Abstract
Study Design
Human Cadaveric Study.
Objective
This study aims to explore the feasibility of using preoperative magnetic resonance imaging (MRI), zero-time-echo (ZTE) and spoiled gradient echo (SPGR), as source data for robotic-assisted spine surgery and assess the accuracy of pedicle screws.
Methods
Zero-time-echo and SPGR MRI scans were conducted on a human cadaver. These images were manually post-processed, producing a computed tomography (CT)-like contrast. The Mazor X robot was used for lumbar pedicle screw-place navigating of MRI. The cadaver underwent a postoperative CT scan to determine the actual position of the navigated screws.
Results
Ten lumbar pedicle screws were robotically navigated of MRI (4 ZTE; 6 SPGR). All MR-navigated screws were graded A on the Gertzbein-Robbins scale. Comparing preoperative robotic planning to postoperative CT scan trajectories: The screws showed a median deviation of overall 0.25 mm (0.0; 1.3), in the axial plane 0.27 mm (0.0; 1.3), and in the sagittal plane 0.24 mm (0.0; 0.7).
Conclusion
This study demonstrates the first successful registration of MRI sequences, ZTE and SPGR, in robotic spine surgery here used for intraoperative navigation of lumbar pedicle screws achieving sufficient accuracy, showcasing potential progress toward radiation-free spine surgery.
Keywords
Introduction
Fusion surgery of the lumbar spine may require 2 preoperative imaging techniques: the most common is magnetic resonance imaging (MRI) for diagnostic assessment of soft tissue and neural structures and computed tomography (CT) for visualization of the bony anatomy. Additionally, CT images are currently used routinely in pre-operative planning of pedicle screw placement and intra-operative spinal navigation and demand high accuracy for screw placement and to avoid potential iatrogenic injury to neural structures.
Leading to the separation of essential anatomic information across 2 different imaging modalities, with CT scans providing optimal visualization of bone structures and MRIs excelling in capturing soft tissue details. Still, in spine surgery, the reliance on bony landmarks is pivotal in guiding precise decompression of soft tissue structures and preventing injury during screw placement. 1 To address the issue of distributed information over 2 imaging modalities, there have been efforts to combine preoperative CT and MR images, showing their use for intraoperative navigation. 1 However, acquiring a CT scan and exposure to ionizing radiation remains.
Therefore, the majority of patients undergoing instrumentation necessitates lumbar spine CT administration, exposing them to radiation doses of approximately 7.5 mSv. 2 While this exposure is a matter of clinical consideration, it’s important to note that evidence suggests a link between low-level ionizing radiation, commonly encountered in medical imaging, and an increase in cancer risk. 3 This concern is especially relevant in CT scans, where radiation exposure may marginally elevate a patient’s long-term cancer risk, especially with repeated scanning. 4 Consequently, mitigating radiation exposure, including reducing the number of scans, remains paramount in ensuring patient safety.
New technologies for enhanced bone visualization on MRI have emerged to make the acquisition of additional CT imaging next to MRI unnecessary, thereby minimizing exposure to ionizing radiation and improving efficacy. Software applications have been developed to post-process MR images to synthesize CT-like images through deep-learning algorithms. 5 Nevertheless, post-processing may introduce misinformation into the artificial images, posing a risk of potential misguidance for surgeons. Deep learning techniques that transfer contrast from 1 imaging modality to another have the potential for hallucination, ie, inaccurate structures may be created if they were not part of the training set. 6 Additionally, soft tissue structures are lost during synthetic CT, which may be valuable for spinal navigation. Furthermore, MRI sequences may not have adequate contrast between short-T2 soft tissues, such as ligaments and tendons, vs air and cortical bone; there is a potential for inaccurate inference during the contrast transfer.
Alternatively, it may be viable to directly utilize 3D MRI acquisitions that are sensitive to bone contrast, for screw placement planning and intraoperative registration, without utilizing deep learning inference algorithms. These bone contrast-sensitive MRI sequences, such as zero-time-echo (ZTE) and spoiled gradient echo (SPGR), already have high signal contrast between soft tissues and cortical bone, and require a signal inversion and masking of air boundaries to create CT-like images. 7 These imaging techniques obviate the need for additional deep learning algorithms, thereby diminishing its associated risks.
Utilizing ZTE and SPGR techniques has undergone extensive investigation within diverse surgical domains, encompassing spine and brain surgeries, where it has been shown that ZTE is a robust alternative to CT for imaging of cortical bone without ionizing radiation. 8 Incorporating ZTE or SPGR in navigated spine surgery can potentially reduce the necessity for preoperative CT scans to a minimum, thereby substantively diminishing radiation exposure for patients undergoing spine interventions.
