Abstract
Study Design
Retrospective.
Objective
Severe curves >100° in adolescent idiopathic scoliosis (AIS) are rare and require careful operative planning. The aim of this study was to assess baseline, perioperative, and 2-year differences between anterior release with posterior instrumentation (AP), posterior instrumentation with posterior column osteotomies (P), and posterior instrumentation with 3-column vertebral osteotomies (VCR).
Methods
Two scoliosis datasets were queried for primary cases of severe thoracic AIS (≥100°) with 2-year follow-up. Pre- and 2-year postoperative radiographic measures (2D and estimated 3D kyphosis), clinical measurements, and SRS-22 outcomes were compared between three approaches.
Results
Sixty-one patients were included: 16 AP (26%), 38 P (62%), 7 VCR (11%). Average age was 14.4 ± 2.0 years; 75.4% were female. Preoperative thoracic curve magnitude (AP: 112°, P: 115°, VCR: 126°, P = 0.09) and T5-T12 kyphosis (AP: 38°, P: 59°, VCR: 70°, P = 0.057) were similar between groups. Estimated 3D kyphosis was less in AP vs P (−12° vs 4°, P = 0.016). Main thoracic curves corrected to 36° in AP vs 49° and 48° for P and VCR, respectively (P = 0.02). Change in estimated 3D kyphosis was greater in AP vs P and VCR (34° vs 13°, P = 0.009; 34° vs 7°, P = 0.046). One incomplete spinal cord injury had residual deficits (P; 1/61, 1.6%). All SRS-22 domains improved postoperatively.
Conclusion
All approaches obtained satisfactory coronal and sagittal correction, but AP had smaller residual coronal deformity and greater kyphosis restoration than the other approaches. This information may help inform the decision of whether to include an anterior release for large thoracic AIS curves.
Keywords
Introduction
Adolescent idiopathic scoliosis (AIS) affects 0.5%-5.2% of the population. 1 Approximately 0.1% ultimately undergo surgery, a decision based in part on maturity at the time of the main coronal curve measuring greater than 45-50°.2,3 Posterior pedicle screw instrumentation with or without posterior column osteotomies (P) is the primary operative technique for modern pediatric scoliosis surgery.4-7 However, large, stiff curves (variably defined as approximately >80° or with <20° flexibility) are traditionally seen as distinct entities that may require more extensive corrective maneuvers.6,7 Combined anterior release with posterior instrumentation (AP) and posterior vertebral column resections (VCR) allow for increased correction via creation of greater three column flexibility.8-12 These procedures are also considered options for significant sagittal deformity; relative hypokyphosis or frank thoracic lordosis a component of AIS that is exacerbated with more severe coronal and axial deformity. Measurement of sagittal deformity is also difficult in 2D particularly as the deformity in the other planes increases. Thoracic kyphosis is underestimated by traditional 2D imaging. 13
Larger AIS curves may lose flexibility as the curve progresses and traction radiographs can give a sense of coronal curve flexibility. For some patients, preoperative halo-gravity traction may be considered as well. Ultimately a decision regarding the surgical approach will be required. In decades past an anterior release was common for curves that did not bend below 50°. The additional power of correction that was enabled by thoracic pedicle screw fixation and posterior column osteotomies, reduced the indications for anterior release. Similarly, the VCR procedure became safer with more advanced intraoperative neuromonitoring and its use grew particularly in more angular deformity.
Given the potential for all 3 of these approaches to be utilized in the more severe cases of AIS, we sought to compare the radiographic, clinical, or patient reported outcomes between AP, VCR, and P for thoracic AIS curves >100°. The purpose of this study is to assess the baseline, peri-operative, and 2-year differences between these three approaches for index AIS cases with severe thoracic curves.
Materials and Methods
Two prospective, multicenter, scoliosis studies were queried for patients undergoing primary surgical treatment for AIS. Written consent was obtained from all patients for inclusion into the studies. Patients ≤21 years old who underwent index surgery for idiopathic scoliosis with a main thoracic curve >100° were included. Only patients with 2-year follow-up were included. Patients with prior spinal surgery where excluded. Patients that may have been enrolled in both registries were identified and included only once based on matching site, surgeon, date of birth, and date of surgery. Standard demographic, radiographic data, and Scoliosis Research Society-22r (SRS-22r) outcome questionnaires were collected at baseline and 2-year post-operative. Deformity measurements included: curve magnitude, curve flexibility (baseline only), T5-T12 kyphosis, and estimated 3D kyphosis calculated from 2D radiographic measurements as described by Parvaresh et al. 21 Intraoperative variables including surgical approach, estimated blood loss (EBL), and complications were collected.
Baseline, intraoperative, and 2-year post-operative outcomes were compared between the 3 approaches with ANOVA for continuous variables and chi square test for categorical variables. Non-parametric data was analyzed with Kruskal Wallis and Mann Whitney testing and medians were reported. Bonferroni post hoc comparisons were performed in the event of main effect significance. Significance was set at P < 0.05.
Results
Demographics.
