Abstract
Study Design
Systematic Review.
Objective
To review the literature for complications and outcomes after the implantation of cellular bone matrix (CBM) during spine fusion.
Methods
The PubMed database was queried from inception to January 31, 2023 for any articles that discussed the role of and identified a specific CBM in spinal fusion procedures. Adverse events, reoperations, methods, and fusion rates were collected from all studies and reported.
Results
Six hundred articles were identified, of which 19 were included that reported outcomes of 7 different CBM products. Seven studies evaluated lumbar fusion, 11 evaluated cervical fusion, and 1 study reported adverse events of a single CBM product. Only 4 studies were comparative studies while others were limited to case series. Fusion rates ranged from 68% to 98.7% in the lumbar spine and 87% to 100% in the cervical spine, although criteria for radiographic fusion was variable. While 7 studies reported no adverse events, there was no strict consensus on what constituted a complication. One study reported catastrophic disseminated tuberculosis from donor contaminated CBM. The authors of 14 studies had conflicts of interest with either the manufacturer or distributor for their analyzed CBM.
Conclusions
Current evidence regarding the use of cellular bone matrix as an osteobiologic during spine surgery is weak and limited to low-grade non-comparative studies subject to industry funding. While reported fusion rates are high, the risk of severe complications should not be overlooked. Further large clinical trials are required to elucidate whether the CBMs offer any benefits that outweigh the risks.
Introduction
Two of the most common reasons for revision spine surgery are infection and pseudarthrosis. 1 Therefore, optimization of surgical conditions and biomaterials to enhance fusion rates, without increasing infection, has been a target of recent translational and clinical research. New research and innovation in this area can broadly be divided into 2 distinct platforms: (1) novel designs and/or developments in interbody cage materials and (2) growth-factor carrier/cell-based technologies. 2 In this review, we will focus on the advances in cell-based technologies with a specific emphasis on outcomes and adverse events that must be considered when deciding whether to implement this technology into practice.
Despite osteobiologic incorporation into lumbar fusions, more than 4-20% of lumbar spinal fusions result in pseudarthrosis. 3 The wide range of fusion success can be partly attributed to the differing biologics used for spinal arthrodesis, such as allograft and demineralized bone matrix, which have high variability in their efficacy of bone formation. While some pseudarthrosis may be asymptomatic, symptomatic pseudarthrosis represents a significant source of patient morbidity and health care expenditure and often results in suboptimal patient outcomes even after revision surgery.4,5 One of the earliest biomaterials developed was recombinant human bone morphogenic protein type 2 (rhBMP-2). However, numerous potential complications, including ectopic bone formation and a local pro-inflammatory response have limited its potential use, particularly in the cervical spine and posterior lumbar spine. 6 One of the most serious concerns with rhBMP-2 use is the high rates of local soft tissue inflammation. This can result in radiculitis, seroma and hematoma formation, and surrounding tissue edema potentially resulting in wound dehiscence or complication. 5 Therefore, the United States Food and Drug Administration (FDA) has only approved the use of rhBMP-2 in anterior lumbar interbody fusion (ALIF) titanium cylindrical cages (although most surgeons have expanded this indication) due to potential adverse events with other spine fusion approaches. 7
To mitigate risks associated with rhBMP-2, recent research has focused on cell-based technologies, including targeted cellular bone matrices (CBMs), which are composed of cadaveric allogeneic bone with retained mesenchymal stromal cells (MSCs). One of the benefits of these products include their reduced activation of cytokines compared to rhBMP-2, which minimizes surrounding soft tissue inflammation. 8 Theoretically, cellular bone matrices provide living cells that can promote spinal fusion without inflammatory side effects. Among products available currently in the market, there is a wide variation among CBMs regarding average donor age, total cellular concentration, percentage of MSCs, shelf life, and cell viability – partly owing to the limited efficacy data required prior to commercialization due to their regulatory classification. 2 Moreover, limited safety data leaves a potential for catastrophic complications, as recently observed in the dissemination of tuberculosis from contaminated CBM in over 100 spine fusion patients.9,10 While a systematic review previously attempted to evaluate all available data, an updated review on this topic is warranted in light of the recent events.
Methods
Search Strategy
A search of the MEDLINE/PubMed database was performed on January 31, 2023 for studies evaluating cellular bone matrix in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The search was conducted using the following terms, including commercially available CBMs to ensure capture of all relevant articles: ((cellular bone matrix) OR (map3) OR (Osteocel) OR (Trinity) OR (Bio4) OR (Via graft) OR (Cellentra) OR (ViviGen)) AND ((spinal fusion) OR ((spine) AND (fusion))). No minimum/maximum number of studies was determined prior to conducting the study. The reference lists of review articles, clinical trials, and meta-analyses were reviewed to collect additional relevant studies. Studies were included if they evaluated human subjects and if they specified the CBM utilized in their procedures. Animal, biomechanical, and non-clinical studies were excluded. Similarly, manuscripts that were narrative reviews, letters to the editor, or systematic reviews were also excluded. More stringent criteria could not be established due to the low level of evidence and scarcity of existing literature.
