivable that patients with DCM may have additional co-morbidities that carry with it an increased risk of operative treatment for these other conditions. Alternatively, the association may be causal. In patients with known DCM, intubation proceeds with cervical immobilisation (avoiding extension which will narrow the spinal canal and worsen compression), 16 and typically ensures a higher mean arterial pressure to maintain spinal cord perfusion. 17 These steps are not necessarily employed in other operative settings, and it is conceivable that repetitive attempts at intubation (especially after encountering a ‘difficult’ airway), intraoperative hypotension, or surgical positioning could compound spinal cord loading and injury.
Given the potential biological rationale for this patient-reported association, this study examines the hypothesis that prior surgical exposure could be causally related to the development of DCM. Secondary sensitivity analyses were conducted to explore potentially relevant mediating mechanisms (such as positioning) that could explain any observed association between preceding surgery and diagnosis of DCM.
Methods
Data Access and Approvals
Data were accessed under the terms of a Material Transfer Agreement provided by UK Biobank. Ethics review was provided by UK Biobank (Approved Research ID 73666), 18 with no Institutional Review Board approval required. All participants provided written and ongoing informed consent to UK Biobank; participants who withdrew their data during analysis were excluded. Therefore, no further specific informed consent was gained for this study. All data analysis was performed using R version 4.3.1. 19 All R scripts are available. 20
Study Design
To estimate the effect of a surgical exposure on the development of DCM, we conducted a case-control study (Figure 1) nested within the UK Biobank cohort. This design was chosen in order to maximise statistical efficiency whilst avoiding the disadvantages of retrospective study design.
21
In this section, we first describe the Biobank cohort as it pertains to our study (Setting), and then the participants and variables used. Nested case-control study design. Cases are defined by a hospital admission for DCM. Controls are defined by a non-DCM hospital admission. Exposure is defined as an operation within the 18 month sampling window, 6-24 months prior to the admission.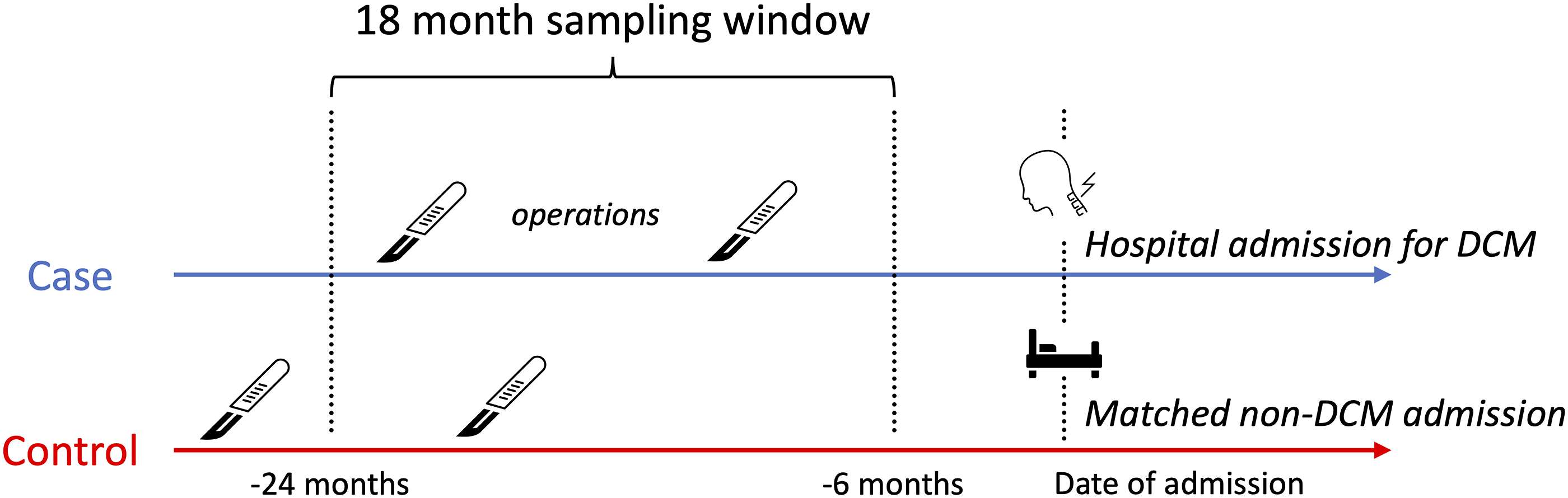
UK Biobank is a large-scale biomedical database and research resource containing genetic, lifestyle and health information from half a million UK participants, which has been previously described. 22 This research was prospectively approved by UK Biobank (application ID 73666), with a lay summary available online. 23 Reporting is in line with STROBE and RECORD checklists (Supplemental 5). 24
Setting & Data Sources
Location, Recruitment, Follow-Up
UK Biobank recruited approximately 500,000 people across the country aged between 40 and 69 years from 2006 to 2010. Assessments were undertaken in 22 centres in Scotland, England and Wales. As part of the assessment process, participants completed touch screen questionnaires, face-to-face interviews, physical measurements, and sample collection of blood, urine, and saliva. Approximately 20,000 of these participants undertook repeat measures.
Data Collection
Participants were linked with their NHS hospital inpatient data for England (Hospital Episode Statistics Admitted Patient Care), Wales (Secure Anonymised Information Linkage Databank Admitted Patient Care), and Scotland (General Acute Inpatient and Day Case – Scottish Morbidity Record).