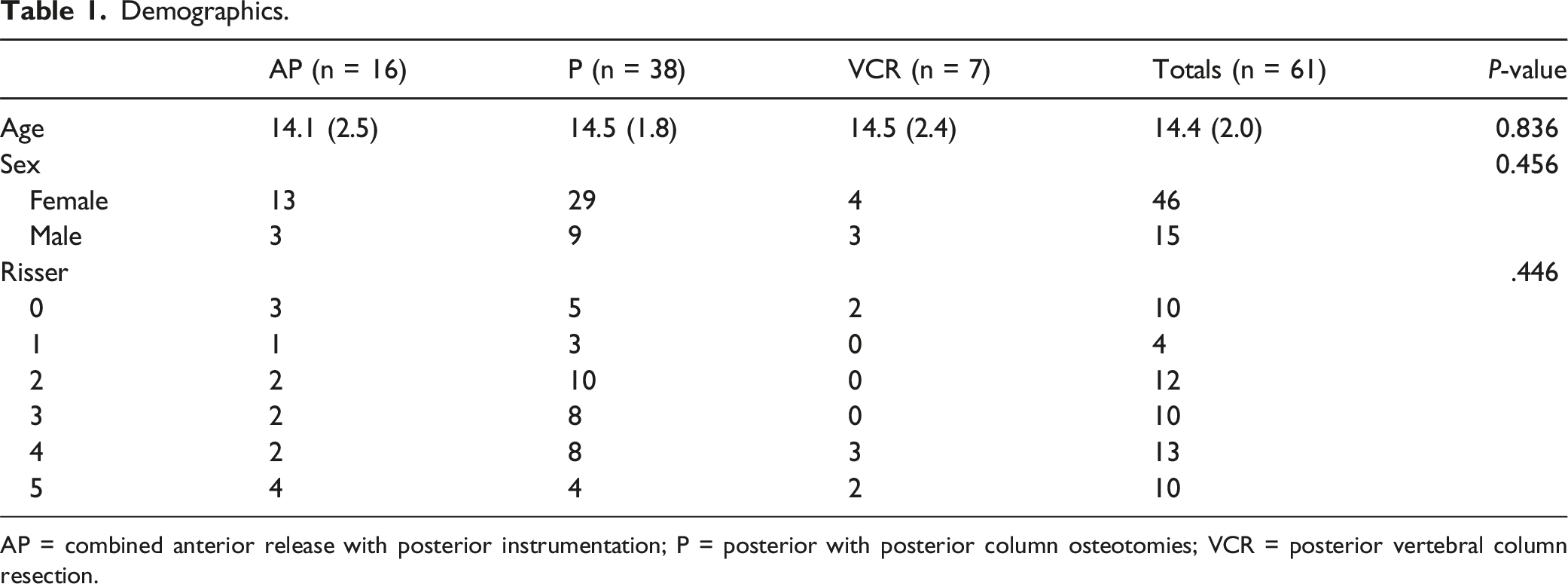
AP = combined anterior release with posterior instrumentation; P = posterior with posterior column osteotomies; VCR = posterior vertebral column resection.
Pre- and Two Year Post-operative Radiographic Measurements.
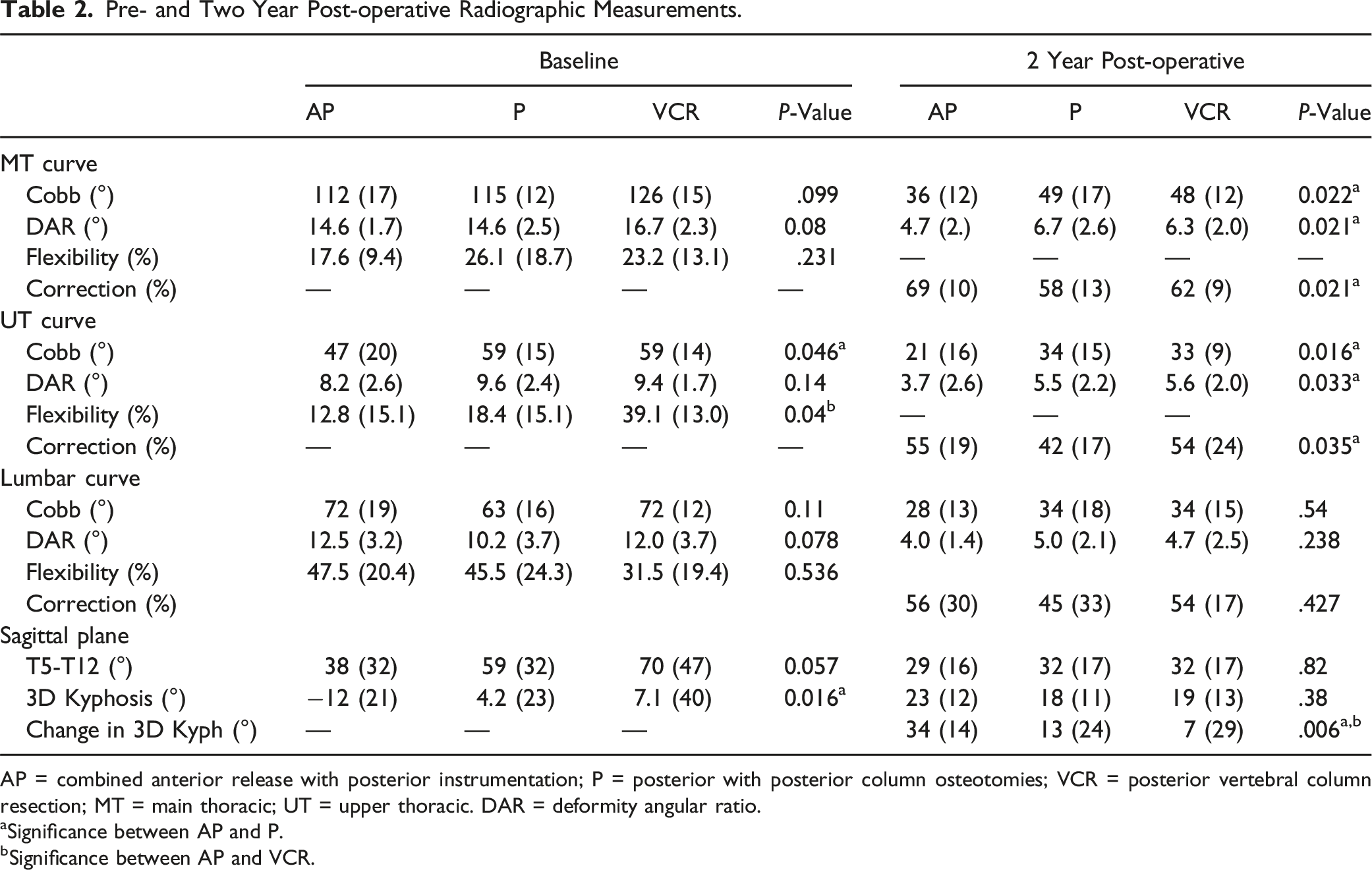
AP = combined anterior release with posterior instrumentation; P = posterior with posterior column osteotomies; VCR = posterior vertebral column resection; MT = main thoracic; UT = upper thoracic. DAR = deformity angular ratio.