Two reviewers independently screened the identified articles and selected studies for full-text review after screening of the title and abstract. Any articles with discrepancies at this stage were automatically included for full-text review. At the full-text stage, articles were screened for inclusion based on the predetermined inclusion/exclusion criteria. For any articles deemed to meet the inclusion criteria, a search of the references was performed to identify any potentially missed studies. For studies where article inclusion was unclear, a more senior author was consulted.
Data Extraction
All articles included in the final analysis were manually reviewed and analyzed to extract the following data: type of cellular bone matrix utilized, number of patients included in the study, type of fusion, prospective or retrospective nature of the study, complication rate, fusion rates, and length of follow up.
Assessment of Bias
Non-randomized studies were assessed using the Newcastle-Ottawa Scale (NOS), with a high quality study defined with a score greater than or equal to 7. 11 The presence of industry funding was also collected based on review of disclosures listed on the article page and from funding to authors listed on the Centers for Medicare and Medicaid Services Open Payments Data website to assess potential bias from industry. All collected data was reported in a self-designed table.
Results
The initial search resulted in 600 manuscripts. After title and abstract review, 21 articles underwent full text review. After full text review, 2 studies were excluded because they were database studies that did not report the specific CBM utilized or long-term fusion rates.12,13 Nineteen articles were included (Figure 1). The most commonly included CBM was Osteocel Plus,14-19 which was one of the first CBMs on the market. Trinity Evolution,20-23 Vivigen,24-27 map3,