Records date back to 1997 for England, 1998 for Wales and 1981 for Scotland and contain coded data on admissions (ICD codes), operations and procedures (OPCS codes). Not every participant will have a hospital inpatient record, as not all have been admitted to hospital within the period covered. UK Biobank receives hospital inpatient data from each data provider on an approximately annual basis. 25
We downloaded the Biobank dataset on 10 August 2022. Hence, our dataset contains records for all recruited participants, except those who subsequently withdrew their data from Biobank. 26 It contains linked hospital data from inception until 30 September 2021.
Participants
Case Ascertainment
We defined cases as hospital episodes with a primary diagnosis (ICD-10) code compatible with DCM. We used the following codes, as previously published
1
: • M4712: Other spondylosis with myelopathy (Cervical region) • M500: Cervical disk disorder with myelopathy • M9931: Osseous stenosis of neural canal (Cervical region) • M9941: Connective tissue stenosis of neural canal (Cervical region) • M9951: Intervertebral disc stenosis of neural canal (Cervical region)
The primary diagnosis code is the main condition treated or investigated during the hospital episode. We chose this method of case ascertainment to maximise specificity, as many hospital episodes had DCM coded as a secondary diagnosis with an unrelated primary diagnosis (Supplemental 1). Where a single participant had multiple hospital episodes with a primary diagnosis of DCM, we used the first episode only, as this was felt to be most biologically relevant.
Control Selection
We defined controls as all other hospital episodes within the Biobank dataset, from participants with no DCM episodes. This method was chosen to maximise statistical power whilst also allowing matching.
We used propensity score matching of cases and controls, using matching weights to create balanced case and control groups. 27 We matched on age at episode, participant sex, and date of episode (Supplemental 2). The latter variable was chosen to control for time-based effects such as changes in medical practice and the reduced rates of operative procedures during the COVID-19 pandemic, across the time period covered by the linked health records (1981 to 2021 as above).
Study Size
In the absence of any pilot data on which to base a power calculation, we utilised the largest practically-available dataset with the variables of interest, UK Biobank. We maximised case and control numbers within this dataset (see Participants).
Statistical Methods
Outcome and Exposure Definition
The outcome of interest in cases was a hospital admission for DCM.
We defined the exposure as any procedure (OPCS-4) code compatible with general or regional anaesthetic, occurring within a sampling window of 6-24 months prior to the relevant hospital episode. This sampling window was based on existing literature showing a mean time from symptom onset to neurosurgical review of 16 months. 6 The OPCS-4 code list was chosen to maximise biological relevance and was derived from a local list of procedures with an anaesthetist in attendance, identified from ongoing approved research studies at our institution (ref: 19/L0/1648).
Biobank holds no data on intraoperative events (e.g. airway manipulation or intraoperative physiology). However, we used available OPCS information to identify subsets of procedures where patients would be reliably ‘non-supine’ – a surrogate for more awkward positioning that could, arguably, result in greater external stress being placed on a vulnerable individuals neck. For this sensitivity analysis, we defined the exposure as any procedure (OPCS-4) code compatible with an operation conducted in a non-supine position (including shoulder surgery (sitting), hip replacement (lateral)). Furthermore, we identified individuals having prior spinal (any level) and non-spinal operations. All OPCS-4 code lists are available as supplementary data (Supplemental 3).
Confounder Adjustment and Definition
To minimise over-correction bias (inclusion of non-confounding covariates), we used a Directed Acyclic Graph (DAG, Figure 2).
28
This is recognised as the gold-standard way to visualise a priori causal relationships between variables and to attempt to isolate a specific causal effect (estimand).
29
Potential confounders were identified based on published literature,30,31 edge inclusion and direction were made based on considerations of biological plausibility and temporality.
32
We used dagitty
33
to identify the minimum adjustment set based on our DAG – a set of variables to be fully conditioned on to provide an unbiased effect estimate.