aSignificance between AP and P.
bSignificance between AP and VCR.
Pre- and Two Year Post-operative Radiographic Parameters Patient Reported Outcomes and Clinical Measurements.
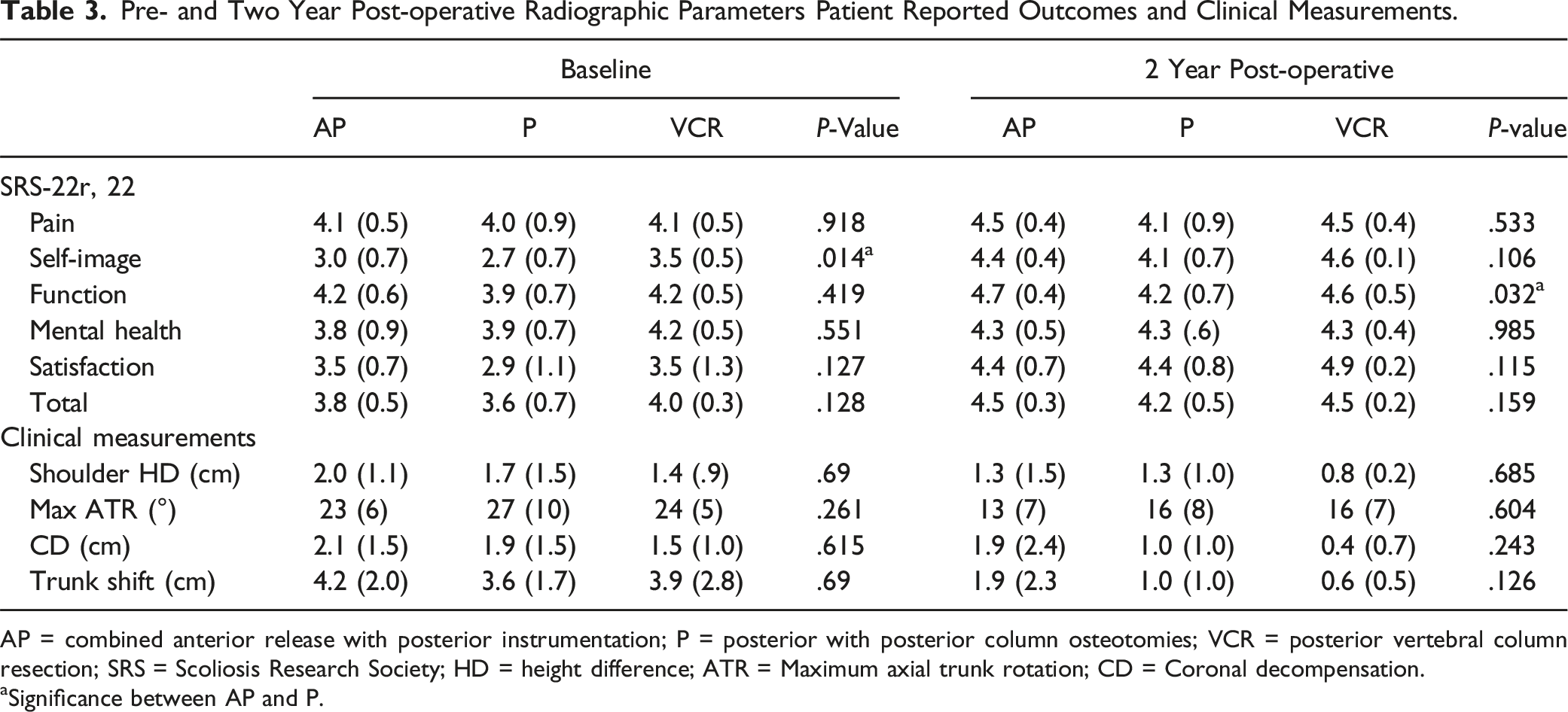
AP = combined anterior release with posterior instrumentation; P = posterior with posterior column osteotomies; VCR = posterior vertebral column resection; SRS = Scoliosis Research Society; HD = height difference; ATR = Maximum axial trunk rotation; CD = Coronal decompensation.
aSignificance between AP and P.
