Abstract
Study Design
Retrospective observational cohort study.
Objective
En bloc resection for primary tumours of the spine is associated with a high rate of adverse events (AEs). The objective was to explore the relationship between frailty/sarcopenia and major perioperative AEs, length of stay (LOS), and unplanned reoperation following en bloc resection of primary spinal tumours.
Methods
This is a unicentre study consisting of adult patients undergoing en bloc resection for a primary spine tumor. Frailty was calculated with the modified frailty index (mFI) and spine tumour frailty index (STFI). Sarcopenia was quantified with the total psoas area/vertebral body area ratio (TPA/VB) at L3 and L4. Univariable regression analysis was used to quantify the association between frailty/sarcopenia and major perioperative AEs, LOS and unplanned reoperation.
Results
95 patients met the inclusion criteria. The mFI and STFI identified a frailty prevalence of 3% and 18%. Mean CT TPA/VB ratios were 1.47 (SD ± .05) and 1.83 (SD ± .06) at L3 and L4. Inter-observer reliability was .93 and .99 for CT and MRI L3 and L4 TPA/VB ratios. Unadjusted analysis demonstrated sarcopenia and mFI did not predict perioperative AEs, LOS or unplanned reoperation. Frailty defined by an STFI score ≥2 predicted unplanned reoperation for surgical site infection (SSI) (P < .05).
Conclusions
The STFI was only associated with unplanned reoperation for SSI on unadjusted analysis, while the mFI and sarcopenia were not predictive of any outcome. Further studies are needed to investigate the relationship between frailty, sarcopenia and perioperative outcomes following en bloc resection of primary spinal tumors.
Keywords
Introduction
En bloc resections for tumours of the spine are surgically complex and associated with a high rate of adverse events (AEs). 1 Our research group previously demonstrated that 79% of patients undergoing en bloc surgery for a primary bone tumour or a solitary metastasis of the spine experienced at least 1 perioperative AEs. 1 Approximately one-third of the cohort required admission to the intensive care unit (ICU), while one-fifth required an unplanned reoperation. In this analysis, the only predictors of AEs were age and surgical duration. 1 The latter reflects the degree of surgical invasiveness and does not inform regarding patient factors contributing to poor outcomes. The impact of patient factors on AEs, length of stay and unplanned reoperation remains largely unknown in patients undergoing oncologic spinal resection.
Frailty is a state of increased vulnerability to adverse health outcomes due to the age-associated loss of physiological reserve and inability to maintain homeostasis after a stressor event.2-4 Within the literature, frailty is recognized as an independent risk factor for postoperative AEs in patients undergoing spine surgery.5-7 The most common frailty index reported in the spine literature is the modified frailty index (mFI), 5 derived from the theory of accumulated deficits. 8 While this index has performed well at predicting outcomes in various spinal pathologies, it was not predictive of AEs in previous analyses of the spinal oncology population.1,9-11 However, these studies were limited by small sample sizes and the inherent limitations of the frailty indices. Accordingly, the Spinal Tumor Frailty Index (STFI) was developed by Ahmed et al to overcome the limitations of the mFI. 12 Specifically designed for primary spinal tumours, this frailty index strongly predicted complications. 12 However, this study reported various spinal tumours, including intra-dural lesions, limiting its application to en bloc resection.
Sarcopenia, defined as a progressive loss of skeletal muscle strength, is associated with the loss of muscle mass, quality and physical performance.13,14 The psoas cross-sectional area has been postulated to be a surrogate for sarcopenia. 14 In the metastatic spine population, the ratio of the psoas cross-sectional area over the vertebral body area at the L3 level is a predictor of perioperative AEs.9,15 Zakaria et al also reported an association between sarcopenia and postoperative AEs in the metastatic spine population when the psoas cross-sectional area was measured at the L4 level.16,17 However, the most accurate level for measuring the psoas cross-sectional area to determine sarcopenia is unknown, 18 and its association with primary tumors of the spine has not been previously reported.
The primary objective of this study was to describe the relationship between frailty/sarcopenia and major perioperative AEs following en bloc resection for primary tumours of the spine. Secondary objectives were to estimate the prevalence and distribution of frailty and sarcopenia and the relationship between sarcopenia and frailty on in-hospital length of stay (LOS) and unplanned reoperation for patients with primary tumours of the spine undergoing en bloc resection.
Methods
Study Design
This is a retrospective study of prospectively collected data from patients at a single quaternary academic teaching centre undergoing elective spine surgery for a primary tumour of the spine. Institutional ethics approval was obtained (H20-01012). The study was conducted in accordance with the principles of the Helsinki Declaration. The requirement for informed consent was waived because of the anonymous nature of the data.
Patient Population
Patients were included in this study if they underwent an en bloc resection for a primary tumour of the spine (benign or malignant) between January 1st, 2009, and February 28th, 2020. Patients were excluded if they underwent en bloc resection for isolated spinal metastases, intra-lesional resection, or if the pathology was intra-dural. En bloc resection is a surgical procedure that aims to remove a tumour as 1 intact piece.