28
Trinity Elite,
29
FiberCel,
9
and VIA30,31 are alternative CBMs that have recently undergone clinical trials and were also included by articles in this study. Only four included articles were comparative studies,18,23,27,27,28 while all other included studies were case series (Table 1). Seven studies evaluated the cervical spine,17-19,21,22,24,27 and eleven evaluated outcomes of the lumbar spine.14-16,20,23,25,26,28-31 One study did not longitudinally follow patients but provided a case series of patients presenting with complications of a specific lot of FiberCel cellular bone matrix.
9
PRISMA Flowchart. Included Studies and Characteristics. ** indicates the study was a case series so risk of bias could not be calculated.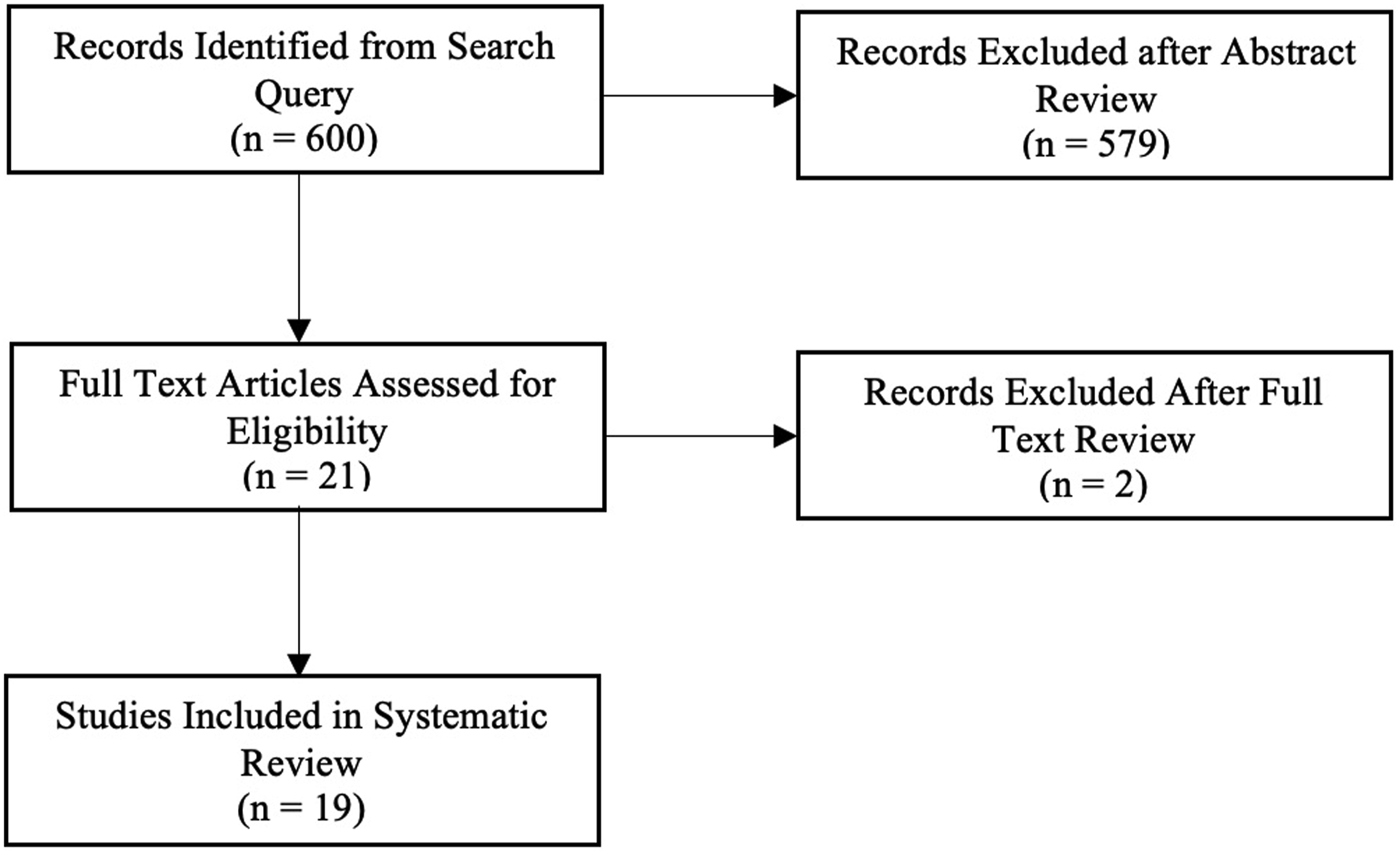
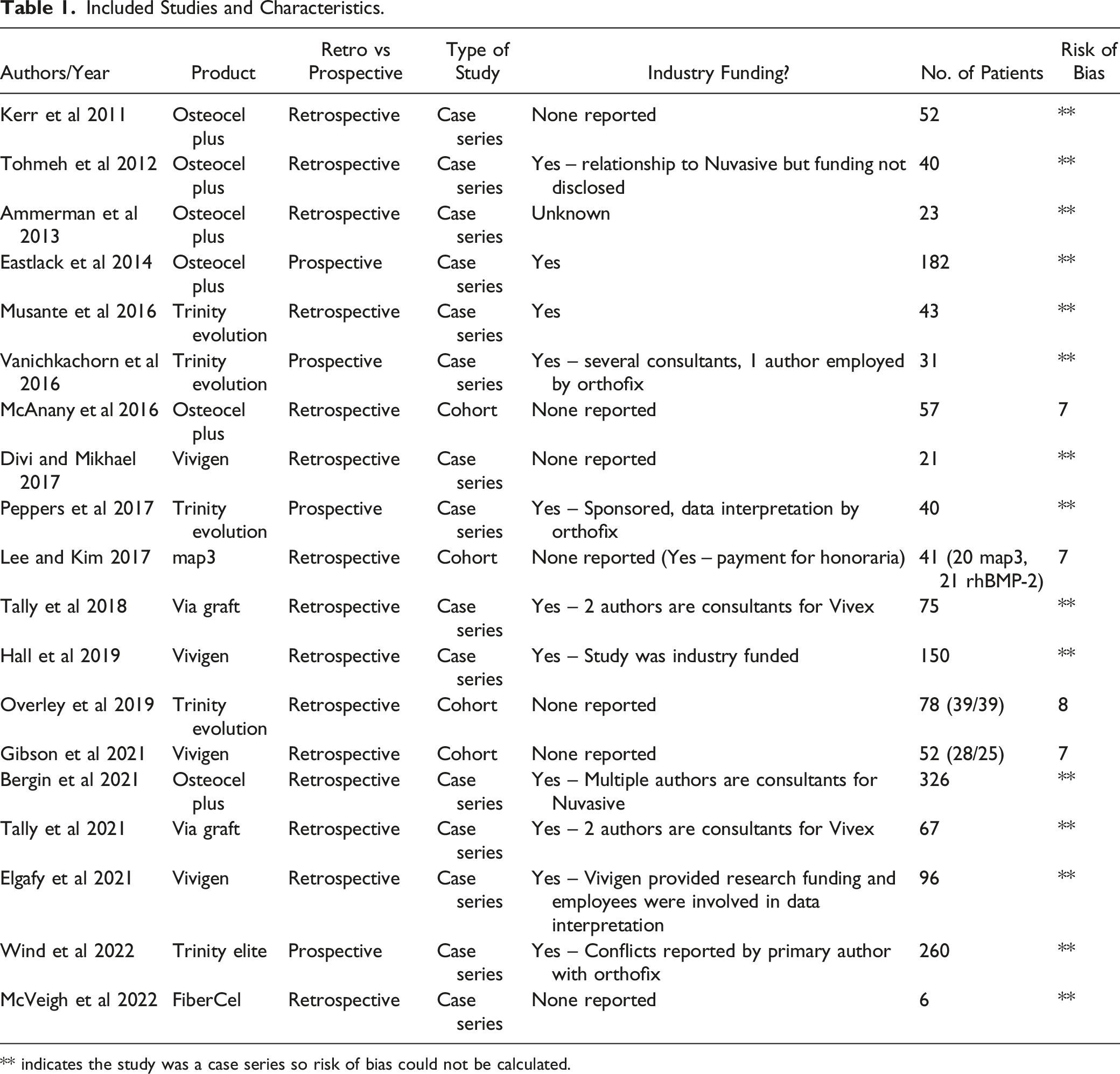
Assessment of Bias
The quality of the 4 comparative studies was deemed to be high utilizing the Newcastle-Ottawa Scale.18,23,27,27,28 The authors of 14 of the studies had conflicts of interest with either the manufacturer or distributor for their analyzed CBM. Conflicts of interest for one study could not be identified since publication was prior to creation of the Open Payments Data website. Of studies with potential industry bias, three were industry-funded,22,23,26 and two had authors involved in data interpretation who were employees of the CBM company.21,26
Fusion Rates
Reported Fusion Rates of Cellular Bone Matrix.