33
Variables within this adjustment set were included in our final statistical model (either through matching or inclusion as an additional independent variable) to attempt to isolate the causal effect of prior surgical exposure on the development of DCM. Directed Acyclic Graph. Hypothesised causal links flow from left to right. Resultant variables forming adjustment set highlighted in red – these variables were conditioned within the logistic regression model.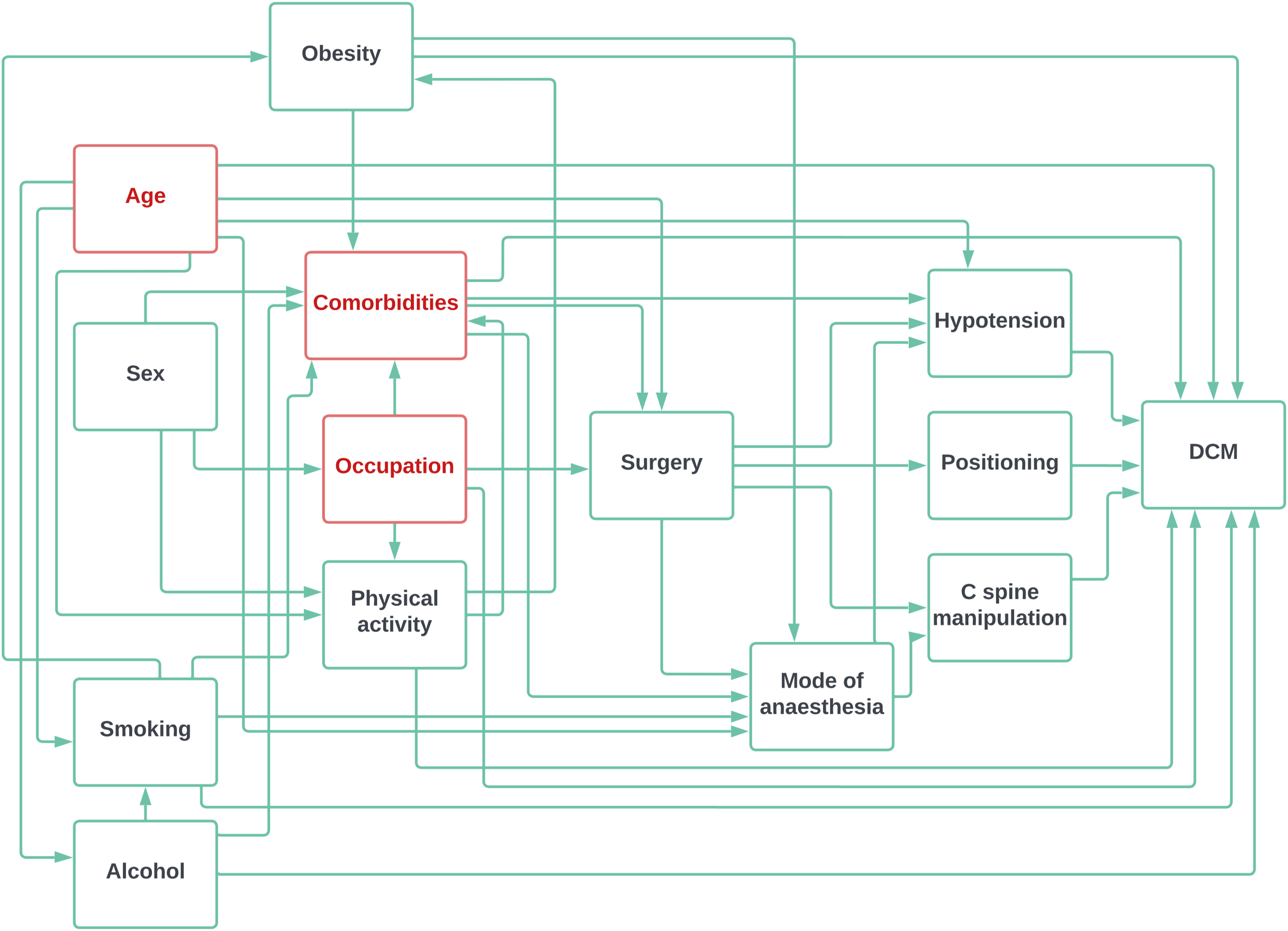
We defined comorbidities potentially relevant to DCM development using the Charlson Comorbidity Index, which is an established tool for quantifying comorbidity in registry-based research. 34 For each hospital episode, we counted instances of ICD-10 codes compatible with congestive heart failure, cerebrovascular disease, diabetes mellitus, myocardial infarction, and peripheral vascular disease, in that participant, prior to that episode. We generated new data columns for these comorbidity counts for each episode.
We defined occupation as the extent to which the participants’ job involved heavy manual or physical work. 35 We coded non-employment as a distinct value.
Matching
We generated a dataset where each row represented a unique hospital episode, with corresponding ICD-10 and OPCS-4 codes. Participant characteristics (as measured at Biobank assessment centres) for the patients in those hospital episodes were included. We excluded non-DCM hospital episodes from participants who also had a DCM hospital episode (so that a participant could not be their own control), and subsequent DCM episodes after an initial DCM episode (for the same reason).
We generated new data columns for DCM (a binary variable, positive if any of the specified ICD-10 codes were present for that episode), age at episode (date of episode minus participant date of birth), and number of exposures within the sampling window. As a sensitivity analysis, we processed the raw number of exposures (a count of procedures compatible with general or regional anaesthetic) into a binarised variable (0 vs ≥ 1 procedures within sampling window) and categorised variable (0 vs 1 and 0 vs ≥ 2 procedures within sampling window) to provide an exploratory analysis as to any potential ‘dosing’ effect. Categorised exposures are reported with 2 separate odds ratios (0 vs 1 and 0 vs ≥ 2), except for non-supine exposures where there were no instances of ≥2 exposures in the case group.
We used the MatchIt package 27 to perform propensity score matching. We then performed a multivariable logistic regression with DCM as the outcome and surgical exposure, covariates and their interaction as predictors and included the full matching weights in the estimation. Included covariates were defined by the DAG adjustment set (Figure 2) – since age had already been matched upon, we used occupation and comorbidities as covariates.
Results
Participants
Our original downloaded UK Biobank dataset contained 502,409 participants. 40 participants withdrew their data during the analysis stage, leaving 502,369 participants eligible for inclusion. Of these, 806 (0.16%) participants had a hospital episode with a primary diagnosis code compatible with DCM, a prevalence in line with previous estimates. 1
These participants had 2,296,283 hospital episodes in total. Of these, 833 were DCM episodes and 2,295,450 were non-DCM episodes. There were 7979 non-DCM episodes from participants who also had a DCM episode – these were excluded. There were 39 non-DCM episodes where the participant year of birth was not available – these were excluded. This left 2,287,432 non-DCM episodes as controls for analysis. There were 27 subsequent DCM episodes from participants who had already had a ‘first’ DCM episode – these were excluded, leaving 806 DCM episodes as cases for analysis. All cases and controls were used for matching. Matching statistics are shown in Supplemental 4.