Patient and Surgical Data
Patient demographic data was prospectively collected in our local database. Tumour histopathological diagnosis, location, previous treatment (systemic treatment, radiation therapy and surgery), preoperative neurologic status (International Standards for Neurological and Functional Classification of Spinal Cord Injury by the American Spinal Injury Association (INSCI ASIA) impairment scale), surgical margins assessed by the pathologist (wide, marginal, intralesional) and surgical procedural details (staged procedure, complex plastic surgery closure, intraoperative blood loss and operative duration) were recorded. The Enneking Appropriateness (EA) was also determined as previously described. 19
Measurement of Frailty
Two frailty measures were used: the modified frailty index (mFI) and the Spinal Tumor Frailty Index (STFI). The mFI is a generic index derived from the cumulative deficit theory of frailty. 20 It includes 11 variables: dependent functional status, diabetes mellitus, lung problems, congestive heart failure, myocardial infarction, cardiac problems, hypertension, impaired sensorium, prior transient ischemic attack, history of stroke, and peripheral vascular disease. 20 The mFI was calculated retrospectively by adding the number of deficits present divided by 11 (n/11). The index range is 0-1, and frailty is defined as greater than or equal to .21 (mFI ≥.21). 21
The STFI developed by Ahmed et al consists of 9 distinct items: anemia, congestive heart failure, chronic obstructive pulmonary disease, coagulopathy, electrolyte abnormalities, pulmonary circulation disorders, renal failure, malnutrition, and pathological fractures. 12 Retrospectively calculated, patients with 0 comorbidities are categorized as non-frail, whereas patients with 2 or more comorbidities are considered frail (STFI ≥2). 12
Measurement of Sarcopenia
Sarcopenia, defined as low muscle strength, is confirmed in the presence of low-performance status or low muscle quantity or quality.13,14 Psoas volume measurement has been used as a surrogate of sarcopenia.22,23 We elected to measure the total psoas area/vertebral body ratio rather than the normalized total psoas area (NTPA),24,25 as the latter requires patient height. This ratio is calculated by the sum of the left and right psoas muscle area (Total Psoas Area (TPA) divided by the area of the vertebral body [TPA/VB]) measured on the first image in the craniocaudal direction in which the 2 transverse processes are visible, as previously described.9,15,16,26 Previous studies have measured the TPA/VB ratio at the L3 and L4 levels on magnetic resonance imaging (MRI) and computed tomography (CT).9,15,16,26 The ratio was retrospectively measured at L3 and L4 on MRI and CT images. Lesions that significantly altered the vertebral body or paravertebral musculature anatomy, precluding accurate morphometric calculations, were removed from the analysis. Measurements were performed on the preoperative imaging done within 3-months of the surgery. MRI measurements were taken on axial T1 weighted images, while CT axial images were adjusted -30 and 110 Hounsfield units (HU) to exclude vasculature, bony structures and fat infiltrating the muscle borders.
To assess the inter-observer reliability of the various ratio measurements (L3 CT TPA/VB, L4 CT TPA/VB, L3 MRI TPA/VB, and L4 MRI TPA/VB), 3 investigators with various training levels (spine surgeon, resident, and a research assistant) independently measured the ratios of 20 CT/MRI images, respectively.
Outcome Measures
The primary outcome was major perioperative adverse events (AEs), which included cardiac event, spinal cord injury, nerve root injury, postoperative neurologic deterioration, cerebrospinal fluid (CSF) leakage requiring a return to the operating room, hardware failure requiring revision, massive blood loss (>5 L in 24 hours or >2 L in 3 hours), vascular injury, thromboembolic event, wound dehiscence, and deep wound infection. AEs are prospectively collected using the Spine AdVerse Events Severity system [SAVES], version 2 containing 14 specific intraoperative and 22 postoperative AEs. 27 Secondary outcomes were collected in our local database, including in-hospital LOS and unplanned reoperation.
Statistical Analysis
Descriptive statistics were used to describe the characteristics of the cohort using means (standard deviation [SD]), medians (interquartile range [IQR]) or percentages, as appropriate. The relationship between frailty, sarcopenia and our primary outcome was analyzed using logistic regression and quantified with unadjusted odds ratios (OR). We then created a multivariate logistic regression model, including variables associated with the primary outcome (P < .2). Other secondary analyses included examining the relationship between frailty, sarcopenia, in-hospital LOS and unplanned reoperation. Unadjusted OR were generated using logistic regression for unplanned reoperation and Poisson regression to determine the Incidence Rate Ratio (IRR) for LOS. Inter-observer reliability measurements were evaluated using the intraclass correlation coefficient (ICC). Based on the 95% confidence interval of the ICC estimate, values <.5; .5 - .75; .75 - .9, and >.90 indicate poor, moderate, good, and excellent reliability, respectively. 28
All data analysis was performed using SAS Version 9.4 (SAS Institute, Cary, North Carolina) and STATA 12.1 (StataCorp, Texas, USA). All tests were two-tailed, and a P-value <.05 was considered statistically significant.