The AP group had an average of 5.0 ± 1.7 discs removed. The approach was open in 8 and thoracoscopic in 8 of the 16 patients. The posterior procedure was performed on the same day in 9 cases and staged in 7. During the posterior portion of the procedure, intraoperative traction was utilized in 43% (7/16 = 43%) and posterior column osteotomies were performed in 81% of cases. Similarly, in the P group, 53% were placed in traction during the procedure and the majority had posterior column osteotomies at multiple levels (5.7 ± 3.3). The VCR procedures were all performed from a posterior only approach and an average of 1.7 vertebrae (range 1-3) were resected. For each of the 3 approaches, an average of 13 posterior levels were instrumented and fused.
Two-year postoperative thoracic curve magnitude was significantly smaller in AP: 36 ± 12° vs P: 49 ± 17°, (P = 0.021, VCR: 48 ± 12° not different than either). In addition, there was a greater percentage correction of the MT curve in the AP: 69 ± 10% compared to the P: 58 ± 14% group, (P = 0.021, VCR: 62 ± 9% not different than either). Postoperative lateral 2D T5-T12 kyphosis (AP: 29 ± 16°, P: 32 ± 17°, VCR: 32 ± 17°, P = 0.82) and estimated 3D kyphosis (AP: 22 ± 12°, P: 18 ± 11°, VCR: 19 ± 13°, P = 0.82) were not different between groups. However, the preoperative to 2-year postoperative change in estimated 3D kyphosis was significantly greater in AP: 34 ± 14° vs both P: 13 ± 24°, P = 0.009 and VCR: 7 ± 29°, P = 0.046, (Tables 2 and 3).