Descriptive Data
Characteristics of Study Participants
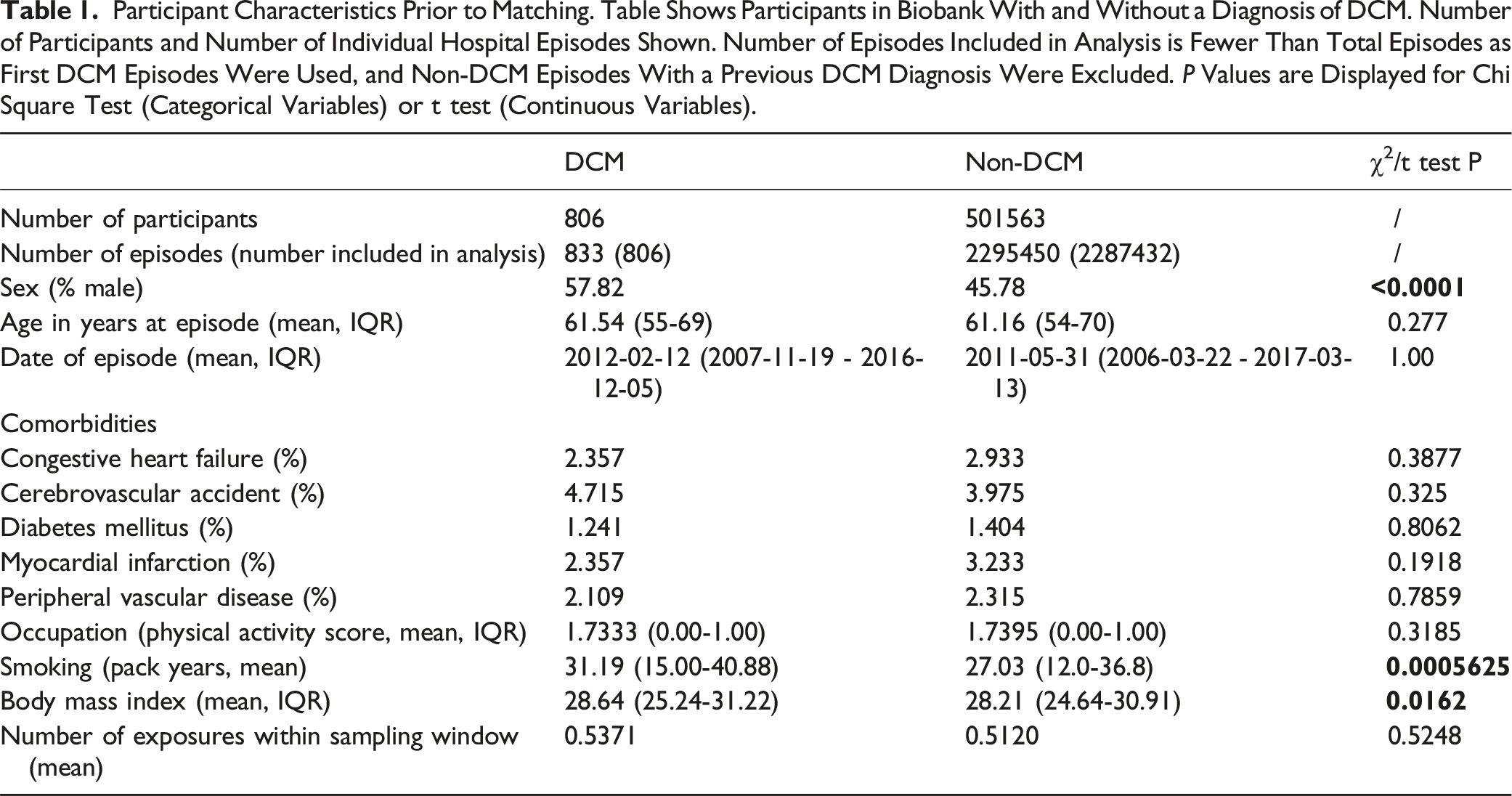
Participant Characteristics Prior to Matching. Table Shows Participants in Biobank With and Without a Diagnosis of DCM. Number of Participants and Number of Individual Hospital Episodes Shown. Number of Episodes Included in Analysis is Fewer Than Total Episodes as First DCM Episodes Were Used, and Non-DCM Episodes With a Previous DCM Diagnosis Were Excluded. P Values are Displayed for Chi Square Test (Categorical Variables) or t test (Continuous Variables).
Missing Data
There were no missing data for outcome variables, as all episodes either had a relevant ICD-10 code (hence were cases) or did not (hence were controls). There was no missing data for age at episode (since those episodes had already been excluded) or date of episode. There was no missing data for comorbidity status or occupation. There were missing data for number of exposures (11 cases and 33,033 controls) – given their small number, these episodes were excluded from analysis.
Main Analysis
Number of Exposures. Table Shows the Number of DCM and Non-DCM Episodes With a Given Number of Exposures (OPCS-4 Codes Compatible With Surgery Under General or Regional Anaesthetic) Within the Sampling Window.