Results
One hundred and forty-one patients were initially identified from our database. Of these, 46 patients were excluded (3 intra-dural tumours, 1 non-tumour-related surgery, 25 intralesional resections, and 17 undergoing en bloc resection for isolated spinal metastases). Thus, 95 patients undergoing en bloc surgical resection for a primary spinal tumour were available for analysis.
Clinical, Tumour and Surgical Characteristics
Study Population Characteristics.

American Spinal Injury Association (ASIA); Aneurysmal Bone Cyst (ABC); computed tomography (CT); Giant Cell Tumour (GCT); Interquartile Range (IQR); Magnetic Resonance Imaging (MRI); Malignant Peripheral Nerve Sheath Tumour (MPNST); modified frailty index (mFI); Spinal Tumour Frailty Index (STFI); Standard deviation (SD); Total Psoas Area (TPA); Vertebral Body (VB).
In terms of tumor histological characteristics, chordoma and chondrosarcoma represented the most common tumours (n = 41, 43.16%). The mean surgical duration was 535.65 minutes (SD ± 23.32 minutes), and the mean intraoperative blood loss was 1718.34 mL (mL) (SD ± 219.26 mL). Closure by plastic surgery was employed in 29 patients (30.53%). Wide or marginal margins were achieved in 83 patients (88%). An Enneking Appropriate procedure was achieved in 83 patients (87.18%). Patients, tumoral and surgical characteristics are detailed in Table 1.