Operative time was significantly shorter for P with a median of 350 (range 348-453 minutes) compared to both AP: 623 (range 520-800, P < 0.001) and VCR: 790 (range 452-942 minutes, P = 0.006). Intraoperative neuromonitoring changes were frequent (AP: 19%, P: 45%, VCR: 43%, P = 0.215). There were no differences in length of stay or estimated blood loss (Table 2). Figures 1-3 demonstrate the correction that can be achieved with each procedure. A patient with a preoperative main thoracic curve of 106°, 29° of 2D T5-T12 kyphosis, and −16° of estimated 3D T5-T12 kyphosis (left) who was treated with a combined anterior release with posterior instrumentation and corrected to 20° of scoliosis, 30° of 2D T5-T12 kyphosis, and 32° of estimated 3D kyphosis at 2 years (right). A patient with a preoperative main thoracic curve of 107°, 33° of 2D T5-T12 kyphosis, and −13° of estimated 3D T5-T12 kyphosis (left) who was treated with a posterior instrumentation fusion and corrected to 40° of scoliosis, 29° of 2D T5-T12 kyphosis, and 20° of estimated 3D kyphosis at 2 years (right). A patient with a preoperative main thoracic curve of 110°, 88° of 2D T5-T12 kyphosis, and 30° of estimated 3D T5-T12 kyphosis (left) who was treated with a posterior instrumentation with 3 column vertebral osteotomies and corrected to 46° of scoliosis, 31° of 2D T5-T12 kyphosis, and 18° of estimated 3D kyphosis at 2 years (right).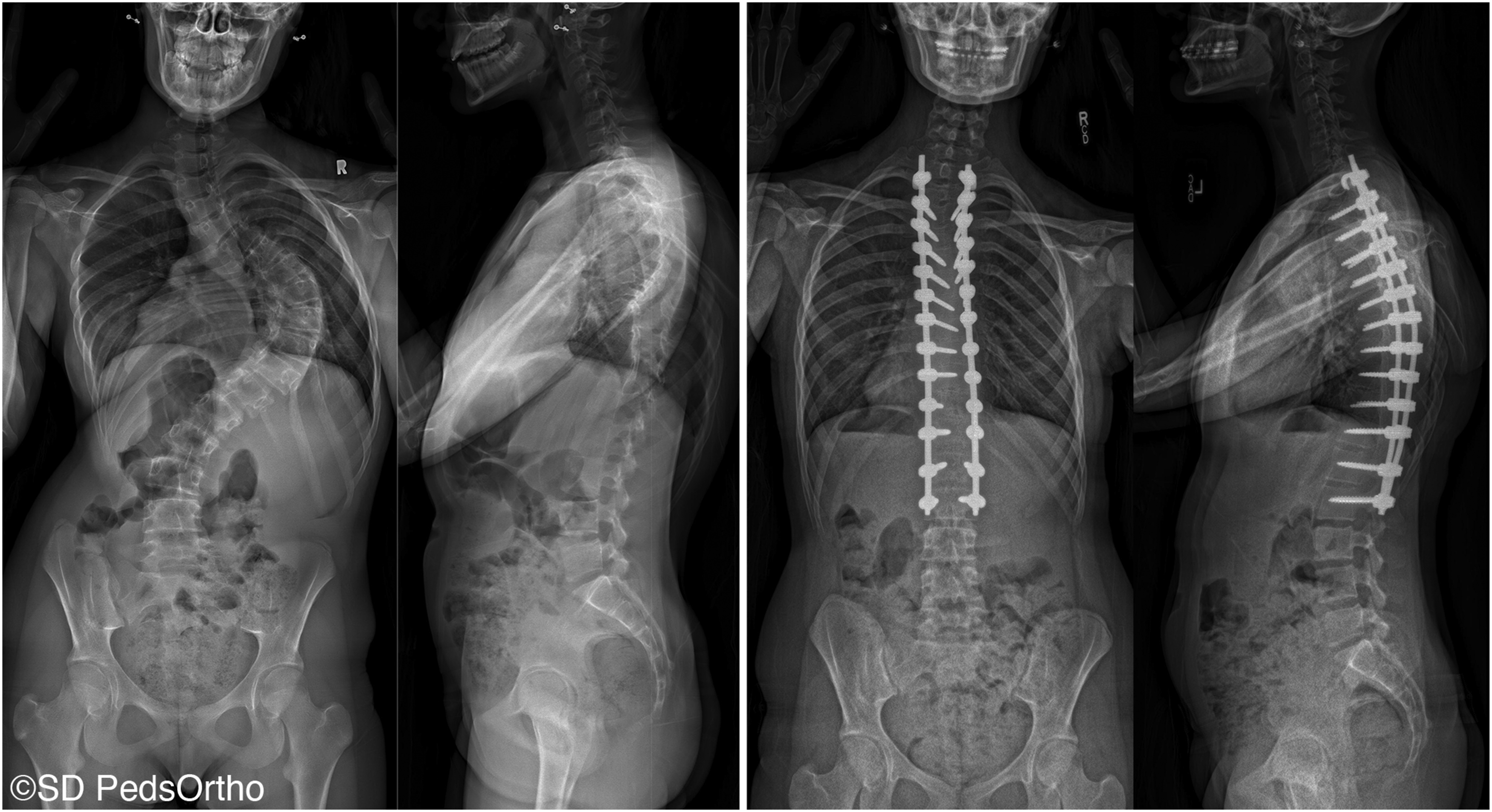