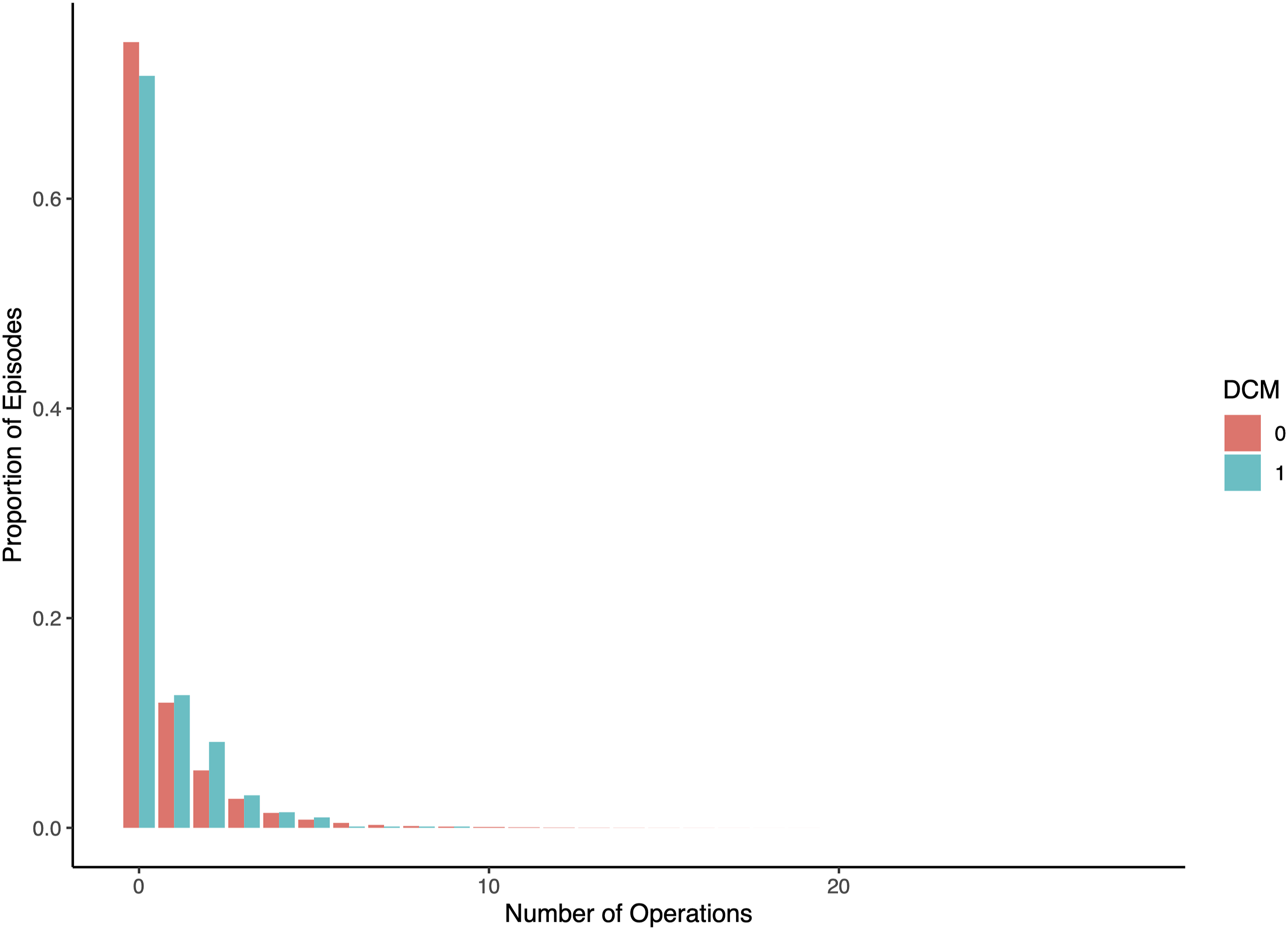
Frequency distribution. Plot shows number of number of operations in the two years to six months prior to episode, for DCM (blue) and non-DCM (red) episodes. Frequencies normalised to proportion of total number of episodes. It is observed that cases are less likely to have zero exposures, and more likely to have ≥1 exposures, than controls.
Results of Logistic Regression Models. Table Shows Hypothesised Predictors of DCM Diagnosis. Chi-Square Tests for Confounders are Shown First. Results of Univariable and Multivariable Logistic Regression Models are Then Shown, With Count, Binarised and Categorised Exposure Definitions. Categorised Exposures Display Statistics for 0 vs 1 and 0 vs 2+ Exposures, Unless There Were No Cases of 2+ Exposures in the DCM Group (Nonsupine). OR, Odds Ratio; CI, Confidence Interval; CHF, Congestive Heart Failure; CVA, Cerebrovascular Accident; DM, Diabetes Mellitus; MI, Myocardial Infarction; PVD, Peripheral Vascular Disease.