Prevalence and Distribution of Frailty
The mean and median mFI were .04 (SD ± .007) and 0 [IQR 0-.09]. Stratified with the mFI, 63 patients (66%) were not frail (mFI = 0), 29 patients (31%) were pre-frail (mFI = .01-.20), and 3 patients (3%) were frail (mFI ≥.21). The mean and median STFI were .65 (SD ± .094) and 0 [IQR 1]. Stratified using the STFI, 56 patients (59%) were not frail (STFI = 0), 22 patients (23%) were mildly frail (STFI of 1), and 17 patients (18%) were considered frail (STFI ≥2). The frailty distribution is illustrated in Figure 1(A) and (B). (A) Frailty Distribution with the modified Frailty Index (mFI). (B) Frailty Distribution with the Spine Tumor Frailty Index (STFI).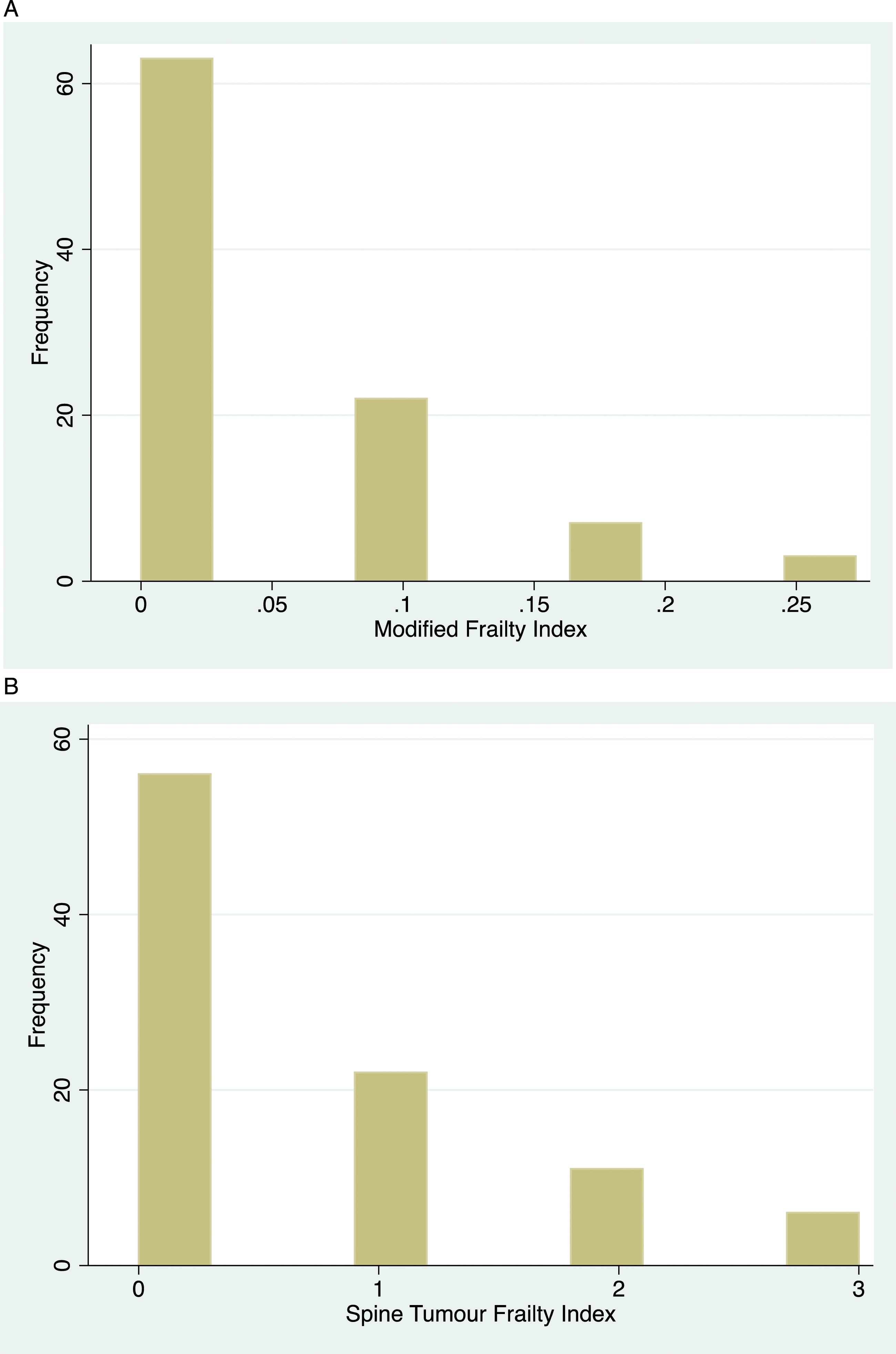
Prevalence and Distribution of Sarcopenia
The CT TPA/VB ratio at L3 was available for 63 patients. At L4, the CT ratio was measured in 64 patients. For the MRI TPA/VB ratio, measurements were obtained in 31 patients for L3 and 32 patients for L4. The mean CT ratio at L3 was 1.47 (SD ± .05) (male 1.53 [SD ± .06] and female 1.37 [SD ± .08]), and the mean CT ratio at L4 was 1.83 (SD ± .06) (male 1.89 [SD ± .07] and female 1.72 [SD ± .11]). The mean MRI ratios were 1.57 (SD ± .10) and 1.89 (SD ± .12) at L3 and L4. Figure 2(A) and (B) illustrates the CT L3 and L4 TPA/VB ratio distribution. The