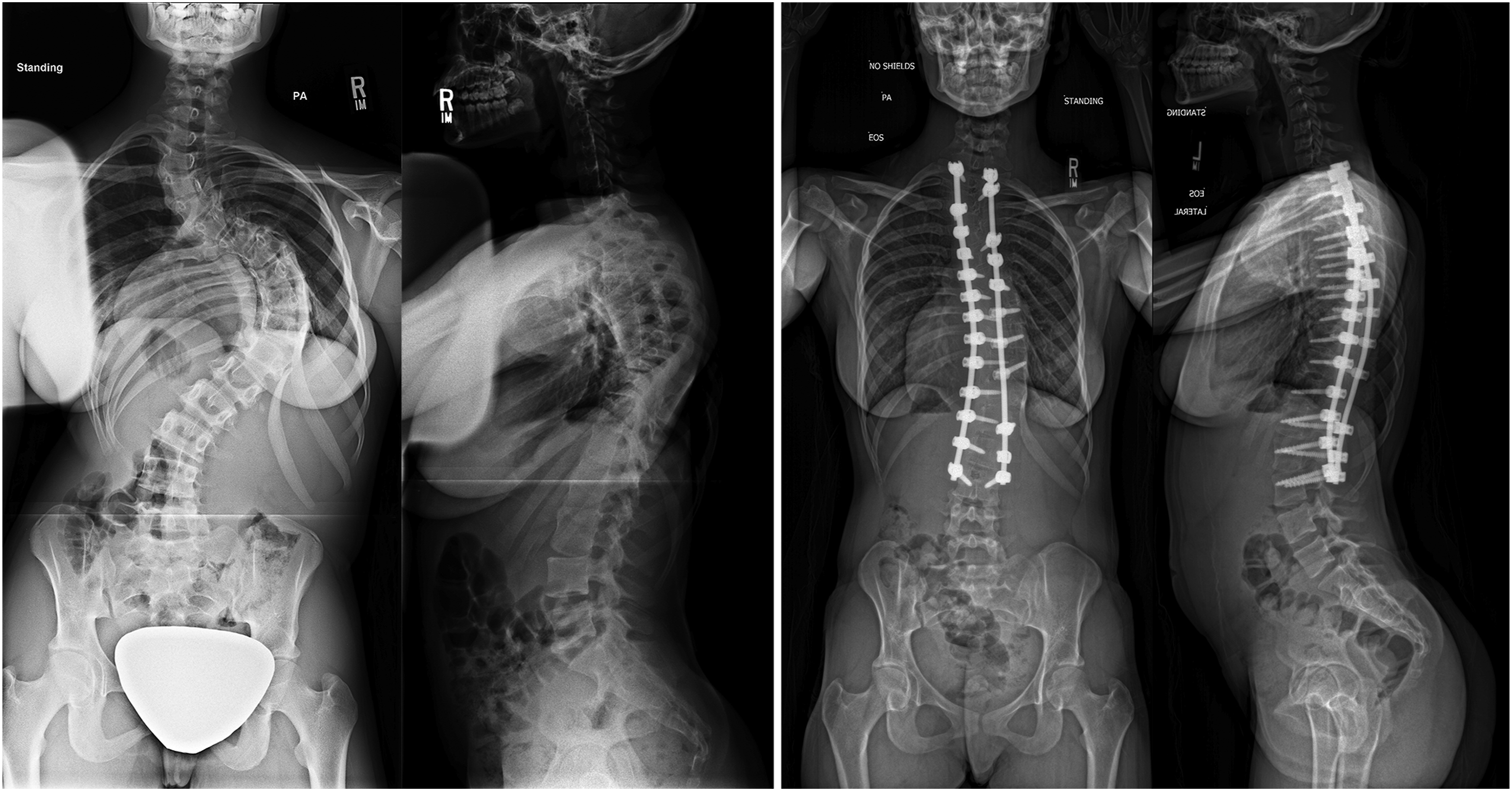
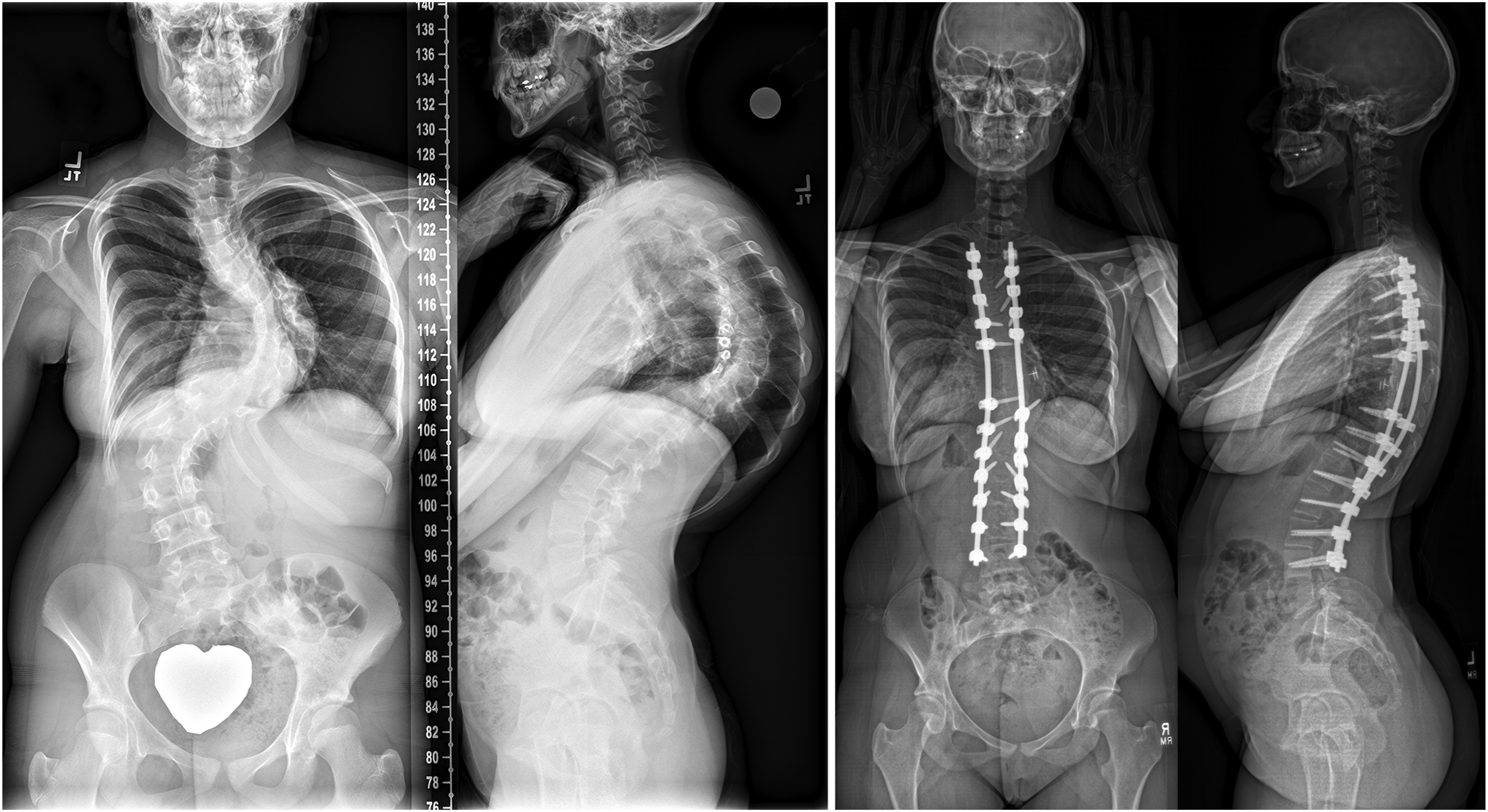
Operative Variables and Complications.

AP = combined anterior release with posterior instrumentation; P = posterior with posterior column osteotomies; VCR = posterior vertebral column resection OR = operating room; EBL = estimated blood loss; NM = neuromonitoring.
aSignificance between AP and P.
bSignificance between VCR and P.
cMedian and range reported, data not parametric.
Discussion
Severe AIS with a coronal curve greater than 100° are rare in regions with advanced health care delivery systems, yet patients do reach these levels of deformity for a variety of reasons. 3 Curves greater than 70-80° are associated with compromised lung function emphasizing the medical need for surgical correction. 15 However, the approach that obtains maximal correction in all three planes with minimal complications is not well established in the literature for this patient population. In our study, combined anterior release with posterior instrumentation created more coronal correction and greater increases in kyphosis than posterior instrumentation with osteotomies and compared to VCR, restored more thoracic kyphosis than a posterior approach with a VCR. The AP approach resulted in the smallest residual coronal MT deformity with correction from 112 ± 17° to 36 ± 12° at 2 years postoperative. This compares to residual coronal curves of 49 ± 17 in the posterior only group and 48 ± 12° in the VCR group.