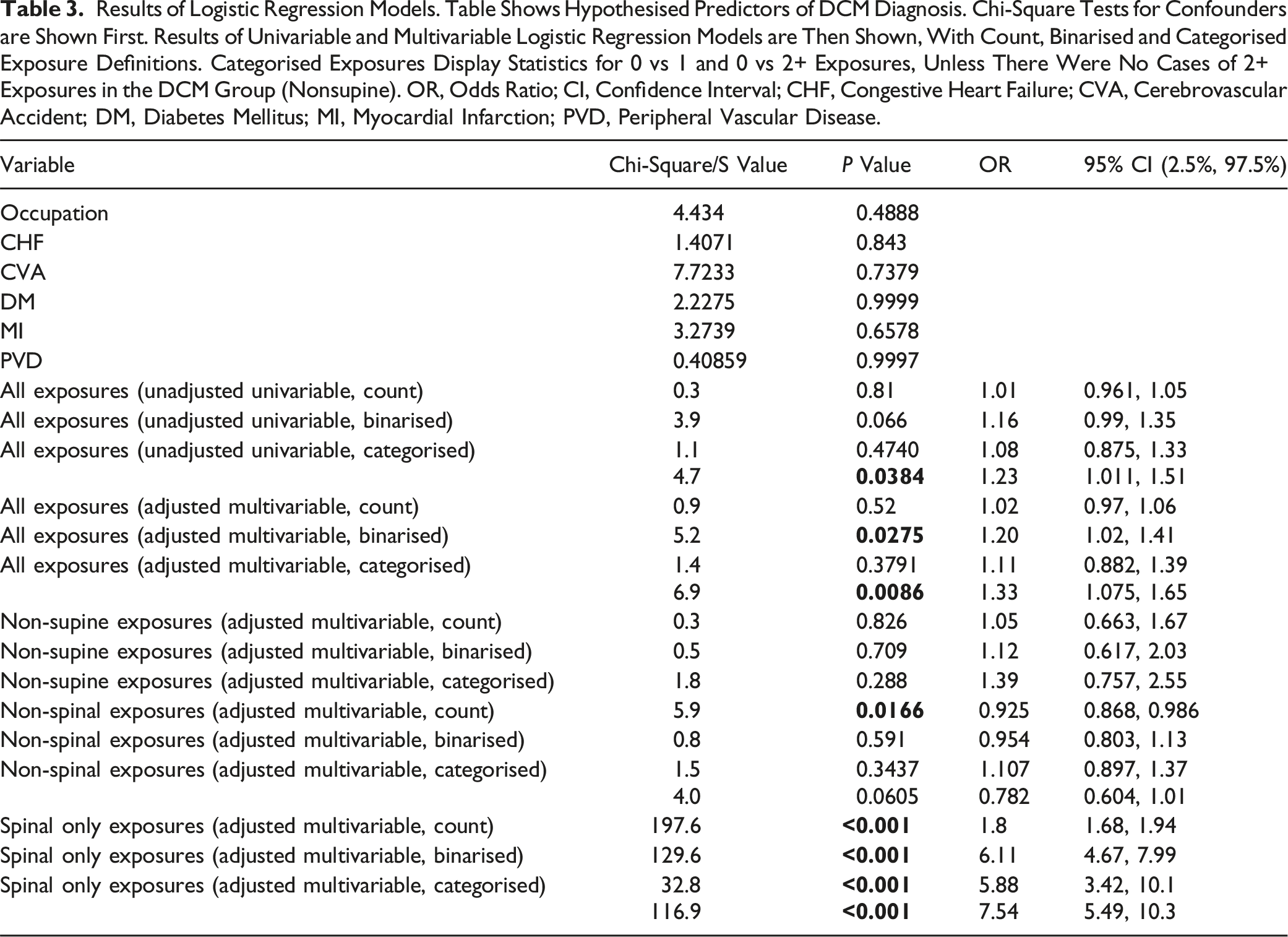
The adjusted multivariable logistic regression model using all variables from our minimum adjustment set gave an odds ratio of 1.20 (95% CI = 1.02-1.41, P = 0.0275) for a binarised exposure, and 1.11 (95% CI = 0.882-1.39, P = 0.3791) & 1.33 (95% CI = 1.075-1.65, P = 0.0086) for a categorised exposure (Figure 4). Sensitivity analysis. Odds ratios for all operations under regional / general anaesthetic, non-supine operations (e.g., shoulder surgery, hip replacement), non-spinal operations and spinal only operations are shown. Odds ratios and 95% confidence intervals for binarised (0 vs ≥1) and categorised (0 vs ≥2) exposures are shown.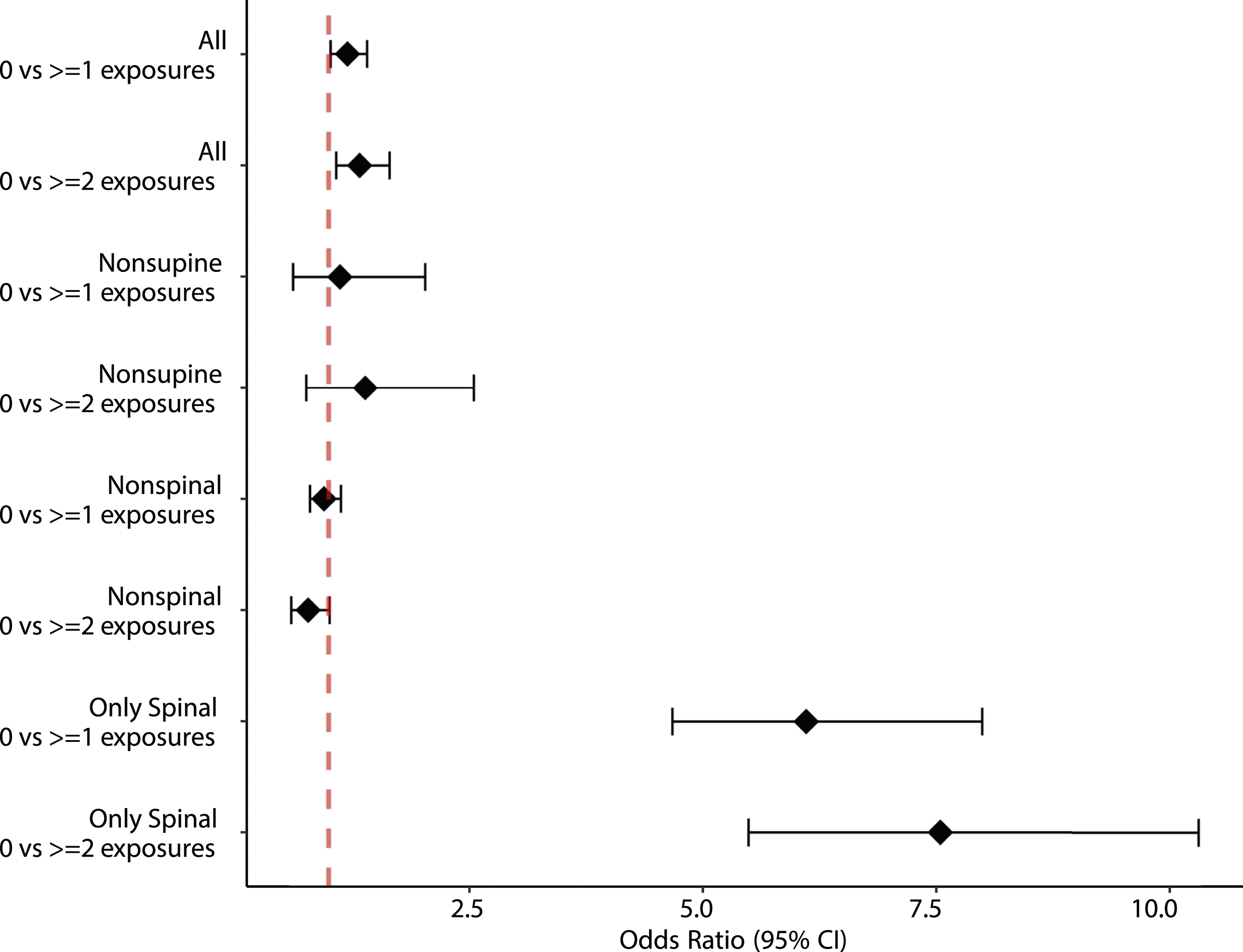
Sensitivity Analyses
Non-supine Positioning
Defining the exposure as surgical procedures in a non-supine position (shoulder surgery, hip replacement), both controls and cases had a mean of 0.01368 exposures within the sampling window. The adjusted multivariable logistic regression model gave an odds ratio of 1.12 (95% CI = 0.617-2.03, P = 0.709) for a binarised exposure, and 1.39 (95% CI = 0.757-2.55, P = 0.288) for a categorised exposure (Figure 4).
Spinal vs Non-spinal Procedures
Defining the exposure as any non-spinal procedure under general/regional anaesthetic (i.e. the primary exposure definition, with spinal procedures removed), controls had a mean of 0.4902 exposures within the sampling window, whilst cases had a mean of 0.4015 exposures. The adjusted multivariable logistic regression model gave an odds ratio of 0.954 (95% CI = 0.803-1.13, P = 0.591) for a binarised exposure, and 1.107 (95% CI = 0.897-1.37, P = 0.3437) & 0.782 (95% CI = 0.604-1.01, P = 0.0605) for a categorised exposure (Figure 4).