Pearson correlation ratio was high (r = .79, 95% CI 0.55-.91, P < .01) for the L3 CT and MRI measurements and high (r = .74, 95% CI 0.47-.88, P < .01) for the L4 CT and MRI measurements. Inter-observer reliability was excellent (.93 and .99) for the CT and MRI L3 and L4 TPA/VB ratios. (A) Distribution of the CT L3 TPA/VB ratio. (B) Distribution of the CT L4 TPA/VB ratio. Total psoas area (TPA); vertebral body (VB).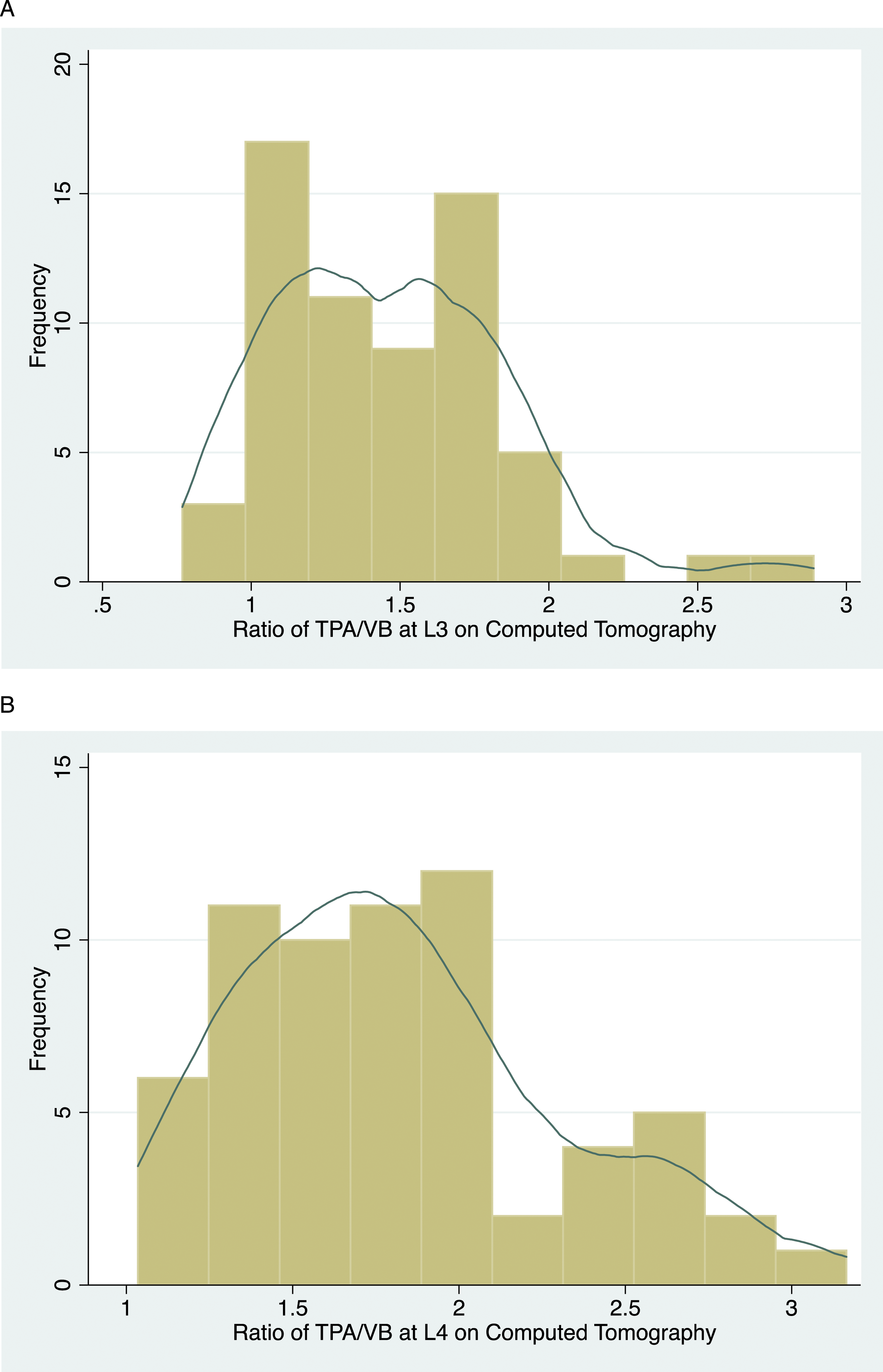
Major Perioperative Adverse-Events
Univariate Predictors of Major Perioperative Adverse Events (AEs).
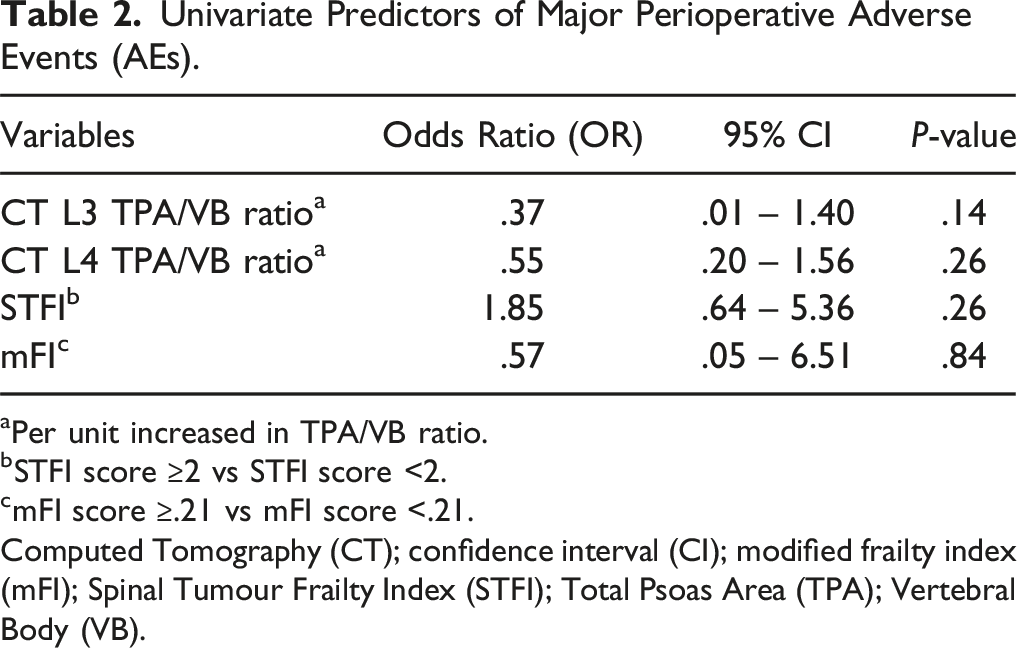
aPer unit increased in TPA/VB ratio.
bSTFI score ≥2 vs STFI score <2.
cmFI score ≥.21 vs mFI score <.21.
Computed Tomography (CT); confidence interval (CI); modified frailty index (mFI); Spinal Tumour Frailty Index (STFI); Total Psoas Area (TPA); Vertebral Body (VB).
In-Hospital Length of Stay
Univariate Predictors of Prolonged In-Hospital Length of Stay (LOS).

aper unit increased in TPA/VB ratio.
bSTFI score ≥2 vs STFI score <2.
cmFI score ≥.21 vs mFI score <.21.
Computed Tomography (CT); confidence interval (CI); modified frailty index (mFI); Spinal Tumour Frailty Index (STFI); Total Psoas Area (TPA); Vertebral Body (VB).
Unplanned Reoperation Within the Same Admission
Univariate Predictors of Unplanned Reoperation During Index Admission.