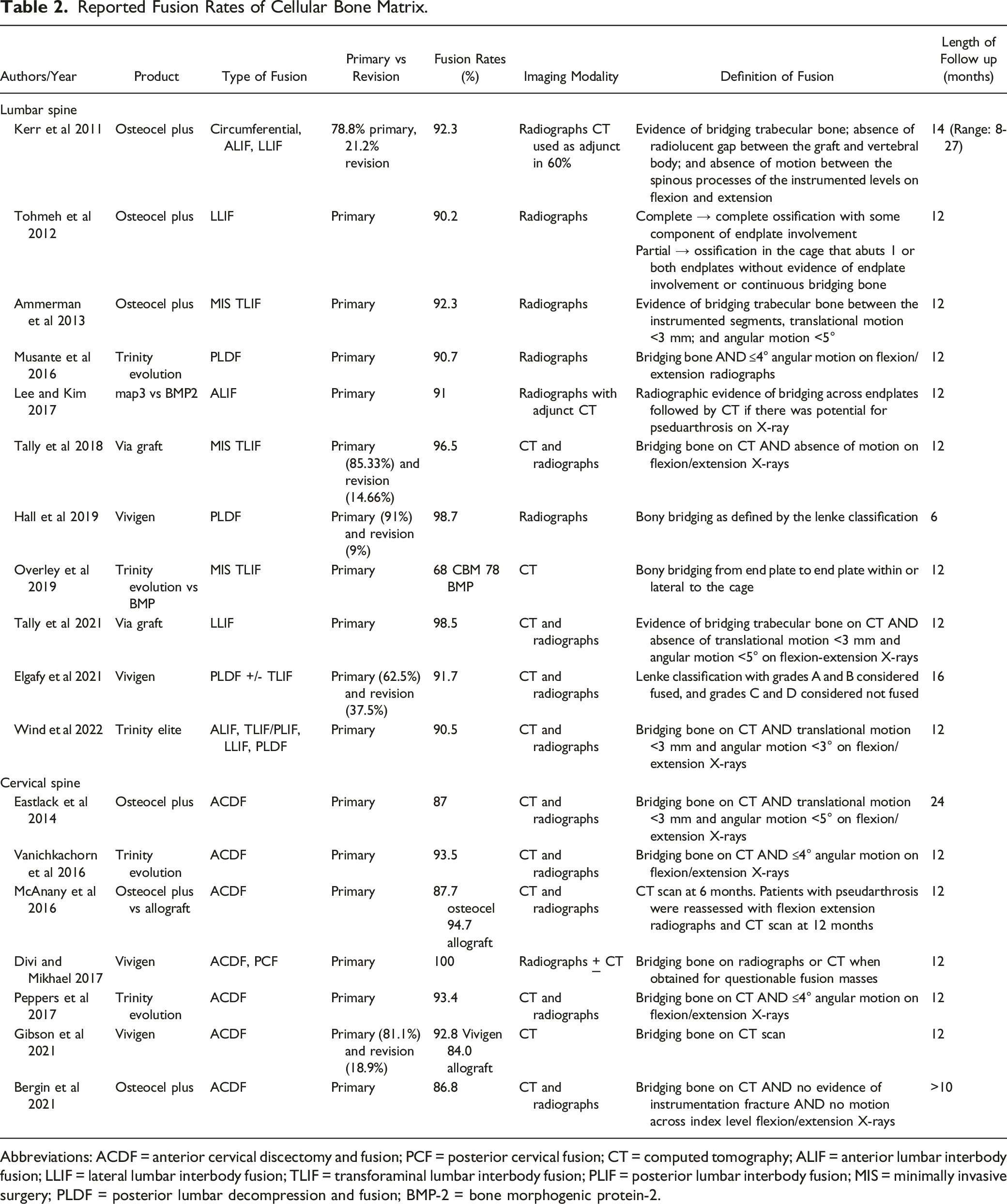
Abbreviations: ACDF = anterior cervical discectomy and fusion; PCF = posterior cervical fusion; CT = computed tomography; ALIF = anterior lumbar interbody fusion; LLIF = lateral lumbar interbody fusion; TLIF = transforaminal lumbar interbody fusion; PLIF = posterior lumbar interbody fusion; MIS = minimally invasive surgery; PLDF = posterior lumbar decompression and fusion; BMP-2 = bone morphogenic protein-2.
Among the comparative analyses directly comparing a cellular bone matrix to a control cohort, no differences were found in fusion rates. Lee and Kim reported that 12-month fusion rates of map3 were similar to rh-BMP2 after anterior lumbar interbody fusion (91% vs 91%, P = .89). 28 Overley et al. compared the Trinity Evolution CBM to standard allograft for patients undergoing minimally invasive transforaminal lumbar interbody fusion (TLIF) and similarly identified no difference in CT-confirmed fusion rates (68% vs 78%, P = .35). 23 In patients undergoing ACDF, Gibson et al. similarly identified no difference in fusion rates between Vivigen CBM and decellularized allograft at 1-year. 27 1 study did not compare fusion rates of cellular bone matrix to another biologic but found that Osteocel implanted in conjunction with allograft during primary ACDF resulted in significantly lower pseudarthrosis rate than when implanted with a PEEK cage (8.4% vs 16.4%, P = .04). 19 While Tohmeh and colleagues did not conduct a comparative study, they suggested that the rate of fusion for Osteocel Plus was higher than selected studies in the literature reporting fusion rates with autograft. 15
Complications
All but one study reported complication and revision rates. 26 Seven of the included studies reported no adverse events or graft-related complications.15,16,18,20,22,30,31 Complication rates were highly variable as no strict definitions of what constitutes a complication existed. Therefore, it was up to the authors’ discretion to decide whether adjacent segment disease and graft related issues were complications or the natural history of degenerative disease.