This methodology exhibits promise in overcoming the inherent challenges associated with conventional imaging modalities, presenting a safer alternative characterized by diminished radiation exposure and displaying relevant information in 1 imaging modality. Therefore, leading to improved patient safety. Nonetheless, the applicability and precision of this preoperative MRI for navigation and robotics remain uninvestigated to date.
The aim of this study was to explore the feasibility of preoperative MRI for navigation in robotic-assisted spine surgery and to assess the accuracy of pedicle screw placement.
Material and Methods
In this study, MRI-based robotic surgery was performed on a human lumbar cadaver. Institutional Review Board (IRB#2019-1402) obtained ethical approval. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013).
Cadaver
A total of 5 lumbar vertebrae sourced from 1 73-year-old female fresh-frozen cadaver without osteoporosis, history of cancer, or previous spine surgery was utilized for this study to perform MRI-navigated robotic pedicle screw placement.
Preoperative Imaging
The cadaver was preoperatively scanned on a clinical 3.0 T scanner (Premier; GE Healthcare, Waukesha, WI) using a 30-channel anterior and 60-channel table coil. Two 3D sequences were acquired in the lumbar spine - a 3D ZTE and a 3D SPGR.
ZTE Acquisition and Post-processing
Unlike regular MRI, ZTE MRI provides CT-like contrast and adequate signal intensity in cortical bone. 9 It is a novel imaging technique that utilizes ultrafast readouts to capture signal from short-T2 tissues. 8 This technique leads to an image quality, achieved post-bias correction and normalization, sufficient to facilitate bone segmentation. In the typical histogram distribution of ZTE signal, discernible peaks for air and soft tissue are evident, with a broader intervening peak representing bone. Intensity thresholding coupled with morphological operations to address partial volume effects allows for the segmentation of air, tissue, and bone segments. 8
An isotropic three-dimensional (3D), proton-density weighted ZTE sequence was acquired with the following parameters: coronal field-of-view = 28 cm, spatial resolution of 0.9 × 0.9 × 0.9 mm, acquisition matrix = 320 × 320 × 280, TR/TE = 506/0 ms, 6 averages, bandwidth = 326 Hz/pixel, 7.5 minutes.
Zero-time-echo-image data were post-processed in order to flatten further signal intensities associated with soft tissue, enhance osseous conspicuity, and ultimately produce images with CT-like contrast. An N4 bias-correction algorithm (default on the scanner software) was applied to ZTE image data, followed by an inverse-logarithmic rescaling to produce positive contrast within the bone. 10
SPGR Acquisition and Post-processing
Spoiled gradient echo also known as incoherent gradient echo is a fat suppressed three-dimensional image, which has been shown to be reliable for the detection of cartilage lesions especially in the knee. 11 The sequence parameters included a sagittal field-of-view = 26 cm, spatial resolution of 1.0 × 1.0 × 1.0 mm, acquisition matrix = 260 × 260 × 276, TR/TE = 5/1.2 ms, bandwidth = 244 Hz/pixel, 5.5 minutes). The images were also inverted, similar to ZTE, but without N4 bias-correction.
Image to Robot Software
Preoperative ZTE and SPGR were uploaded in the robotic software (Mazor X, Version 5.0; Medtronic, Ireland) as 2 individual surgical cases. First the screws were planned on ZTE navigation and used for surgery. After doing so, the ZTE case was closed and the SPGR images were uploaded to the robotic software, starting a new surgical case.
Screw Planning
MRI Navigation
During the preoperative phase, bilateral pedicle screws were planned on L1 to L5 vertebrae for both imaging modalities. Every screw was planned manually for trajectories, including entry points as well as thickness based on the pedicle’s diameter. To address a known limitation of the SPGR imaging technique, which can exhibit reduced robustness in the presence of patient motion, SPGR was strategically employed specifically for the navigation process in the upper lumbar region (L1-L3). It is important to note that this choice was made knowing that no motion artifacts would be present in a cadaver. For the lower lumbar levels, L4 and L5, ZTE imaging was chosen for navigation.
Surgical Set-Up
Before transferring the cadaver to the operation table, a bed frame was incorporated, offering essential support for the robotic arm. It was then placed on the Jackson table.
Robotic Set Up
To optimize the ergonomic setup, the robotic base was strategically placed at the foot of the bed on the right side. This arrangement allowed space for both the robotic arm mount and the C-arm, strategically positioned on the opposite side to maintain sterility during intraoperative imaging.
To attach the robotic system to the bony anatomy, an incision over the right posterior superior iliac spine (PSIS), was made, followed by placing a Schanz pin and connecting it to a screwball adaptor. This step marked the initiation of the registration process, effectively tethering the robotic arm to the anatomical landmarks. Full synchronization between the C-arm and the robotic platform was achieved by integrating the Mazor X Software Robotics image adaptor with the C-arm image intensifier before surgery. Once the robotic system was firmly integrated with the bed, the necessary draping procedures could be carried out.