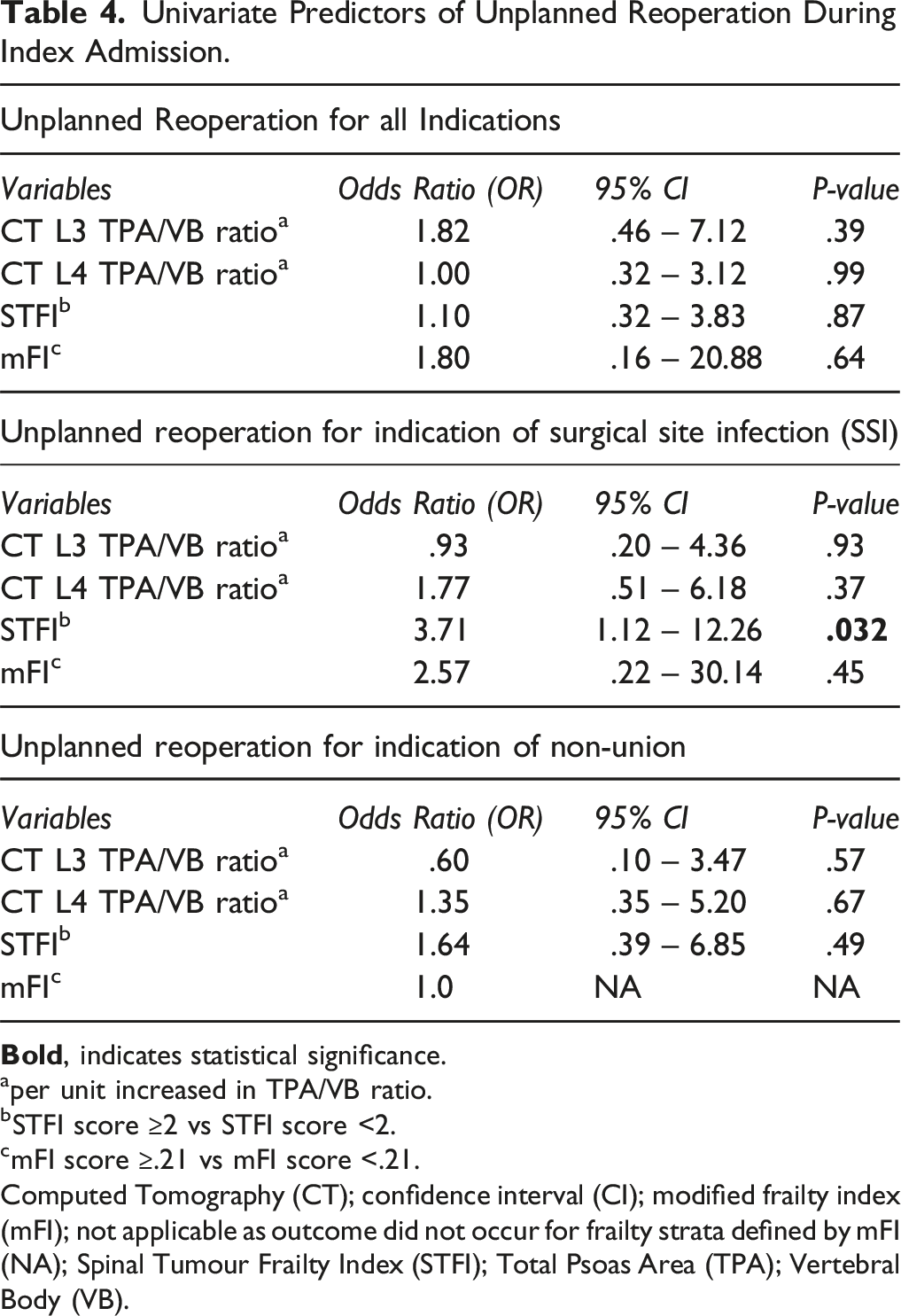
aper unit increased in TPA/VB ratio.
bSTFI score ≥2 vs STFI score <2.
cmFI score ≥.21 vs mFI score <.21.
Computed Tomography (CT); confidence interval (CI); modified frailty index (mFI); not applicable as outcome did not occur for frailty strata defined by mFI (NA); Spinal Tumour Frailty Index (STFI); Total Psoas Area (TPA); Vertebral Body (VB).
Multivariable Analysis
A multivariate logistical regression model was developed to further explore the relationship between sarcopenia, frailty and major perioperative AEs. Variables included within this model were age, gender, tumour location (fixed or mobile spine), malignant history, previous treatment (systemic treatment, radiation therapy and surgery) and the presence of neurologic deficit (ASIA A-B-C-D). This model revealed large 95% CI intervals, suggesting imprecise measurements from either a lack of power or the absence of association with major perioperative AEs, LOS or unplanned reoperation (Supplemental Material).