Reported Complication and Revision Rates of Cellular Bone Matrix.
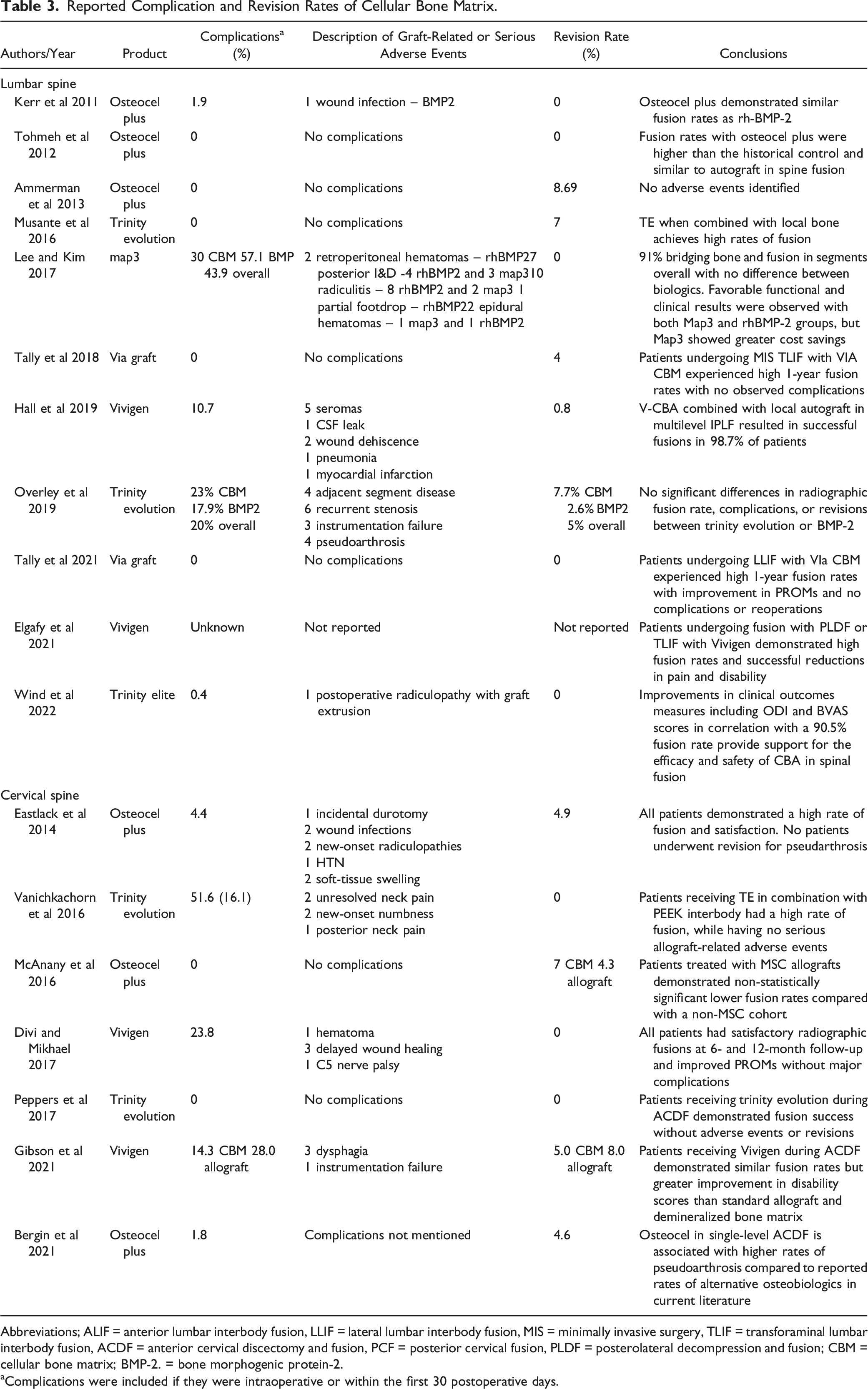
Abbreviations; ALIF = anterior lumbar interbody fusion, LLIF = lateral lumbar interbody fusion, MIS = minimally invasive surgery, TLIF = transforaminal lumbar interbody fusion, ACDF = anterior cervical discectomy and fusion, PCF = posterior cervical fusion, PLDF = posterolateral decompression and fusion; CBM = cellular bone matrix; BMP-2. = bone morphogenic protein-2.
aComplications were included if they were intraoperative or within the first 30 postoperative days.
McVeigh et al presented a case series of 6 cervical spine surgery patients presenting with tuberculosis that were found to be inoculated from the donor source cadaver for the FiberCel CBM. 9 All patients in their series tested positive for spinal tuberculosis. Two patients presented with wound dehiscence, 2 with prevertebral abscess, 1 with an epidural abscess, and 1 was asymptomatic and presented after the recall was issued. Four patients underwent incision and drainage, and 1 required a subsequent revision spine fusion. Five patients did well postoperatively for up to 1-year follow-up and completed medical tuberculosis therapy while 1 died at 6 months from hypoxemic respiratory failure due to COVID-19.