Imaging Registration
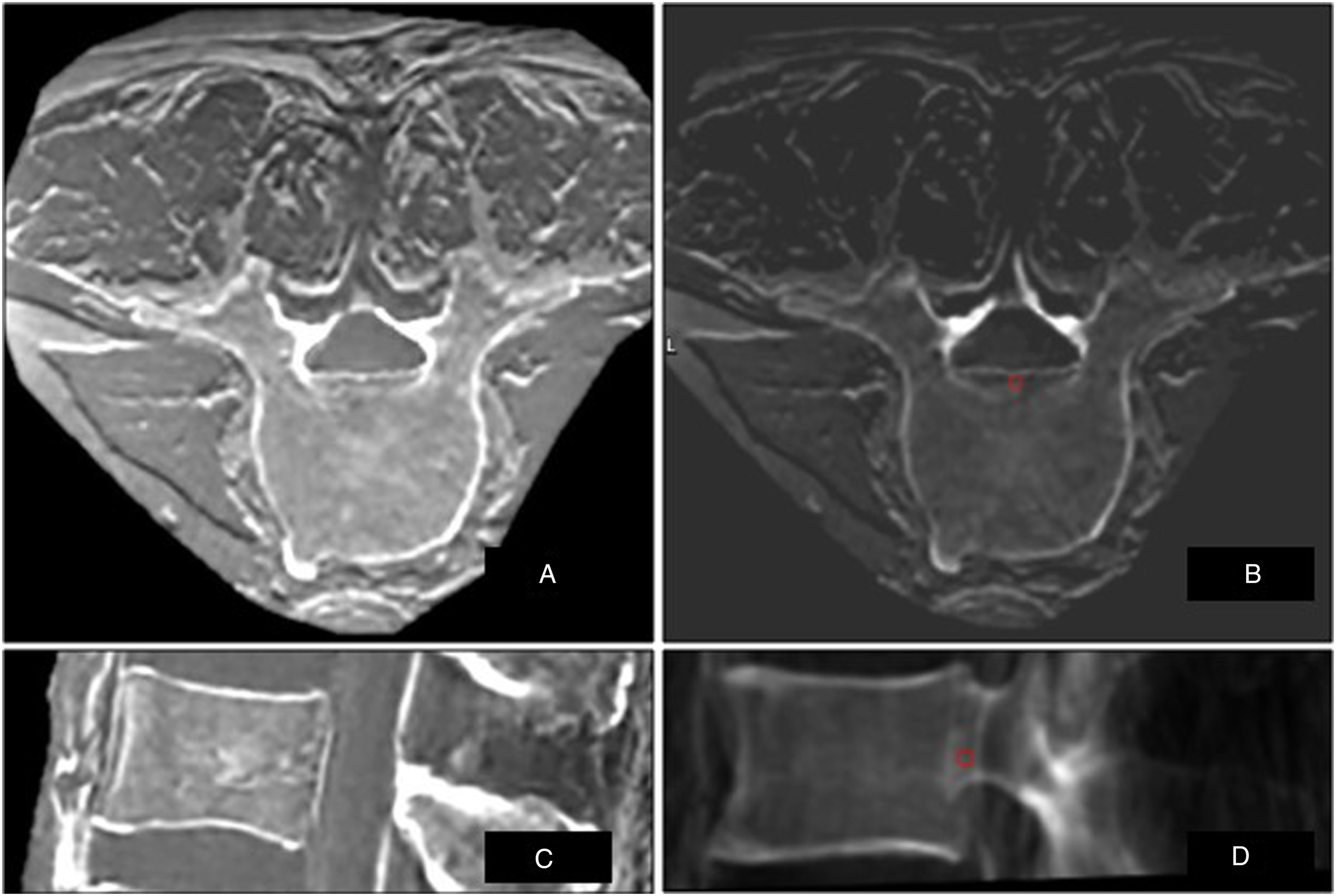
Two biplanar fluoroscopic images (anterior-posterior and lateral-oblique projection (30° to 60°)) were taken using a mobile C-arm (OEC 9900 Elite; GE Healthcare, USA) and then merged with the preoperative MRI being ZTE (Figure 1) or SPGR (Figure 2) in the Mazor X Software Robotics. Registration was confirmed by the robotic software and robotic instruments were successfully registered. ZTE MRI in (A) axial and sagittal (C) view, with the same images inverted before loading into the robotic navigation software - (B) axial and (D) sagittal views - showing surrounding muscles including fascia and aorta in front. SPGR MRI in (A) axial and sagittal (C) view, same images in the robot software for navigation in (B) axial and (D) sagittal showing surrounding muscles incl fascia and aorta in front.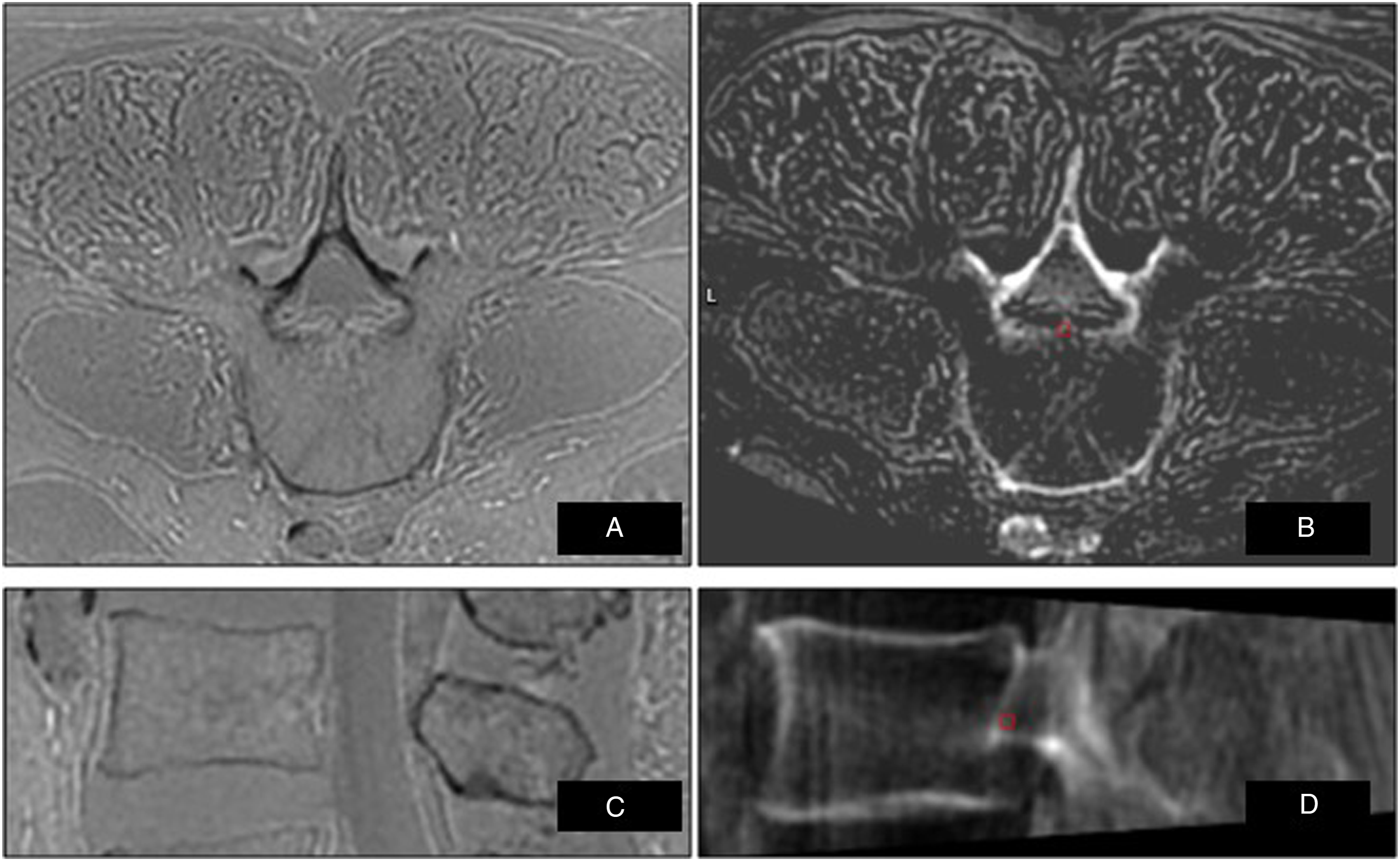
Screw Positioning
MRI-Navigation on ZTE
First the screws planned on the ZTE MRI were selected. The screw trajectory of the left L5 was selected and the robotic arm was sent there. Based on the position of the robotic arm, a paramedian Wiltse incision approximately 3 cm away from the midline was made. Soft tissue and muscle were dissected until the bony cortex was reached. A navigated dilator and cannula were then inserted into the robotic arm, and the position was confirmed using a navigated tapping device. The cortex at the entry point was decorticated with a navigated power burr, followed by cannulation of the pedicle along the predefined path using a navigated drill. Finally, the predefined pedicle screw was placed under real-time robotic navigation. The same workflow based on ZTE images was repeated for the right L5 pedicle screw, along with both pedicle screws in L4.