Discussion
Given the relationship between frailty, sarcopenia and postoperative AEs in spine surgery, 5 we sought to determine and compare the ability of these measures as risk stratification tools in a subgroup of the surgical spine oncology population, those undergoing en bloc resection for primary spinal tumours. This study focused on en bloc resection as these surgeries are associated with a high risk of perioperative AEs. 1 On univariate analysis, only frailty defined by the STFI was significantly predictive of unplanned reoperation for the indication of surgical site infection. However, the STFI failed to remain statistically significant on multivariable analysis. Sarcopenia and frailty were otherwise not predictors of major perioperative AEs, length of stay, or unplanned reoperation. Descriptive analysis identified that this unique surgical population consisted of mainly non-frail or pre-frail patients with little to no comorbid burden when assessed with the mFI and STFI. Accordingly, these results highlight the challenge of defining and assessing frailty and sarcopenia for patients undergoing en bloc resection for primary bone tumours of the spine.
Understanding and identifying factors associated with adverse perioperative outcomes is fundamental when counselling patients on the risk of operative intervention. Frailty tools such as the STFI and mFI may be helpful risk stratification tools for determining surgical risk and candidacy. 5 Our results suggest that the mFI is not the appropriate tool to measure frailty in this unique oncologic population. The mFI was not a predictor of perioperative AEs following en bloc resection of spinal tumours, in keeping with previously published results within the spine oncology literature.1,9-12,29 Within the metastatic spine tumour population, the mFI poorly predicted postoperative AEs and LOS. 9 Similarly, the STFI has been a variable predictor of postoperative AEs. Initially validated by Ahmed et al, the STFI independently predicted major postoperative AEs with moderate sensitivity in their study cohort of patients undergoing surgery for primary tumours of the spine. 12 However, only frailty defined by an STFI score ≥2 predicted unplanned reoperation for SSI in our study despite a higher prevalence of frailty (18% vs 2.2%), greater incidence of perioperative AEs (46.3% vs 10.6%) and longer postoperative LOS (median LOS 17 days [IQR 8-36] vs median LOS 5 days [IQR 3-9]) compared to Ahmed et al. 12 This variability is attributable to differences in the study population, reporting and defining perioperative AEs, and methodology for establishing frailty. Compared to our study, Ahmed et al included a heterogeneous cohort of patients with benign or malignant spinal tumours requiring spinal decompression and/or fusion. 12 They also failed to describe a valid and reliable approach to defining, reporting and collecting perioperative AEs, subsequently increasing the risk of reporting and coding biases. As frailty measures, the STFI and mFI are imprecise tools for estimating frailty. These tools assess each health deficit dichotomously without adjusting the individual score based on prevalence or association with frailty and adverse health outcomes. 30 Consequently, the mFI and STFI may underestimate or overestimate patient frailty as equal scores are awarded to deficits that potentially do not contribute to frailty or complication risk. 30 Regardless, the heterogeneity observed within the spine oncology population between the mFI, STFI and major perioperative AEs raises concerns regarding the validity of these measures as risk stratification tools.
A limited number of clinical frailty instruments are validated risk stratification tools in the spine oncology population undergoing surgical resection of primary or metastatic spinal tumours.5,7 Inherent construct limitations of the mFI and STFI may have contributed to the absence or poor relationship between patient frailty and perioperative outcomes observed in our study.31,32 The mFI and STFI determine frailty by predominately assessing the absence or presence of specified comorbidities.1,31 In our study, the cohort consisted of patients with primary spinal tumours, a population of younger individuals with minimal comorbidities or systemic disease. Consequently, only 3% and 18% of our study population were considered frail, while 66% and 59% were non-frail when assessed with either the mFI or STFI. Given the design of these instruments, the mFI and STFI are unlikely to identify frail patients in younger and healthier populations, therefore reducing the association between frailty and adverse outcome.1,31,32 Patients undergoing en bloc surgical resection of primary spinal tumours must be physiologically fit to tolerate prolonged surgical duration and greater surgical invasiveness. 1 The STFI is derived from the metastatic spine tumour frailty index (MSTFI)12,33 that assesses physiological parameters of metastatic disease, consequently reducing the sensitivity of the STFI to assess frailty in our population. Additionally, instruments such as mFI do not account for the physiological effects associated with oncological disease, such as tumour burden and adjunctive therapy. 32 These factors affect underlying physiological reserve and confound the relationship between frailty and postoperative AEs.
Given the current limitations of frailty measures utilized in the spine oncology population, sarcopenia is becoming more commonly recognized as a risk stratification tool.15,17 As a marker of functional decline and physical frailty, sarcopenia is easily assessable in an objective and consistent manner.34-36 Previously published literature has demonstrated that morphometric analysis of the psoas area is an important predictor of perioperative AEs following spine surgery.9,15-17,25,37 We observed that sarcopenia was not predictive of perioperative major AEs, LOS, or unplanned reoperation, inconsistent with previously reported findings in the spine oncology population.9,15 Bourassa-Moreau et al reported that sarcopenia significantly predicted perioperative AEs in patients undergoing emergent spine surgery for metastatic spine tumours. 9 However, the authors observed a far greater severity of sarcopenia in their population (median L3 TPA/VB 0.93 [IQR .48-2.62]) 9 compared to ours (mean L3 TPA/VB 1.47 [SD ± .05]). The variability in our study is potentially reflective of a floor effect whereby the L3 and L4 TPA/VB ratio has a lower limit to the data values it can reliably specify. Given the characteristics of our study population, the degree of sarcopenia is inherently lower, raising the question of whether sarcopenia is an appropriate risk stratification instrument in this population. Ultimately, further studies are needed to validate and determine the role of sarcopenia measures as risk stratification tools in the primary spinal tumour population undergoing en bloc resection.