Discussion
Improving the local biological healing environment has been a long-standing goal of spine surgeons wishing to optimize spinal fusion and reduce complication rates, namely pseudarthrosis. Historically, this has included incorporating iliac crest autograft, which was considered the gold standard for bone grafting, but donor site morbidity, increased operative length, and potentially equally efficacious bone allograft and osteobiologic options have shifted surgeons away from iliac crest autograft.3,32 The earliest CBM available was Osteocel Plus,
14
derived from MSCs which are multilineage, pluripotent cells that can differentiate into osteoblasts in the setting of the correct cellular microenvironment. These characteristics differentiate CBMs from other osteobiologics that focus solely on providing growth factors or osteoconductive alternatives for promotion of bony fusion (Figure 2). While bone marrow has varying amounts of MSCs depending on a number of factors, including age, it is estimated that the average healthy adult has 1 MSC for every 50,000 nucleated cells.
33
Until recently, concerns with sterilization, cost, ex vivo expansion, and culture time limited its potential for clinical impact. In our systematic review, we identified that while a growing body of literature has highlighted the successful utilization of CBM in spinal fusion, the literature consists of low quality evidence that largely lacks comparative effectiveness of CBM to bone allograft, autograft, or rhBMP-2. Due to this limitation, a meta-analysis of the available data is not possible at this time. Further, CBM has the potential for catastrophic adverse events as demonstrated by the recent report on 6 patients infected with TB from a contaminated lot of CBM that resulted in multiple deaths and over 100 infected individuals in a widespread outbreak raising serious questions about the risk-reward profile for including this adjuvant to promote bone healing. Illustration of the progression of mesenchymal stem cells to osteoblast progenitor cells with the support of various transcription factors. Republished with permission of Elsevier Books, from Mesenchymal Stem Cells and Osteoblast Differentiation. Aubin J. In Principles of Bone Biology, third ed. 2008, permission conveyed through Copyright Clearance Center, Inc.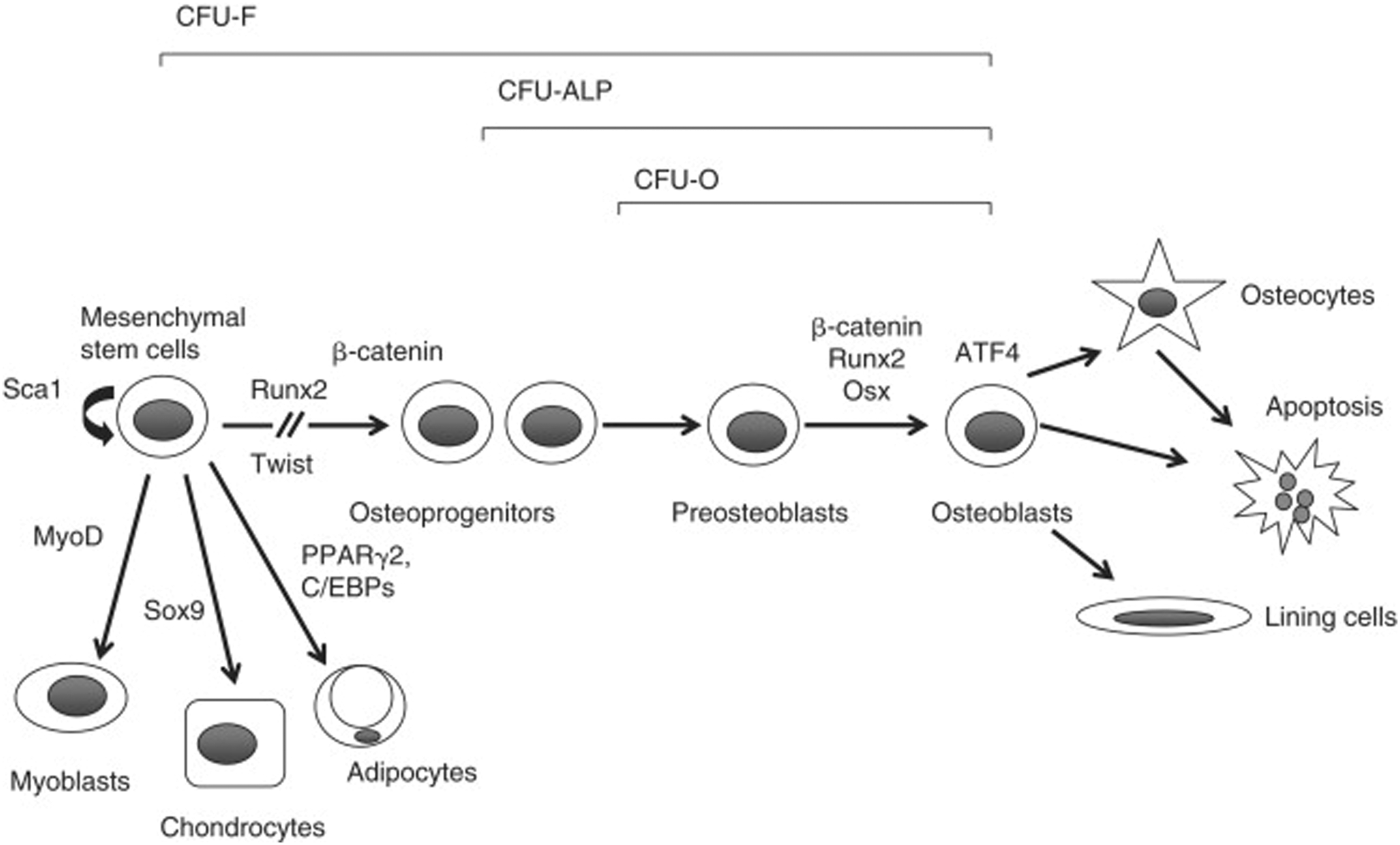
The Regulatory Environment
In the United States, Section 361 of the Public Health Service (PHS) Act states that the Secretary of Health and Human Services has the autonomy to take appropriate measures to prevent the introduction and spread of transmissible disease from both foreign and state threats. 34 The Code of Federal Regulations determines the exact criteria for the “human cells, tissues, and cellular and tissue-based products” (HCT/P) to be solely regulated under Section 361 of the PHS: (I) the tissue is minimally manipulated; (II) the tissue is only intended for homologous use; (III) no additives including live cells or additional tissues are included in the HCT/P unless they are water or a sterilizing or preserving agent; and (IV) the primary function of the HCT/P does not depend on the metabolic activity of living cells and there is no systemic effect from the HCT/P or the systemic effect from the HCT/P is because the HCT/P depends on the activity of the living cells to fulfill its function and the HCT/P is for autologous use, reproductive use, or allogeneic use in a first- or second-degree relative. In summary, HCT/Ps consist of human cells or tissues that are intended for implantation, transplantation, infusion or transfer into a recipient patient and whose processing has not altered their utility for reconstruction, repair, or replacement. These guidelines regarding homologous use and minimal manipulation ultimately allow the FDA to determine which products receive the 361 HCT/P product designation and are permissible for human implantation without the more stringent regulatory clearance—through the premarket approval (PMA) or 501(k) pathways— required for biologics designation.