MRI Navigation on SPGR
The SPGR images of the lumbar spine and the previously planned screws were selected. First the robotic arm was sent to L3 on the left. Again, based on the position of the robotic arm, a paramedian incision and dissection up to the until the bony cortex was made. The tapping, drilling, the use of the navigated power burr and the insertion of the pedicle screw did not differ from the previously described technique. The identical workflow based on SPGR images was repeated for the contralateral pedicle screw of L3 as well as both L2 and L1 pedicle screws.
The screws were meticulously positioned, and the robotic system verified their placement accuracy through check marks visibly presented.
Postoperative Imaging
Postoperative the cadaver was scanned in a routine CT (GE Discovery/LightSpeed; GE Healthcare, Waukesha, WI) with the standard department protocol of 0.75-mm axial slices.
Quantitative Analysis
The position of all robotic placed screws was assessed based on the postoperative CT-scan for the MRI navigated screws. Measurements in millimeters (mm) were taken within the pedicle, both in axial and sagittal planes, for each instrumented pedicle as shown in Figure 3. The precision of pedicle screw placement was assessed using the Gertzbein and Robbins scale (GRS), and classified into 4 groups: A) fully intra-pedicular without pedicle cortex breach, B) breach <2 mm, C) breach between 2 and 4 mm, and D) exceeding the pedicle cortex by 4 to 6 mm. Critical breaches were defined as those surpassing 2 mm (GRS C and D).
12
(A) Preoperative MRI in the robotic planning software in axial and sagittal view and (B) postoperative CT scans, including measurements of the shortest distance to pedicle cortices in both planes.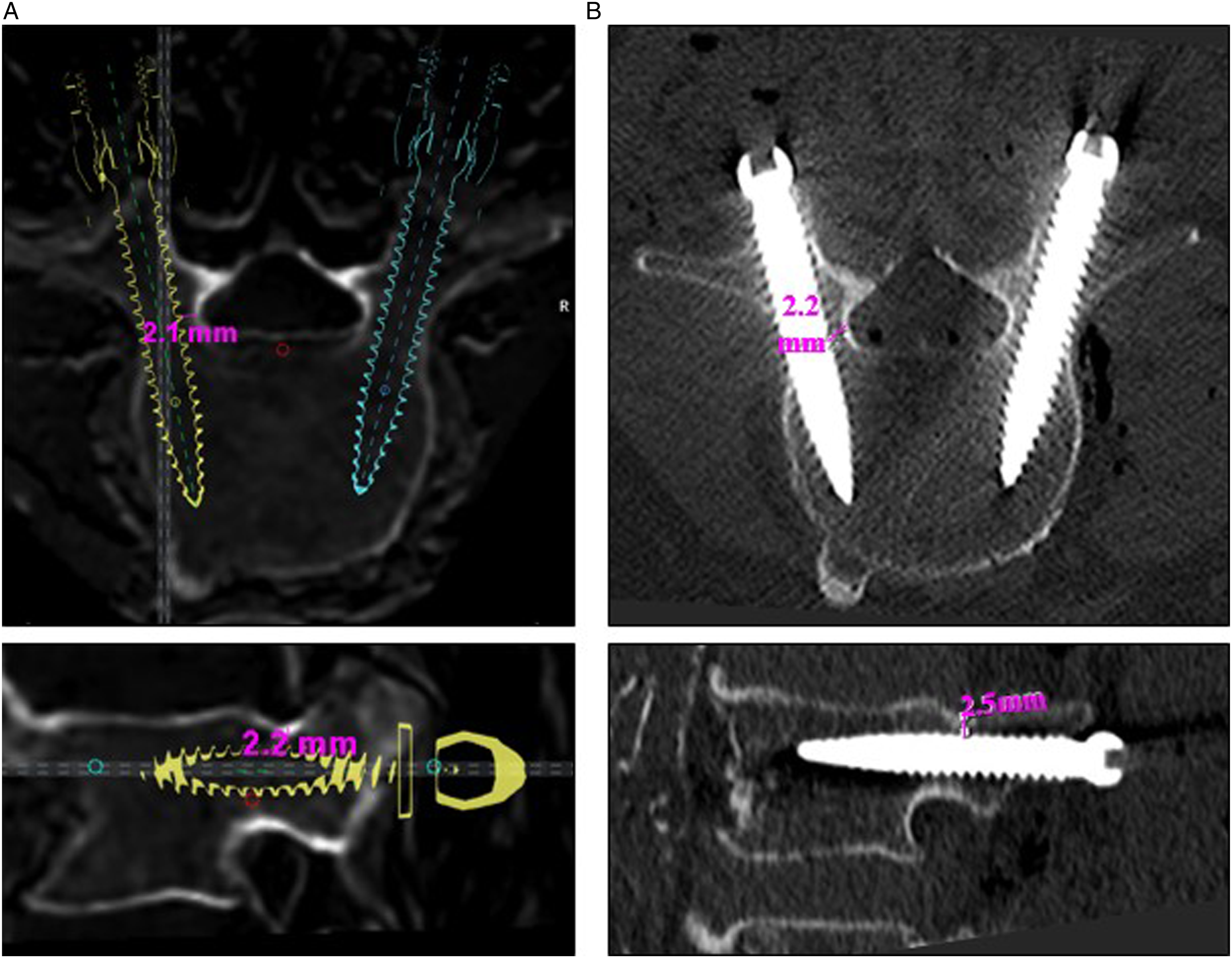
To analyze the screw accuracy the preoperatively planned screw trajectories within the robotic software were compared to the actual screw position on the postoperative CT-scan. Discrepancies from the preoperative planning template were assessed in the medial or lateral direction in the axial plane and in the cranial or caudal direction in the sagittal plane.