Defining the exposure solely as spinal procedures, controls had a mean of 0.0253 exposures within the sampling window, whilst cases had a mean of 0.1572 exposures. The adjusted multivariable logistic regression model gave an odds ratio of 6.11 (95% CI = 4.67-7.99, P = <0.001) for a binarised exposure, and 5.88 (95% CI = 3.42-10.1, P = <0.001) & 7.54 (95% CI = 5.49-10.3, P = <0.001) for a categorised exposure (Figure 4).
Discussion
Key Results
Our results show that surgical exposure under general or regional anaesthetic is associated with subsequent development of DCM. This association is particularly strong for spinal procedures. By guiding our multivariable adjustment with a DAG, our analysis, within the limitations of our proposed causal model and variable definitions, suggests that this association may be causal. Further work to triangulate or corroborate this finding is vital but, to our knowledge, this is the first time that such an association has been reported in the literature. Importantly, it adds quantitative backing to a core theme of patient experience identified through earlier work.
Limitations and Generalisability
DCM lacks a specific disease code. 36 Case ascertainment has therefore relied on a selection of previously-published diagnosis codes, 1 but the true sensitivity and specificity of this method is unknown.
The study has been conducted using observational routinely-collected health data. This has several challenges. The UK Biobank cohort is not necessarily representative of the wider population, with evidence of a ‘healthy volunteer’ selection bias with a lower prevalence of obesity and smoking, both relevant covariates for DCM.30,37 This could affect our identified prevalence, although reassuringly this is similar to previous estimates based on the entire HES dataset. 1 This potential selection bias38,39 could also limit the external validity of our results, meaning the findings will need to be validated in other cohorts.