Coronal and sagittal plane correction is created differently by each surgical approach. Standard posterior approaches lack anterior column shortening options and thus works via multi-segmental osteotomies to loosen and lengthen the posterior column. Traditional posterior column releases (such as Ponte osteotomies) have been associated with several degrees of increase in thoracic kyphosis and coronal correction per level. This may or may not be sufficient given the coronal and axial correction maneuvers tend to “reposition” the lengthened anterior column back into the global sagittal plane.16-19 The anterior approach on the other hand is associated with anterior column shortening secondary to the removal of the intervertebral discs. 17 VCR of course removes an entire spinal segment, translating the spinal column up 35-60° in the plane(s) of choice.11,12,16
We compared these three approaches in our cohort, but with little precedent for reference in the literature. Lenke et al. 11 reported outcomes for 35 consecutive pediatric scoliosis patients undergoing VCR, but with a mixed cohort, including severe kyphosis, that is not directly generalizable to AIS. Suk et al 20 ’s posterior-only pedicle instrumentation for severe curves up to 105° reports kyphosis restoration from 22° ± 10° to 29° ± 10° post-operatively (P = 0.0001). Dobbs et al. 6 conversely reports thoracic flattening in both AP and P approaches, the former decreasing from 28° to 18° post-operatively, the latter 21° to 15°. Like Suk, the patients in our study achieved kyphosis on 2D lateral T5-T12 measurements of ∼30° without significant difference between groups (Table 2).
Novel 3D reconstructive technology has unmasked the loss of thoracic kyphosis typical of AIS, yet difficult to appreciate and quantify on 2D radiographs.13,22,23 This introduces a measurement bias in 2D imaging, estimated to undershoot 3D T5-T12 kyphosis measured in the local sagittal reference plane of each vertebrae, rather than the 2D plane of the radiograph, by an average of 11° to as much as 40°. 13 This “measurement error” of kyphosis increases as coronal deformity increases, introducing substantial error in a cohort of large curves as is this series. Considering this, our analysis included 3D kyphosis calculations from Parvaresh et al 22 ’s validated formula, defined as 18.1 + (0.81*2D T5-T12 sagittal Cobb) - (0.54*2D coronal Cobb). In the AP patient group, T5-12 kyphosis averaged 38° on 2D radiographs, but 12° of lordosis by 3D calculation. Similarly, 3D calculations captured the substantial 34° increase (restoration) of thoracic kyphosis obtained by AP approach vs just 13° by P and 7° by VCR. Overall, these results suggest that the removal of multiple intervertebral discs anteriorly followed by posterior release and instrumentation allows for superior multiplanar deformity reconstruction in the thoracic spine.
Intraoperative Profile and Complications
The intraoperative and complication profiles between groups were real, but largely similar between approaches. Notably, the oft reported and feared complications associated with specific approaches were not borne out in this study. Early VCR series described the technique as “safe but challenging,” reporting significant blood loss and long operating times while stressing the necessity of neuromonitoring due heightened post-operative neurologic deficits.11,12 VCR in our study was not an outlier over other approaches in any of these domains, although operating time was greater than standard posterior with osteotomies limited to the posterior column. Permanent neurologic deficit in our study also did not occur in VCR: the one reported case of permanent deficit underwent the standard posterior approach, underscoring the high-risk nature of these cases regardless of approach.
The AP and VCR groups are uniquely associated with pulmonary complications due to direct manipulation of the pleural space.23,24 While one AP patient experienced reportable atelectasis in our study (1/16, 6.3%), the VCR group reported the most pulmonary events requiring procedural intervention (chest tubes, thoracentesis; 3/7, 43%). This percentage may be inflated due to small sample size (3/7, 43%), but pleural effusion is a noted complication of VCR in large pediatric cohorts. 25
Patient Reported Outcomes
The lowest of all recorded SRS-22r scores was preoperative Self-Image for posterior group at 2.7 ± 0.6. Large operative AIS cohorts cite preoperative Self-Image closer to 3.3.27-29 The association of curve magnitude with Self-Image is a well-studied questioned with inconsistent results. Belli et al 30 found significant decrements in Self-Image reporting with increasing coronal curve size, with a nadir of 3.3 for 50° to 60° curves. However, other studies suggest that the relationship is more complicated, with multiple papers evaluating surface topography, psychological function, and demographics as possible influencers.26,28,31-34 There is little consensus between these, other than that most tend to improve post-operatively. Self-Image also improved from pre-to postoperatively for all groups in our study, (2.9 ± 0.6 to 4.2 ± 0.6) as did all domain scores for each group (Table 3).
Limitations
There are several limitations to this study. First, the choice of approach was of course not randomized and selection bias and unequal distribution of the approaches was present - Posterior group comprised nearly two-thirds of the study population with VCR contributing only 7 patients. As such, several variables that did not reach significance between groups and we may be underpowered to do so. Limited sample size is an unavoidable limitation due to the rarity of severe scoliosis and the choice of approach may potentially be influenced by a lack of surgeon familiarity/comfort for AP and VCR approaches.11,24,25 Second, we did not have 3D imaging for these cases and severe coronal and apical rotation of the spine make lateral radiographic measurements particularly difficult.21,22 As such, details of the deformities are missed in this analysis (axial plane) but to gain a better understanding than what 2D radiographs alone can provide, we used surrogate measures of deformity, including a calculated estimation of 3D kyphosis 22 and clinical angle of trunk rotation for the axial plane. Lastly, surgeon preference and institutional/regional bias may influence the chosen surgical approach. 35 If inconsistent between subjects, parameters determining who receives what surgery may differ and thus conceal confounders that placed patients in specific operative categories. This potential is an inherent challenge in a retrospective review that lacks data to explain decision making. Nonetheless, the variables captured in this study found minimal preoperative differences between groups.
We chose to compare three approaches when the more obvious might have been to compare posterior only to anterior plus posterior to isolate the effect of the anterior release; or to compare the 2 posterior-only approaches. Given that VCR and AP are both being done in this population, we felt it was important to include all 3 approaches. Specifically looking at the effect of the anterior release, the AP vs P comparisons identify greater coronal and sagittal plane correction when an anterior release is included. The extent of the additional correction in exchange for a longer procedure may continue to be debated, but we hope the information will inform shared decision making with future patients and surgeons facing the decisions surrounding the management of thoracic AIS >100°. We were unable to identify specific advantages of VCR over Posterior in this cohort.