It also bears consideration that part IV of 1271.10 states that the HCT/P should not be dependent on “living cells” to fulfill its function for it to qualify for inclusion under section 361. However, MSCs are the key component of CBMs. The MSCs are specifically included in CBMs due to their theoretical osteogenic ability and are combined with allogeneic bone matrix. The current treatment of CBMs as HCT/Ps under PHS Act Section 361 despite the premise of “living cells” does warrant further consideration. In the last year, the FDA has reported that they are exploring the creation of new regulatory pathways for certain cellular products. 35
Complication Profile
In our analysis, complication rates were highly variable as there was no strict alignment in reporting complications amongst the included studies, thus, what may classify as a “serious adverse event” or “graft-related” complication varied among publications. Seven of the included studies reported no adverse events or graft-related complications.15,16,18,20,22,30,31 Even though 8 studies reported no reoperations at final follow-up, there was a trend towards higher reoperation rates in patients given CBMs compared to rhBMP-2 and traditional structural allograft. 23 However, this difference was not statistically significant and emphasizes the need for high quality larger-cohort comparative effectiveness studies.
Importantly, the risk of communicable disease transmission does warrant further consideration. CBM products stratified into the HCT/P category are deemed to have minimal potential communicable disease transmission risk by the FDA. 34 While potential donors are screened based on a combination of reviews of medical and social history with physical exams and testing for several lab-based communicable diseases, tuberculosis has not historically been mandated by the FDA. In 2021, over 100 patients were potentially exposed to mycobacterium tuberculosis (TB) from contaminated FiberCel allograft from a single donor. While an immediate urgent recall of all potentially infected graft was issued, patients at numerous institutions required significant surgical and medical management for spinal tuberculosis and wound complications. 10
Regardless of donor history, after cells are obtained, they must undergo sterilization. Gamma irradiation with cobalt-60 is widely used for commercial allografts, however, this may affect cellular viability.36,37 While alternative sterilization methods include thermal heat, ethylene oxide, and chemical processing,36,38 these may affect cellular viability, an area that requires further study for CBM. If CBM is to increase in utilization, large studies must evaluate its safety and further research should evaluate the effectiveness of various sterility methods while preserving the cellular viability necessary for the efficacy of CBM.
Fusion Rates
Current evidence regarding the effectiveness of CBMs in spinal fusion have predominantly been limited to translational research and prospective or retrospective case studies. Translational research has indicated CBMs provide similar fusion rates compared to iliac crest autograft without increasing complication rates. 39 Our analysis found that 10 out of 11 studies reported fusion rates above 90% in the lumbar spine with CBM augmentation, while 1 reported a fusion rate of 68%. In the cervical spine, reported fusion rates ranging from 87-100% with CBM augmentation.