Missing data is another challenge. This occurs as a result of incomplete capture – e.g. incomplete linkage between UK Biobank and HES – but also due to the limitations of the Biobank dataset. The UK Biobank dataset does not, for instance, capture intraoperative blood pressure, positioning, or airway management. This means that whilst a confounder-controlled association was able to be found, there is limited scope to uncover ‘why’ this occurs through detailed causal mediation analysis.
Case ascertainment bias is another issue. Our case definition only included the relevant ICD-10 codes in the ‘primary diagnosis’ field, because exploratory analysis showed that many hospital episodes had DCM coded as a secondary diagnosis with an unrelated primary diagnosis (Supplemental 1). This will have improved our specificity but at the expense of sensitivity. Further, the HES database captures only inpatient diagnoses, representing another possible loss of sensitivity. However, our previously published work has shown that outpatient attendances represent just over a tenth the incidence of inpatient episodes. 1 Further, the diagnosis of DCM requires secondary care assessment, 6 so this is unlikely to be a substantial source of case ascertainment bias.
Finally, the choices made in this novel analysis represent a source of potential measurement error. The 18 month sampling window is based on existing literature 6 and has biological plausibility, but time to diagnosis varies widely. 6 Likewise, our DAG is based on published work 30 with temporally plausible links, but other nodes and arcs are possible.
Interpretation
Whilst erroneous surgical treatment has been reported during the diagnostic workup for DCM, 40 the finding that repeated surgical exposure further increases the risk of developing DCM (i.e., a dose-response relationship) points towards a more fundamental biological link.
The precise link however remains unclear. DCM is proposed to be a function of 3 factors: mechanical stress (e.g. mechanical loading of the spinal cord from degenerative pathology), vulnerability (e.g. propensity to develop spinal cord compression and/or spinal cord injury in response to mechanical loading), and time. 11 Sensitivity analysis demonstrated that operative positioning is unlikely to be the key mediating factor, with an insignificant odds ratio for non-supine operations. By contrast, spinal procedures seem to be particularly high risk for subsequent DCM. This points away from ‘mechanical stress’ being important and instead towards vulnerability. Vulnerability as a domain encompasses a range of potentially relevant mechanisms, such as spinal cord hypoperfusion, advanced biological age and genetics. The association with prior spinal surgery here is striking in this regard. A number of contemporary studies have started to show a relationship between lumbar canal stenosis - but not lumbar disc herniation - and DCM, and to explore the value of screening.41,42 Conceivably genetic factors that predispose either to the onset of spinal degeneration, or the propensity to develop symptoms from spinal degeneration, may affect the entire spine. 31 Further, lumbar canal stenosis (but not lumbar disc herniation) is associated with advanced age. There could also be an effect of the health care system itself – namely, that patients who have undergone prior spinal surgery will likely be under followup and may have had whole spine imaging, and therefore more likely to have subsequent cervical myelopathy diagnosed and treated in a timely manner.
Further mechanistic exploration is likely to require a different form of research. For instance, genetic studies could shed further light on mediators of spinal vulnerability. Such studies could be conducted using the Biobank cohort. However, examination of intra-operative factors such as neck positioning and the use of cervical immobilisation, airway manipulation, and intra-operative hypotension is likely to require a local-level approach using individual hospital records. This is likely to come with significant challenges especially if such data is to be manually recorded.
Future research should also focus on how the risk of developing DCM from asymptomatic spinal cord compression could be mitigated. Asymptomatic spinal cord compression is estimated to affect 1 in 4 adults, 9 but routine MRI imaging for those undergoing general anaesthesia would be impractical. Development of risk stratification models (perhaps incorporating vulnerable patient factors, at-risk procedures, or intraoperative events) may be a logical route forward. If appropriately validated such a model could enable appropriate shared decision making and intraoperative precautions to prevent the development of DCM in those undergoing surgery.
Conclusions
Recent surgery is an independent risk factor for the development of DCM. The biological mechanism and clinical significance for this remain to be defined. However, the potential for prior anaesthesia to be a triggering and/or exacerbating factor for this disabling condition warrants further investigation, as prevention is better than current cures.
Supplemental Material
Supplemental Material - Previous Surgical Exposure and the Onset of Degenerative Cervical Myelopathy: A Propensity-Matched Case-Control Analysis Nested Within the UK Biobank Cohort
Supplemental Material for Previous Surgical Exposure and the Onset of Degenerative Cervical Myelopathy: A Propensity-Matched Case-Control Analysis Nested Within the UK Biobank Cohort by Ben Grodzinski, Daniel J. Stubbs, and Benjamin M. Davies in Global Spine Journal.
Supplemental Material
Supplemental Material - Previous Surgical Exposure and the Onset of Degenerative Cervical Myelopathy: A Propensity-Matched Case-Control Analysis Nested Within the UK Biobank Cohort
Supplemental Material for Previous Surgical Exposure and the Onset of Degenerative Cervical Myelopathy: A Propensity-Matched Case-Control Analysis Nested Within the UK Biobank Cohort by Ben Grodzinski, Daniel J. Stubbs, and Benjamin M. Davies in Global Spine Journal.
Footnotes
Acknowledgments
This research has been conducted using the UK Biobank Resource under application number 73666.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the BG received funding from Christ’s College in the University of Cambridge for access to the UK Biobank Resource. BMD is supported by a National Institute for Health Research (NIHR) Clinical Doctoral Research Fellowship. DS is supported by a Wellcome Trust Doctoral Research Fellowship.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