Conclusion
Severe scoliosis greater than 100° is rare and requires thoughtful planning to obtain maximal safe correction in all planes. Combined anterior release with posterior instrumentation, posterior instrumentation with osteotomies, and posterior vertebral column resection each obtain satisfactory coronal correction with similar complication profiles. However, there was more thoracic kyphosis created with greater coronal curve correction with the anterior/posterior combined approach compared to a posterior only approach. VCR was the least commonly applied approach to this population, and although there may be exceptions, it is likely more appropriately reserved for spinal pathologies other than primary AIS.
Footnotes
Authors Note
Study Conducted at Rady Children’s Hospital, San Diego.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the in part by a grant from the John and Marcella Fox Research Fund and in part by grants to the Setting Scoliosis Straight Foundation in support of Harms Study Group research from DePuy Synthes Spine, EOS imaging, Stryker Spine, Medtronic, NuVasive, Zimmer Biomet and the Food and Drug Administration.
Ethical Statement
Harms Study Group Investigators
Aaron Buckland, MD; Melbourne Orthopaedic Group & Royal Children’s Hospital
Amer Samdani, MD; Shriners Hospitals for Children—Philadelphia
Amit Jain, MD; Johns Hopkins Hospital
Baron Lonner, MD; Mount Sinai Hospital
Benjamin Roye, MD; Columbia University
Burt Yaszay, MD; Seattle Children’s Hospital
Chris Reilly, MD; BC Children’s Hospital
Daniel Hedequist, MD; Boston Children’s Hospital
Daniel Sucato, MD; Texas Scottish Rite Hospital
David Clements, MD; Cooper Bone & Joint Institute New Jersey
Firoz Miyanji, MD; BC Children’s Hospital
Harry Shufflebarger, MD; Paley Orthopedic & Spine Institute
Jack Flynn, MD; Children’s Hospital of Philadelphia
John Asghar, MD; Paley Orthopedic & Spine Institute
Jean Marc Mac Thiong, MD; CHU Sainte-Justine
Joshua Pahys, MD; Shriners Hospitals for Children—Philadelphia
Juergen Harms, MD; Klinikum Karlsbad-Langensteinbach, Karlsbad
Keith Bachmann, MD; University of Virginia
Lawrence Lenke, MD; Columbia University
Lori Karol, MD; Children’s Hospital, Denver Colorado
Mark Abel, MD; University of Virginia
Mark Erickson, MD; Children’s Hospital, Denver Colorado
Michael Glotzbecker, MD; Rainbow Children’s Hospital, Cleveland
Michael Kelly, MD; Rady Children’s Hospital
Michael Vitale, MD; Columbia University
Michelle Marks, PT, MA; Setting Scoliosis Straight Foundation
Munish C Gupta, MD; Washington University
Nicholas Fletcher, MD; Emory University
Noelle Larson, MD; Mayo Clinic Rochester Minnesota
Patrick Cahill, MD; Children’s Hospital of Philadelphia
Paul Sponseller, MD; Johns Hopkins Hospital
Peter Gabos, MD: Nemours/Alfred I. duPont Hospital for Children
Peter Newton, MD; Rady Children’s Hospital
Peter Sturm, MD; Cincinnati Children’s Hospital
Randal Betz, MD; Institute for Spine & Scoliosis
Stefan Parent, MD: CHU Sainte-Justine
Stephen George, MD; Nicklaus Children's Hospital
Steven Hwang, MD; Shriners Hospitals for Children—Philadelphia
Suken Shah, MD; Nemours/Alfred I. duPont Hospital for Children
Sumeet Garg, MD; Children’s Hospital, Denver Colorado
Tom Errico, MD; Nicklaus Children's Hospital
Vidyadhar Upasani, MD; Rady Children’s Hospital.
Fox Study Group Investigators
Amer Samdani, MD; Shriners Hospitals for Children—Philadelphia
Amit Jain, MD; Johns Hopkins Hospital
Baron Lonner, MD; Mount Sinai Hospital
Burt Yaszay, MD; Seattle Children’s Hospital
Daniel Sucato, MD; Texas Scottish Rite Hospital
David Bumpass, MD; Arkansas Children’s Hospital
David Polly, MD; University of Minnesota Medical Center
Han Jo Kim, MD; Hospital for Special Surgery
Harry Shufflebarger, MD; Paley Orthopedic & Spine Institute
John Asghar, MD; Paley Orthopedic & Spine Institute
John Emans, MD; Boston Children’s Hospital
Joshua Pahys, MD; Shriners Hospitals for Children—Philadelphia
Lawrence Lenke, MD; Columbia University
Mark Erickson, MD; Children’s Hospital, Denver Colorado
Michael Kelly, MD; Rady Children’s Hospital
Munish C Gupta, MD; Washington University
Oheneba Boachie-Adjei, MD; FOCOS Orthopedic Hospital
Paul Sponseller, MD; Johns Hopkins Hospital
Peter Newton, MD; Rady Children’s Hospital
Peter Sturm, MD; Cincinnati Children’s Hospital
Richard McCarthy, MD; Arkansas Children’s Hospital
Suken Shah, MD; Nemours/Alfred I. duPont Hospital for Children
Sumeet Garg, MD; Children’s Hospital, Denver Colorado
Vidyadhar Upasani, MD; Rady Children’s Hospital.