Only 4 comparative studies are available in the literature comparing CBM to another standard of care. Comparing CBM to standard allograft during ACDF, McAnany et al found a nonsignificantly higher rate of pseudoarthrosis in patients with CBM allografts, 18 while Gibson et al. reported similar long-term fusion rates. 27 Notably, these both were not industry-sponsored studies that failed to report benefit to CBM usage. Overley et al. evaluated the 1 year radiographic fusion outcomes for 78 patients undergoing MI-TLIF with either rhBMP-2 or CBM placed in an interbody cage. 23 While the overall fusion rate was 68% in the CBM group and 78% in the rhBMP-2 group, this did not produce a statistically significant difference. However, CBM did not appear to provide superior outcomes compared to rhBMP-2. Similarly, Lee and Kim compared radiographic and clinical outcomes of CBM vs rhBMP-2, but in a cohort undergoing ALIF. 28 CBM allograft demonstrated equivalent fusion rates to rhBMP-2 in 1 to 3 level ALIF, but with a potentially lower rate of wound complications. These 4 studies underscore the low-quality of evidence that has previously supported the use of CBM. None of these studies provided substantial clinical benefit to utilization of CBM to other standard and lower-cost alternatives.
In studies that used radiographs plus adjunct CT, an average 92.0% fusion rate was reported. Studies that used radiographs only reported an average fusion rate of 94.4%. In all but 2 studies, bony fusion was assessed at greater than 1 year follow-up.19,25 However, the specific criteria for nonunion varied significantly between studies. Some followed the FDA guidelines of radiographically defined successful lumbar fusion as less than 3 mm of translational motion and less than 5° of angular motion on flexion and extension radiographs. 40 But controversy still exists regarding both the degree of motion utilized to define pseudoarthrosis and the optimal method of assessment. Moreover, while CT scans demonstrate the greatest accuracy, they still demonstrate inaccuracy compared to surgical exploration.41,42 High-quality comparative studies are required to demonstrate the superior fusion rates that these case series suggest.
Cost Effectiveness of CBMs
Despite limited evidence documenting improvements in clinical outcomes or spinal fusion rates when incorporating CBM into a fusion construct, CBMs are associated with significantly higher costs compared to other available products. 43 The estimated cost of CBM is $525 per cm3 compared to $17 for cancellous bone chips and $151 for DBM. 43 It should be noted that rhBMP-2 also carries a large cost, estimated at $10,444 per primary fusion and its cost-effectiveness for primary lumbar fusions is questionable until the product’s cost is reduced by 50%. 44 Further analysis of the incremental cost-effectiveness of rhBMP-2, iliac crest autograft, local bone autograft, allograft chips and DBM were analyzed using a Markov decision model in patients undergoing one-level lumbar fusion for degenerative spondylolisthesis. RhBMP-2 was found to be the most cost-effective option ($16,595 per quality adjusted life year (QALY)), while cancellous allograft was least cost-effective ($28,153 per QALY), with the remaining options all costing approximately $21,000 per QALY. 45 The above studies attempting to identify the cost-effectiveness of biologics demonstrate the heterogeneity present in the currently available literature. To date, no high-quality studies demonstrate the superiority of one bone graft alternative over another. Although limited data exists regarding the cost-effectiveness of CBM, one retrospective study has demonstrated its use is at least mildly cost-effective when compared to rhBMP-2.12,28
Industry Funding and Potential Influence
Studies currently available on CBMs are heavily influenced by industry. To date, most clinical evidence surrounding CBMs are supported by industry funding, thus, the results of the trials must be met with some skepticism. For example, complications are uncommon in most industry sponsored studies. In some instances, the list of complications were nebulous, making it difficult to confidently determine if complications were related to CBM or were due to unresolved perioperative symptoms. 17 Industry’s influence on complication rates can best be seen when comparing studies with conflicts of interest compared to those without. Studies with conflicts of interest reported no complications or much lower complication rates (0-10.7%) compared to those without conflicts of interest (23.8-30%). Regardless of conflict of interest, fusion rates are routinely greater than 90%, but it should be noted the previous CBM studies are mostly exploratory without comparative groups to determine if CBMs are more effective at promoting fusion.
Limitations
As addressed above, the current systematic review does have several limitations due to the nature of the existing literature. While there has been an increase in the literature on CBM, most of the studies were smaller cases series and did not compare the effectiveness of CBM to more widely-used alternatives, such as allograft or rhBMP-2, making a meta-analysis untenable. Although most of the included studies did report complication rates, these were not standardized, making comparisons difficult. Moreover, as there were only 4 comparative studies found to be high quality as per the Newcastle-Ottawa Scale and most of the studies had authors with industry-related conflicts of interest, not enough literature exists at this time for an unbiased review of CBM use.
Conclusions
While cellular bone matrices may play an adjunct role with limited indications for use in the promotion of spinal fusion, current evidence does not support its usage in routine clinical care due to the risk of catastrophic events with limited safety and efficacy data in the current regulatory environment. There is insufficient high-quality evidence to demonstrate that use of CBMs is more cost-effective, provides superior clinical outcomes or reduces pseudoarthrosis, compared to other bone graft alternatives. Moreover, the available evidence remains heavily influenced by industry conflict of interest and funding. There is need for unbiased prospective large cohort clinical trials to elucidate whether the CBMs offer any benefits compared to other alternatives that outweigh the risks.
Footnotes
Author’s Contribution
All authors significantly contributed to the document and have reviewed the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