Statistical Analysis
Statistical analysis was performed with IBM SPSS Statistics Version 29.0.1.0 (171). Data were non-normally distributed as assessed with the Shapiro-Wilk test. Data are presented as medians and interquartile range (IQR). Mann-Whitney U tests were performed for comparison of MRI placed screws. Statistical significance was set to P ≤ .05.
Results
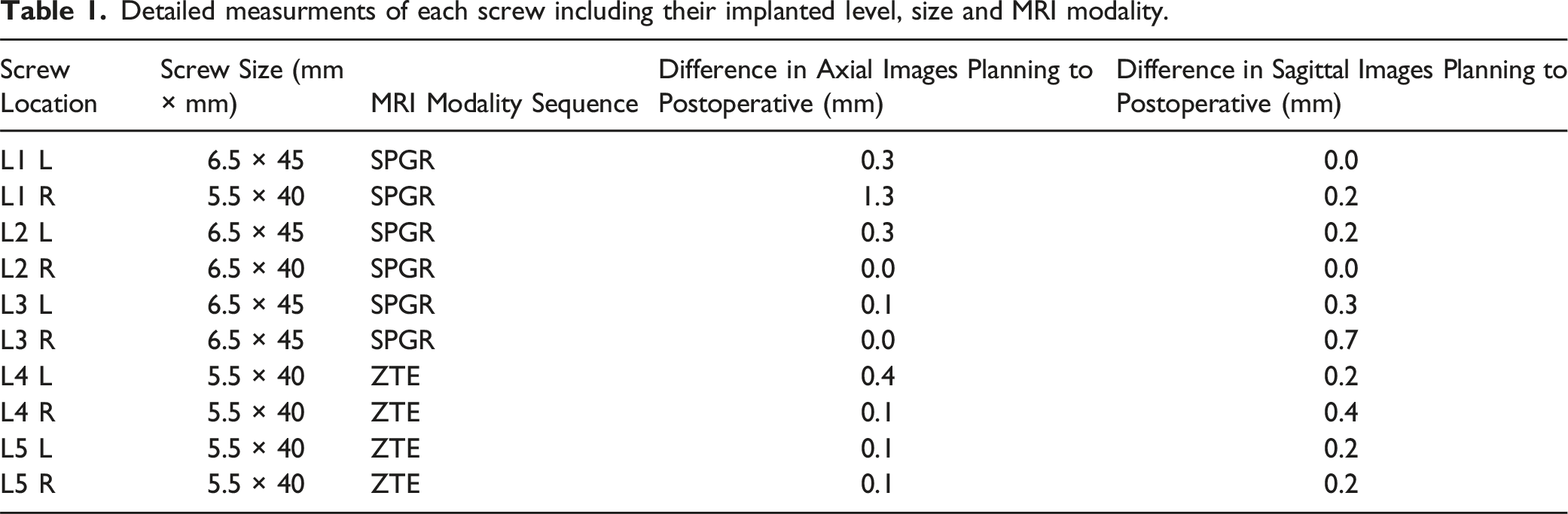
Detailed measurments of each screw including their implanted level, size and MRI modality.