Aside from the spine oncology population, sarcopenia has also been a highly variable predictor of postoperative AEs within the degenerative spine population. 38 However, it is challenging to compare sarcopenia between the degenerative and oncological populations due to differences in the assessment method and underlying disease process. Within the degenerative spine literature, sarcopenia has been examined using the NTPA, 38 which is not validated in the spinal oncology population. Additionally, the development of sarcopenia in the degenerative population is primarily mediated by pain, which alters spine biomechanics and promotes muscle wasting at the localized level of symptomatic disease. 39 This differs vastly from the oncology population, whereby sarcopenia results from local, systemic, and treatment processes, including chronic cytokine response, chemotoxicity, localized pain, and poor nutritional intake. 40
Similar to frailty, many measures for screening, assessing and quantifying sarcopenia have been described ranging from anthropometric measurements to clinical tests such as grip strength and questionnaires.34-36 The L3 and L4 TPA/VB ratios were used to assess sarcopenia given their practicality and previous reports validating their use for predicting short-term and long-term postoperative outcomes in the spinal oncology population.9,15-17 Additionally, we observed a high degree of inter-rater reliability, suggesting that the L3 TPA/VB and L4 TPA/VB can be consistently assessed with minimal error. A high degree of concordance between CT and MRI assessments of L3 and L4 TPA/VB was also observed, suggesting sarcopenia can be readily measured from preoperative CT or MRI imaging. However, the usability of the L3 and L4 TPA/VB ratio depends on acquiring timely preoperative imaging. As seen in our study, many patients had unavailable imaging to calculate the L3 or L4 TPA/VB. Accordingly, obtaining timely preoperative imaging may be a significant barrier to using radiographic sarcopenia analysis for risk stratification.
Strengths and Limitations
One of the major strengths of our study is the utilization of a prospective and spine-specific adverse-event dataset allowing for transparent and accurate reporting of perioperative AEs. Furthermore, the inter-rater reliability analysis ensures that sarcopenia was consistently calculated, reducing the risk of bias. Despite our strengths, the limitations observed in our study are directly related to the specific patient population. As previously mentioned, en bloc resection of spine tumours is rarely performed. Consequently, this results in an inherently small study population, reducing the power of association between frailty, sarcopenia and perioperative AEs and increasing type II error. This was observed in our univariate and multivariate analyses that demonstrated large 95% confidence intervals and missing data as the outcomes did not occur in specific frailty strata. The small sample size also precluded the ability to perform a formal survival analysis, as we could not adjust for potential confounders through a multivariate analysis. While the associations found in univariate analysis are of interest, we could not adjust for potential confounders, and further confirmation in a larger cohort is required.
Conclusion
En bloc resection of primary bone tumours of the spine with appropriate oncological margins is associated with a high rate of perioperative morbidity and adverse events (AEs). Sarcopenia assessed with the CT L3 TPA/VB and L4 TPA/VB ratios and frailty defined by the mFI were not predictive of any clinical outcomes in this oncological spine population. Frailty assessed with the STFI only predicted unplanned reoperation for SSI on univariate analysis, but not major perioperative AEs, LOS or any other outcomes. Accordingly, further investigation is needed to characterize the relationship between frailty, sarcopenia and perioperative outcomes, and identify the optimal tool for assessing frailty and sarcopenia in the spine oncology population undergoing en bloc surgical resection for primary spinal tumours.
Supplemental Material
Supplemental Material - The Role of Frailty and Sarcopenia in Predicting Major Adverse Events, Length of Stay and Reoperation Following En Bloc Resection of Primary Tumours of the Spine
Supplemental Material for The Role of Frailty and Sarcopenia in Predicting Major Adverse Events, Length of Stay and Reoperation Following En Bloc Resection of Primary Tumours of the Spine by Eryck Moskven, MD, Oliver Lasry, MD, PhD, FRCSC, Supriya Singh, MD, FRCSC, Alana M. Flexman, MD, FRCPC, John T. Street, MD, PhD, FRCSC, FRCS (Tr and Orth), Nicolas Dea, MD, MSc, FRCSC, Charles G. Fisher, MD, MHSc, FRCSC, Tamir Ailon, MD, MPH, FRCSC, Marcel F. Dvorak, MD, MBA, FRCSC, Brian K. Kwon, MD, PhD, FRCSC, Scott J. Paquette, MD, MEd, FRCSC, and Raphaële Charest-Morin, MD, FRCSC in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Previous Presentation
No portion of the work has been published. A podium presentation was presented at the Annual Canadian Spine Society virtual meeting, February 2021.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