Navigation
Robotic navigation was successfully completed for both imaging modalities SPGR and ZTE. There was no need for Re-registration. Throughout the procedure, all instruments inserted were monitored by the robotic navigation, and there was no abnormal movement or loss of registration. Figure 4 shows a 3D reconstruction of the bony anatomy as well as parts of the surrounding soft-tissue based on the data acquired in ZTE as well as SPGR in comparison to the regular preoperative CT scan. Three-dimensional reconstruction in sagittal and posterior view of the (A) preoperative CT, (B) ZTE and (C) SPGR.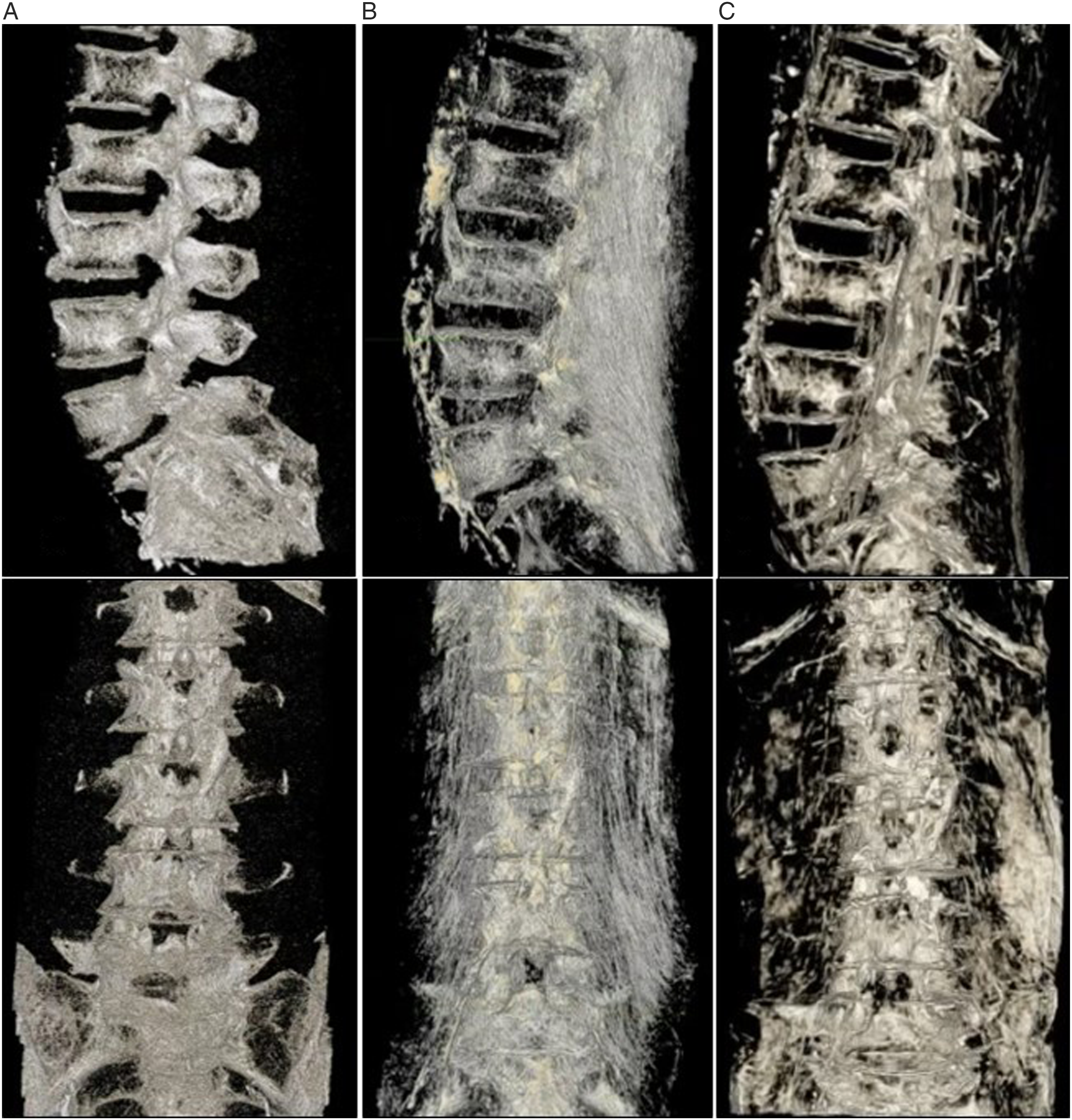
Screw Accuracy
All the placed screws were uniformly classified as Gertzbein and Robbins Grade A. There was no breach and there were no instances of screw revisions observed throughout the entirety of the procedure. The screws deployed exhibited consistent dimensions, ranging between 5.5 and 6.5 mm in width and 40 to 45 mm in length.
When comparing the preoperative planning in the robotic software to the postoperative CT scan no difference could be detected for 2 screws in the axial plane and for 2 screws in the sagittal plane, 1 screw showed no deviation in both planes being visually matched to the preoperative plan. The majority of screws showed a minimal deviation in all planes with a median deviation of 0.25 mm (0.0; 1.3). Whereas the median deviation in the axial plane was 0.27 mm (0.0; 1.3) and in the sagittal plane was 0.24 mm (0.0; 0.7).
Discussion
This study demonstrates the first successful registration of MRI sequences, ZTE and SPGR, in robotic spine surgery here used for intraoperative navigation of lumbar pedicle screws. The findings from this cadaver study show a sufficient accuracy of robotic navigation based on MRI marking a significant step toward radiation-free spine surgery underscoring the potential transformative role of MR-images in enhancing the safety and accuracy of spinal interventions.
Previous research has been done to validate robotic navigation based on a synthetic CT, showing accurate navigation of 7 lumbosacral screws. 13 However, the creation of a synthetic CT based on MRI requires an additional software, not available in every institution, to post-process images using a deep learning algorithm which was derived using paired MRI and CT data. 14 Potential artifacts may arise within this learning paradigm, stemming from constraints such as a restricted variety in the images employed for training. This limitation could result in a constrained ability to accurately identify specific pathologies. 6
It has been shown that there have been significant differences in the performance and robustness of these deep learning models for synthetic CT generation. 14 Further, synthetic MRI are generated from MRI input, therefore being vulnerable for reconstruction artifacts due to limited source MRI contrast. 6 All of these factors can potentially change the accuracy of the image used for navigation and therefore navigation becoming a false-friend. The used MRI-sequences, ZTE and SPGR, used in this study do not require an additional software after their acquisition and therefore the risk of potential inaccuracy due to algorithmic modification does not exist.
Comparing these two MRI sequences: SPGR has higher signal to noise ratio than ZTE, and stronger cortical bone to soft tissue contrast. However, ligaments can appear similar to cortical bone in SPGR, more so than on ZTE. Also, the readout used with ZTE is more robust to patient motion, so certain levels such as L1 and L2 are more prone to motion artifacts than L3 and below. Since ZTE has been shown to be a robust alternative to CT for imaging of cortical bone without the use of ionizing radiation offering the advantages of rapid examination times, silent scanning, and artifact resistance. 8
When comparing ZTE and SPGR it has been shown that for example in the knee ZTE improved the assessment of osseous abnormalities in comparison to SPGR. 15 Further, high intramodality and interrater agreement between ZTE MRI and CT were also found when evaluating cervical neuroforaminal stenosis, lumbar degenerative changes, and sacroiliac joint and structural lesions. 16 In addition, ZTE MRI can be used to evaluate ossification of the posterior longitudinal ligament of the spine.9,17,18 Offering similar bony reconstructions as 3D CT reconstructions is a significant advantage of ZTE and SPGR. However, a previous limitation of these 2 types of MRI has been that they did not provide quantitative Hounsfield Unit (HU) maps. 19 This issue has been addressed in our study by converting the MRI sequences into HU.
For example, combining the essential information of bony structures as well as soft-tissue details in 1 image for intraoperative navigation has been tried before by combining preoperative CT and MRI Images however, the acquisition of a CT scan and, therefore, exposure to ionizing radiation remained. 1 During the robotic placement of pedicle screws with navigation using MRI images, the procedure now provides not only accurate bony information but also details about surrounding soft tissues such as vessels. This added dimension of information is instrumental in preventing inadvertent injury to these critical structures during the surgical intervention. Therefore, combining the strengths of conventional MRI, soft-tissue contrast, and ZTE-MRI, cortical bone detail, the benefits associated with acquisition of concurrent CT imaging data may no longer outweigh its costs including ionizing radiation exposure in some cases. 9
The maximum translational error tolerance for image guided screw placement was found in a mathematical model to be 1 mm for each spine level in our study a deviation of actual postoperative screw placement to the planed trajectories based on MRI was on average 0.25 mm. 20 In addition, a pedicle breach up to 2 mm is considered as safe and acceptable by some reports, whereas all our MRI based navigated screws were positioned with a maximal deviation of 1.3 mm. 21
However, we had a limited sample size, involving only 1 cadaver, thereby restricting the generalizability of findings to the broader lumbar spine surgery population. The experimental conditions of the preoperative imaging as well as the surgery did not replicate normal surgical circumstances, including respiration potentially leading to more inaccuracy of the acquired images.
Future studies should create validation for inpatient navigation involving the cervical and thoracic spine as well as the MRIs application in patients with previous spinal instrumentation in context of revision surgery. Further investigation should focus on the benefits of visualizing the surrounding soft-tissue besides the accuracy of bony structures while navigating more complex surgical steps in close distance to critical structures.
Conclusion
Preoperative ZTE and SPGR MRI can be successfully registered for intraoperative navigation and robotic-assisted pedicle screw placement and potentially representing a radiation-free equivalent to preoperative CT. The outcomes of our cadaver study suggest that the accuracy of robotic navigation using MRI is sufficient. Our findings represent a step towards radiation-free spine surgery, emphasizing the potential using of MR-images in elevating the safety and precision of spinal interventions. However, further investigation is required to assess application in patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded with a grant-support from Rama and Shashi Marda Foundation.
